Abstract
Treatment patterns in migraine patients with cardiovascular risk factors are largely unknown. A retrospective observational study was conducted to characterize the baseline cardiovascular risk profile of new users of specific abortive migraine drugs, and to investigate treatment choices and patterns in patients with and without a known cardiovascular risk profile. New users of a triptan, ergotamine or Migrafin® (n = 36 839) from 1 January 1990 to 31 December 2006 were included. Approximately 90 of all new users did not have a clinically recognized cardiovascular risk profile. The percentage of new users with a cardiovascular risk profile did not differ between new users of a triptan, ergotamine or Migrafin® and also did not change during the study period of 17 years. Differences in treatment choices and patterns between migraine patients with and without a known cardiovascular risk profile reveal a certain reticence in prescribing vasoconstrictive antimigraine drugs to patients at cardiovascular risk.
Introduction
It is well known that ergotamine can lead to serious ischaemic adverse effects such as peripheral ischaemia, arterial stenosis, myocardial infarction and cerebral ischaemia (1–4), probably due to its broad pharmacological activity involving serotonin (5HT1 and 5HT2), dopamine and α-adrenoceptors. For a long time ergotamine was the only specific migraine abortive drug available, and the potential for these complications has always been a limitation to its use, especially in patients with a cardiovascular risk profile. The selective 5-HT1B/1D agonists, known as triptans and available from the 1990s onwards, have improved the quality of acute migraine treatment by providing a higher degree of efficacy and a more favourable side-effect profile than ergotamine. However, soon after the introduction of sumatriptan, reports began to appear of angina-like chest symptoms (5, 6). Data from long-term open-label clinical trials and large postmarketing studies suggested that the vast majority of triptan-related chest symptoms were non-cardiac in origin (7–9). Nevertheless, case reports of serious ischaemic complications such as myocardial infarction (10–13), ischaemic stroke (14, 15) and ischaemic colitis (16–18) were published related to triptan use, mostly in patients with cardiovascular disease risk factors. Therefore, the use of triptans, like ergotamine, is contraindicated in these patients.
Subsequent observational evidence has shown that the incidence of cardiovascular adverse events is low when triptans are used appropriately (19–21). A recent case–control study has shown that regarding vasoconstrictive complications, triptan use and even its overuse did not increase the risk for these complications, even in patients simultaneously using cardiovascular drugs. Overuse of ergotamine, however, significantly increased the risk for these complications, especially in those simultaneously using cardiovascular drugs (22).
Prescribers' concerns about the cardiovascular safety of ergotamine and triptans may lead them to choose other treatment options first, such as non-steroidal anti-inflammatory drugs (NSAIDs) or migraine prophylaxis, before prescribing the specific migraine abortive drugs to patients at cardiovascular risk. In addition, treatment patterns of antimigraine drugs themselves, such as the intensity of use, may differ. Due to growing evidence that the incidence of triptan-associated serious cardiovascular adverse events in both clinical trials and clinical practice appears to be extremely low, this concern, and therefore also prescribing behaviour, may have changed over time. However, treatment patterns in migraine patients with cardiovascular risk factors remain largely unknown.
This retrospective observational study was conducted to characterize the baseline cardiovascular risk profile of new users of specific abortive migraine drugs and its change over time. Treatment choices and patterns were investigated in patients with and without a clinically recognized cardiovascular risk profile.
Methods
Setting
We reviewed data from the PHARMO record linkage system, which includes several databases and links drug dispensing records and hospital records from currently more than two million individuals in defined areas in the Netherlands (23). The pharmacy database consists of a sample of > 200 pharmacies in > 50 regions scattered over the Netherlands and is representative of the Netherlands with respect to age, gender and healthcare consumption. Currently, it covers data of more than 2 million residents regardless of type of insurance, corresponding to 12% of the Dutch population (24). Since virtually all patients in the Netherlands are registered with a single community pharmacy, independent of prescriber, pharmacy records are virtually complete with regard to prescription drugs. Members of the PHARMO population enter the database with the first prescription filled in a PHARMO community pharmacy and are followed until the last prescription.
The computerized drug dispensing histories contain information concerning the dispensed drug, dispensing date, the prescriber, amount dispensed, prescribed dosage regimen and the estimated duration of use. The duration of use of each dispensed drug is estimated by dividing the number of dispensed units by the prescribed number of units to be used per day. All drugs are coded according to the Anatomical Therapeutic Chemical Classification. Patient information per prescribed medicine includes gender and date of birth. The database does not provide information concerning the indications for use of the medicines, in this case the diagnosis of migraine vs. cluster headache, or accurate registration of non-prescription medicines [e.g. over-the-counter (OTC) use of salicylates, NSAIDs or paracetamol].
The hospital discharge records were obtained from PRISMANT, previously known as the Dutch Centre for Healthcare Information (LMR database), an institute that collects nationwide all hospital discharge records in the Netherlands since the 1960s in a standardized format (24). These records include detailed information concerning the discharge diagnoses, diagnostic, surgical and treatment procedures, type and frequency of consultations with medical specialists and dates of hospital admission and discharge. All diagnoses are coded according to the International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM).
Study population
For this study, all patients with at least one dispensed prescription for either a triptan, ergotamine (alone or in combination with caffeine and/or cyclizine), or Migrafin® (a combination drug of 900 mg acetylsalicylic acid and 10 mg metoclopramide, approved for acute migraine treatment since 1996 in the Netherlands) from 1 January 1990 to 31 December 2006, were initially identified from the PHARMO system. To identify ‘new users’ of these specific migraine abortive drugs the date of the first dispensed prescription for a triptan, ergotamine or Migrafin® in that time period was termed the ‘start date’. Patients were only included in the present study if, at the start date, the patient had at least 1 year's history in PHARMO and if the patient was ≥ 18 years old.
The following characteristics were evaluated at the start date: gender, age, prescriber, use of NSAIDs during the year preceding the start date, and use of migraine prophylactics [propranolol, metoprolol (since in the Netherlands propranolol and metoprolol are the only β-blockers mentioned in guidelines for migraine prophylaxis), valproic acid, clonidine, methysergide, pizotifen, flunarizine, topiramate] during the year before the start date.
Patients who used both ergotamine and triptans or Migrafin® were categorized as ‘switchers’ only if the last dispensing date of the drug type (ergotamine, triptan or Migrafin®) dispensed on the start date was before the first dispensing date of the drug type were one switched to. Therefore, during the period before switching, only one drug type (ergotamine, triptan or Migrafin®) was used. For switchers the date of the first presented prescription of the drug, were one switched to, was termed the ‘switch date’.
Cardiovascular risk profile
The determinant of interest was a clinically recognized cardiovascular risk profile at the start of the treatment with migraine abortive drugs. This was determined on the basis of the use of cardiovascular drugs as well as hospitalizations for cardiovascular reasons.
The use of cardiovascular drugs was assessed during the 6 months before the start date. This window was set to indicate simultaneous use of the various types of drugs. A 6-month time window is long enough to observe at least one dispensing in chronic medication, but not too long for drugs used sequentially, rather than simultaneously.
Dispensing of the following drugs during the 6 months before the start date was identified: renin–angiotensin–aldosterone system inhibitors, β-blockers—except propranolol and metoprolol—calcium channel blockers, diuretics, nitrates, cardiac glycosides, oral anticoagulants, antiplatelet therapy, lipid-lowering therapy and oral antidiabetics. Except for dispensing of the individual drugs, combination therapy was be identified: zero, one, two, three and four or more drugs in the above-mentioned categories.
In addition, hospitalizations due to a cardiovascular event during 1 year preceding the start date were assessed. The included discharge diagnoses were: hypertensive disease [essential hypertension (ICD-9-CM code 401), hypertensive heart disease (ICD-9-CM code 402), secondary hypertension (ICD-9-CM code 405)], ischaemic heart disease (ICD-9-CM codes 410-414), heart failure (ICD-9-CM code 428), cerebral ischaemia (ICD-9-CM codes 433-436, 437.0, 437.1 and 437.6) and atherosclerosis (ICD-9-CM code 440).
For switchers, in the same way, the cardiovascular risk profile was also determined preceding the switch date.
Treatment choices and patterns
Different treatment patterns between new users were identified, all stratified to patients with and without a cardiovascular risk profile. The use of other migraine treatment options before the start of a specific abortive migraine drug was assessed by determining the use of NSAIDs or migraine prophylactics (see section on Study population) during the year before the start date.
After the start date we looked at different patterns of specific abortive migraine drug use. First, the prevalence of single use (only one prescription) was determined. Second, for non-incident users (repeated prescriptions) with at least 1 year's follow-up after the start date, the intensity of triptan, ergotamine and Migrafin® use during the first year after the start of the antimigraine drug was determined. To express the intensity of use, for each patient the total consumption of drugs was estimated by the sum of defined daily doses (DDDs) of triptans, ergotamine and Migrafin® dispensed during this first year (25). One DDD was defined as 6 mg sumatriptan parenteral, 50 mg sumatriptan oral, 25 mg sumatriptan rectal, 20 mg sumatriptan nasal, 2.5 mg naratriptan, 2.5 mg zolmitriptan, 10 mg rizatriptan, 12.5 mg almotriptan, 40 mg eletriptan, 2.5 mg frovatriptan, 4 mg ergotamine single preparation by any route, 2 mg ergotamine combination preparation by any route and one tablet Migrafin®. Patients were subsequently categorized according to the intensity of use: > 0–< 12, 12–< 36, 36–< 72 and ≥ 72 DDDs dispensed during this year. Finally, we assessed switch behaviour of new users: switch from ergotamine or Migrafin® to a triptan, switch from a triptan or Migrafin® to ergotamine and switch from ergotamine or a triptan to Migrafin®. For definition of ‘switchers’ we refer to the section on Study population.
Data analysis
For all new users of antimigraine drugs the prevalence of each characteristic was determined on the start date. Logistic regression was used to estimate the strength of the differences in treatment characteristics between migraine patients with and without cardiovascular risk, expressed as odds ratios (ORs), crude and adjusted for age, with 95% confidence interval (CI). Microsoft Access, a relational database software package, was used for database management and internal quality procedures. All statistical analyses were performed with
Results
A total of 72 743 patients had been dispensed a triptan, ergotamine or Migrafin® during the study period 1990–2006. Patients with < 1 year of medication history (n = 33 432), patients < 18 years old (n = 2376) and patients who started two types of drug (e.g. ergotamine and a triptan) on the same date (n = 96) were excluded, resulting in a final study population of 36 839 new users of specific abortive migraine drugs. Characteristics of the study population at the start date are described in Table 1. There were no remarkable differences between new users of triptans, ergotamine or Migrafin® with respect to age distribution, gender, prescriber and history of prescription NSAID use or history of migraine prophylaxis.
Baseline characteristics of new users (n = 36 839) of triptans, ergotamine or Migrafin® ∗

∗A combination drug of 900 mg acetylsalicylic acid and 10 mg metoclopramide, approved for acute migraine treatment in 1996 in the Netherlands.
NSAID, non-steroidal anti-inflammatory drug.
Table 2a shows that 88–90% of all new users did not have a clinically recognized cardiovascular risk profile; 6.4–7.1% used one drug and 3.7–5.4% used more than one drug for cardiovascular risk management during the period of 6 months before the start date. Only 0.2–0.4% of all new users had experienced a hospitalization due to a cardiovascular event during 1 year preceding the start date. For switchers the cardiovascular risk profile was determined preceding the switch date: 2408 persons switched from ergotamine or Migrafin® to a triptan, 137 switched to ergotamine and 545 switched to Migrafin®. Cardiovascular risk profiles of these switchers were comparable to the profiles of new users (data not shown). Table 2b shows that the use of drugs for cardiovascular risk management was age dependent. There were no major differences between the three treatment groups, except that in the group > 60 years old ergotamine users had 1.3 times more frequently no use of drugs for cardiovascular risk management than starters of triptans or Migrafin®.
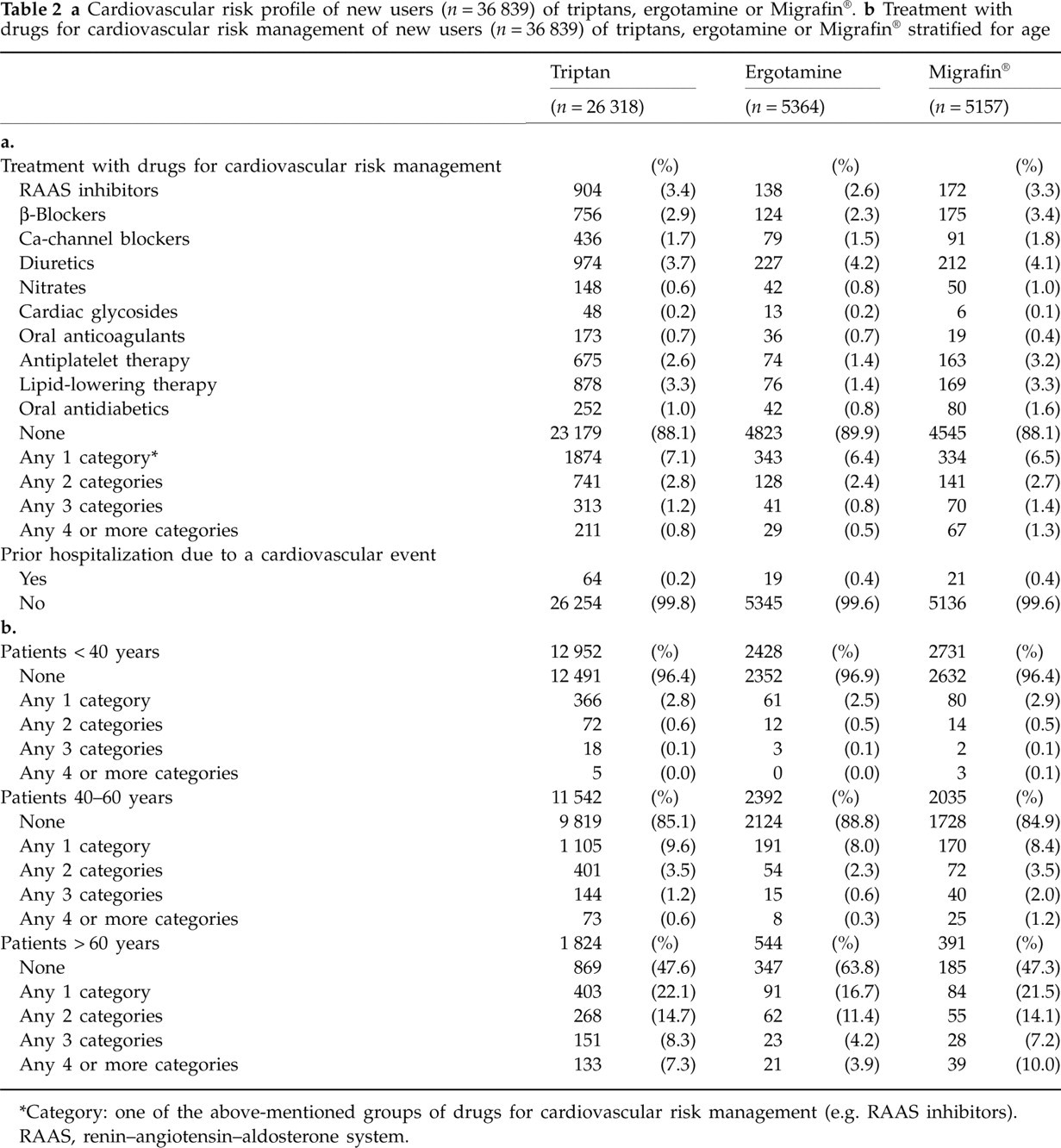
∗Category: one of the above-mentioned groups of drugs for cardiovascular risk management (e.g. RAAS inhibitors).
RAAS, renin–angiotensin–aldosterone system.
Figure 1 shows that the percentage of new users with a known cardiovascular risk profile at the moment a triptan, ergotamine or Migrafin® was started did not change over calendar time. This was also the case when these three groups were observed separately (figures not shown). The average age of patients starting one of the study drugs remained constant over the entire study period (38.8–42.8 years). During the years we observed an almost complete substitution from ergotamine to triptans. In 1991 28.2% of the new users started with a triptan, 71.8% with ergotamine and 0% with Migrafin®, compared with, respectively, 94.9, 0.4 and 4.7% in 2006. After its introduction in 1996, Migrafin® showed in 1997 and 2000 a maximum of 28% market share, declining rapidly afterwards to < 5% in 2006.
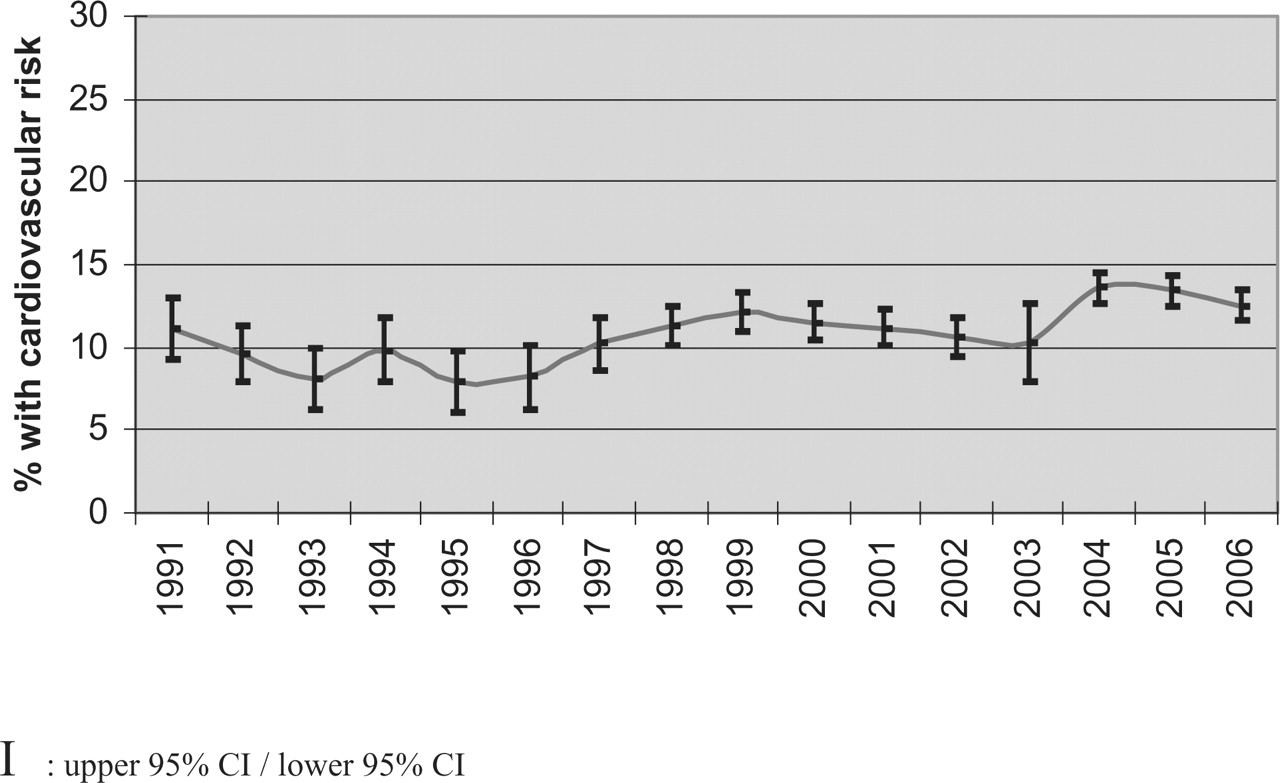
Percentage new users of specific abortive migraine drugs with a known cardiovascular risk profile: change over time. I, upper 95% CI/lower 95% CI.
Treatment choices and patterns are shown in Table 3. During the year preceding the first prescription of an abortive migraine drug, patients with a cardiovascular risk profile used 2.3 (95% CI 2.1, 2.5) times more often drugs for migraine prophylaxis and 1.4 (95% CI 1.3, 1.5) times more often NSAIDs than patients without a known cardiovascular risk profile. Single use of an abortive migraine drug was more frequently seen in patients with cardiovascular risk. The intensity of use of abortive migraine drugs was not different between patients with and without cardiovascular risk. Switch to a triptan was less often seen in patients with cardiovascular risk than in patients without cardiovascular risk (OR 0.67, 95% CI 0.56, 0.79).
Treatment choices and patterns of migraine patients with and without a known cardiovascular risk profile

∗Adjusted for age.
Single use is defined as delivery of just one prescription (first prescription = last prescription).
DDD, defined daily dose; intensity of use of non-incident users (repeated prescriptions) is presented; NSAID, non-steroidal anti-inflammatory drug.
Discussion
Our research has shown that since the introduction of sumatriptan in 1991 in the Netherlands, almost 90% of new users of triptans and ergotamine have no clinically recognized cardiovascular risk profile. This pattern did not change during the study period. Comparing treatment choices and patterns between patients with and without a cardiovascular risk profile, the more frequent history of use of drugs for migraine prophylaxis and NSAIDs in patients with cardiovascular risk factors during the year preceding the start of an abortive migraine drug reveals a certain reticence in prescribing vasoconstrictive antimigraine drugs to these patients. Once treatment with a specific antimigraine drug was accepted by the doctor and patient, the intensity of use did not differ between patients with and without a cardiovascular risk profile.
Our data about prescribing triptans to patients with a clinically recognized cardiovascular risk profile are of the same order as findings from a population-based study in the UK: 5.9% hypertension, 3.1% cardiac disease and 0.8% diabetes among triptan-treated migraine patients (21). A population-based study in France showed a higher percentage: 16% of new triptan users had known cardiovascular risk factors (26). The study from the UK also showed that triptans were selectively prescribed to those less at risk for cardiovascular events (21). No difference in intensity of use after accepting the treatment was also seen in a recent nested case–control study, which showed that patients who used cardiovascular drugs were dispensed as much specific antimigraine drugs as those without cardiovascular drug use (22).
Our analysis is limited by the fact that, due to several reasons, the cardiovascular risk profile in our population may be underestimated. First, we considered only clinically recognized cardiovascular risk, since cardiovascular disease unknown to the treating physician cannot influence treatment decisions. Population-based studies show that a great part of cardiovascular disease—such as hypertension and diabetes—are undetected and untreated (27, 28). Other cardiovascular risk factors such as smoking, family history, obesity and physical condition were unknown. However, this misclassification is likely to be non-differential over time and non-differential between different exposure groups. Furthermore, different validity studies indicate that certain conditions may not be accurately reflected by discharge ICD-9 codes. However, studies investigating the sensitivity and the positive predictive value (PPV) of ICD-9 codes 434 and 436 have found sensitivity rates of 76 and 82% and PPVs between 71 and 85% (29, 30). ICD-9 code 433 showed a low PPV of 15% (30). In our final study population only three patients showed hospitalization for an event diagnosed by code 433 1 year before the start of an antimigraine drug. Considering our low hospitalization percentages, this means that, at least for these ICD codes, our data are hardly influenced by this degree of invalidity. Dutch validity studies for the ICD-9 discharge codes we used have not been published so far.
Second, due to fact that our database does not contain information about the indication for use of medicines, in a proportion of patients propranolol and metoprolol will be used as cardiovascular drugs and incorrectly categorized as migraine prophylactics. This might have led to overestimation of prophylactic use in both the cardiovascular and non-cardiovascular group (propranolol and metoprolol monotherapy). Therefore, the OR of 2.3, indicating a reticence in prescribing vasoconstrictive antimigraine drugs to migraine patients with a cardiovascular risk profile, has to be interpreted carefully. Furthermore, since the PHARMO record linkage system is based upon dispensings from community pharmacies, it does not provide an accurate registration of non-prescription (i.e. OTC) NSAIDs. This means that the frequency of NSAID use in both the cardiovascular and the non-cardiovascular group will be underestimated. It is, however, unlikely that this misclassification is differential. We believe therefore that the risk estimate is still valid.
Another limitation is the fact that our study population consisted of patients who started with a specific abortive migraine drug. One should be aware of this when comparing the results with data concerning migraineurs clinically diagnosed in accordance with International Headache Society standardized diagnostic criteria for migraine with and without aura. Furthermore, due to the selection of ‘new users’, our study cannot give information about the cardiovascular risk profile in migraineurs who are not treated with specific abortive migraine drugs. This was the main reason for including the group Migrafin® users as a control group, since the components of this drug have no vasoconstrictive properties and therefore the official label does not contraindicate the use in patients with known or suspected coronary artery disease.
Recently, in the Netherlands, the GEM population-based study has shown that migraineurs, particularly with aura, have a higher cardiovascular risk profile than individuals without migraine (31). Among migraineurs, the prevalence of high total cholesterol was 16%, total cholesterol:high-density lipoprotein ratio > 5 was 22%, history of diagnosed hypertension 33% and hypertension at examination 18%. This study did not investigate whether these patients with cardiovascular risk factors were treated with triptans or ergotamine. However, these higher prevalences of cardiovascular risk factors, compared with our data, also suggest reticence in prescribing these specific antimigraine drugs to migraineurs with these risk factors, or underestimation due to underdiagnosis of hypercholesterolaemia and hypertension. In our study we were not able to distinguish between migraineurs with and without aura. Therefore we do not know if prescribing patterns differ between these two forms of migraine.
Recent data from two prospective cohort studies suggest also an association between migraine and ischaemic cardiovascular disease (32, 33). Findings from the Women's Health Study, in which information on migraine aura was recorded, indicate that this association is limited to migraineurs with aura (32). From this point of view, it is very important to investigate thoroughly the cardiovascular safety of triptans, since withholding these drugs from migraine patients with a cardiovascular risk profile may influence the quality of life.
Most clinical trials and clinical practice data on triptans are derived from patients without known coronary artery disease. The lack of information about cardiovascular safety of triptans in this specific population probably explains the observed constant prescribing pattern of triptans to patients with known cardiovascular risk. No consensus exists among family practitioners or headache specialists about when to avoid using a triptan due to excessive cardiac risk factors (34). Cardiovascular risk assessment should be applied to decisions for prescribing triptans.
Apparently prescribers did not choose Migrafin® as the first alternative for patients with cardiovascular risk factors. We would then have seen a higher percentage of patients with cardiovascular risk in new Migrafin® users compared with triptans and ergotamine, which was not the case. One might wonder if NSAIDs are attractive alternative drugs for triptans in patients with cardiovascular disease. Due to adverse effects on cardio-renal function, use of NSAIDs is not without risk in patients with cardiovascular disease. There are complications arising from alteration of renal haemodynamics such as worsening of congestive heart failure, oedema and increased blood pressure (35, 36). In combination with platelet aggregation inhibitors or oral anticoagulants, the bleeding risk increases (37–39). Moreover, use of NSAIDs at high frequency or dose has been associated with a significantly increased risk for major cardiovascular events (40).
Single use of an abortive migraine drug, indicating non-acceptance of the treatment, was more frequently seen in patients with cardiovascular risk. A questionnaire study has shown that fear of side-effects and occurrence of side-effects are the main reasons for non-acceptance (single prescription) of selective serotonin reuptake inhibitor treatment (41). Although non-cardiac in origin, it might be the case that patients with cardiovascular disease receive more information about chest symptoms such as burning, tingling or tightness. This information may cause fear of side-effects, preventing patients from starting or continuing therapy. Fear itself may even cause side-effect-like symptoms. This, of course, does not mean that information on side-effects should not be disclosed to the patient.
In conclusion, during the past 16 years the percentage of migraine patients with known cardiovascular risk factors to whom ergotamine or triptans were prescribed remained low and constant. Growing evidence that the incidence of triptan-associated serious cardiovascular adverse events in both clinical trials and clinical practice appears to be extremely low did not change prescribing patterns over time. Available evidence that concerns about cardiovascular safety of triptans are unwarranted suggests that a prospective trial of triptans is justified in patients who would previously have been excluded from treatment (42). An earlier choice for optimal treatment in these patients might improve their quality of life.
Footnotes
Acknowledgements
The authors thank Patrick Souverein for his assistance in data processing for this study.
