Abstract
Early treatment and combining a triptan with a non-steroidal anti-inflammatory drug (NSAID) are thought to improve outcome during migraine attacks, possibly by counteracting the negative influence of cutaneous allodynia. The aim of this multicentre, double-blind pilot study was to evaluate the prevalence of brush allodynia and its relative influence on the efficacy of a triptan-NSAID combination compared with headache intensity at the time of treatment. In a randomized, cross-over design, 112 migraineurs treated two moderate or severe attacks with almotriptan 12.5 mg combined with either aceclofenac 100 mg or placebo. Patients used a 2-cm brush to assess cutaneous allodynia. Allodynia was reported in 34.4± of attacks. The almotriptan-aceclofenac combination was numerically superior to triptan-placebo on 2-24-h sustained pain-free (P = 0.07), 2-h pain-free (P = 0.07) and headache recurrence (P = 0.05) rates, but not on 1-h headache relief. Allodynia numerically reduced treatment success overall, but this effect was not significant for the primary outcome measures. Headache intensity had a significant negative influence on 1-h relief in both attacks (P = 0.0001 and 0.0008, X 2) and on 2-24-h sustained pain-free rates in triptanplacebo-treated attacks (P = 0.013). Multivariate logistic regression analysis confirmed that headache intensity at treatment intake, rather than allodynia, significantly influenced most outcome measures, predominantly so in attacks treated with almotriptan and aceclofenac. In the latter, severe compared with moderate headache intensity reduced the likelihood of achieving the primary efficacy end-points [odds ratios (OR) 0.12 and 0.33], whereas allodynia was not a significant explanatory variable (OR 0.76 and 0.65). The results apply to the protocol used here and need to be confirmed in larger studies using quantitative sensory testing.
Introduction
Allodynia, a common symptom during a migraine attack, is defined as an abnormal, unpleasant or painful perception of a normally innocuous stimulus (e.g. combing hair, washing the face). It is more frequently experienced by patients with moderate to severe migraine pain. In a laboratory setting, allodynia can be found by sensory testing to at least one static/dynamic mechanical or thermal stimulus in up to 80% of patients during a migraine attack (1–3). Mechanical and cold allodynia develop early in the attack on the side of the headache and culminate at 2 h, whereas heat allodynia lags behind by approximately 1 h (1, 2). Evidence from animal studies suggests that allodynia involves a transient increase in the responsiveness or sensitization of second-order nociceptive neurons in the spinal trigeminal nucleus that process information from intracranial structures and skin (4).
To date, the incidence and time course of allodynia during migraine attacks have not been assessed through auto-evaluation by patients themselves without intervention by an investigator, nor in a double-blind, randomized clinical trial.
Although allodynia may not contribute significantly to the disability experienced by patients during a migraine attack per se, several unblinded studies have suggested that its presence does reduce the efficacy of acute antimigraine treatments. For example, in the pioneer study by Burstein et al. (5) pain-free rates following subcutaneous injection of sumatriptan were reduced by > 80% if allodynia was present when treatment was initiated. In contrast, it has been reported by others that the subcutaneous administration of sumatriptan is an effective treatment at any time in the course of a migraine attack (6) and that it may even cause transient allodynia on its own (7).
Superiority of early treatment with triptans at a stage when the pain is still mild has been claimed for many years on the basis of circumstantial evidence and the intuitive deduction that mild pain is more easily treatable than severe pain (8). It has only recently been proven by randomized, placebo-controlled trials for sumatriptan (9), rizatriptan (10) and almotriptan (11), but the gain in efficacy compared with placebo is, as in previous open studies, greater for pain-free rates than for headache relief. Concordantly, it has been argued that allodynia, and thus central sensitization, might be primarily responsible for reduced pain-free and sustained pain-free rates (12), but this remains to be demonstrated in a clinical trial. As the migraine attack is most often a gradual process where pain, like allodynia (1–3), accrues over time, the question arises whether headache intensity or allodynia, or both, are the most relevant predictive factors for treatment outcome, especially when attacks are treated early.
Although the triptans represent an important part of the armamentarium employed by clinicians to treat migraine attacks symptomatically, some studies have suggested that oral formulations may not be superior or may even be inferior to high doses of commonly used analgesics (13, 14). Furthermore, it has been shown in animal studies (15, 16) and in allodynic migraineurs (16) that intravenous administration of a non-steroidal anti-inflammatory drug (NSAID) (ketorolac/indomethacin) is able to terminate the sensitization of second-order trigeminal nociceptors and the migraine attack, respectively, unlike sumatriptan.
Therefore, a combination of a triptan and an NSAID may represent a more effective treatment for the acute management of migraine than a triptan alone. Until now, this has been confirmed only by results from two open-label studies combining rizatriptan with rofecoxib or tolfenamic acid (17, 18), and in two double-blind trials of sumatriptan combined with naproxen (19, 20). These studies reported increased headache relief and reduced recurrence rates in migraineurs treated with the triptan–NSAID combination.
We found it of interest to study the influence of cutaneous allodynia on various outcome measures in a randomized, double-blind, comparative trial of a triptan, almotriptan, combined with either an NSAID, aceclofenac, or placebo. We also wanted to evaluate the relative impact on treatment outcome of allodynia compared with headache intensity at the time of dosing. In addition, we believed that it would be clinically more accurate for the patients to evaluate allodynia themselves using a brush at various time points before and after treatment, thereby avoiding their attendance at a medical centre, which could have potentially influenced the investigator and the study outcomes. The study was investigator-initiated by members of The Belgian Headache Society. Laboratorios Almirall (Barcelona, Spain) provided the study material and supported data analysis for a pilot trial.
Methods
Study design
This was a double-blind, double-dummy, cross-over investigator-initiated study conducted at eight centres throughout Belgium under the auspices of The Belgian Headache Society. The study was conducted over a period of 60 days, with patients receiving two randomly assigned consecutive treatments. One migraine attack was treated with oral almotriptan (almo, 12.5 mg) and acelofenac (aceclo, 100 mg)—an arylalkanoic acid inhibiting cyclooxygenases 1 and 2—and the other attack with almotriptan (12.5 mg) and placebo with a wash-out period of at least 1 week between the two attacks. Two similar tablets were taken by each patient per attack. The treatment sequence of almo–aceclo and almo–placebo or almo–placebo and almo–aceclo was assigned randomly at a 2:1 ratio.
Patients were asked to treat moderate or severe migraine attacks and to assess the presence of cutaneous allodynia by using a 2-cm long brush with one row of soft nylon hairs in five zones on each side of the head: supraorbital, ±2 cm above the eyebrows; infraorbital, ±2 cm below the eye; mandibular; hair of parietal cranium, ±1 cm above the ear; and occipital cranium, ±4 cm behind the ear (Fig. 1). Patients were asked to brush each area at least three times by moving the brush slowly along its long axis, and to record in a diary whether the brushing sensation was normal, abnormal but not unpleasant, unpleasant or painful. Any report of abnormal sensation was considered to reflect the presence of allodynia. The diary also recorded headache intensity on a scale of none, mild (no reduction of normal activities), moderate (reduction in performance, but no interruption of activities) or severe (prevention of normal activities) (21), as well as duration of headache and associated symptoms. Recordings were requested just prior to taking the trial medication (time 0) and 0.5, 1, 2 and 24 h later.

The brush and the zones on the head used by the patients to assess the presence of cutaneous allodynia (for details see Methods).
Patients
Male or female patients aged 18–65 years were eligible for inclusion if they had a minimum 12 months' history of migraine with or without aura (International Classification of Headache Disorders, 2nd edn, criteria), experienced two to six migraine attacks in each of the 2 months preceding trial entry, and if the onset of migraine occurred before 50 years of age. Patients who were pregnant, currently on an NSAID regimen, unable to distinguish between migraine and non-migraine headaches, had a history or evidence of substance abuse or addiction, or any concurrent illness, including dermatological disease, likely to jeopardize trial participation were excluded. Approvals were obtained from the local regulatory authority and ethical review committees, and each patient who participated provided written informed consent. The study conformed to Good Clinical Research Practice.
Concomitant medication
Any existing prophylactic migraine treatment, except NSAIDs, was permitted provided there was no change to the patient's regimen during the study. Patients must not have taken NSAIDs or any other acute antimigraine treatment within 24 h prior to study treatment.
Efficacy parameters
The primary efficacy parameters were sustained pain free (pain free at 2 h following treatment with no rescue medication in 24 h and no pain recurrence within 24 h) and headache relief (from severe or moderate to mild or none) at 1 h post treatment. Secondary parameters included: pain free at 0.5, 1.0, 2.0 and 24 h, headache relief at 0.5, 2.0 and 24 h, and headache recurrence between 2 h and 24 h post treatment. As in most published trials of acute antimigraine drug treatment, pain-free data include some of the attacks considered to show headache relief (see (21)). Other outcome measures were the prevalence of allodynia in the overall patient population and across the two migraine attacks, the influence of migraine attack severity on the prevalence of allodynia at baseline, the influence of allodynia and pain intensity at time 0 on headache relief rates at 1 and 2 h, and on 2-h and sustained pain-free rates. The prevalence (and disappearance) of allodynia at 0.5, 1.0, 2.0 and 24 h post treatment for both attacks was also assessed, as well as the influence of the delay between the beginning of the attack and treatment (time 0) on the prevalence of allodynia and treatment efficacy.
Safety and tolerability
Safety/tolerability assessments included adverse-event recording, vital signs, physical examinations and the use of concomitant medications.
Statistical analysis
A non-parametric analysis for two-period cross-over clinical trials was used to evaluate differences in efficacy measures between the two treatment periods (22). The Mann–Whitney U-test and Fisher's exact test were employed to assess the influence of the number of allodynic areas and the occurrence of allodynia at time 0 on headache relief and sustained pain-free rates, respectively. The same tests were used to analyse the influence of the time before treatment on the various outcome measures and on the occurrence of allodynia at time 0. Data from almo–aceclo or almo–placebo-treated attacks were analysed with the χ2 test to assess the influence of allodynia on treatment outcome measures for all attacks and separately for those treated when the headache was moderate or severe, as well as the influence of headache severity on outcome. On attacks treated with either combination, multivariate logistic regression analysis, including a likelihood ratio test, was performed to evaluate the respective impact of allodynia and headache severity as response variables on primary and secondary outcome measures.
Results
Patients
Overall, 112 patients were randomized to the two treatments with a total of 90 patients forming the intent-to-treat population: 57 patients in the almo–aceclo/almo–placebo treatment group and 33 patients in the almo–placebo/almo–aceclo treatment group. Across both treatment groups, age, height, weight and body mass index were comparable. Only nine patients had migraine with aura in addition to migraine without aura; during the study, six of these patients treated an attack preceded by an aura, resulting in a total of 11 treated attacks with aura, five with almo–aceclo and six with almo–placebo. Patient demographics and disposition are presented in Table 1.
Patient demographics and disposition (ITT population)

ITT, intention-to-treat.
Allodynia and efficacy measures
Allodynia
Overall, allodynia was reported by patients in 34.4% of migraine attacks; it was experienced by 38.2% of patients in at least one attack and by 23.3% during both attacks (Fig. 2). The cumulative incidence of allodynia across the two migraine attacks was 61.8%. The incidence of allodynia at time 0 was proportional to pain severity: 24% in attacks of moderate intensity and 45.8% in severe attacks. The majority of allodynic patients (49.2%) perceived the brushing sensation as abnormal, 41% as unpleasant and 9.8% as clearly painful. Most patients reported allodynia in one or two zones (35.9% and 33.3%, respectively), most frequently in the supraorbital area and hair of parietal cranium on the side of the headache (if unilateral) or bilaterally.

Incidence of allodynia (a) in all, moderate and severe attacks and (b) across both treated migraine attacks.
After dosing, allodynia disappeared over time at comparable rates in both treatment groups (Fig. 3). After 2 h, allodynia persisted in 21% of attacks treated with the almo–placebo combination and in 17% of those treated with the almo–aceclo combination; respective values at 24 h were 11 and 6%. The differences between the two treatment groups in the rate of allodynia disappearance were not significant.
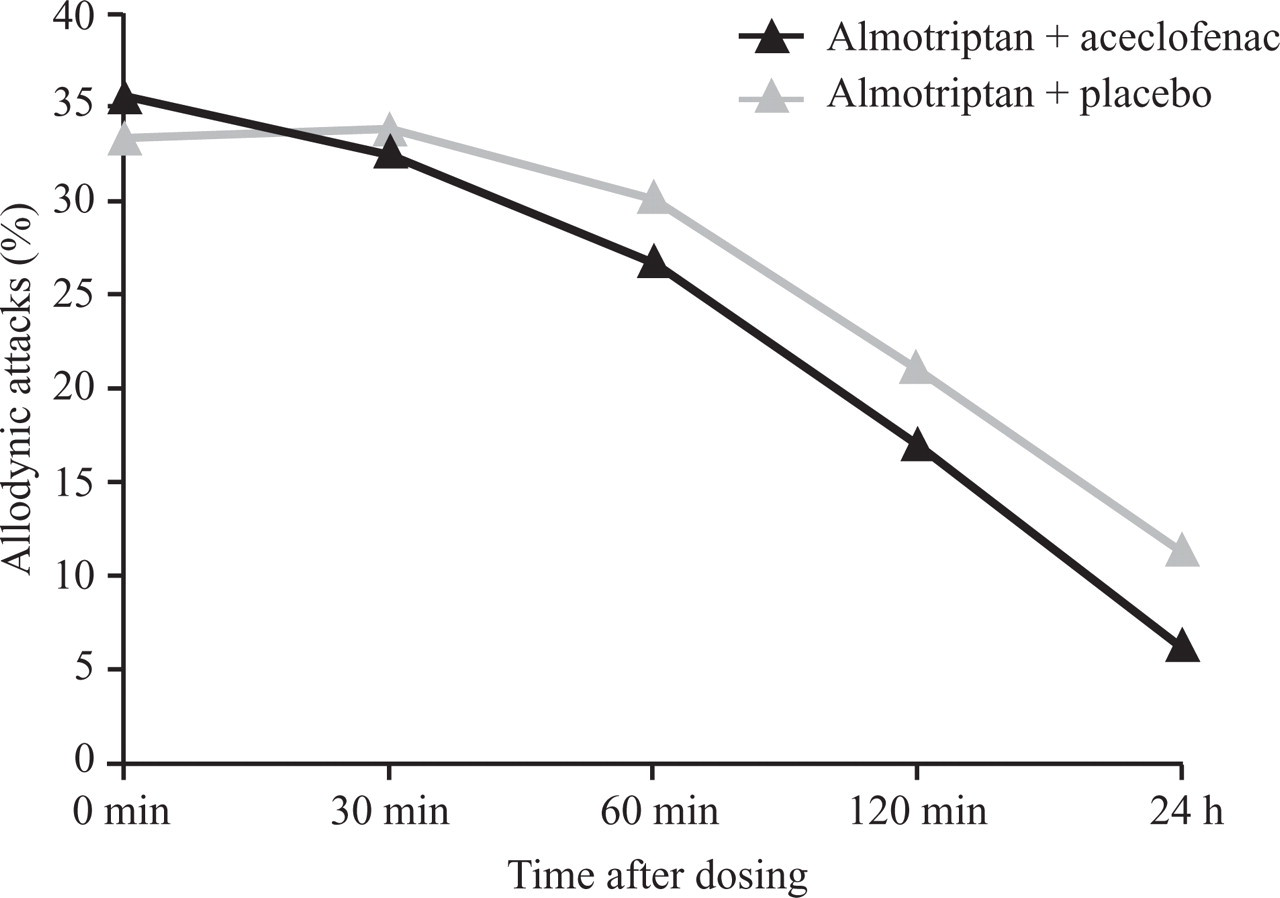
Decrease in incidence of allodynia with time in all treated allodynic attacks.
Treatment efficacy
The almo–aceclo combination was numerically more effective than the almo–placebo combination on all outcome measures except headache relief at 1 h (35.5% vs. 38.2%, respectively). For headache recurrence, this difference was statistically significant (15.2% vs. 29.2%; P = 0.05). There was a clear trend towards statistical significance for 2–24-h sustained pain-free rates (31.4% vs. 19.8%; P = 0.07) and pain-free rates at 2 h (40.7% vs. 29.1%; P = 0.07) (Fig. 4). As regards migraine-associated symptoms, the incidence of nausea and vomiting at time 0 was slightly higher in the almo–aceclo-treated attacks (53.75 and 7.5%, respectively) compared with the almo–placebo combination (50.62 and 6.17%). However, at 1 h and 2 h post treatment, nausea and vomiting were proportionally less frequent in the almo–aceclo-treated attacks (46.67 and 5% at 1 h; 42.86 and 7.14% at 2 h) than in the almo–placebo combination (61.90 and 6.35% at 1 h; 59.57 and 12.77% at 2 h). These differences were not significant, and there were no numerical differences of incidence or post-treatment evolution in photophobia and phonophobia between the two treatment arms.
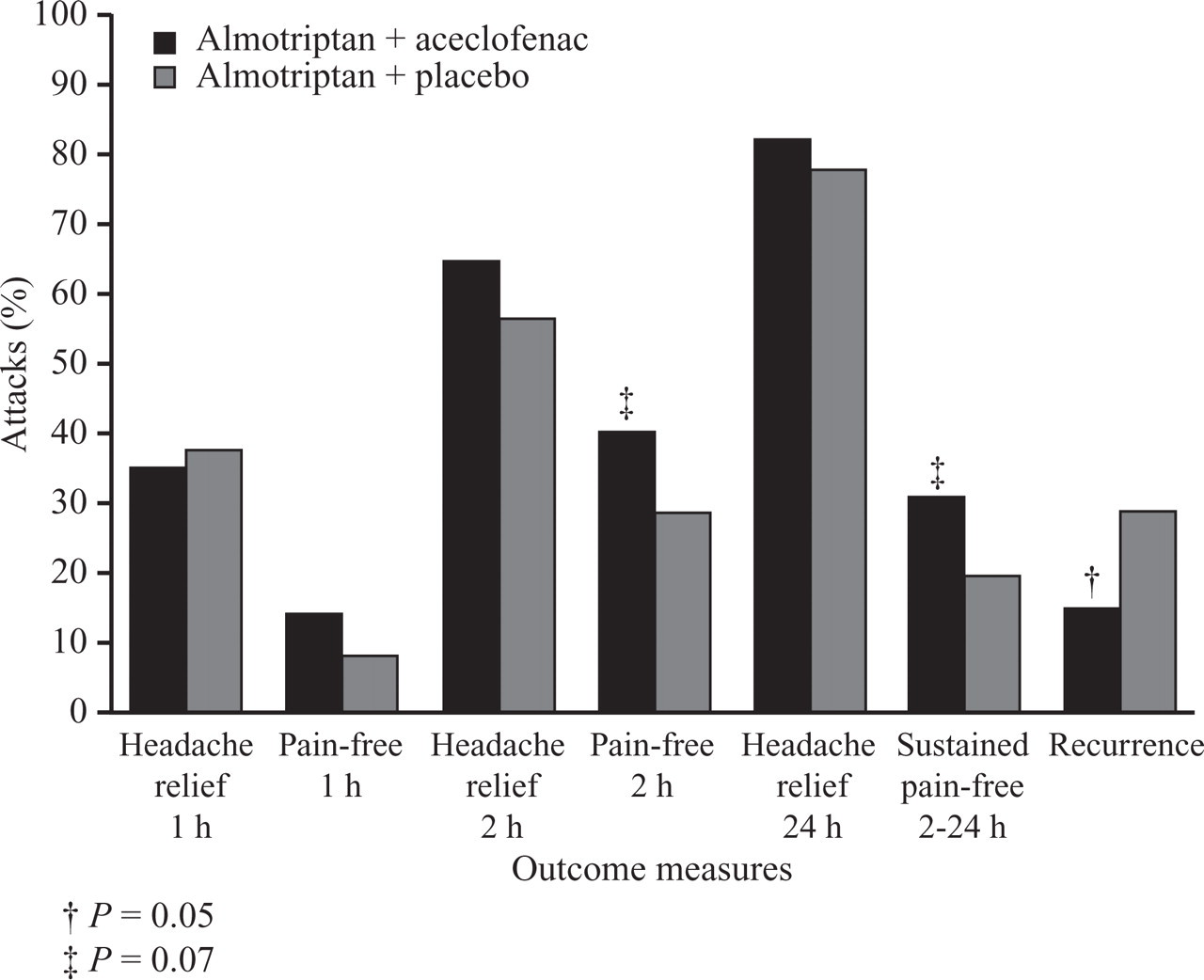
Treatment efficacy across outcome measures in attacks treated with the almotriptan–aceclofenac (dark bars) or the almotriptan–placebo (light bars) combination.
Influence of allodynia and headache intensity on efficacy
For all efficacy measures, the outcome tended to be worse in allodynic than in non-allodynic attacks. This is illustrated in Fig. 5 for sustained pain-free rates in the two treatment groups; whereas the almo–aceclo combination rendered patients pain free at 2 h for at least 24 h in 38% of attacks without allodynia, it did so in only 22% of attacks when allodynia was present at time 0—a relative efficacy reduction of 42%. With the almo–placebo combination, the presence of allodynia reduced the sustained pain-free rate by 52% (Fig. 5). However, the difference between the two treatment arms in the allodynia-induced reduction of efficacy did not reach the level of statistical significance.

Sustained pain-free rates by treatment group in allodynic (dark bars) and non-allodynic (light bars) attacks. The numerical differences do not reach the level of significance.
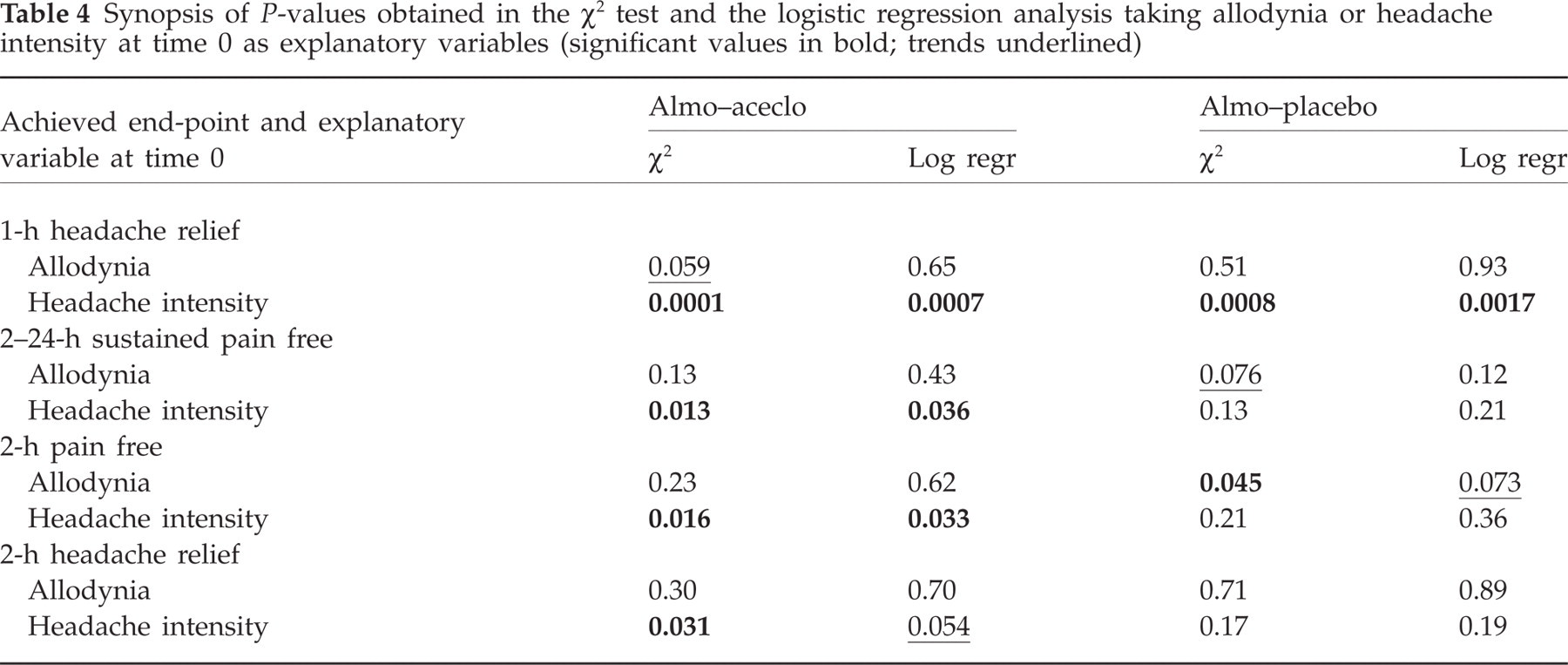
The intensity of headache at the time of treatment also influenced outcome parameters. The respective influence on outcome measures of allodynia and headache intensity is numerically illustrated in Table 2 for almo–aceclo-treated attacks and in Table 3 for almo–placebo-treated attacks. Using the χ2 test (Table 4), the negative relationship between allodynia and outcome only showed some statistical trends for the primary efficacy parameters. Allodynia significantly reduced 2-h pain-free rates in attacks treated with the almo–placebo combination (35% vs. 14.3%, P = 0.045). When controlled by pain intensity in the logistic regression model (see Table 4), this remained significant for attacks of moderate intensity (424% vs. 9%, P = 0.043), but not for the total number of attacks (P = 0.073). By contrast, using the χ2 test, severe headache intensity significantly reduced all outcomes in almo–aceclo-treated attacks as well as 1-h relief rates in the almo–placebo group (see Table 4). When the logistic regression model was applied to include the confounding role of allodynia, the negative effect of headache intensity remained significant, except for 2-h pain-relief rates (P = 0.054). The presence of a severe headache, and not of allodynia, at time 0 seemed therefore to be the most significant factor which decreased the likelihood of achieving relief at 1 h in the almo–aceclo [odds ratio (OR) 0.12, 95% confidence interval (CI) 0.03, 0.40 vs. 0.76, 95% CI 0.24, 2.44] and almo–placebo groups (OR 0.18, 95% CI 0.06, 0.53 vs. 0.95, 95% CI 0.32, 2.82), or 2–24 h sustained pain free in almo–aceclo-treated attacks (OR 0.33, 95% CI 0.12, 0.93 vs. 0.65, 95% CI 0.23, 1.89).
Influence of allodynia and headache intensity at time 0 on outcome measures in attacks treated with the almotriptan–aceclofenac combination

Influence of allodynia and headache intensity at time 0 on outcome measures in attacks treated with the almotriptan–placebo combination
Synopsis of P-values obtained in the χ2 test and the logistic regression analysis taking allodynia or headache intensity at time 0 as explanatory variables (significant values in bold; trends underlined)
The delay between the beginning of an attack and treatment (time 0) was highly variable between attacks (123.52 ± 186.12 min). Although it tended to be slightly shorter in successfully treated attacks, it did not differ significantly between attacks in which headache relief at 1 h and 2–24 h sustained pain free, the primary efficacy parameters, were achieved and those in which they were not.
Safety and tolerability
The proportion of patients with adverse events was similar across both treatment groups (12.24%) and no serious adverse events were reported. The proportion of patients with adverse events considered related to treatment was also similar across treatment groups (almo–aceclo, 7.14% vs. almo–placebo, 5.1%). All adverse events resolved completely, and there were no clinically relevant changes in vital signs in either treatment group.
Discussion
This pilot study represents the first double-blind study to examine the efficacy of almotriptan combined with aceclofenac and the detrimental effect of allodynia on therapeutic outcome in migraine attacks. Brush (dynamic) allodynia was auto-assessed by the patients themselves, which allowed evaluation of the occurrence of allodynia during an attack within the migraineurs' natural environment and without an investigator's intervention. Approximately one-third of moderate to severe migraine attacks were accompanied by brush allodynia. Allodynia was consistent across both migraine attacks in 23.3% of patients, with its prevalence proportional to the severity of pain experienced by patients. It was most frequently located in the territory of the ophthalmic division of the trigeminal nerve and, in unilateral attacks, on the side of the headache.
The allodynia prevalence figures found in our study are at first sight lower than those reported in other studies, including 79% of patients in the pivotal study by Burstein et al. (1), which used quantitative sensory testing (QST), and 54, 75 and 42% of attacks in the studies by Mathew et al. (23), Jakubowski et al. (24) and Landy et al. (25), respectively, who used structured questionnaires to assess allodynia. This difference could be explained by the fact that QST (1) is a more sensitive method of detecting allodynia and that both QST and questionnaires explore several sensory modalities, whereas in our study only dynamic mechanical allodynia was evaluated. However, it is possible that in previous studies the prevalence of allodynia was overestimated because of an investigator-related placebo effect and/or recall bias. In fact, in a more recent study (26), prevalence rates for dynamic (brush) and static (von Frey hairs) mechanical allodynia were 16 and 20%, respectively, in episodic migraine—the migraine type included in our study—compared with 48.7 and 50% in chronic migraine. A higher prevalence of brush allodynia (42.7%) in chronic migraineurs has also been reported in another study by the same group (27). As already suggested by the first reports of Burstein et al. (1, 2), mechanical allodynia is more prevalent in migraine with aura (57.1%) than in migraine without aura (17.6%) (26). As only 11 of 176 treated attacks in our study were preceded by an aura, the overall incidence of brush allodynia of 34.4% is within the range reported in the literature. Finally, the role of preventive antimigraine treatment was not analysed in our study, but it remains to be determined if it can directly influence the incidence of allodynia during an attack.
One could argue that we included as allodynic attacks those during which the patient reported an abnormal brushing perception without clear discomfort or pain, which is beyond the strict definition of allodynia, which is ‘pain resulting from non-noxious stimulus to normal skin’. It is common clinical experience, however, that many patients during the migraine attack report, on touching or combing hairs, an abnormal sensation that is not painful (28). Such abnormal perception could be the first stage of central sensitization, which is supposed to underlie allodynia and probably corresponds to the lowest allodynia ratings on visual analogue scales graded from 0 to 100, such as that used by Ashkenazi et al. (27). If a stricter definition had been applied, the prevalence of allodynia in our study would be 18%—a low rate similar to that reported by LoPinto et al. (26)—which would not have allowed any conclusion about its influence on treatment outcome and suggested that it has little clinical relevance. Before the conclusions and correlations established here can be generalized, a study validating auto-evaluation of brush allodynia and its different levels by quantitative sensory testing is nevertheless necessary.
The almotriptan–aceclofenac combination was numerically superior to the almotriptan–placebo combination for all outcome measures except headache relief at 1 h, but this difference was significant only for headache recurrence. The lack of statistical significance (only trends) for 2-h pain-free and 2–24-h sustained pain-free rates is likely to be due to underpowering of this pilot study. The benefit of adding an NSAID to an oral triptan was less pronounced in our study compared with previous open-label trials (17, 18), but appears to be in agreement with the two recent double-blinded studies (19, 20), which examined the efficacy of a sumatriptan (50 and 85 mg)–naproxen sodium (500 mg) combination against sumatriptan or naproxen alone and placebo using a much larger patient population (972 and 2956 patients). Two-hour pain-relief rates for the triptan–NSAID combination were similar (65%) in our study and the two previous trials (19, 20). However, 2-h pain-free and 2–24-h sustained pain-free rates were greater in our trial for the triptan–NSAID combination (40.7% vs. 34% and 30%; 31.4% vs. 25% and 23%, respectively) and the triptan monotherapy (29.1% vs. 20% and 25%; 19.8% vs.11% and 16%, respectively). It is unlikely that these differences can be explained by encapsulation of the sumatriptan tablets, which may lead to decreased absorption during a migraine attack (29), since it was used in the study by Smith et al. (19), but not in the Brandes et al. (20) trial. A difference in potency between sumatriptan and almotriptan used in our study is a more likely explanation.
Both the presence of cutaneous brush allodynia and severe headache at the time of treatment had numerically a negative impact on treatment efficacy. However, on the χ2 test, the effect of allodynia did not reach the level of statistical significance. The only exception to this was the 2-h pain-free end-point in almotriptan–placebo-treated attacks, where the P-value was 0.045. However, this was not a predefined primary efficacy measure, and P increased to 0.073 when the logistic regression model was applied to control for headache intensity. This result contrasts with previous studies showing that investigator-assessed allodynia is associated with poorer treatment outcome (5), although in these trials the comparative influence of headache severity was not assessed. That the effect of allodynia was significant for 2-h pain-free rates when placebo, and not aceclofenac, was combined with the triptan may suggest that the NSAID is able to reduce the negative impact of allodynia. This suggestion, however, which was drawn from the open use of an intravenous NSAID (16), is not supported in our study for the other outcome measures and needs thus to be confirmed by adequate larger trials.
By contrast, the negative impact of headache severity on primary outcome measures was clearly significant both with the χ2 test and when its effect was weighted against allodynia in the logistic regression analysis. This was the case for 1-h headache relief in both treatment groups and for 2–2-h sustained pain free in almotriptan–placebo-treated attacks. Severe headache compared with moderate headache, and not allodynia, was thus found at treatment onset to be the determinant variable that decreased the likelihood of achieving relief at 1 h after almotriptan alone (82% decrease) or combined with aceclofenac (88%), sustained pain free (67%) or pain free at 2 h (65%) after the combined treatment. Allodynia decreased these probabilities, respectively, by 4, 24, 35 and 22%. We have no explanation as to why for various outcome measures significant effects were found when aceclofenac rather than placebo was combined with almotriptan. One explanation may be that numbers of effectively treated attacks were smaller in the almotriptan–placebo group, at least for pain-free outcomes. The rather small numbers of patients this pilot study allowed us to include may also explain why some results discussed above have not reached the level of significance.
This is not the first observation suggesting that cutaneous allodynia per se may not be a major or sufficient cause for resistance to acute-migraine therapy. Others have reported that subcutaneous sumatriptan is effective for migraine attacks despite the presence of allodynia (6) and that 100-mg sumatriptan tablets are more efficacious after early (< 1 h) than late (> 4 h) treatment, whether allodynia is present or not (25).
It could be argued that there was not sufficient time for allodynia to develop, as in one study (1) mechanical allodynia reached its maximum 2 h after the beginning of attacks with aura. Since in our study the median time to treatment was 50 min, it is possible that the peak incidence of allodynia was not reached. However, this may also be true for the headache, as in Burstein et al.'s study (1) the migraine pain also peaked at 2 h. These dynamic changes at the beginning of an attack had thus probably no determinant influence on the regression analysis, but they underline that the conclusion that pain intensity is a better predictor of treatment outcome than allodynia is valid for our study protocol, but needs to be confirmed in other trials and with different protocols.
To conclude, our study suggests that mechanical (brush) cutaneous allodynia can be adequately assessed by the patients themselves during a migraine attack, although this needs to be validated by quantitative sensory testing. Prevalence rates of auto-assessed allodynia (34.4% for any abnormal perception, 18% for unpleasant/painful perception) were proportional to headache severity and within the range of those reported in episodic migraine without aura using questionnaires or clinical testing. The almotriptan–aceclofenac combination was numerically superior to the almotriptan–placebo combination in all efficacy measures, but statistical significance was limited to only a few of them because the study, sponsored as a pilot study, was underpowered. The presence of cutaneous allodynia at the time of treatment tended numerically to be associated with poorer outcome in both treatment groups, but its effect did not reach statistical significance. By contrast, treatment outcome was significantly worse when headache intensity at the time of dosing was severe, including for the primary outcome measures 1-h pain relief and 2–24-h sustained pain free (the latter only in almotriptan–aceclofenac-treated attacks). Logistic regression analysis confirmed that, for most outcome measures, headache intensity, compared with allodynia, had the most significant negative impact on treatment outcome, decreasing by 67–82% the likelihood of achieving the primary end-points. Taken together, these results indicate a complex relationship between headache intensity, allodynia and treatment outcome. For clinical practice and future trials, they suggest that allodynia could be driven by headache severity and that the latter might be a better criterion than the former in terms of optimizing acute antimigraine treatment and predicting its outcome.
