Abstract
Introduction
The aim of this study was to evaluate the prevalence and clinical impact of interictal microembolic signals (MES) in patients suffering from migraine with higher cortical dysfunction (HCD), such as language and memory impairment, during an aura.
Patients and methods
This study was carried out on 34 migraineurs with language and memory impairment during aura (HCD group), 31 migraineurs with only visual or visual and somatosensory symptoms during aura (Control group I), and 34 healthy controls (Control group II). We used a Doppler instrument to detect microemboli. Demographic data, disease features and the detection of MES between these groups, as well as the predictors of HCD during the aura, were analyzed.
Results
The duration of aura was longer and the frequency of aura was higher among patients with language and memory impairment during aura compared to Control group I. MES was detected in 29.4% patients from the HCD group, which was significantly higher compared to 3.2% in Control group I and 5.9% in Control group II. Regarding the absence or presence of MES, demographic and aura features were not different in the HCD subgroups. A longer duration of aura, the presence of somatosensory symptoms during the aura and the presence of interictal MES were independent predictors of HCD during the aura.
Conclusion
The present findings indicate that HCD and MES are related in patients with migraine with aura. Further research is needed to better understand the exact pathophysiological mechanism.
Introduction
The impairment of higher cortical function during aura in migraine patients occurs more frequently than previously thought (1,2). Specifically, migraine patients have reported speech and memory difficulties during aura (3,4). Although the mechanism of migraine is regarded as a functional disorder of the brain, numerous studies have reported that migraine is closely associated with abnormalities in the vascular system (5). Furthermore, a study proposed that microemboli can play a pivotal role in the development of migraine attacks (6). The origin of microemboli can vary (7). Carotid artery stenosis is a well-known source of cerebral microembolic signals (MES) (8). Additionally, MES frequently occur in patients with various cardiac sources of embolism (9). Moreover, patent foramen ovale (PFO), frequently found in migraine patients with aura, may be linked to the role of microemboli in the pathophysiology of the aura (10). Although these microemboli do not cause immediate symptoms, growing evidence suggests that they may cause delayed cognitive impairment if they enter the cerebral circulation in significant numbers (11).
Transcranial Doppler (TCD) ultrasound is a sensitive technique for the real-time detection of clinically silent MES (12). Although technology in the area of MES detection has improved recently, it remains impossible to distinguish the composition of emboli with certainty (12,13). The detection of MES in patients with cerebral vascular diseases is valuable in establishing diagnoses and prognoses (7); however, the applicability of TCD in the evaluation of the influence of MES on migraineurs remains unexplored.
In patients with memory and language impairment during migraine aura, the cerebral cortex may be affected by cortical spreading depression (CSD) in regions other than the occipital lobe, and microemboli may trigger CSD (14), contributing to the pathophysiology of migraine aura. Therefore, using TCD in patients with higher cortical dysfunction (HCD) migraine aura, we decided to test our hypothesis that the complexity of migraine aura is dependent on hypoperfusion caused by microemboli in various brain regions. The aim of this study was to evaluate the prevalence and clinical impact of interictal MES in migraine patients with HCD during aura.
Patients and methods
Patient selection
All migraineurs with visual and somatosensory aura treated from the beginning of 2005 to the end of 2013 (nine years) at the Headache Center, Neurology Clinic, Clinical Center of Serbia, were invited to participate in this study, yielding a total of 156 patients. The diagnosis of migraine with aura was based on the International Classification of Headache Disorders criteria (15,16). Exclusion criteria were: other neurological diseases, heart diseases and vascular diseases. Seventy-two patients met the inclusion criteria and agreed to participate in the study. They were all referred to the ultrasound laboratory in the Neurology Clinic. After standard TCD examination of the cerebral arteries and Doppler ultrasonography of the carotid arteries, 68 patients without carotid or intracranial artery stenosis were selected. These patients were divided into two groups according to the presence of HCD estimated by a specially designed questionnaire in order to collect data on HCD (presence of dysphasia, dysgnosia, dyspraxia and/or memory impairment) during the aura, as explained in detail elsewhere (2). The diagnosis of migraine with HCD during aura was established in 36 patients. These patients comprised the HCD group. The patients without HCD during visual or visual and somatosensory aura were included in Control group I (32 patients). Both groups were evaluated with TCD to detect microemboli in the interictal phase. Afterward, two patients from the HCD group and one from Control group I were excluded because of inadequate acoustical temporal bone windows. Subsequently, monitoring for the detection of right-to-left shunts (RLS) by contrast TCD, called the “bubble test,” was performed in order to evaluate the relationship of RLS and HCD during aura in the migraineurs. Additionally, to compose Control group II, 34 healthy controls were voluntarily recruited from hospital staff and matched with migraineurs with HCD in terms of gender and age. The exclusion criterion for participation in the study was previous diagnosis or treatment of some other neurological, cardiological or vascular disorder. In all patients and controls, carotid or intracranial artery stenosis was excluded by standard TCD examination of the cerebral arteries and Doppler ultrasonography of the carotid arteries. Demographic data, MES findings and aura features were compared between the groups. Additionally, we analyzed the independent predictors for the occurrence of HCD in patients with migraine aura. The research protocol of this study was approved by the Review Board of the Neurology Clinic, Clinical Center of Serbia.
Detection of TCD MES
RIMED Digi-Lite (RIMED, Israel), a dual-channel TCD system equipped with special software for the detection of microemboli, was used in the present study. Insonation was performed throughout the temporal acoustic bone windows according to a standard approach using 2 MHz transducers to display flow through the middle cerebral artery (MCA) (17). Bilateral monitoring of the MCA was performed with each probe held in place over the temporal bone by the head frame. MES were continuously recorded and counted, and the data were stored on a computer hard disc. All embolic tracks were counted in the bilaterally insonated MCA from a depth of 45 mm to 65 mm for 30 minutes. A velocity scale between 100 and 150 cm/s was used. We used a 128-point fast Fourier transform resolution and set the high-pass filter at 100 Hz. The power was 160 mW/cm2 and the pulse repetition frequency was 6500 Hz. A detection threshold of 9 dB was used to identify the microembolic events. Two examiners performed this part of the study without knowledge of the diagnosis of migraine. MES were determined and detected according to the criteria of the International Consensus Group on Microembolus Detection (17). All patients with detected MES were thoroughly evaluated by ultrasound examination of the heart and aortic arch, and the potential source of microemboli was not found.
TCD bubble test
Considering that RLS may be a major cause of MES, we decided to include the TCD bubble test in our research in order to gather more information about the origin of MES. This test represents a sensitive technique for identifying RLS by combining the injection of agitated saline (containing tiny bubbles) with ultrasound of the brain arteries (TCD). The TCD bubble test was conducted with the same TCD device used for the detection of TCD MES. In all patients, the TCD bubble test was performed in the supine position with the head inclined forward at 30 degrees. At least two contrast bolus injections with 9 ml of agitated saline mixed with 1 ml of air and a small amount of the patient’s blood were administered into an antecubital vein. The first injection was performed during normal respiration and the second was performed during the non-calibrated Valsalva maneuver. The TCD bubble test was considered to be positive if at least one microbubble was detected during the first 12 seconds after injection within the MCA on either side (18).
Statistical analysis
The data are presented as arithmetic means or as percentages. The Mann-Whitney U test was used to compare demographic data between the HCD group, Control group I and Control group II, as well as to compare aura features between the HCD group and Control group I. The chi-squared test was used to compare these groups in terms of gender and the results of the bubble test, as well as to compare the presence of somatosensory aura between the HCD group and Control group I and the number of patients with detected MES. Fisher’s exact test was used to compare the presence of HCD and results of the bubble test in MES and non-MES subgroups. Spearman’s test was used to assess the correlation between the frequency of aura and the number of MES detected, as well as between the duration of the aura and the number of MES detected in the MES subgroup. A binary logistic regression model was applied to determine the predictors of the occurrence of HCD during a migraine aura. The significance level was set at 5% (p < 0.05) prior to statistical analysis.
Results
Comparison of demographic data and aura features between HCD group, Control group I and Control group II.

Comparison of demographic data and aura features between HCD group and Control group I; Control group II, matched with migraineurs with HCD in terms of gender and age.
Comparison between HCD group and Control group II.
HCD: higher cortical dysfunction; MES: microembolic signals.
Demographic and clinical data of patients with detected MES.

Control group I. bControl group II.
HCD: higher cortical dysfunction; MES: microembolic signals; MCA: middle cerebral artery; F: female; M: male; L: left; R: right.
Predictors for HCD during migraine aura, binary logistic regression.
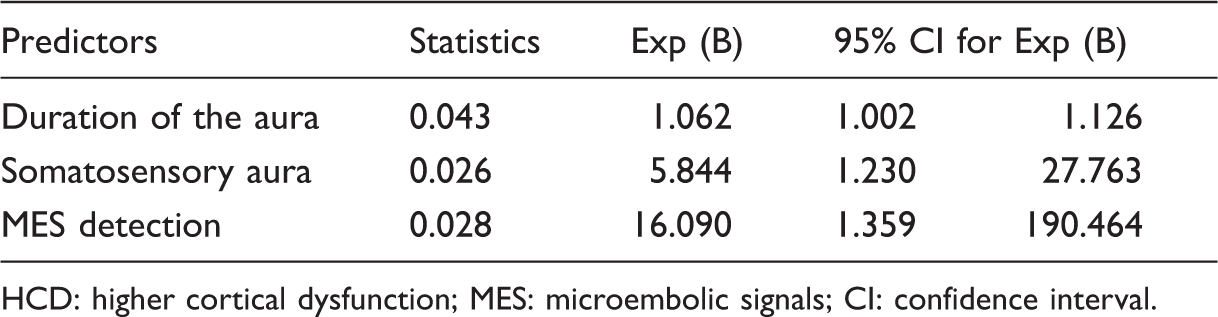
HCD: higher cortical dysfunction; MES: microembolic signals; CI: confidence interval.
Discussion
The main finding of the present study was that migraineurs with HCD during aura more frequently exhibited MES, with a higher number of detected MES compared to migraine patients without HCD during the aura and to individuals without migraine. Namely, MES was interictally detected in 29% of migraineurs experiencing HCD during the aura and in only 3% of the patients with visual and/or somatosensory aura. Moreover, the number of detected MES in a single patient was as high as 85 in the HCD group, compared to eight detected in other examined patients and healthy controls. All patients selected for this study were free of carotid artery stenosis or discovered cardiac sources of embolism.
The detection of microemboli by TCD sonography in stroke patients has been well studied (19,20). However, knowledge of the role of microemboli in the pathophysiology of migraine, especially of the aura, is still insufficient. In one published case, migraine with aura and MES disappeared simultaneously after removal of the pseudoaneurysm originating from the left vertebral artery (6). Studies investigating the presence of MES in migraine patients with aura are lacking. To the best of our knowledge, our study is the first that investigates the prevalence of interictal MES and their impact on aura in migraineurs.
The origin of microemboli in migraine patients is difficult to determine. Besides artery stenosis and formed plaques or intracranial aneurysms, RLS may be a possible origin of microemboli. Examined patients without carotid artery disease were selected for the study, and the correlation between the presence of MES and a positive bubble test has not been found. Despite extensive discussions, the role of RLS in the pathophysiology of migraine remains unclear (21). In our study, we did not find a direct connection of RLS with the appearance of HCD during aura or the detection of MES, but we cannot exclude the potential contribution to the pathophysiology of migraine (22). Although there was no significant difference between the HCD group and Control group I in the number of detected RLS, our results show greater levels of detection in each group than previous studies (11,23). This finding demands further investigation of other links between MES and memory and/or language impairment during migraine aura. Nevertheless, intravenous injection of agitated saline with air microbubbles has been reported to be a migraine trigger in patients with a large right-to-left shunt (14).
A recent study in 1456 women showed an increased risk of migraine with aura in carriers of thrombophilic factors, such as a procoagulant state due to platelet hyperaggregability or to a higher prevalence of inherited coagulation abnormalities, strongly supporting an association between thrombophilia and migraine (24). Moreover, a higher prevalence of a hypercoagulable state was found in stroke patients with migraine when compared to non-migraineurs, providing further support for this association (25). In addition to our results, those findings suggest that there is a need for thrombophilia screening in migraineurs with aura and detected MES. Further investigations should address a link between thrombophilic factors and MES detection in migraineurs in order to gain more information about the pathophysiology of complex migraine aura. Moreover, the detection of MES and the investigation of thrombophilic factors could be a valuable combination of tools for screening migraineurs with aura for ischemic stroke risk and the investigation of links between migraine with aura and ischemic stroke, which have been previously established (26).
The clinical manifestations of MES in one patient may not correlate well with symptoms of microemboli in another patient despite similar loads and locations, as seen during cardiac, vascular, and orthopedic operative procedures (27). In our study, patients with detected MES reported heterogeneous HCD: the inability to properly name familiar people and objects, difficulties with reading, difficulties in recalling past events, and manual imprecision (Table 2). Presently, there is not enough evidence to describe a causal link between interictal MES and HCD during aura. However, it could be hypothesized that HCD is related to persistent or intermittent microemboli, particularly cortical regions involved in aura by cortical CSD (28). Moreover, many studies (7,29) showed that the detection of MES may be intermittent and that they can be more easily recorded with serial registration, suggesting that MES could be detected even more frequently in the HCD group than estimated interictally.
Another possibility is that repetitive microembolism in migraine patients with aura could be influenced by neuron-glial interaction and network modulation. The role of glial cells in the propagation of the CSD wave cannot be discounted, as it plays a major role in the regulation of the extracellular potassium (K+), calcium (Ca2+), and glutamate levels during and after neuronal excitation (30,31). Moreover, energy failure in astrocytes increases the vulnerability of neurons to CSD (32).
Several limitations must be taken into consideration when the results of this study are interpreted. Limitations in study design include the limited monitoring time for MES detection and the fact that the examination was performed only on the MCA. There were also technical limitations in determining the detection threshold. A recommended monitoring time of only 30 minutes per patient (9) may be too short to detect intermittent MES and may actually produce a false-negative result. Additionally, posterior circulation was not monitored by TCD for MES detection. Silent brain infarctions and hyperintense lesions, possibly due to embolism, were identified in the posterior circulation in migraine patients with aura (33). MES studies performed on posterior circulation should be included in future studies. Last but not least, the selection of an MES intensity of 9 dB likely introduced a bias in the study sample toward large particles, although values ranging from 3 to 9 dB have been recommended to discriminate MES from general background noise (17).
In summary, higher prevalence and frequency of MES were detected in migraine patients with HCD during aura. The exact pathophysiological mechanism of this finding is not clear and requires additional research.
Article highlights
Higher prevalence and frequency of interictal microembolic signals (MES) were detected in migraine patients with higher cortical dysfunction during aura. MES detection and investigation of the origin of microembolism could be a valuable tool for screening migraineurs with aura for ischemic stroke risk and the investigation of links between migraine with aura and ischemic stroke. Further research should include analysis of the influence of microemboli on the neuron-glial interaction or the network modulation and pathophysiology of cortical spreading depression.
Footnotes
Funding
This work was supported by a grant from the Ministry of Education, Science and Technological development of the Republic of Serbia (grant number 175022).
Acknowledgements
The authors express their gratitude to A. Pavlović, MD, PhD, M. Mijajlović, MD, PhD, T. Švabić-Međedović, MD, and N. Veselinović, MD, and ultrasound technicians B. Kovačević, B. Minić and G. Poček for their contribution in the clinical and ultrasound examination of patients.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
