Abstract
Background
The socio-economic impact of migraine is mostly related to work loss either by absenteeism or decreased work performance. Migraine-associated cognitive dysfunction during an attack may contribute to these difficulties.
Objective
The objective of this article is to analyze the presence and relevance of cognitive symptoms during migraine attacks and to relate their intensity and symptom-related disability with other migraine-defining symptoms.
Methods
Consecutive migraine patients of a headache clinic completed diaries scoring each migraine symptom (including cognitive symptoms) intensity and symptom-related disability.
Results
Of 100 consecutive patients included in this study, 34 (all females, age average 31.8 ± 8.8 years) returned information on 229 attacks, on average 6.7 per participant. Every symptom’s intensity was always rated slightly higher than the disability it caused. Pain was the symptom scored with the highest intensity and disability, followed by cognitive symptoms (difficulty in thinking and worsening with mental effort) and photo- and phonophobia. Scoring was independent of any of the clinical variables. Attack intensity and disability scores correlated with intensity and disability from pain and from worsening with mental effort.
Conclusions
Attack-related cognitive symptoms are intense and disabling. Some attack-related cognitive symptoms correlate to intensity and disability subjectively attributed to the migraine attack. Cognitive performance should be addressed as a valuable secondary endpoint in trials of acute migraine treatment.
Introduction
Cognitive symptoms, although reported frequently by patients during migraine attacks (1–7), are not considered as core symptoms of the migraine diagnosis (8). Patients self-report being only 46% effective when working during migraine (9), but which part of this disability is related to cognitive dysfunction is undetermined.
Cognitive symptoms often precede migraine attacks, being very frequent in the premonitory phase of migraine (10–12) and being highly predictable of an attack (1). Disturbances of speech or thought are also described in the time gap between the end of the aura and the onset of pain (13). Cognitive difficulties can persist after the headache phase as postdromes (1,6,11,12,14) and may not be relieved by acute migraine medication (5,15). Cognitive symptoms are not a usual endpoint of acute treatment trials in migraine.
Cognitive dysfunction during migraine has been documented in some studies (16–20), with involvement of the domains of processing speed (20), working memory, visual-spatial processing (16–18), immediate and sustained attention and verbal learning (19,20). This is suggestive of preferential dysfunction of the pre-frontal and temporal cortices during migraine attacks.
These migraine-related clinical manifestations on cognition are important evidence of functional brain changes underling migraine pathophysiology and should be valued as therapeutic targets to be dealt with when evaluating acute treatment drugs.
Our objective was to analyze the presence and subjective relevance of cognitive symptoms during migraine attacks, by collecting data prospectively on paper diaries regarding the intensity and disability of cognitive and migraine-defining symptoms (8) in each attack of migraine patients.
Participants and methods
Population
Participants were recruited consecutively at a headache outpatient clinic, during either first or follow-up visits during the first semester of 2013 and invited to participate. Inclusion criteria were: a) age between 18 and 55 years; b) minimum education of nine years; c) minimum headache frequency of one monthly attack in the three months preceding inclusion; d) written informed consent; and e) diagnosis of definite episodic migraine with or without aura according to the International Classification of Headache Disorders, second edition (ICHD-II) (21).
Exclusion criteria were simultaneous presence of migraine (either with or without aura) and other headache types that could present with attack-related or non-attack-related cognitive symptoms, including tension-type headache, chronic migraine with or without medication overuse and migraine aura without headache. A history of past or current alcohol or drug dependence or abuse and the presence of a severe or uncontrolled medical or psychiatric disorder were also exclusion factors. The study protocol was approved by the institution’s review board and ethics committee.
Study design
Recruitment and inclusion were carried out at a regular clinic visit by a headache specialist who verified the patient eligibility criteria and carried out a standard clinical evaluation. After informed consent had been obtained, data were collected including verification of ICHD-II criteria for diagnosis, gender, age, literacy, disease duration, current attack frequency, duration and intensity, attack and aura characterization, use of prophylactic treatment and detailed medical and pharmacological history. At the end of the routine appointment, the patient was asked to complete the subjective cognitive impairment scale for migraine attacks (Mig-SCog) (7) and Headache Impact Test-6 (HIT-6) (22) scales. The Mig-SCog (7) is a nine-item questionnaire that quantifies self-reported subjective cognitive symptoms during migraine attacks and its score can run from 0 to 18, the higher scores representing higher frequency of cognitive symptoms; the HIT-6 (22) is a six-item standardized questionnaire that measures the impact of migraine on functional status and well-being. Patients were also given 10 headache diaries, one for each migraine attack, including information about timing of the attack (start and end of pain, timing of medication and of completion of the diary), medication use (acute and rescue medication) and about intensity and disability related to each attack. Attack treatments were grouped into four categories: triptans, nonsteroidal anti-inflammatory drugs (NSAIDs), analgesics (including combination analgesics with codeine, caffeine or ergots) and antiemetic drugs in combination (either with triptan, NSAID or analgesic). On one side of the sheet, patients were asked to rate each migraine symptom’s intensity on a 0–10 visual analog scale (VAS) for that specific migraine attack (intensity of pain, nausea, photophobia, phonophobia, kinesiophobia (23), worsening with physical effort, worsening with mental effort, difficulty in thinking and global attack intensity). On the reverse side of the sheet, patients were asked to rate each migraine symptom-related disability on a 0–10 VAS scale for that specific migraine attack (disability attributed to pain, nausea, photophobia, phonophobia, kinesiophobia (23), worsening with physical effort, worsening with mental effort, difficulty in thinking and global attack disability).
Diaries were returned at follow-up appointments occurring before December 31, 2013.
Statistical analyses
Statistical analysis used Stata release 12 (Stata Corp., College Station, TX, USA). Generalized estimating equation (GEE) with gamma family and a log link, with an AR(1) correlation structure and robust standard errors based on the Huber-White-sandwich variance estimator was used to analyze the relationships between scores of the different migraine symptoms adjusted by all other symptoms. A correlation coefficient between scores of any two symptoms was obtained with Spearman’s rank correlation after averaging each patient’s symptom scores over all the episodes. GEE analyses using global attack intensity and global attack-related disability as the dependent variables were performed. Significance was set at the 5% level (p < 0.05). The Holm-Bonferroni procedure was used to correct p-values for multiple testing.
Results
Population
One hundred patients were included in this study, eight males, with an age average of 31.2 ± 7.5 years, of whom 13 had migraine with aura and 87 without aura. There were nine (14%) dropouts for several reasons (losing their diaries, forgetting to bring the diary to the scheduled follow-up, not having had time to fill out the diaries or returning very incomplete diaries that were not included; one patient emigrated); 57 patients failed to attend the scheduled follow-up.
Clinical characteristics of participants and non-participants.

HIT-6: Headache Impact Test; Mig-SCog: subjective cognitive impairment scale for migraine attacks; SD: standard deviation; n.s.: non-significant. p > 0.010.
The study sample consisted of 34 females, one left-handed, of whom six had migraine with aura and 28 without aura, with an age average of 31.8 ± 8.8 years. Average HIT-6 score was 63.4 ± 4.4 (range 50 to 70), reflecting a moderate to severe impact of migraine although 68% of the sample had five or fewer headache days in the month preceding inclusion. Mig-SCog score average was 8.6 ± 4.1 (range 2 to 18), a medium score of subjective cognitive complaints.
Medical comorbidities were present in 11 (32%) patients, mostly vascular risk factors (high cholesterol and obesity, two patients each), followed by thyroid dysfunction (two), asthma or allergies (two), mild anxiety or depression (two) and others (congenital glaucoma and esophageal reflux or gastritis). Twenty (67%) of these patients were on birth control methods (19 oral contraception, one local hormonal device) and 13 (43%) on prophylactic headache treatment (amitriptyline two, topiramate six, propranolol three, valproic acid two). Five (15%) patients were on other medical treatments (lower-dose selective serotonin re-uptake inhibitor (SSRI) antidepressants two; asthma treatment, thyroid hormone, statin and topic glaucoma treatment, one patient each).
Headache diaries were completed on average 14.2 ± 7.5 hours (range 4.1 to 26.4) after the end of each attack. The average time interval from inclusion to handing back of the diaries was 96.1 ± 64.2 days (range 19 to 270 days, 8.8 months).
Migraine attack characteristics
Average duration of the studied attacks was 20.0 ± 14.3 hours (range 4.2 to 67.2 hours). Analyzing attack clinical features, 207 (90.4%) fulfilled the ICHD-II criteria for migraine, 19 (8.3%) for probable migraine and only two (0.9%) could be classified as probable tension-type headache. On average, patients waited 3.0 ± 4.5 hours (range 5 minutes to 20 hours and 45 minutes) before taking their acute medication.
All the patients took acute medication in at least one of their attacks; most of the patients (18, 52.9%) took it in all reported attacks and 12 (25%) took it in more than two-thirds of attacks.
Of the 229 attacks studied, 221 (96.5%) were treated. The first choice of abortive treatment in this sample were triptans (48.4% of treated attacks), either alone (38.5%) or in combination with an antiemetic drug (5.9%) or with an NSAID (4.1%). NSAIDs were the second choice in 40.7% of patients (alone 29.9% or with an antiemetic 6.8%). Analgesics, combination analgesics with codeine, caffeine or ergots were used in 9% of attacks, in 1.8% adding an antiemetic.
Rescue medication was used in 45.7% (101) of initially treated attacks and taken on average 4.5 ± 5.2 hours (range 15 minutes to 20.5 hours) after the initial therapy. The first choice of rescue treatment were NSAIDs (43.6%, with 5% adding an antiemetic and 5.9% a triptan). Triptans were chosen secondly as rescue therapy by 35.6%, adding an antiemetic in 3.0%. Thirteen percent of attacks required repeated rescue medication. Analgesics, combination analgesics with codeine, caffeine or ergots represented the choice in 10.9% of attacks, in 1% adding an antiemetic.
Symptom intensity and disability
Average scores of symptom intensity and symptom-related disability. On a 0–10 VAS scale.
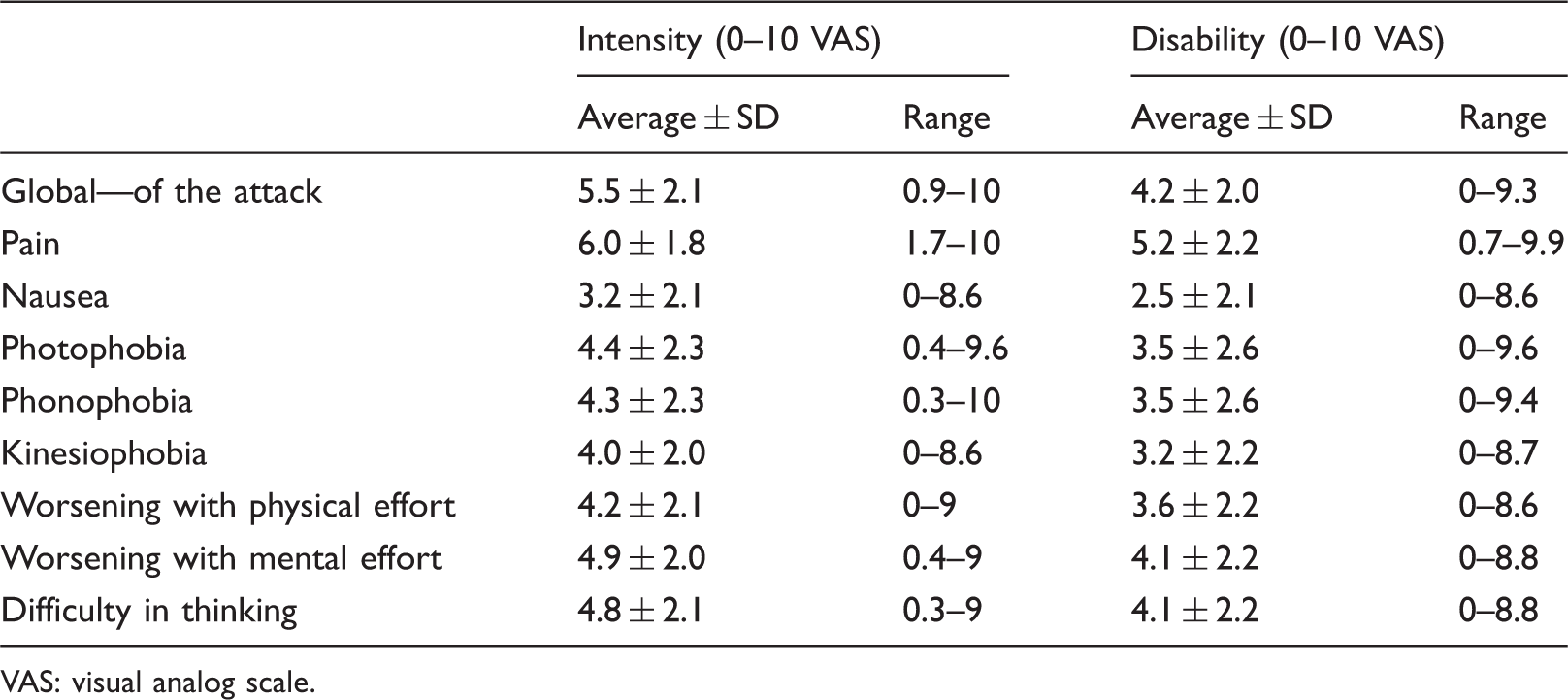
VAS: visual analog scale.
Correlations and regression analysis
Age was found to correlate with disease duration (Spearman’s rho 0.659, p < 0.0001) and inversely with attack frequency (Spearman’s rho –0.475, p = 0.005). Reported attack duration correlated with time to take rescue medication (Spearman’s rho 0.645, p < 0.0001) after initial acute treatment, but not with time to take initial acute treatment. There were no other significant correlations between disease-related variables (age, literacy, disease duration, attack frequency, reported attack duration and number of attacks reported, HIT-6 and Mig-SCog scores).
Initial acute treatment correlations with attack-related cognitive symptoms.

NSAIDs: nonsteroidal anti-inflammatory drugs. aSignificant correlation, p < 0.010.
Correlations found between intensities of each migraine symptom were scarce—intensity of pain correlated only with intensity of worsening with mental effort (p = 0.02), intensity of nausea with intensity of kinesiophobia (p < 0.001), intensity of photophobia with that of phonophobia (p < 0.001), intensity of kinesiophobia with worsening with physical effort (p < 0.001), the degree of worsening with physical effort related to worsening with mental effort (p < 0.001) and with intensity of difficulties in thinking (p = 0.002); finally the degree of difficulty in thinking correlated with worsening with mental effort (p < 0.001). Global attack intensity was correlated with pain intensity and worsening with mental effort (Figure 1).
Correlation of each symptom’s intensity with global attack intensity.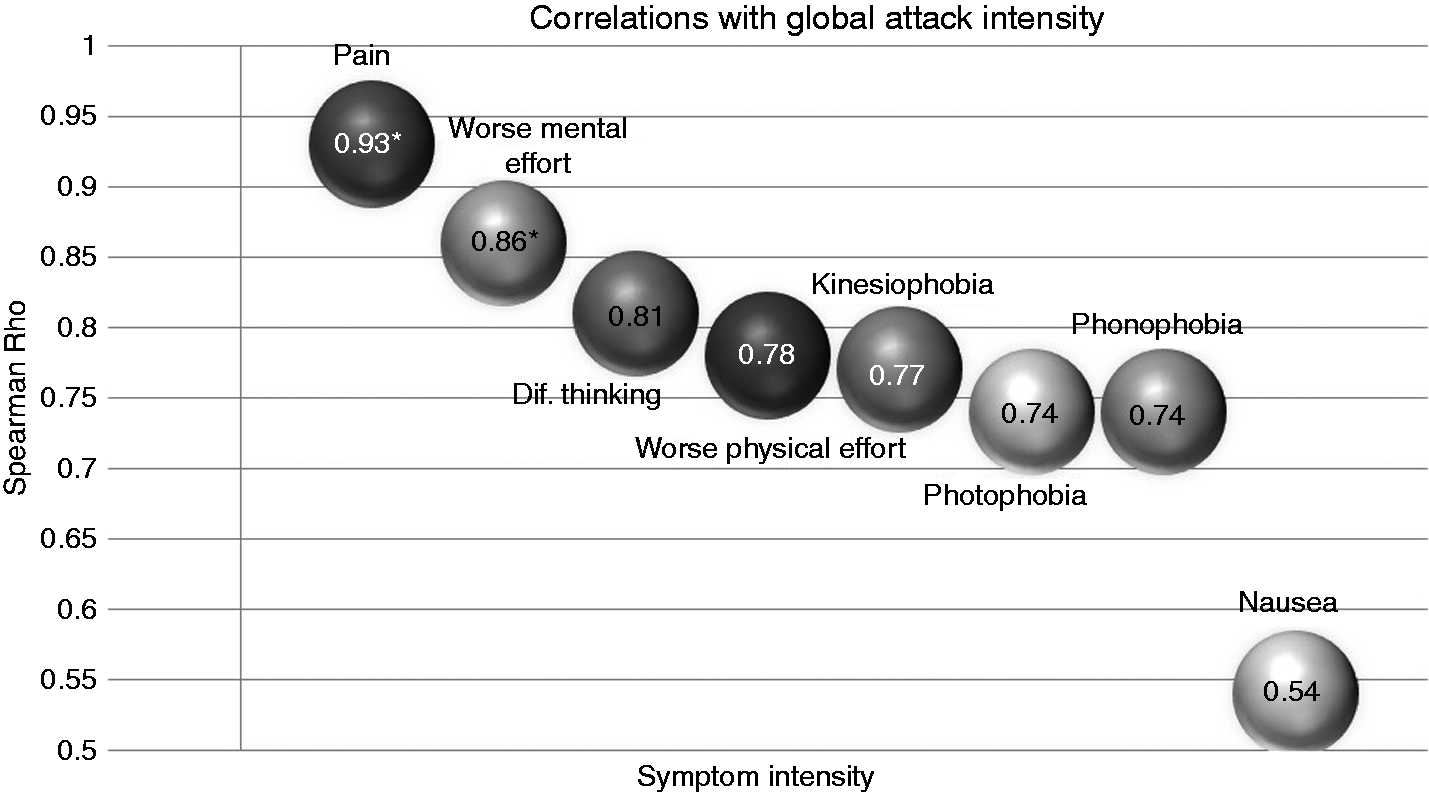
Correlations between disability scoring for each migraine symptom were also analyzed; disability of pain had no significant correlation with any other symptom. Disability due to nausea correlated with disability of kinesiophobia (p < 0.0001), disability of photophobia correlated with that of phonophobia (p = 0.002), phonophobia also correlated with the disability attributed to difficulty in thinking (p = 0.003), and disability of kinesiophobia with that of phonophobia (p < 0.001). Disability of worsening with physical effort correlated with disability due to kinesiophobia (p = 0.03) and disabilities of difficulty in thinking and worsening with mental effort were also correlated (p < 0.001). Attack disability correlated to pain and worsening with mental effort (Figure 2).
Correlation of each symptom’s related disability with global attack disability.
The correlation of each symptom-related disability with symptom intensity revealed that disability of nausea correlated with intensity of nausea (p < 0.001), intensity of worsening with mental effort (p = 0.01) and intensity of difficulty in thinking (p = 0.006). Disability of some symptoms correlated only with disability of the same symptom, such as with photophobia (p < 0.001), phonophobia (p < 0.001), kinesiophobia (p = 0.004) and worsening with physical effort (p = 0.004). Global attack disability also correlated with global attack intensity (p = 0.008). Disability due to pain, worsening with mental effort and difficulty in thinking had no correlations with any of the migraine symptoms’ intensity.
GEE analysis using global attack disability as the dependent variable failed to show an association with any of the studied variables (age, literacy, comorbidities, current medication, current migraine prophylactics, migraine diagnosis, disease duration, attack frequency and duration, HIT-6 and Mig-SCog scores) with the exception of global attack-related intensity. Gender was not included in the analysis because all the individuals were females.
Discussion
This prospective study of 229 migraine attacks revealed that patients are able to score intensity and disability of specific migraine attack-related symptoms independently. Patients’ scores of each symptom’s intensity were higher than the corresponding symptom-related disability scores, possibly reflecting self-perceived individual coping mechanisms during attacks although we cannot exclude a chance association. Among all migraine symptoms, pain was rated with the highest intensity, had the highest disability during attacks and was correlated with global attack intensity and attack-related disability, which supports the current view that pain control measures (pain freedom, pain relief and sustained pain freedom) are an appropriate primary endpoint of drug trials for the treatment of acute migraine attacks (24).
Cognitive symptoms sought (worsening with mental effort and difficulty in thinking during attacks) were scored in the second rank after pain both in intensity and attack-related disability, followed by photophobia, phonophobia, worsening with physical effort, and kinesiophobia. Unexpected findings were the low scores attributed to nausea intensity and associated disability that we speculate to be related to the early use of an antiemetic drug (in around 13% of treated attacks), associated with an analgesic, although our study design did not allow us to test this hypothesis.
Attack-related disability was highly correlated to both pain and worsening with mental effort, suggesting that some aspects of cognitive dysfunction have a role in migraine disability, independently of other symptoms. However, with our study design, we were unable to estimate the proportion of the variability of the attack-related disability that might be explained by cognitive symptoms.
Current guidelines include some other migraine-associated symptoms, such as nausea, photophobia and phonophobia and a measure of total migraine freedom (instead of simple pain freedom) as secondary endpoints in acute migraine treatment trials (24). These guidelines do not include cognitive endpoints nor other migraine-related symptoms. This fact reflects not only the lack of therapeutic agents that are able to treat all migraine symptoms but also influences the perceived lack of control of symptoms other than pain—if we are not measuring, we will never know. The inclusion of impact and quality of life measures, medication needs and other unconventional endpoints in prophylactic drug trials has allowed the evaluation of subtle benefits in difficult populations (25). The inclusion of measurements of cognitive impact or disability could help establish differences between acute medication profiles. To our knowledge, only two studies evaluated cognitive performance during an attack and after treatment (26,27) as outcome measures.
In our study, none of the clinical variables analyzed had an influence on symptom intensity or disability scoring. For most symptoms, their related disability correlated with its intensity, reflecting that intensity is related to the disability or symptom impact. Disability due to pain and to cognitive symptoms (worsening with mental effort and difficulty in thinking) failed to demonstrate a relationship to each symptom’s intensity, suggesting that the symptoms themselves are not the major factor determining their related disability.
Intensity of some symptoms was, however, related. Examples include kinesiophobia and worsening with physical effort, which is understandable if we acknowledge that kinesiophobia is a part of the avoidance behavior observed during migraine attacks, due to the aggravation of pain or enhancement of its throbbing character by head movement and/or psychical effort (23); nausea and kinesiophobia intensity were also correlated, which can be related to increased prevalence of motion sickness in migraine patients (28), as is supported by the fact that disability of kinesiophobia and disability of nausea and worsening with physical effort were also correlated. The degree of worsening with physical effort was associated with the degree of worsening with mental effort and of difficulty in thinking, probably reflecting the patients’ perception of the need to stop all activity.
Intensity of photo- and phonophobia being related reflects the fact that sensitivity to light and sound are believed to be the clinical expression of impairment of sensory processing during attacks (29), which is supported by the fact that their associated disabilities are also correlated; using the same line of thought difficulty in thinking and worsening with mental effort intensities and disabilities are also related as both can be interpreted as cognitive dysfunction-related phenomena (7,20).
Interesting enough, pain intensity was associated with the degree of worsening with mental effort, which may reflect higher impact of the attack, as pain intensity has been shown to have some influence in attack-related cognitive changes (20). The fact that we analyzed treated attacks is an obvious limitation of this study, as treatment influences symptom intensity and symptom-related perceived disability. Treatments can also act as confounders, for cognitive symptoms and nausea can be side effects of migraine acute medications (15,30), although in some reports enhancement of cognitive function was reported after treatment (31,32). We were able to identify some associations between some of the initial acute treatments used and cognitive symptoms, although the interpretation of these data is conflicting. The association of the use of triptans and combination analgesics with a higher intensity of cognitive symptoms might be either a consequence of drug effects or of the choice of such more effective drugs in face of a higher intensity attack. Taking antiemetics doesn’t seem to correlate with cognitive symptom impact yet this group was heterogeneous (including patients taking triptans, NSAIDs and analgesics). Our study design does not allow an accurate determination of the effect of each treatment in cognitive functioning, although this is an important topic for future research.
Our patient sample was clinic-based, reflecting episodic migraine patients with some comorbidities and a moderate to high impact disease (high HIT-6 score), some (38%) requiring migraine prophylactic medication. Migraine prophylactics and other chronic medications can influence cognitive performance and subjective symptom reporting (33). The comorbidities and concomitant treatments of our study population were scarce and mild and we assumed they had little influence on cognitive or other migraine-related symptoms, as reflected in the GEE analysis. Although the sample was clinic-based, it does not represent the usual population of tertiary headache centers (high-frequency attacks, high need for prophylactics, medication overuse and frequent comorbidities) nor does it apply to the population-based low-impact migraineur, which are limitations to the generalization of our findings. On the other hand, it was useful to select the migraine patients in whom disability is almost exclusively related to the attacks (i.e. having low interictal impact) and who have a high probability of being active and employed, strengthening the view that episodic attack-related cognitive dysfunction contributes to disability. The possibility of a recall bias is also a limitation that we attempted to minimize by having the patients report as soon as possible (on average 14 hours) after each migraine attack.
The most important limitation of this study is the high attrition, as only 35% of the patients returned their diaries. However, participating patients were similar to non-participating patients in all clinical variables, including disease impact and Mig-SCog scores. There were 14% dropouts, and other potential reasons for the high attrition include the lack of financial compensation for the participants and the demography of the sample that consisted of young working adults with a moderate impact long standing disease who failed to return to the follow-up appointment within the estimated time frame.
The benefits of treating the attacks early (first two hours, when the pain is mild (34)) are common knowledge for sufferers and have been documented in clinical trials and cost-effectiveness studies (35), leading to better medical counseling especially in headache clinics such as ours. An interesting observation of this study was that our population, despite having a high-impact migraine and being treated and coached in a headache clinic, still waited on average three hours before taking their acute medication after the onset of the attack. There may be many reasons explaining this delay such as fear of side effects, availability of medication or cost (34), yet another speculative contributing factor could relate to cognitive dysfunction, such as lack of initiative or mental slowing, to explain this delay.
There is some evidence that reversible cognitive dysfunction occurs during migraine attacks (36,37), corroborating patients’ spontaneous and subjective complaints (1,7). Migraine subtypes and disease severity may influence the expression of such symptoms (37). The mechanisms explaining these symptoms are still elusive yet functional imaging has contributed to document changes in the human cingulated and pre-frontal cortex (38) during migraine attacks, as well as in the insula and temporal lobe (39). Interictal functional connectivity changes on executive resting state and salience networks have been documented (40,41), as well as cortical gray matter differences of migrainous brains (42–44).
One aspect of cognitive dysfunction during attacks (worsening with mental effort) was found to correlate with attack-related disability, supplanting disability caused by nausea, photo- and phonophobia, which is a reflex of its importance to migraine sufferers.
In conclusion, cognitive symptoms are important contributors to migraine attack-related disability. The cutting edge of new acute migraine drugs should evaluate the return to normal function as a primary endpoint, including cognitive-related measures to evaluate the efficacy of such drugs in the return to normal cognitive performance, instead of simple freedom of pain.
Clinical implications
Pain and cognitive dysfunction during the migraine attack are correlated with total attack disability. According to the perception of migraineurs, pain is the symptom that most contributes to attack-related disability, followed by cognitive symptoms that produce higher disability than nausea, photo-, phonophobia and kinesiophobia. Cognitive performance should be valued as a secondary endpoint in clinical trials of acute migraine drugs.
Footnotes
Acknowledgements
Author contributions: Raquel Gil-Gouveia, Isabel Martins and António Oliveira were responsible for conception and study design, Raquel Gil-Gouveia for data acquisition, Raquel Gil-Gouveia and António Oliveira for data analysis, and Raquel Gil-Gouveia and Isabel Martins for data interpretation. Raquel Gil-Gouveia drafted the manuscript, António Oliveira and Isabel Martins revised it critically and all the authors agreed to the final version submitted. All authors agree to all aspects of the work and revised it for integrity and accuracy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
