Abstract
Background
A model for the testing of novel antimigraine drugs should ideally use healthy volunteers for ease of recruiting. Cilostazol provokes headache in healthy volunteers with some migraine features such as pulsating pain quality and aggravation by physical activity. Therefore, this headache might respond to sumatriptan, a requirement for validation. The hypothesis of the present study was that sumatriptan but not placebo is effective in cilostazol-induced headache in healthy individuals.
Methods
In a double-blind, randomized, cross-over design, 30 healthy volunteers of both sexes received cilostazol 200 mg on two separate days, each day followed by oral self-administered placebo or sumatriptan 50 mg. Headache response and accompanying symptoms were registered in a questionnaire by the participants themselves.
Results
Cilostazol induced a reproducible headache in 90% of the participants. The headache had several migraine-like features in most individuals. Median peak headache score was 2 on the sumatriptan day and 3 on the placebo day (p = 0.17). There was no reduction in headache intensity two hours after sumatriptan (p = 0.97) and difference in AUC 0 to four hours between two experimental days was not significant (p = 0.18). On the placebo day eight participants took rescue medication compared to 3 on the sumatriptan day (p = 0.13).
Conclusion
Despite similarities with migraine headache, cilostazol-induced headache in healthy volunteers does not respond to sumatriptan.
Introduction
Over the last 15 years human models of migraine have proved useful in the analysis of signaling pathways involved in the induction of headache, and likely of migraine pathogenesis (1–3). They have also suggested novel antimigraine drug targets (4,5). Useful models for the testing of new antimigraine drugs have, however, not yet been developed.
The treatment options for acute migraine attacks are acceptable, but there remains a need for more effective drugs with fewer side effects and in particular, for new classes of specific drugs that may be effective in triptan non-responders. In the prophylactic treatment of migraine there is an even greater need for new drugs. Unfortunately, the pharmaceutical industry has become risk aversive and several interesting targets are not pursued. Experimental models that can promote the development of new antimigraine drugs are therefore needed. In previous human models naturally occurring signaling substances or their precursors are infused intravenously and the headache is recorded at regular intervals together with a number of other biological variables (5–8). This model has shown some utility in drug testing but it is too resource demanding for widespread use. For a model to be useful, it should preferable be effective in healthy volunteers who are easy to recruit. It should ideally use tablet provocation and should respond to triptans given in tablet formulation, because companies primarily develop tablets, and because this allows outpatient administration. Furthermore, it should be repeatable in each participant, allowing for cross-over designs. The present study is the first of a series aiming at developing and validating such a model.
Cilostazol is a selective inhibitor of phosphodiesterase type 3 (PDE3), which is one of the two quantitatively most important cyclic adenosine monophosphate (cAMP)-degrading enzymes (9). In a previous study oral cilostazol 200 mg induced a reproducible mild-moderate headache in healthy volunteers. The headache had similarities with migraine in most volunteers (1). Thus, cilostazol seems to be a suitable provoking drug in healthy individuals. Our hypothesis was, therefore, that sumatriptan would have a significant effect against cilostazol-induced headache in a double-blinded, cross-over experiment in healthy volunteers.
Material and methods
Design
This was a double-blind, randomized, balanced, placebo-controlled, cross-over study in which cilostazol 200 mg was given orally on two separate days five days or more apart, each day followed by oral self-administration of placebo or sumatriptan 50 mg.
The central pharmacy of the Capital Region of Copenhagen performed the randomization of the experimental drug in a balanced fashion. The randomization code remained in the hospital during the study and was not available to the investigators until the study was completed. It remained unbroken until data management took place.
Participants
Thirty healthy volunteers (15 female (F)/15 male (M)) with no history of migraine were included. Inclusion criteria were: healthy individuals of both sexes aged 18–60 years and weighing 50–90 kg. Females were requested to use effective contraception.
Exclusion criteria were: any type of headache (except episodic tension-type headache < 1 day per week), serious somatic or psychiatric disease, pregnancy, and intake of daily medication (except oral contraceptives). The participants were informed that cilostazol might induce headache or migraine in some individuals. One participant dropped out for personal reasons. He was replaced with a new participant.
Standard protocol approvals
All participants gave written, informed consent to participate in the study. The study was approved by the Ethics Committee of Copenhagen (H-4-2013-174), the Danish Data Protection Agency, and the Danish Medicines Agency and was conducted according to the Helsinki II declaration of 1964, as revised in 2008.
All participants were enrolled via the website: www.forsøgsperson.dk.
Study procedure
Full medical history, physical examination, electrocardiography (ECG) and vital signs were collected on the day of recruitment. All participants had two study days at least five days apart. They arrived non-fasting at the clinic between 8:30 a.m. and 12:00 p.m. They had to be headache free 48 hours prior to the study and to not have taken any type of painkillers 12 hours before the study day. A pregnancy test was taken at the beginning of each study day on all female participants. All participants received cilostazol 200 mg orally on both study days and went home immediately after. When the participants reached headache intensity 3 on the verbal rating scale (VRS) or four hours after cilostazol, placebo or sumatriptan was taken. In case of severe headache not responding to the experimental drug, the participants were offered rescue with paracetamol or nonsteroidal anti-inflammatory drugs (NSAIDs) but not before two hours after placebo or sumatriptan. During the study, an emergency phone was always open where patients could call if they experienced severe headaches or discomfort.
Headache parameters
Headache parameters and accompanying symptoms were recorded by the investigator in the clinic before study start based on a headache questionnaire. Afterward headache intensity was scored on a self-administered questionnaire every 30 minutes the first six hours after cilostazol and thereafter every hour until 12 hours after cilostazol. The intensity was scored on a VRS from 0 to 10, 1 representing a very mild headache (including feeling of pressing or pulsation), 5 a headache of medium severity and 10 the worst possible headache (10). Headache characteristics (unilateral/bilateral, quality and aggravation by physical activity) and accompanying symptoms (nausea/vomiting, phono- and photophobia) were also recorded in the questionnaire in order to compare with the International Classifications of Headache Disorders, third edition beta (ICHD-3 beta) for migraine without aura (MO) (11). Two participants went to sleep before 12 hours after cilostazol and missing data were filled in using last observation carried forward.
Statistical analysis
Headache intensity scores are presented as medians (range).
Calculation of sample size was based on the detection of a difference between two experimental days in changes of the head pain intensity (headache intensity before versus headache intensity after sumatriptan/placebo) at 5% significance with 90% power. We estimated that the effect on the placebo days was 20% and on the sumatriptan day 60%. Standard deviation was estimated based on previous data. The correlation within the two days was estimated conservatively at 0.5. We also assumed no carry-over effect. We estimated that at least 18 participants should complete both experimental days. Due to uncertainty regarding these assumptions we decided to include 30 participants. The area under the curve (AUC) for headache score was used as a summary measure for analyzing differences between the groups and was calculated according to the trapezium rule (12).
One primary endpoint was median headache score (regardless of type) at two hours after sumatriptan/placebo. We compared pre- and post-treatment headache scores within each day. Another primary endpoint was the difference in the area under the headache score curve (AUC) 0 to four hours after sumatriptan/placebo between the two experimental days. Secondary endpoints were nausea, photo- and photophobia, median peak headache score, median headache score four hours after sumatriptan/placebo and AUC for headache 0 to two hours after sumatriptan/placebo.
Headache intensity scores are presented as medians (range). Differences in AUC for headache intensity scores were tested using the Wilcoxon signed rank test.
The incidence of headache and associated symptoms were analyzed as binary categorical data with McNemar’s test. Reproducibility of the headache was tested with a Bland-Altman plot. All analyses were performed with SPSS for Windows 11.5 (Chicago, IL, USA), or GraphPad Prism version 6.0. A p < 0.05 was considered significant.
Results
Thirty healthy volunteers (15 F, 15 M), mean age 25 years (range 18–50 years) and mean weight 71.5 kg (range 50–90 kg) with no history of migraine completed both study days. Cilostazol induced a reproducible mild to moderate headache (range 1–7 on VRS) in all but three participants.
Response to sumatriptan
The primary endpoint, median headache score two hours after treatment, is illustrated in Figure 1. There was no reduction in headache intensity after either sumatriptan or placebo. Reduction in median headache score for four hours was not significant either (sumatriptan: p = 0.17, placebo: p = 0.37).
Box-and-whisker plot representing medians and quartiles of headache score 0 and two hours after treatment. There was no statistical reduction in headache intensity two hours after treatment (sumatriptan: p = 0.99, placebo: p = 0.42).
The other primary endpoint, difference in area under the headache score curves (AUC) 0 to four hours after treatment was not statistically significant (p = 0.18). AUC 0 to four hours is illustrated in Figure 2. Our secondary endpoint, difference in AUC 0 to two hours after treatment, was not significant either (p = 0.42).
Box-and-whisker plot representing medians and quartiles of area under the headache score curve 0 to four hours after treatment. We found no difference between the two treatment groups (p = 0.18).
Clinical characteristics of headache and associated symptoms after cilostazol.
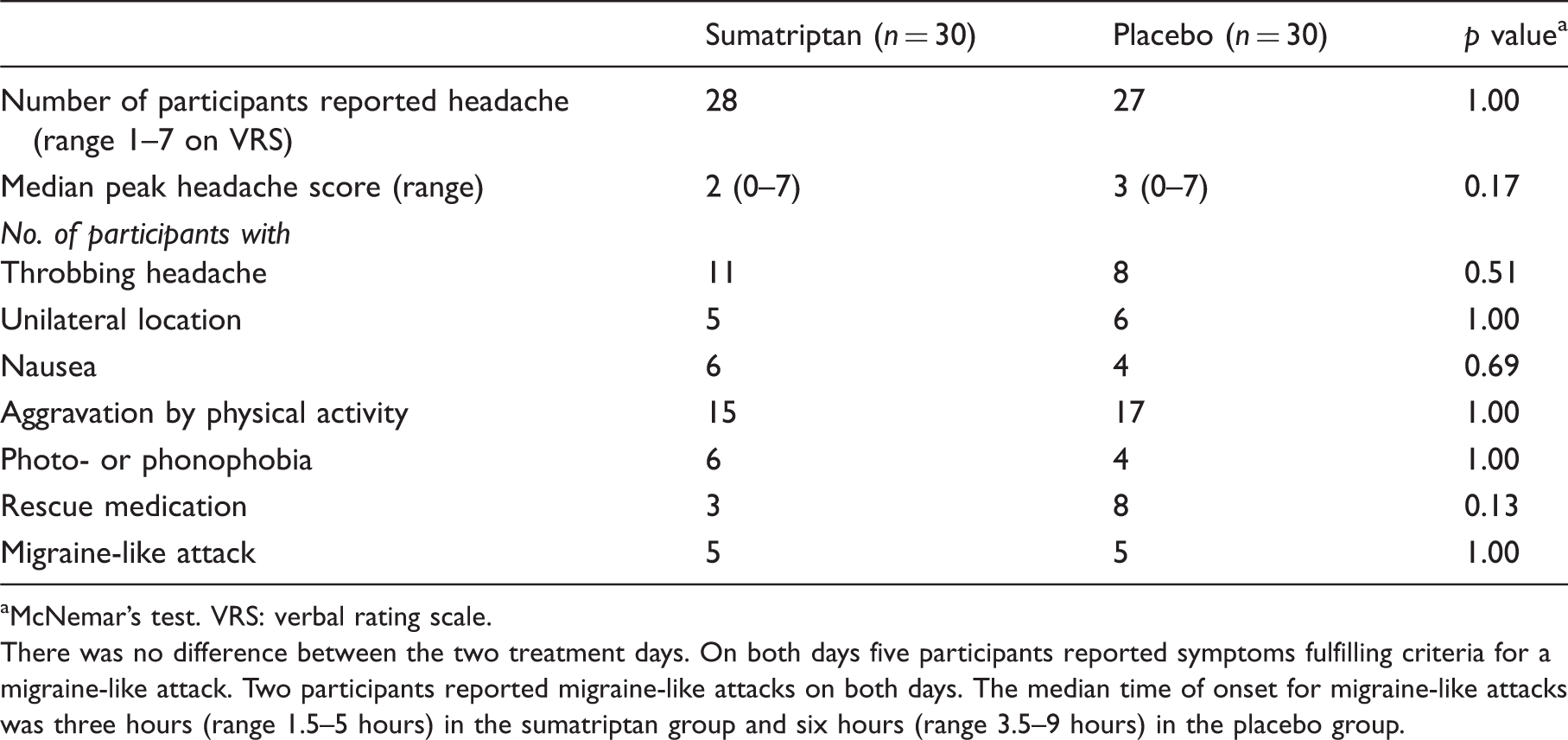
McNemar’s test. VRS: verbal rating scale.
There was no difference between the two treatment days. On both days five participants reported symptoms fulfilling criteria for a migraine-like attack. Two participants reported migraine-like attacks on both days. The median time of onset for migraine-like attacks was three hours (range 1.5–5 hours) in the sumatriptan group and six hours (range 3.5–9 hours) in the placebo group.
Only five (sumatriptan group) and seven (placebo group) participants reached headache intensity 3 on the VRS before four hours after cilostazol (and therefore treated before four hours). Median time for treatment was four hours (range two to four hours). Median headache score at treatment was 1.5 on the sumatriptan day and 2 on the placebo day. Median headache scores 0 to 12 hours after cilostazol for the two treatment groups are illustrated in Figure 3. There was no difference in treatment response on the two days (p = 0.078 for difference in AUC 0 to eight hours after the two treatments).
Median headache score for the two treatment groups 0–12 hours after cilostazol intake. Median time for treatment was four hours (range two to four hours) illustrated with the thin dotted line. Median headache intensity at treatment was 1.5 (sumatriptan day) and 2 (placebo day) on the verbal rating scale (VRS). There was no difference in treatment response between the two days (p = 0.078 for difference in area under the curve (AUC) 0 to eight hours after the two treatments). Note that the headache is reproducible on the two days until time of treatment.
Seven of the 30 participants had a first-degree relative with migraine (reported by the participants themselves). There were no differences in headache intensity or treatment response between predisposed and non-predisposed individuals. There were overall no differences in response between sexes either.
In a secondary analysis, the five participants fulfilling criteria for migraine-like attacks in the sumatriptan group had a median headache score of 3 at the time of treatment. Two hours after sumatriptan the headache intensity had increased to 4 and thus did not respond to sumatriptan.
Discussion
In line with a previous study (1), the present study found that cilostazol induces a headache in the majority of healthy participants. The headache was relatively reproducible on the two study days and had several migraine features such as pulsating pain quality and aggravation by physical activity. Nevertheless, the headache did not respond to sumatriptan tablets in a reasonably sized cohort. Therefore, cilostazol-induced headache in healthy individuals is not a practical model for the testing of new migraine drugs.
Why a human migraine model and how
Human experimental models are valuable in the early-phase development of new acute and prophylactic antimigraine drugs. For acute treatment the headache is induced with a substance and treated with active drug or placebo (8,13). Testing prophylactic drugs could be performed the other way around: The active drug or placebo is given for a period of time followed by the headache-inducing substance (14,15).
Experimental human models have several advantages compared to spontaneous migraine attacks. It is our experience that disability inhibits the migraineurs from traveling to the hospital during a migraine attack (16), which makes it difficult to study novel drug candidates. This is avoided using a model. Experimental models allow the study of migraine and its treatment under standardized circumstances and carefully controlled conditions where it is possible to register vital signs, take blood samples for pharmacokinetics and add imaging. Furthermore, a model makes it possible to test new drugs in a short period of time since participants do not need to get a spontaneous attack. Prophylactic drug trials in migraine are long-lasting and expensive and require long-term toxicology information (three to five months) (17). In addition to the above-mentioned benefits, a human migraine model for the testing of prophylactic drugs probably needs only two weeks of toxicology. It is preferable to develop a model that is valid in healthy volunteers since they are easy to recruit but, as discussed later, it seems difficult to develop such a model. If we do not succeed in finding a model useful in healthy participants, MO volunteers would also be useful. The latter requires the establishment of a register of migraineurs who are willing to participate in studies against payment.
Only a few previous attempts at developing a human model for drug testing have been made. Tvedskov et al. used glyceryl trinitrate (GTN) infusion in healthy volunteers and found that tonabersat, aspirin, zolmitriptan and a calcitonin gene-related peptide (CGRP) antagonist had no effect on GTN-induced headache (13,18,19). The same group also observed no preventive effect of propranolol on GNT-induced headache in MO patients (15). On the other hand valproate reduced the number of patients with GTN-induced migraine compared to placebo (14). Tfelt-Hansen et al. showed that prednisolone decreased the intensity of delayed GTN-induced headache in MO patients (20). Another study found that GTN-induced headache responded to pretreatment with a subcutaneous injection of 6 mg sumatriptan (8).
Could modifications improve the present model?
It was unexpected that sumatriptan did not have an effect on cilostazol-induced headache. An explanation could be that cilostazol induces another headache than migraine that, just like episodic tension-type headache, does not respond to sumatriptan (21). Another reason for the lack of response might be that treatment was given before migraine mechanisms were activated (two to four hours after cilostazol). We are not optimistic about a later time of treatment. First of all, some studies have indicated that sumatriptan is more effective when taken early in a migraine attack without aura while the headache is still mild (22,23). Secondly, median time for peak headache score was 3.5 h in the sumatriptan group and 4.8 hours in the placebo group. Thereafter, the headache in both groups began to decrease and thus it could give other problems if we treated later. Peak headache corresponded to peak plasma concentration of cilostazol at 2.8 hours (range 1.5–4.0 hours) (24). The majority of the participants in the present study reached only a mild to moderate headache intensity, and migraine is characterized by moderate-severe pain intensity (minimum 4 on the VRS). Median headache score at time of treatment with sumatriptan was 1.5 (six participants reported headache score 0 at sumatriptan intake). Since headache has been reported as a dose-related adverse event in the clinical use of cilostazol (25), another modification to the model could be to induce a more severe headache with a higher dose of cilostazol. However, administration of a higher dose might result in more and intolerable side effects and it would not be possible for participants to treat at home making the model impractical and expensive. One could also treat only if the headache reaches at least 2 on the VRS (same intensity at which migraineurs often begin to treat). This is, however, not a promising possibility since participants with headache score ≥2 at the time of treatment did not respond better to sumatriptan than participants with headache score <2. Triptans hyperpolarize nerve terminals and cause vasoconstriction (26,27). Cilostazol acts mainly downstream by blocking PDE3 and thereby increases intracellular cAMP level. An explanation for the absent response of sumatriptan might therefore be that cilostazol bypasses the effect of sumatriptan.
Finally, it might be possible that a larger number of participants would have given a positive result. Our study was, however, amply powered. Furthermore, the model would be impractical if more participants were needed.
In summary, we do not think that modifications might lead to a more useful model using cilostazol in healthy individuals.
Future development of a human migraine model
The present study is the first in a series needed to develop and validate a pragmatic human model for the testing of novel migraine drugs. It was our hope that cilostazol-induced headache in healthy volunteers would respond to sumatriptan, a highly specific antimigraine treatment. That would have validated the headache in healthy volunteers as migraine like, and one would subsequently be able to validate further using a CGRP antagonist. Unfortunately it did not work. We particularly would like a model in healthy individuals because they are easy to recruit if paid appropriately. Since the cilostazol model did not fulfill our requirements, we next want to test a long-acting nitro preparation in healthy subjects. If that also does not respond to sumatriptan then we shall move to a model in MO patients. Our recent studies suggest that cilostazol-induced headache in patients with MO responds to triptans (28). It is, therefore, tempting to repeat the present double-blinded study in MO patients. Before we indefinitely conclude that cilostazol-induced headache is non-responsive to sumatriptan, one should test oral sumatriptan at 100 mg or a subcutaneous dose at 6 mg.
Another possibility is to use other migraine-provoking substances in healthy volunteers or migraine patients. It is demanding to test 30 individuals in a cross-over design, 60 days in the laboratory, but the reward for migraine drug development may be considerable if a valid model can be developed.
Clinical implications
Drug development for migraine is not sufficient. This study presents a pragmatic human model for the testing of new drugs. Unfortunately, it has to be modified in the future to respond to sumatriptan. Several possibilities for such modifications are suggested.
Footnotes
Funding
This work was supported by the European Union’s Seventh Framework program (2007–2013) under the grant agreement no. 602633 (FP7-Euroheadpain).
Conflict of interest
None declared.
