Abstract
Background
Migraine with visual aura (MA) is associated with distinct visual disturbances preceding migraine attacks, but shares other visual deficits in between attacks with migraine without aura (MO). Here, we seek to determine if abnormalities specific to interictal MA patients exist in functional brain connectivity of intrinsic cognitive networks. In particular, these networks are involved in top-down modulation of visual processing.
Methods
Using resting-state functional magnetic resonance imaging, whole-brain functional connectivity maps were derived from seeds placed in the anterior insula and the middle frontal gyrus, key nodes of the salience and dorsal attention networks, respectively. Twenty-six interictal MA patients were compared with 26 matched MO patients and 26 healthy matched controls.
Results
The major findings were: connectivity between the anterior insula and occipital areas, including area V3A, was reduced in MA but not in MO. Connectivity changes between the anterior insula and occipital areas further correlated with the headache severity in MA only.
Conclusions
The unique pattern of connectivity changes found in interictal MA patients involved area V3A, an area previously implicated in aura generation. Hypoconnectivity to this and other occipital regions may either represent a compensatory response to occipital dysfunctions or predispose MA patients to the development of aura.
Introduction
In up to 30% of migraine patients, the headache is preceded by aura sensations typically lasting less than one hour (1). Visual aura, the most common type of aura sensations, occurs in more than 90% of aura patients (1,2). Prior studies established that the generation of visual aura is linked to cortical spreading depression, a slowly propagating wave of neuronal depolarization originating from the occipital cortex, followed by a sustained suppression of spontaneous neuronal activity (3). Other deficits in the visual system have been found in the interictal phase both in migraine with visual aura (MA) and migraine without aura (MO). For example, the visual cortex exhibits abnormal inter-ictal excitability and reduced habituation suggesting deficient regulatory mechanisms of cortical excitability (4–7). Both MA and MO patients also display interictal impairments of visual form and motion perception, which involve areas in the inferior temporal (IT) and middle temporal (MT) cortices (8–10). A visual network related to motion processing has previously been found to be dysregulated in MO patients (11). The presence of interictal cognitive impairments, including impaired visual and sustained attention (12–15), furthermore suggest altered prefrontal processing in both migraine groups (16–18). Interestingly, hyper-responsiveness to visual stimuli has previously been found in cortical regions subserving these functions in MA patients (19). Whether abnormalities in occipital and temporal cortical processing are related to dysfunctions in cognitive control networks remains an open question.
The brain has been shown to be organized into multiple intrinsic functional resting-state networks (RSNs), each exhibiting low-frequency temporal correlations among its constituent areas (20). Two RSNs have been identified linking higher-order cognitive functions to visual processing: the dorsal attention network (DAN) and the salience network (SAL) (17,20,21). Both networks exhibit increased activity during attention-demanding tasks and have been implicated in different aspects of sensory modulation (16,17,21). The frontal eye field, located in the middle frontal gyrus, is the main node within the DAN. It is a region that has the capacity to influence processing of visual form and motion through reciprocal connections to extrastriate visual regions in both the ventral and dorsal visual streams (16,17,21). The anterior insula is a key node within the SAL. It is a region thought to evaluate the impact of sensory stimuli on the body state based on the integration of multimodal sensory input (22,23). The anterior insula is, therefore, well positioned to detect and respond to aberrant signals resulting from either deficient regulation of cortical excitability or the dysfunctional state that elicits cortical spreading depression. Taken together, it is conceivable that the frontal eye field, the anterior insula, and their associated RSNs may exhibit altered functional interactions with regions in the occipital and temporal lobes of migraine patients and that some of these changes may be specific to MA.
To address whether abnormal occipital and temporal cortical processing is related to dysfunctions in cognitive control networks in migraine, resting-state functional magnetic resonance imaging (fMRI) was used to examine the functional connectivity of the anterior insula, within the SAL, and the frontal eye field, within the DAN, since both regions are capable of influencing visual processing. Additionally, connectivity of the posterior cingulate, the main node within the default mode network (DMN), was examined because this region exhibits anticorrelations with the DAN and the SAL, i.e. it is attenuated, among others, by sensory stimuli and attention -demanding tasks. Enhanced connectivity between the DMN and the insula (24) and decreased connectivity within the DMN have previously been found in MO patients (25). As MA patients (1) have aura symptoms that are not observed in MO patients and (2) share symptomatology with MO patients, we hypothesize that abnormalities specific to MA exist and that additional abnormalities common to MA and MO can be found in functional connectivity of these networks. To this aim, MA patients were compared to matched MO patients and healthy matched controls. Since episodic and chronic migraine might involve different pathophysiologies, the present study included only patients with low-frequency migraine to avoid mixed results.
Material and methods
Study population
Patients with episodic migraine were recruited from the Headache Clinic of Taipei Veterans General Hospital and were diagnosed according to the International Classification of Headache Disorders, second edition (2004). Healthy controls were recruited if they did not have any history of migraine; however, infrequent episodic tension-type headache (<1 headache day/month) was allowed. Apart from the migraine diagnosis in the patient groups, both patients and healthy controls were normal in physical and neurological examinations and did not have any systemic diseases or psychiatric disorders. The study was approved by the Institutional Review Board of Taipei Veterans General Hospital and was conducted in accordance with the Declaration of Helsinki (sixth revision). Prior to entering the study, all participants gave their informed consents to participate.
Each patient was required to keep a headache diary and completed a semi-structured questionnaire on demographics, headache and aura profile, past medical history, and usage of medication, during his or her first visit. All the recruited patients underwent MRI scanning (see below) and the time interval between scanning and the most recent migraine attack prior to or after the measurement was determined based on their headache diaries and telephone follow-up. Patients were enrolled in this study if they (1) had one to nine headache days per month, i.e. low-frequency migraine attacks; (2) were in the interictal state, i.e. had no migraine attacks within 48 hours before and after the MRI scanning; (3) had not received any prophylactic medication six months prior to the experiment; and (4) took no acute abortive medications 48 hours prior to the scanning. In addition, all MA patients had to have active visual aura, i.e. more than one migraine with visual aura per month, and aura preceding the headache in more than 50% of all migraine attacks.
Image acquisition
Data were acquired at Taipei Veterans General Hospital on a 3-Tesla GE Discovery MR750 (GE Healthcare, Waukesha, WI, USA) scanner. To diminish motion artifacts during scanning, the participant’s head was immobilized with cushions inside the coil. Functional data were acquired with a T2*-weighted gradient echo planar imaging (EPI) sequence using blood oxygenation level-dependent contrast (BOLD; repetition time (TR)/echo time (TE)/flip angle (FA): 2000 ms/30 ms/90 degrees) with the parameters: matrix, 64 × 64 × 40; field of view (FOV), 230 × 230 mm2, coverage in the slice direction, 160 mm; slice thickness, 4 mm. Two hundred volumes were acquired for each participant. During functional scans, the participants were instructed to remain awake and to keep their eyes open to reduce the influence of the global signal on the RSNs (26). However, no objective measures were implemented to ensure that participants complied with these instructions. The anatomical image was acquired using a T1-weighted, three-dimensional (3D)-gradient echo pulse sequence (fast spoiled gradient echo (FSPGR); TR/inversion time (TI)/FA: 9.156 ms/3.68 ms/12 degrees) with the following parameters: matrix, 256 × 256 × 180; FOV, 230 × 230 mm2; slice thickness, 1 mm. In order to quantify the total volume of white matter lesions, a fluid-attenuated inversion recovery sequence (FLAIR; TR/TE/TI/FA: 6000 ms/ 129.2 ms/ 1864 ms/90 degrees) was applied with the parameters: matrix, 512 × 512 × 180; FOV, 260 × 220 mm2, slice thickness, 1 mm with 1-mm gap.
Data processing
Functional data were preprocessed and analyzed with statistical parametrical mapping (SPM8, Wellcome Trust Centre for Neuroimaging, London, http://www.fil.ion.ucl.ac.uk/spm) and the REST toolbox (27). Using SPM8, scans were slice time corrected, head motion corrected (all participants: translation < 1.5 mm and rotation < 1.5 degrees) and coregistered to the individual anatomical image before spatial normalization to standard space using the Montreal Neurological Institute atlas (MNI 152). Scans were further re-sampled (2-mm3 voxel size) and smoothed with a Gaussian kernel (full width at half-maximum: 8 mm). In REST, this was followed by removal of linear trends and temporal band-pass filtering (0.01–0.08 Hz). To remove nonspecific sources of variance the following averaged time courses were regressed out: (1) the whole brain signal, (2) the cerebrospinal fluid signal, (3) the white matter signal, and (4) the six head motion parameters obtained from the realignment. Although removal of the whole-brain signal from resting-state fMRI data is a widely used pre-processing step, its use is controversial. Hence, the analyses were repeated without removal of the whole-brain signal. A seed-correlation analysis was then performed for selected spherical regions of interest (ROIs). For each individual and each seed-point, r-maps were normalized to Z-maps using Fisher’s r-to-Z transformation. To detect regional differences in functional connectivity, the Z-maps entered into a seed-point specific analysis of variance (ANOVA) model with the three participant groups as a variable.
Seed-points were defined as spheres of 4-mm radius centered at the bilateral middle frontal gyri (MFG) (left DAN: (–29, –9, 54); right DAN: (29, –9, 54)) and anterior insula (left SAL: (–41, 3, 6); right SAL: (41, 3, 6)), and midline posterior cingulate (DMN: (0, –52, 27) (28, 29). Based on the findings from the left anterior insula (see results), we further performed a post-hoc analysis for a seed placed in the right cuneus ((24, –75, 24)). Initially, correlated and anticorrelated statistical maps were generated in the healthy control group to derive the corresponding functional networks. The threshold was set at a voxel-wise q < 0.05, corrected for false-discovery rate (FDR) in the entire brain volume. To examine MA-specific differences in functional connectivity, the MA group was compared to the MO and healthy control groups. For each of the patient groups, regression analyses were also performed for the seed-points using separate one-sample t-tests with mean headache severity (scale: 0–10), migraine frequency (days per month), most recent migraine attack (days), disease duration (years), and scores from the Visual Aura Rating Scale (30) as regressors. In addition, the migraine frequency was further subcategorized into migraines with and without aura in the MA group. For all these tests, the resulting statistical maps were first thresholded at an uncorrected voxel-wise threshold of p < 0.005. Clusters passing an extent threshold of q < 0.05, FDR corrected in the entire brain volume, were deemed significant. Anatomical structures were labeled by transforming the MNI coordinates obtained from SPM into Talairach space (http://imaging.mrc-cbu.cam.ac.uk/imaging/MniTalairach) before entering into the Talairach Client (http://www.talairach.org/client.html).
An optimized voxel-based morphometric (VBM) analysis was performed on the anatomical images to assess the possible influence of structural volumetric changes on differences in functional connectivity. The segmented gray matter images were analyzed in SPM8 according to the procedures described in Tu et al. (31). A similar ANOVA model as implemented for the functional data was set up but with the total intracranial volume and age as covariates of no interest. As for the functional data, clusters were considered significant when passing an FDR-corrected extent threshold of q < 0.05.
The Lesion Segmentation Tool software (http://dbm.neuro.uni-jena.de/software/lst/), running under SPM8, was used for automated calculation of the total volume (in mm3) of hyperintensity lesions. These calculations were based on both the anatomical image and the FLAIR image and used the default settings of the toolbox.
Statistical analysis
The SPSS software package for Windows (version 15.0; SPSS Inc, Chicago, IL, USA) was used for statistical analysis of demographic data and the total lesion volume. Chi-square tests or one-way ANOVA were used for comparisons when appropriate. Statistical significance was set at p < 0.05. All tests were two tailed.
Results
Demographic and clinical characteristics
Demographics of healthy controls and patients (mean ± SD).

MO: migraine without aura; MA: migraine with aura. BDI: Beck Depression Inventory; MIDAS: Migraine Disability Assessment; VARS: Visual Aura Rating Scale; NSAID: nonsteroidal anti-inflammatory drug; na: not applicable, d/m: days/month. aOne-way analysis of variance (ANOVA) test or independent t test, when appropriate. bChi-square test.
The seed-based RSNs
The DMN was derived from a seed placed in the posterior cingulate cortex. The resulting correlation map in healthy controls included regions most commonly associated with the DMN, i.e. the posterior cingulate cortex, the precuneus, the ventral and dorsal medial prefrontal cortices, and bilateral inferior parietal lobes, middle temporal gyri, and dorso-lateral prefrontal cortices (Figure 1(a)). Seeds placed in the left and right middle frontal gyri (frontal eye field) each generated the typical bilateral fronto-parietal pattern associated with the DAN (Figure 1(b)). Seeds placed in the left and right anterior insula resulted in the SAL and included regions throughout the bilateral insulae and the temporal-parietal junctions, and in the medial premotor areas and the anterior cingulate (Figure 1(c)). It is noteworthy to mention that the regions within the DAN and SAL were anticorrelated with the DMN and vice versa (Figure 1(a)–(c)).
Correlated (red) and anticorrelated (blue) networks for seeds placed in (a) the posterior cingulate (PCC), (b) the left middle frontal gyrus (MFG; frontal eye field), and (c) the left anterior insula (AINS) in healthy controls (CTL). The derived networks from the three seeds corresponded to the default mode network (DMN), the dorsal attention network (DAN), and the salience network (SAL), respectively. Regions within the DAN and SAL were anticorrelated with the DMN and vice versa. All regions passed a false-discovery rate (FDR)-corrected extent threshold of q < 0.05.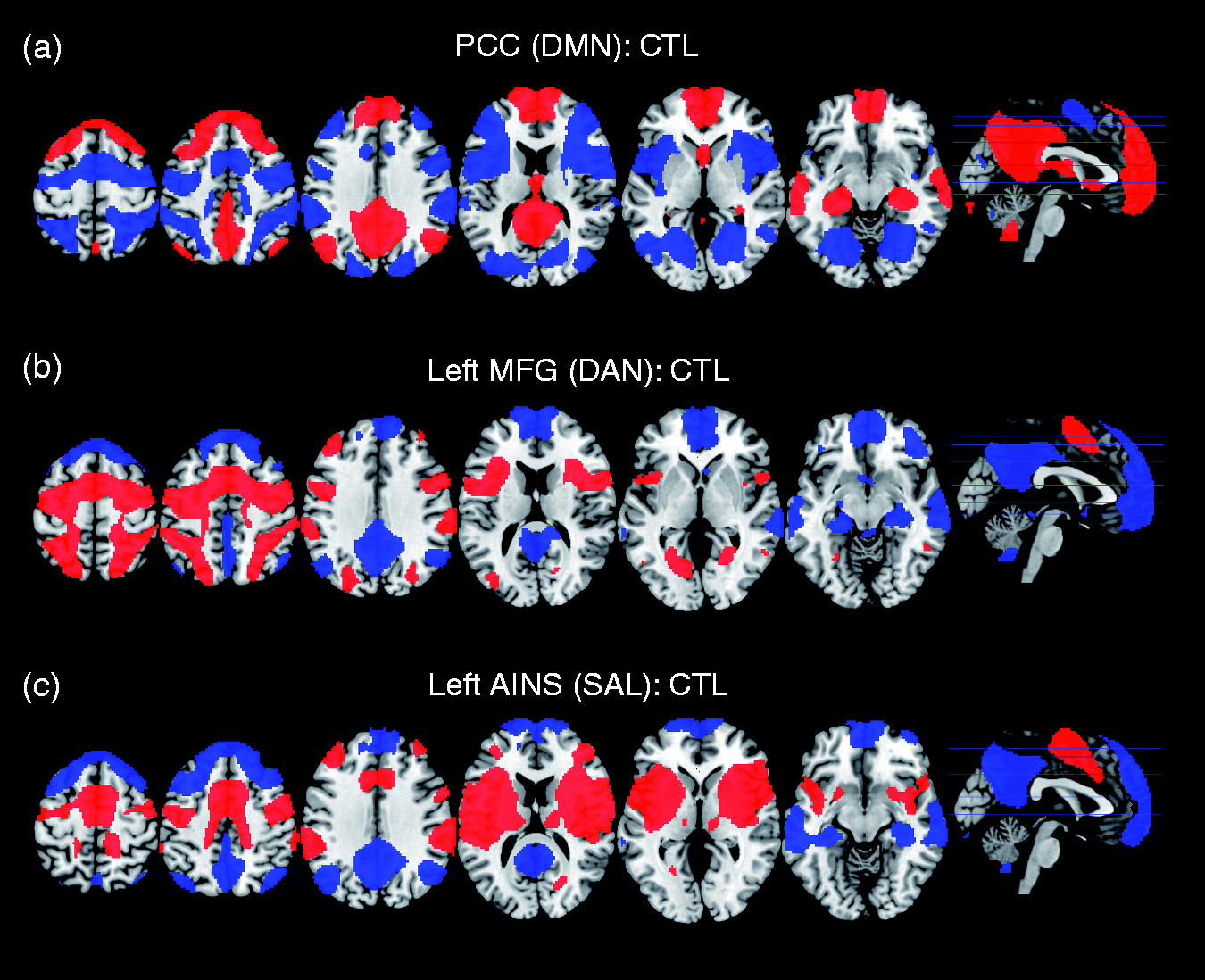
Between-group analysis
Between-group comparisons revealed increased functional connectivity between the left middle frontal gyrus, the seed within the DAN, and two clusters located in the ventral medial prefrontal cortex and the right IT and MT regions in MA compared to healthy controls (Figure 2(a), Table 2). When MO was compared to healthy controls, increased connectivity was also found between the left middle frontal gyrus and the same regions in the right IT and MT (Figure 2(b), Table 2). Regional differences were not found for the seed when comparing MA to MO. Thus, enhanced connectivity to the temporal regions is a feature shared by both MA and MO.
Regions exhibiting altered functional connectivity with the left middle frontal gyrus (MFG; frontal eye field) within the dorsal attention network and with the left anterior insula (AINS) within the salience network. For MFG, increased connectivity (red) was found in (a) migraine with visual aura (MA) and (b) migraine without visual aura (MO) compared to healthy controls (CTL) in the right temporal regions. For AINS, reduced (blue) functional connectivity with occipital regions was found for MA compared to (c) CTL and (d) MO. All regions passed a false-discovery rate (FDR)-corrected extent threshold of q < 0.05. Brain regions with increased and decreased functional connectivity in migraine with (MA) and without (MO) visual aura compared to healthy controls (CTL). Clusters passing a voxel threshold of p < 0.005 (T > 2.64) and a corrected extent threshold (q < 0.05) were considered significant. BA: Brodmann area; FDR: false-discovery rate correction; G: gyrus. Coordinates (x, y, z) are in millimeters.
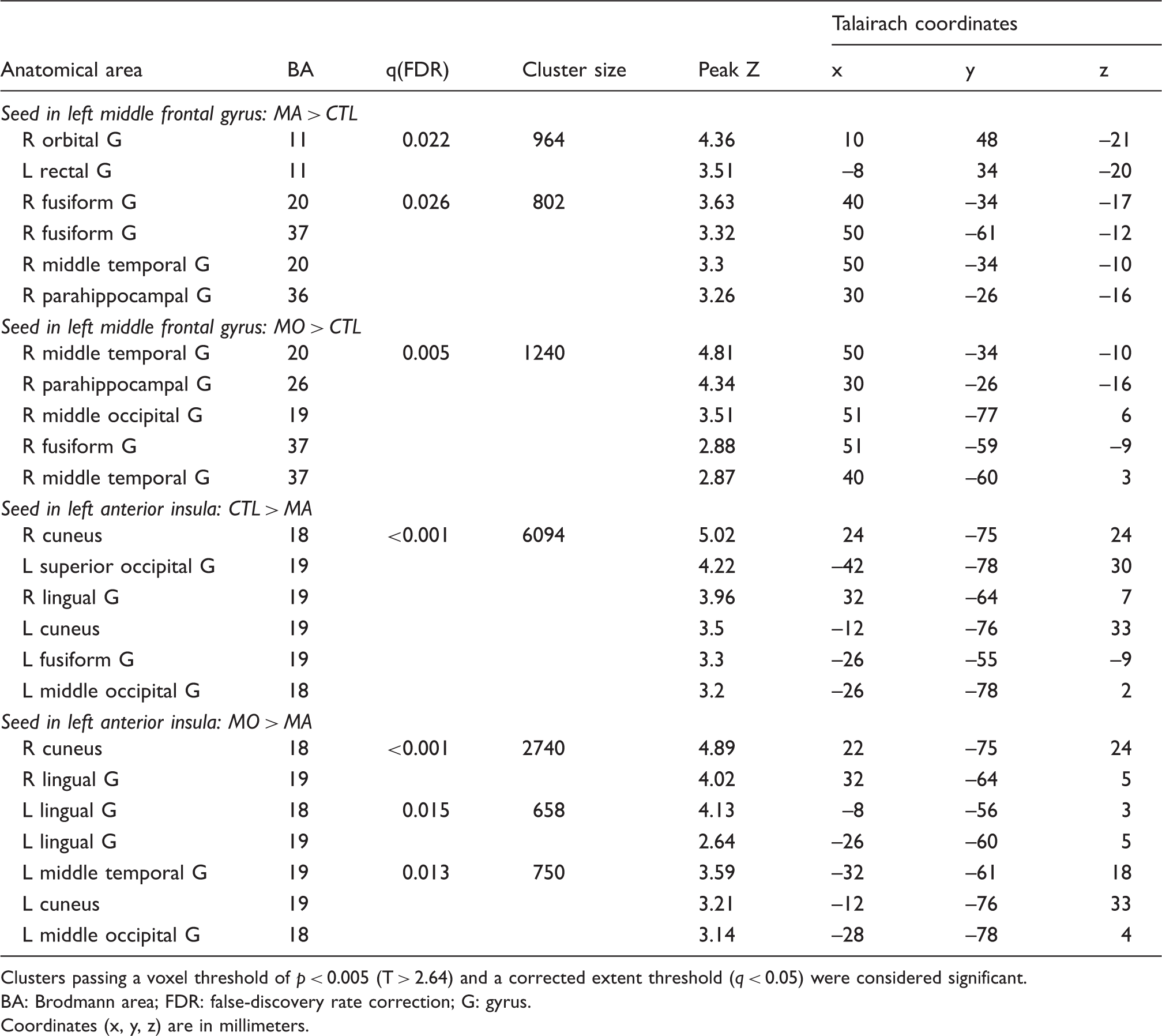
For the seed placed in the left anterior insula within the SAL, decreased functional connectivity was found to extrastriate regions mainly within the bilateral occipital lobes when MA was compared to healthy controls (Figure 2(c), Table 2). Similar regions also exhibited decreased connectivity when MA was compared to MO (Figure 2(d), Table 2). Regional differences were not found for the seed when comparing MO to healthy controls. Thus, decreased connectivity between the key node within the salience network and regions within the occipital lobe was specific to MA patients. Connectivity changes were not found for the other seeds placed within the DMN, the right DAN, and the right SAL.
To further examine the network of regions associated with reduced occipital connectivity in MA, a seed was placed within the dorsal extrastriate visual cortex within the occipital lobe at the principal locus from the previous analysis (right cuneus: (x, y, z) = (24, –75, 24) from Table 2). This seed was also used to examine if the between-group differences in functional connectivity to the occipital lobe involved only the left anterior insula. The derived RSN from the occipital seed in healthy controls corresponded to the intrinsic visual network and encompassed the bilateral striate and extrastriate visual areas (Brodmann areas 17, 18, and 19) and the bilateral frontal eye fields in the middle frontal gyri, as well as the bilateral anterior insula (Figure 3(a)). For the same seed, comparisons of MA with both MO and healthy controls resulted in a widespread decrease in functional connectivity to regions encompassing the right anterior insula and basal ganglia. Decreased connectivity was also found to the medial premotor areas for MA compared to healthy controls. These regions are all integral parts of the SAL (Table 3, Figure 3(b) and 3(c)). Note, statistical values were not reported for the left anterior insula to avoid circular analysis. Additionally, enhanced functional connectivity was found between the occipital seed and regions of the DMN, in particular the posterior cingulate and the precuneus, when comparing MA to both MO and healthy controls (Table 3, Figure 3(b) and 3(c)).
Functional connectivity and connectivity changes of the right cuneus. (a) Regions exhibiting positive correlations (red) with the cuneus in healthy controls (CTL) were mainly located within the occipital lobe. Enhanced (red) and reduced (blue) functional connectivity was found within the default mode and salience networks, respectively, when comparing migraine with visual aura (MA) with either (b) CTL or (c) migraine without visual aura (MO). All regions passed a false-discovery rate (FDR)-corrected extent threshold of q < 0.05. Brain regions with increased and decreased functional connectivity to the right cuneus in migraine with (MA) and without (MO) visual aura compared to healthy controls (CTL). Clusters passing a voxel threshold of p < 0.005 (T > 2.64) and a corrected extent threshold (q < 0.05) were considered significant. Put: putamen; BA: Brodmann area; FDR: false-discovery rate correction; G: gyrus. aStatistical values are not reported for the left anterior insula to avoid circular analysis.

Regression analysis
Regions exhibiting correlations with headache and migraine parameters.

Lb: lobule; +: positive correlation; BA: Brodmann area; FDR: false-discovery rate correction; G: gyrus.
The influence of global signal regression
In the main analysis above, the global signal was removed from the data by regression. Although this pre-processing step is commonly applied, its use remains controversial because global signal removal may introduce artifactual anticorrelations, which may further complicate between-group comparisons. Hence, to examine the influence of global signal regression, the analysis was repeated without removal of the global signal. For the contrast in which MA previously exhibited reduced connectivity with healthy controls, both the cluster size and the significance increased (Supplementary Figure 1(a), Supplementary Table 1). In addition, the seed placed in the right anterior insula now resulted in a significant reduction in functional connectivity to regions within the occipital lobe in MA compared to healthy controls but not compared to MO (Supplementary Figure 1(b), Supplementary Table 1). The contrasts for which both MA and MO exhibited increased connectivity did not result in any significant findings as was the case for the regression analyses. In summary, using global signal regression does not affect the finding of reduced connectivity between the SAL and visual areas in the occipital lobe of MA patients.
Structural changes
A significant decrease in gray matter volume was found only for the MA group relative to healthy controls in the right parahippocampus (cluster peak, (21, –16, –22); peak Z = 4.28; cluster size, 690 voxels; cluster extent corrected q = 0.028). An overlap between structural and functional changes was observed for this locus (not including the IT or MT) in MA.
Discussion
Migraine is a complex neurological disorder that may affect multiple brain systems. Questions remain regarding whether deficits found throughout the visual hierarchy in interictal migraine are interrelated and whether interictal MA is associated with distinct brain alterations within this hierarchy. We addressed these questions by examining the functional connectivity of intrinsic cognitive RSNs in well-matched participant groups and by identifying a specific pattern of connectivity that distinguished MA patients from MO patients. We first showed that the left anterior insula, one of the main nodes within the SAL, had reduced connectivity to extrastriate visual areas in the occipital lobe of interictal MA patients only. We further showed that the principal locus among these visual areas was functionally part of a visual network, mainly located in the occipital lobe, and that the reduced connectivity of this region further encompassed other major regions of the SAL. Additionally, connectivity changes between the anterior insula and extrastriate visual regions in the occipital lobe were associated with headache severity in MA patients. These findings occurred without concomitant changes in regional gray matter volumes in the occipital lobe and the insula. The absence of major structural volumetric changes in patients with low-frequency migraine is congruent with a previous study on a mixed group of low-frequency migraine patients (32).
Brain connectivity changes specific to MA
Reduced connectivity between the anterior insula and extrastriate visual areas in the occipital lobe was found to uniquely characterize MA patients. Since MA patients were in the interictal state, these changes were independent of the presence of aura attacks. However, known interictal occipital dysfunctions are shared between the two patient groups (7,8,10,14). Furthermore, we tried to eliminate potential confounding factors by matching the patient groups with respect to demographics and clinical profiles. It is, thus, likely that our finding should be explained in the context of the cortical dysfunction preceding and leading to elicitation of cortical spreading depression, the cortical homologue of visual aura (3). In support of this, the principal locus among the occipital areas exhibiting reduced connectivity was located in the medial-dorsal portion of the occipital lobe corresponding to the dorsal area V3A (33), where cortical spreading depression most likely is initiated (3).
The connectivity change between the anterior insula and extrastriate visual areas, including area V3A, in MA patients was inversely related to headache severity. This finding suggests an interaction between interictal connectivity changes and headache-generating mechanisms. A causal relationship between migraine aura and headache has previously been established in a migraine animal model (34). Further on, the anterior insula is well positioned to detect and respond to signals from visual and pain-related regions (22,35). The anterior insula is concerned with higher-level integrative processes based on multimodal convergence. The integration of sensory input with visceral, autonomic, and hedonic information has been suggested to subserve the evaluation of the impact of sensory stimuli on the body state (22,23). Moreover, the anterior insula is involved in the anticipation of aversive outcomes and provides information about future aversive states (23,36). Taken together, the anterior insula is capable of responding to a dysfunctional occipital lobe in MA patients. The relationship between aura-generating mechanisms and headache may, therefore, reflect the integration of relevant visual and pain-related information in this region. Our findings were not limited to the anterior insula but included several other important areas within the SAL, including the inferior frontal gyrus, the anterior cingulate/superior frontal gyrus, the basal ganglia and the anterior portion of the middle frontal gyrus. This suggests that our main findings should be interpreted within the general framework of the SAL, i.e. detection and reorientation toward salient and behaviorally relevant stimuli (17,22). It is not, however, possible to discern from RSNs how altered patterns of connectivity influences the processing of sensory stimuli. Our findings may, thus, represent a compensatory response to the occipital dysfunction that renders MA patients susceptible to cortical spreading depression. Alternatively, the altered pattern of connectivity may predispose MA patients to the development of cortical spreading depression.
Increased functional connectivity was also found between the extrastriate visual seed in the occipital lobe and the posterior DMN, including the precuneus and the posterior cingulate, in MA patients, but not in MO patients. Previous resting-state studies on other pain conditions have demonstrated reduced connectivity between the DMN and visual areas (37) and increased connectivity between the DMN and the insula (37–39). The latter was also found in interictal MO patients (24). Together, these reports further confirm the different mode of brain functional organization in interictal MA patients. The increased coupling with the DMN and the decreased coupling with the SAL suggests a shift in the connectivity balance of the extrastriate area. Interestingly, the SAL has been proposed to coordinate such shifts between the DMN and task-relevant/non-task-relevant brain networks (40). It is noteworthy that the connectivity within the DMN, i.e. between the posterior cingulate and the medial prefrontal and medial temporal regions, increased as the time interval to the most recent migraine attack increased. This not only suggests an ictal disruption of the DMN but also that a restorative process occurs in these brain regions in the interictal phase.
Brain connectivity changes shared between MA and MO
Increased functional connectivity of the middle frontal gyrus, the main node within the DAN, was found to the right IT and MT areas of both MA and MO patients. The location of the middle frontal seed at the junction of the superior frontal sulcus and the precentral gyrus corresponds to the frontal eye field, a region exerting top-down modulation on visual processing in the temporal cortex (16,18). The IT and MT participate in visual object recognition and motion perception, respectively. These functions may be impaired in the interictal phase of both MA and MO patients (8–10). Although no structural volumetric changes were found in these regions in our study, others have observed increased cortical thickness and compromised white matter integrity of similar areas in both migraine subgroups (41,42). Together, functional and structural cortical changes in these regions could either underpin or arise as a consequence of neural dysfunction leading to perceptual changes.
The effect of global signal regression
Resting-state fMRI data contain a global component that is mainly found in gray matter and is common to most of the RSNs (28). The global component consists of a mixture of neurophysiological signals, systemic physiological noise, and instrumental noise (43,44). The rationale for the removal of the global signal is that the signal obscures the underlying dynamics of interacting regions with known anatomical connections (28,45). Retaining the global signal in the data may further result in inaccurate hypothesis testing by inflating the estimates of the residual error in the model used to assess the data (46). However, removal of the global signal may also introduce spurious artificial anti-correlations (46,47), which can result in larger spatial extent of the anti-correlated regions (46,47). In our study, the main result of reduced functional coupling between the visual and salience networks not only remained significant with and without global signal regression but the spatial extent of the regions was larger without regression. This suggests that artificial anti-correlations did not contribute to the result and that the global signal regression resulted in the more conservative finding. Since it cannot be ruled out that the other results, demonstrated only when the global signal was regressed out, were influenced by artificial anti-correlations, these should be interpreted with caution.
Study limitations
There are some limitations to our study that must be addressed. Assessments of cognition and visual perceptions were not performed in our study. It is, thus, not possible to elaborate further on the functional significance of our findings. However, we consider the present study to be hypothesis generating, enabling future studies to target the specific brain regions revealed here with tailored behavioral tests. For the same reason, a Bonferroni correction for multiple seeds was not implemented. Nonetheless, our main findings would remain after such a correction. In the present study, the contribution of structural volumetric changes to the observed functional connectivity changes was examined. However, since cortical thickness or white-matter microstructural changes were not assessed, a contribution from these cannot be ruled out.
The hypothesis-driven seed-based approach implemented here requires a priori selection of the seed locations. These locations may not be optimal, thus resulting in reduced statistical sensitivity. This was the case in our study for the seeds placed in the posterior cingulate and the right anterior insula. These seeds did not yield any significant findings. In contrast, the occipital seed yielded significant connectivity changes to both the posterior cingulate and the anterior insula albeit at different loci from the initial locations. The sensitivity of the seed-based approach to the seed location is further highlighted by a recent resting-state study in MA patients in which seeds in extrastriate visual areas and in the posterior cingulate did not yield any significant findings (48). Interestingly, one of the posterior cingulate seeds in the study by Hougaard et al. (48) overlapped in location with our initial seed in the posterior cingulate and resulted in a negative finding in both studies. Since it is difficult to decipher to what extent methodological differences, e.g. longer repetition time, different seed locations and larger ROIs, can account for the discrepant findings, our study would gain in strength by being reproduced in a separate MA population.
Clinical implications
Our findings lend support to the notion that migraine with and without aura are distinct disorders but with overlapping features. Migraine with aura is characterized by specific interictal brain pathophysiology that differentiates it from migraine without aura. Aura-related brain pathophysiology, previously found only just prior to a migraine attack, extends into the symptom-free interictal phase. Interictal pathophysiology involves brain regions previously found to be involved in aura generation.
Footnotes
Funding
This work was supported by grants from the National Science Council, Taiwan (NSC 100-2314-B-010-019-MY2, NSC 100-2314-B-010-018-MY3), Taipei Veterans General Hospital (VGHUST102-G7-6-1, V102C-118, V102E9-001), Ministry of Health and Welfare (MOHW 103-TDU-B-211-113003), NSC support for Center for Dynamical Biomarkers and Translational Medicine, National Central University, Taiwan (NSC101-2911-I-008-001), Brain Research Center, National Yang-Ming University, and a grant from the Ministry of Education, Aim for the Top University Plan.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
