Abstract
Background
Diffusion tensor (DT) magnetic resonance imaging (MRI) provides several quantities with the potential to disclose white matter (WM) microstructural abnormalities. We explored alterations of WM architecture in pediatric migraine patients using DT MRI and two different methods of analysis.
Methods
Dual-echo and DT MRI scans were acquired from 15 pediatric migraine patients and 15 age-matched controls. Whole-brain voxel-wise comparisons of WM DT MRI abnormalities were performed using tract-based-spatial-statistics (TBSS). A DT probabilistic tractography analysis was also run.
Results
Both TBSS and DT tractography analysis showed that, compared to controls, pediatric migraine patients had significant lower mean (MD), axial (AD) and radial (RD) diffusivity of WM tracts located in the brainstem, thalamus and fronto-temporo-occipital lobes, bilaterally. Patients also experienced increased fractional anisotropy (FA) of the optic radiations. No correlation was found between WM tract abnormalities and disease duration and attack frequency.
Conclusions
Pediatric migraine patients harbor diffuse brain WM microstructural abnormalities. High FA and low MD, AD and RD in these patients might be explained by repeated neuronal activation, which may lead to cell swelling and stimulate activity-dependent myelin-modulation, or by increased fiber and dendritic densities. Both these mechanisms might reflect a hyperexcitability of the brain in migraineurs.
Introduction
Migraine affects around 15% of the population and is associated with a substantial personal and social burden. It is the most common acute and recurrent headache syndrome in children, with up to 75% of children reporting a notable headache by the age of 15 years (1).
During the past few years, the extensive application of advanced structural and functional magnetic resonance imaging (MRI) techniques has contributed to improving our understanding of the pathophysiology of migraine and to elucidating novel mechanisms that might become the target of future therapeutic interventions (2). What is now established is that migraine is not simply a disease related to pain occurring intermittently, but rather a process that over time either affects the brain or acts on a predisposed brain (3).
The vast majority of structural MRI studies have demonstrated that adult migraine patients experience a distributed pattern of gray matter (GM) and white matter (WM) abnormalities to regions involved in nociception, visual and sensorimotor processing (3). Whether these morphological abnormalities represent the consequence of attack repetition over time or rather a phenotypic biomarker of the disease is still a matter of debate (2). Although studying pediatric patients with migraine might help to unravel this issue, so far only one study has mapped the distribution of GM and WM volume abnormalities in pediatric migraine patients and found distributed GM abnormalities of brain regions involved in nociceptive and visual processing, but no WM volume abnormalities (4).
Diffusion tensor imaging (DTI) is a quantitative technique that exploits the diffusion of water within biological tissues, allowing the in vivo visualization of microscopic pathological abnormalities occurring in the central nervous system (CNS). From the tensor, it is possible to derive several indexes, which include fractional anisotropy (FA), a dimensionless index that reflects axonal integrity and fiber organization; mean diffusivity (MD), which measures the overall magnitude of diffusion; axial diffusivity (AD), which is associated with fiber density and axon intrinsic characteristics; and radial diffusivity (RD), which mostly reflects the degree of myelination (5).
Against this background, we performed an explanatory DT MRI analysis to assess the presence and extent of brain WM tract abnormalities from a cohort of pediatric patients with migraine. To provide some clues about the nature of the detected changes, we also assessed their correlation with disease duration and attack frequency.
Methods
This study was approved by the local ethics committees on human studies, and all participants’ parents provided written informed consent prior to study participation.
Participants
We included 19 right-handed pediatric patients with episodic migraine and 15 right-handed, gender- and age-matched healthy controls without a familial history of migraine, no history of neurological dysfunction (including migraine), a normal perinatal and child development and a normal neurological exam. Healthy controls were relatives of colleagues or friends who were kindly willing to take part in the study. Migraine patients were in a headache-free state for at least one month prior to the MRI scan. Patients with perinatal or pediatric diseases, hypertension, hypercholesteremia, diabetes mellitus, vascular/heart diseases and other major systemic, neurological or psychiatric conditions were excluded. Patients were recruited consecutively from the migraine population attending the Outpatient Clinic, Department of Neurology, San Raffaele Scientific Institute. All patients met the criteria of the International Classification of Headache Disorders for the diagnosis of migraine (6). None of the patients suffered from episodic syndromes frequently associated with migraine in childhood (e.g. benign paroxysmal vertigo, cyclic vomiting).
Images of four patients were discarded during the analysis because of motion artifacts (the demographic and clinical characteristics of these patients did not differ from those of the remaining group). Eight patients had a diagnosis of migraine with aura (MWA) (seven had visual aura and one sensory and speech disturbances), and seven had migraine without aura (MWoA). Apart from three patients who suffered from a right-sided migraine, all the remaining patients had bilateral migraine. Twelve patients had a familial history of migraine. Apart from three patients who complained of attacks lasting more than 24 hours, the duration of migraine attacks was of a few hours in the majority of the patients. At the time of MRI, five patients were on prophylactic medication for migraine, including flunarizine and ginkgolide B. All participants were assessed clinically a few days prior to the MRI scan by a single neurologist who was unaware of the MRI results.
MRI acquisition
Using a 3.0 Tesla Intera scanner (Philips Medical Systems, Best, the Netherlands), the following sequences of the brain were obtained from all participants: 1) axial T2-weighted turbo-spin echo (repetition time (TR)/echo time (TE) = 3000/120 ms, flip angle (FA) = 90 degrees, matrix size = 512 × 512, field of view (FOV) = 230 mm2, 28 4 mm thick, contiguous slices); 2) axial fluid-attenuated inversion recovery (FLAIR) (TR/TE = 11000/120 ms, inversion time = 2800 ms, FA = 90 degrees, matrix size = 256 × 256, FOV = 230 mm2, 28 4 mm thick, contiguous slices); and 3) pulsed-gradient spin-echo, echo-planar (TE/TR = 58/8775.5 ms; flip angle = 90 degrees; matrix size = 96 × 96; FOV = 240 × 240 mm2; 55 contiguous, 2.3 mm thick axial slices) with sensitivity encoding (SENSE) (acceleration factor = 2) and diffusion gradients applied in 35 non-collinear directions. Two optimized b factors were used for acquiring diffusion-weighted images (b1 = 0, b2 = 900 s/mm2).
MRI analysis
T2-weighted scans were analyzed for the presence of lesions and FLAIR scans were always used to increase confidence in their identification. T2-hyperintense lesion volumes (LV) were measured using a local thresholding segmentation technique (Jim 5.0, Xinapse System, Northants, UK).
DT MRI analysis
Diffusion-weighted images were first corrected for distortions caused by eddy currents and movements. Then, using the FMRIB’s Diffusion Toolbox (FDT tool, FSL 4.1, http://www.fmrib.ox.ac.uk), the DT was estimated in each voxel by linear regression (7) and FA, MD, AD and RD maps derived.
WM microstructure abnormalities were explored using tract-based spatial statistics (TBSS) analysis, which performs a voxel-wise analysis of the whole brain WM DT MRI measures (http://www.fmrib.ox.ac.uk/fsl/tbss/index.html). To confirm TBSS results, we also performed a DT probabilistic tractography analysis, which allows us to investigate DT MRI abnormalities of individual WM tracts.
TBSS voxel-wise analysis
Individual FA images were non-linearly registered to the FMRIB58_FA atlas provided within FSL and averaged to obtain a customized atlas. The resulting mean FA image was thinned to create a WM tract “skeleton,” which was thresholded at an FA > 0.2 to include only WM voxels. Individual-participant normalized FA maps were warped onto the FA skeleton before statistical comparisons, by searching perpendicularly from the skeleton for maximum FA values (8). The individual registration and projection vectors obtained during the above-described process were also applied to MD, AD and RD data.
DT probabilistic tractography analysis
FA, MD, AD and RD average values were estimated in WM tracts, which were derived using an atlas-based approach, as previously described (9,10). In detail, an FA atlas was created based on data from 24 healthy individuals with no previous history of neurological dysfunction (reference group). Then, using DT MRI tractography, probability maps of the left and right corticospinal tract (CST), superior longitudinal fasciculus (SLF), inferior fronto-occipital fasciculus (IFOF), uncinate fasciculus (UNC), optic radiation (OR), cerebellar peduncles (CP) (superior, middle and inferior), fornix (FX), cingulum (CIN) and corpus callosum (CC) were produced. The reference FA atlas was non-linearly transformed to the FA maps from controls and patients. These transformations were also applied to the probability maps of the WM tracts that were thresholded at 40%. Then, using WM tract maps, average FA, MD, AD and RD were derived.
Statistical analysis
Normal distribution assumption was checked for continuous variables by using Kolmogorov-Smirov and Shapiro-Wilk tests, as well as with graphical inspection of Q-Q plots, and between-group comparisons were performed using parametric and nonparametric tests as appropriate. Categorical variables were compared between groups using the Fisher exact test (SPSS software, version 21.0).
Voxel-wise differences in FA, MD, AD and RD values between migraine patients and controls were tested using a permutation-based inference for nonparametric statistical thresholding (“randomise” program within FSL) (11) and two-sample t tests, adjusting for age. The number of permutations was set to 5000. TBSS results were also confirmed with 10,000 permutations. The resulting statistical maps were thresholded at p < 0.05, with correction for multiple comparisons (family-wise error (FWE) corrected) at a cluster level using the threshold-free cluster enhancement (12). The WM tracts were identified using WM atlases, provided within FSL (13).
For tract-specific statistics, between-group comparisons were performed using a general linear model adjusted for age. P values were adjusted for multiple comparisons using the Bonferroni correction (SPSS software, version 21.0).
In migraine patients, correlations between voxel-wise DT MRI abnormalities vs disease duration and attack frequency were assessed using regression models in FSL (p < 0.05, corrected for multiple comparisons at the cluster level).
Results
Main demographic and clinical characteristics of individuals enrolled in the study.
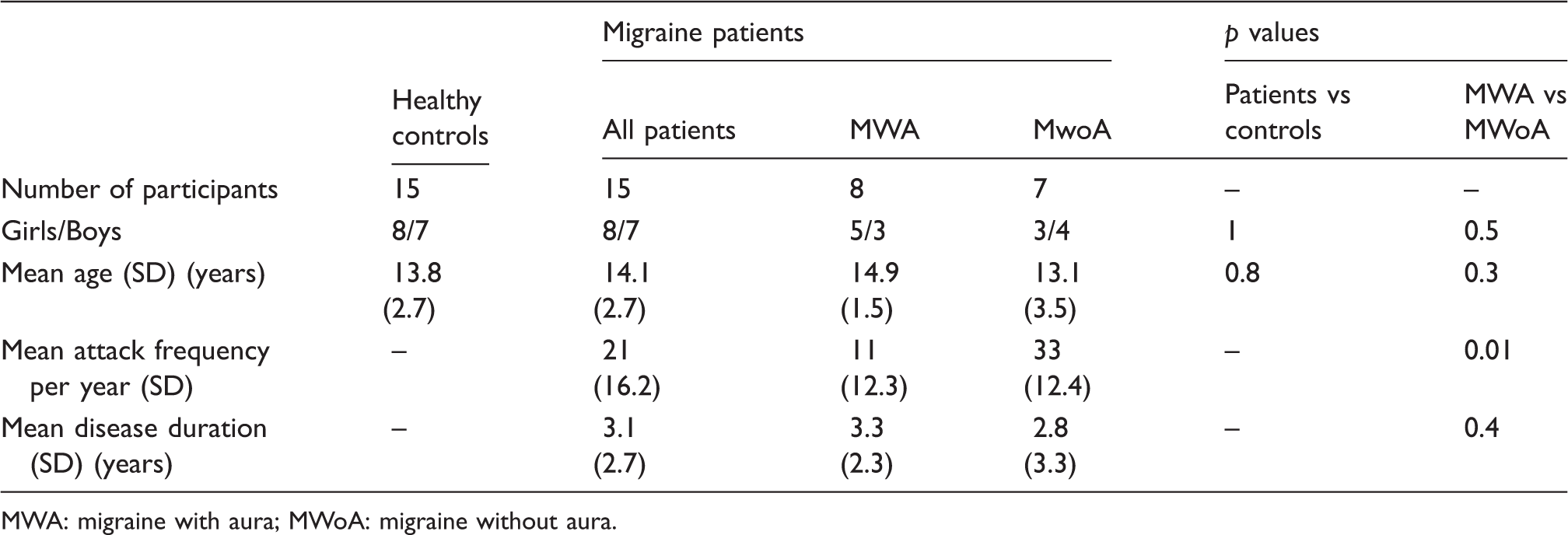
MWA: migraine with aura; MWoA: migraine without aura.
Age (p = 0.8) and gender (p = 1) did not differ between migraine patients and controls or between MWA and MWoA patients. Patients with MWoA had a higher attack frequency (p = 0.01) than those with MWA.
No WM hyperintense lesions were found in healthy controls and 13 (87%) of the 15 pediatric migraine patients. Two pediatric patients with MWoA had a few small, punctate T2 hyperintense lesions in deep and subcortical WM. Mean T2 LV in these patients was 0.08 ml (SD = 0.03 ml).
TBSS voxel-wise analysis
The results of the voxel-wise analysis of WM DT MRI measures are shown in Figure 1. Compared to controls, pediatric migraine patients had a distributed pattern of DT MRI abnormalities of the WM (p < 0.05, FWE corrected), characterized by: 1) a decreased MD of the bilateral optic tract, OR, CST, thalamic radiation, CIN, inferior longitudinal fasciculus (ILF) and IFOF, left SLF and CC; 2) a decreased AD of the bilateral optic tract and OR, left CST, ILF and IFOF, right CIN and CC; 3) a decreased RD of the bilateral OR, trigeminothalamic tract, CST, thalamic radiation, ILF and IFOF, right CIN and CC.
Areas of significant reduced white matter (WM) mean (red), axial (green) and radial (yellow) diffusivity (p < 0.05, FWE corrected) in pediatric patients with migraine vs healthy controls, superimposed on a fractional anisotropy template in the Montreal Neurological Institute space. MD: mean diffusivity, AD: axial diffusivity, RD: radial diffusivity, L: left, R: right; FWE: family-wise error rate.
No WM FA abnormalities were detected in pediatric migraine patients compared to healthy controls. Although this analysis has to be considered exploratory, given the low number of patients/group, no differences were found between MWA and MWoA patients.
DT probabilistic tractography analysis
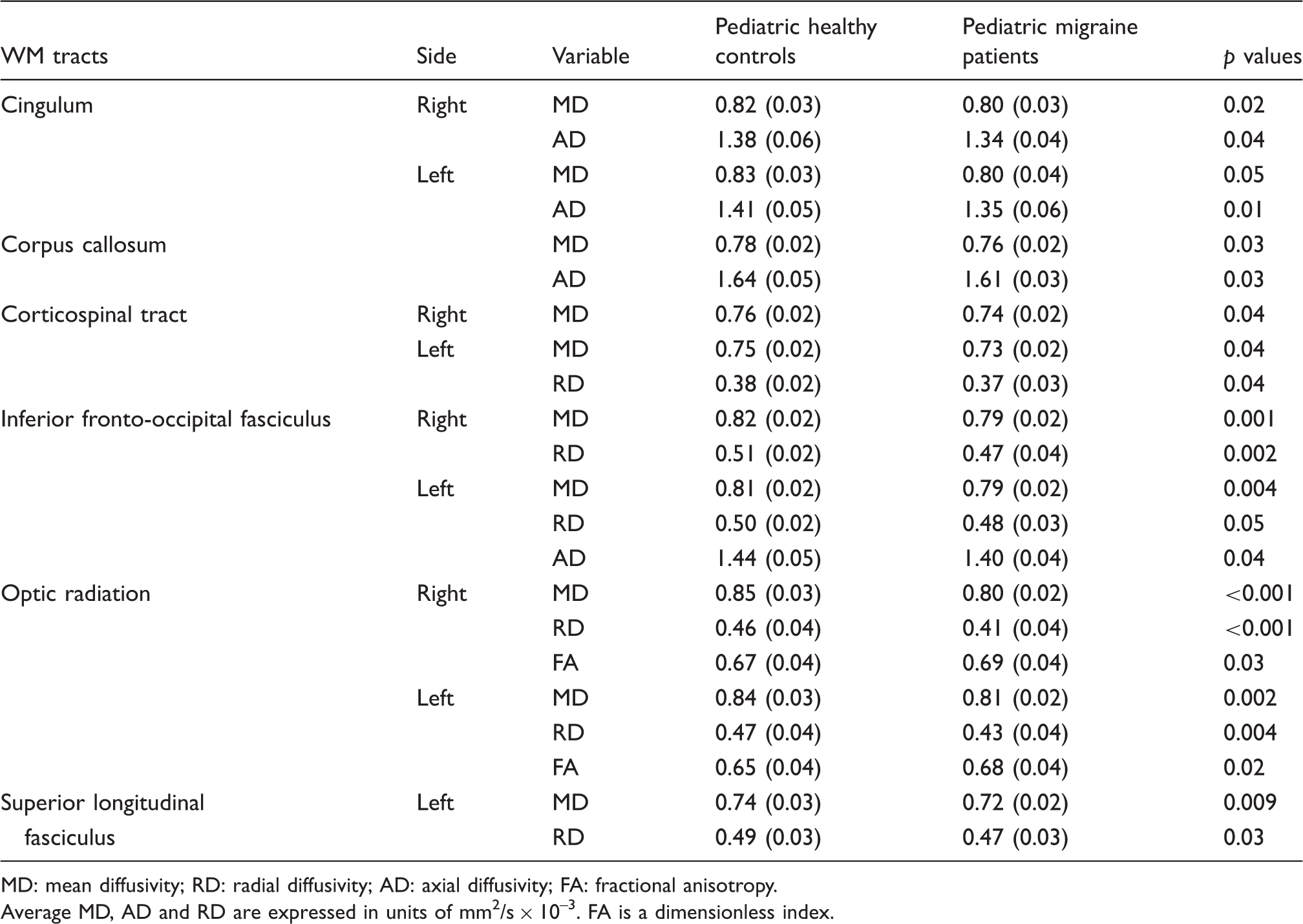
Table 2 summarizes the results of the comparisons of tract-average DT MRI metrics values between pediatric migraine patients and healthy controls (only significant tracts/indexes are reported). This analysis confirmed the presence of a distributed pattern of WM abnormalities in pediatric migraine patients, characterized by decreased MD, RD and AD values when compared to healthy controls. Pediatric migraine patients also showed an increased average FA of the bilateral OR compared to controls (p < 0.05, Bonferroni corrected) (Figure 2).
(a) Probability maps of the bilateral optic radiation (OR) superimposed on the fractional anisotropy (FA) atlas. (b) Box-plots comparing the FA values of the bilateral OR in pediatric migraine patients and controls. L: left, R: right, A: anterior, P: posterior. Diffusion tensor (DT) magnetic resonance imaging (MRI) metrics (mean values and standard deviations (SD)) of white matter (WM) tracts significantly different between pediatric patients with migraine and healthy controls. MD: mean diffusivity; RD: radial diffusivity; AD: axial diffusivity; FA: fractional anisotropy. Average MD, AD and RD are expressed in units of mm2/s × 10–3. FA is a dimensionless index.
Correlation analysis
In pediatric migraine patients, voxel-wise DT MRI abnormalities were not correlated with disease duration and attack frequency.
Discussion
Using different MRI techniques and analysis tools, a distributed pattern of morphological brain abnormalities has been consistently shown in adult patients with migraine (3). DT MRI studies have detected microstructural abnormalities of WM tracts connecting areas that are part of the pain network. However, the biological interpretation of these findings remains elusive. On the one hand, they might be a sign of maladaptive plastic modifications or the accumulation of damage to the WM (14,15) and, on the other, reflect functional alterations secondary to chronic or repetitive stimulation (10,16).
The first intriguing finding of our study is the in vivo demonstration of widespread abnormalities of diffusivity indexes occurring in the WM of pediatric patients with migraine. Such abnormalities were characterized by decreased MD, RD and AD along WM tracts related to nociception. The CC, CIN, CST and SLF connect brain regions known to be part of the sensory, motor, affective and cognitive components of pain. The CC contributes not only to interhemispheric transfer and integration of sensory and motor information (17), but also to attentive and inhibitory functions, suggesting that CC structural abnormalities might be associated with an altered inhibitory pain control (18). The CIN is part of the limbic system and is involved in emotions, attention and memory. The anterior CIN plays a key role in the emotion-affective state of pain and contributes to the modulation of nociceptive transmission during placebo analgesia (19). In our analysis, microstructural abnormalities mostly involved the mid- and posterior CIN, which is involved in skeletomotor orientation of the body to noxious stimuli and in assessing self-relevant sensations (19). Cortical motor efferents through the CST contribute to the descending pain modulatory system. Several neurophysiological studies have corroborated the existence of an interrelationship between pain and the motor system, suggesting that the motor cortex may be effective in reducing the intensity of pain perception (20). Moreover, functional MRI studies of patients with chronic painful conditions, such as fibromyalgia, have shown that activation of motor areas of the brain may influence the affective-motivational and cognitive-evaluative components of pain (21). The SLF interconnects frontal, temporal and parietal association areas. Beyond its central role in language, it contributes to higher cortical functions, such as memory and attention, as well as visuospatial and audiospatial processing, which are involved in different aspects of pain processing (22). These abnormalities might help to explain cognitive deficits described in adult and pediatric patients with migraine (23,24).
Similarly to what has been observed in adults (16,25,26), pediatric patients with migraine exhibited abnormalities of DT MRI indexes in the trigeminal and thalamocortical tracts. A large body of evidence supports the role of these two pathways in migraine pathophysiology. Recent studies have shown that cortical spreading depression (CSD), which is supposed to be the underlying mechanism of both MWA and MWoA (27), can activate trigeminovascular and thalamic neurons (28). In addition, several studies have demonstrated that pain and other typical migrainous symptoms, such as impaired habituation and hypersensitivity to nociceptive and visual stimuli, result from abnormal activation and sensitization of the trigeminovascular and thalamic tracts (28).
The second intriguing finding of this study is the extensive involvement of WM tracts implicated in processing of visual stimuli in pediatric migraine patients, as reflected by decreased MD, RD, AD values of the optic tracts and the OR, and an increased FA of the OR. These results are in agreement with the increasing number of MRI studies suggesting that brain hyper-excitability might explain the copious visual phenomena associated with migraine (2). In particular, the concomitant involvement of visual, trigeminal and thalamic pathways supports the notion that they are commonly involved in the genesis of migraine-related photophobia. Recent evidence suggests that photic signals from the retina converge at the trigeminovascular thalamocortical pathway, leading to exacerbation of migraine headache by light. In addition, the flow of nociceptive signals along the trigeminovascular pathway converges on the visual cortex and alters its responsiveness to visual stimuli (28). Pediatric migraine patients also had WM microstructural abnormalities of the ILF and IFOF, two WM tracts that play an important role in visuospatial processing. The ILF connects the extrastriate occipital cortex to anterior temporal structures, while the IFOF connects the frontal lobe with the posterolateral temporal and occipital lobes (29). Our findings, therefore, suggest an early involvement of the motion-processing visual network in migraine patients.
In the CNS, water diffusion is influenced by the microstructural components of tissues, including cell membranes, macromolecules and organelles. By tracking the principal diffusion directions, DT MRI provides a unique method for assessing the orientation and integrity of WM tracts, thus allowing us to draw inferences on the underlying pathologic substrates. MD is a tensor-derived measure that characterizes the overall magnitude of diffusion, whereas FA is a dimensionless index that quantifies the degree of anisotropy, which is a measure of tissue organization. FA has been associated with variations in axon count, density of axonal packing, and myelination. The diffusivity along the principal axis is called AD and is associated with axon diameter, count, and density, while the diffusivities along the two minor axes are often averaged to produce a measure of RD, which is mostly influenced by the degree of myelination (5). Based on these considerations, it is tempting to speculate that reduced MD, AD, RD and increased FA in pediatric migraine patients might reflect greater axon diameter or higher fiber and dendritic densities, which could support a more efficient transfer of information between brain regions involved in nociception and visual processing. Alternatively, but not mutually exclusive, DT MRI abnormalities we have found might be explained by a repeated neuronal activation that is likely to lead to cells swelling and stimulate activity-dependent myelin-modulation. There is, indeed, growing awareness that neuronal electrical activity may influence myelination, by modulating astrocytes activity (30). Finally, glial proliferation might be another mechanism at work in our patients, consistent with previous studies that reported glial proliferation and neurogenesis following repeated episodes of CSD and/or hypoperfusion (25). Recent studies pointed to migraine as a disorder of brain excitability. It has been hypothesized that a deficient regulation of the cortical excitatory-inhibitory balance may lead to hyperactivity of cortical circuits, thus creating the conditions for CSD ignition and propagation and the hypersensitivity to sensory stimuli (31). As a consequence, combined with recent neurophysiological data (32), our findings contribute to support the notion of such a state of brain hyperexcitability from the early stage of the disease.
Considering the cross-sectional design of our study, it remains difficult to clarify whether our observations are the cause or the consequence of migraine. Structural plasticity of WM tracts has been demonstrated after training (33) and rehabilitative treatments (34). However, the absence of a correlation between WM abnormalities and patients’ clinical characteristics, the positive familial history for migraine in the majority of our patients and the strong effect of genetic components on brain structure that has been reported by several authors (35) support the notion that such abnormalities may represent a phenotypic biomarker of pediatric patients with migraine.
Our findings in pediatric patients with migraine contrast with those of previous DT MRI studies of adult patients suffering from this condition, in whom an increased MD and decreased FA has been almost consistently demonstrated (10,14,16). Apart from aging, which influences the degree of myelination and maturation of WM tracts, and, as a consequence, DT MRI quantities (36), several other factors might contribute to explain this discrepancy. First, morphologic and functional imaging studies suggested that brain abnormalities in patients with migraine may be dynamic and influenced by several factors, including treatment, frequency of attacks, disease duration and stage of the disease (ictal vs iterictal) (2,3). Second, many factors occurring during the course of life, including concomitant diseases and environmental influences, can change the phenotypic expression of migraine (37).
Our study is not without limitations. First, the sample size of the two groups was relatively small. This is why, to reinforce our findings, we decided to report and describe exclusively results corrected for multiple comparisons. Second, we did not have reliable pieces of information concerning the time elapsed from the last attack, nor severity of pain or cognitive deficits, which would have allowed us to better interpret our findings. Finally, five patients were on prophylactic therapies, which might influence DT MRI metrics. Future studies should enroll larger numbers of individuals to confirm our results and should include an assessment of pain severity as well as neuropsychological tests, to ascertain the clinical relevance of WM microstructure abnormalities. Longitudinal studies are also warranted to explore the dynamics of the evolution of WM microstructural abnormalities in pediatric migraine patients.
Clinical implications
Both tract-based spatial statistics and diffusion tensor (DT) tractography analysis showed that, compared to pediatric healthy controls, pediatric migraine patients had significant (p < 0.05) lower mean (MD), axial (AD) and radial (RD) diffusivity of white matter (WM) tracts located in the brainstem, thalamus and fronto-temporo-occipital lobes, bilaterally, including the trigeminal tracts, cingulum, corpus callosum, corticospinal tract, superior and inferior longitudinal fasciculus (ILF), fronto-occipital fasciculus and optic radiation. In pediatric migraine patients, voxel-wise DT magnetic resonance imaging (MRI) abnormalities were not correlated with disease duration and attack frequency. The use of DT MRI to define the presence and distribution of WM damage in patients with migraine contributes to a better characterization of these patients and improves the understanding of the pathophysiological processes underlying this condition.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
None declared.
