Abstract
Aim
Migraine is known to run in families and has long been considered a strongly heritable disorder. This study sought to evaluate the relationship between age at onset of pediatric migraine and family history of migraine.
Methods
Review of the medical files of the headache clinic of a tertiary pediatric medical center yielded 344 children with migraine for whom details on migraine in family members were available.
Results
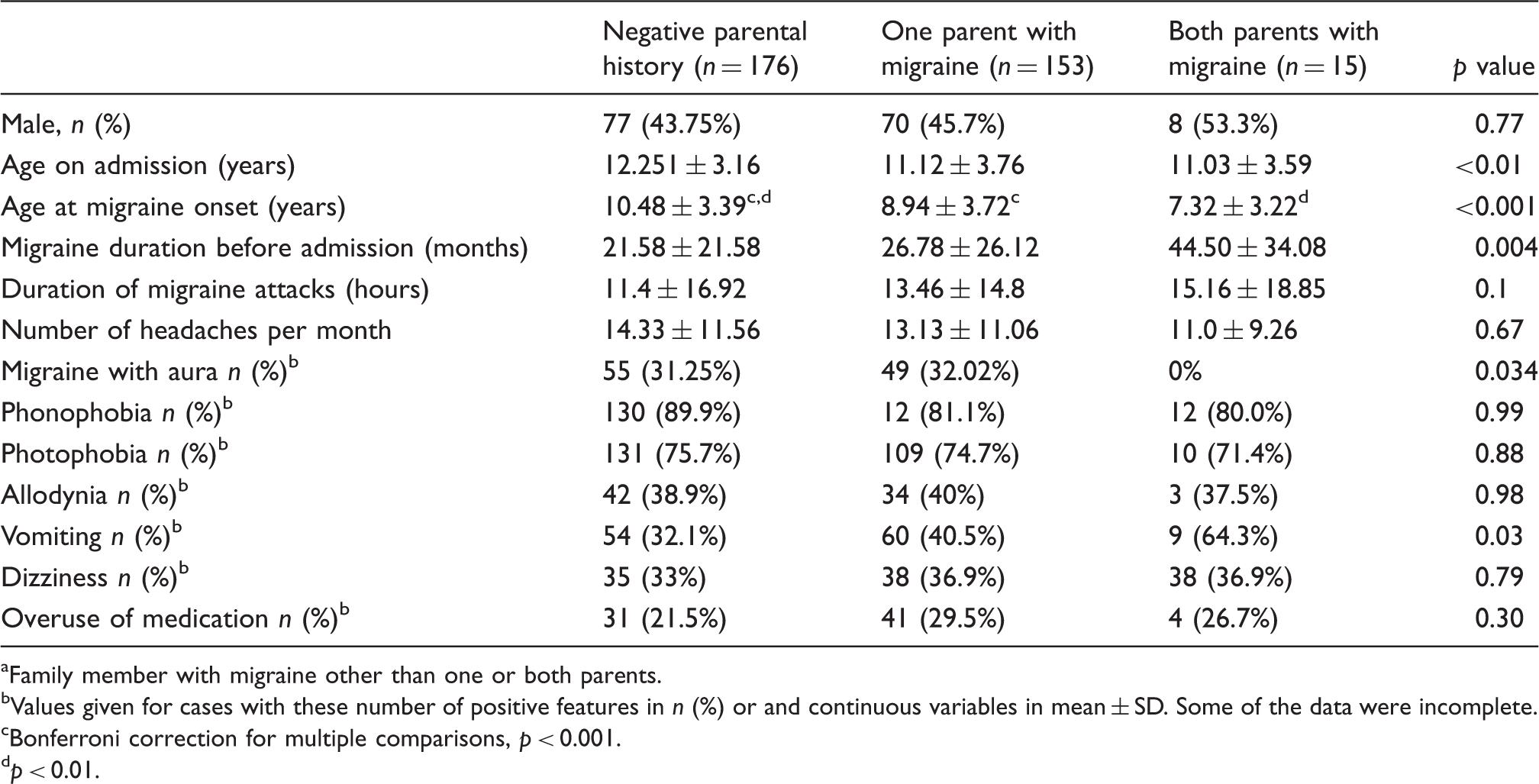
Mean age of the cohort was 11.69 ± 3.49 years, and mean frequency of headache per month, 13.68 ± 11.26. Mean age at migraine onset in patients with a negative parental history was10.48 ± 3.39 years; in patients with one parent with migraine, 8.84 ± 3.72 years; and in patients with both parents with migraine, 7.32 ± 3.22 years (p < 0.001).The duration of migraine attacks (in hours) was significantly longer in patients with any family member with migraine than in those with no family history (p = 0.026).
Conclusions
Among children attending a tertiary pediatric headache clinic, migraine appears at a younger age in those with parental history of migraine than in those with a negative family history. The findings suggest that having a genetic background of migraine makes a child more susceptible to migraine earlier in life than a child without a family history.
Introduction
Migraine is known to run in families and has long been considered a strongly heritable disorder. Migraine with aura and migraine without aura may have distinct familial patterns (1,2). Genetic epidemiologic studies reported that compared to the general population, first-degree relatives of probands with migraine without aura had a 1.9-fold increased risk of migraine without aura and a 1.4-fold increased risk of migraine with aura. Furthermore, first-degree relatives of probands with aura had nearly four times the risk of migraine with aura, with no increased risk for their spouses (1).
Stewart et al. (3), in a population study in the greater London area, found a familial aggregation component in migraine prevalence, severity, and onset. The sample included first-degree relatives of 532 adults (age 18–55 years) who experienced migraine attacks at least five times per year and a matched control group. The prevalence rate of migraine in the family members was 30.1% in the study group compared with 17.3% in the control group. There was a direct association of migraine severity with level of family migraine. Relatives of probands in whom migraine appeared before age 16 years (early onset) had a substantially higher risk of migraine (2.50; 95% confidence interval (CI): 1.63 to 3.79) than relatives of probands in whom migraine appeared later. However, the precise age at migraine presentation was not studied, and the study participants were all adults.
In addition, numerous studies have reported a high prevalence of positive family history of migraine in migraineurs. The risk of migraine was found to be two- to four-fold higher in individuals with a first-degree relative with migraine than in the general population, depending on migraine type (1,3–5).
The aim of the present study was to evaluate the relationship between age at onset and severity of migraine in children with a family history of the disease.
Methods
The database of the Pediatric Headache Clinic of a tertiary hospital was reviewed for all children diagnosed with migraine from 2010 to 2013. The diagnosis was based on the 2004 revised criteria for children of the International Headache Society (6). (Most of the patients were examined before publication of the International Classification of Headache Disorders III beta.) Excluded from the study were patients for whom data on migraine in family members were missing and siblings of the probands (to avoid bias). The study was approved by the hospital’s research ethics board.
Data for the study were collected from the medical files. At the first visit to our clinic, both older children and their parent(s) or only the parent(s) for younger children complete a standard International Headache Society-based questionnaire covering the frequency, duration, and nature of the headaches, duration of headache before presentation at the clinic, and related symptoms (vomiting, photophobia, phonophobia, etc.). For very young children whose verbal communication is limited, headache frequency is determined both by the child’s complaints and the parents’ impression from the child’s behavior. In addition, parents are questioned about the diagnosis of the child’s community physician, personal medical history of migraine, migraine in other family members, possible overuse of medications by the child, social problems in the family (low socioeconomic status, divorce, criminality, etc.), and emotional problems of the child (attention deficit disorder, anxiety, conversion, learning disorders, peer problems, somatization disorder, depression, criminality, depression, conduct disorder, eating disorder, obsessive compulsive disorder, etc.). Migraine in family members is validated by interviewing the patient’s father or mother or both, if possible. Basic questions cover, among other factors, the presence, severity, and duration of headache in the family member, other symptoms such as vomiting, photophobia, or phonophobia, and whether a definitive diagnosis of migraine headache was made by the physician of the affected family member.
Data collection is followed by a complete full physical and neurological examination of the patient. All diagnoses are categorized by the same single pediatrician.
Statistical analysis
Data for the study were managed and analyzed with BMPD software (7). Continuous variables are presented as mean ± SD. Analysis of variance (ANOVA) and two-way ANOVA were used to compare continuous variables between groups, with Bonferroni correction for multiple comparisons. The p value for statistical significance in the Bonferroni correction was adjusted by the number of groups (e.g. p < 0.0167 is required in comparisons of three groups, p < 0.0125 in comparisons of four groups). For parameters with a non-Gaussian distribution (length of attacks in hours, migraine frequency, migraine duration in months before admission), we used the nonparametric Kruskal-Wallis test with multiple comparisons. Discrete variables were compared using Pearson’s chi-square test or Fisher’s exact test, as appropriate. A p value of ≤0.05 was considered significant.
Applying power analysis, we calculated that with a sample size of 150 in each group (with/without a family history of migraine; total 300), the study would have 99.9% power to yield a statistically significant result. This computation assumes a mean difference of 2.0 years in age of onset (corresponding to means of 10.5 years vs 8.5 years) and a common within-group standard deviation of 3.5.
Results
Of the 511 children who attended the clinic during the study period, 167 were excluded from the study because they lacked data on family history (n = 162) or they were younger siblings of patients already included in the study (n = 5). The remaining 344 children formed the study group. There were 155 male patients (45%) and 189 female patients (55%) of mean age 11.69 ± 3.49 years (range 3–18 years).Migraine parameters were as follows: mean frequency of headaches per month, 13.68 ± 11.26 (median eight, range 0.03–30); mean duration of headaches in months before admission, 24.88 ± 25.16 (median 12, range 1–120); mean age at migraine onset in years, 9.66 ± 3.64 (range 1.9–17.92); mean duration of attacks in hours, 12.49 ± 15.97 (median six, range 1–72). There was no statistically significant difference in the duration of migraine attacks in hours by age group (up to 6 years, 6–12 years, 12–18 years; p = 0.18). Migraine without aura was documented in 240 patients (69.9%), and migraine with aura, in 104 (30.1%).
Number of relatives with migraine of 344 affected children.a

The total number of patients with family migraine adds up to more than 223 because some of the patients had more than one affected relative (such as parent and grandparent).
Demographic and clinical parameters in344 pediatric migraineurs with a negative or positive family history of migraine. a

Parent, grandparent, sibling, uncle/aunt, other.
Values given for cases with these number of positive features in n (%) and continuous variables in mean ± SD. Some of the data were incomplete.
Demographic and clinical parameters in 168 pediatric migraineurs with a history of migraine in the parents only. a
Family member with migraine other than one or both parents.
Values given for cases with these number of positive features in n (%) or and continuous variables in mean ± SD. Some of the data were incomplete.
Bonferroni correction for multiple comparisons, p < 0.001.
p < 0.01.
Regarding family members other than parents, we found that having a sibling with migraine (n = 35) was not significantly associated with any of the demographic or clinical parameters listed in Table 2. Patients who had a grandparent (maternal or paternal) with migraine (n = 68) were significantly younger at disease onset than patients who did not (8.5 years vs 9.4 years p = 0.0025), regardless of the migraine status of the other family members. Findings for the other parameters evaluated were not statistically significant. The small numbers in each group precluded the calculation of other associations of age at onset of migraine and history of migraine in specific maternal/paternal grandparents and other between-group comparisons.
Discussion
The present study was conducted in a cohort of children with migraine attending a dedicated headache clinic of a tertiary medical center. The relationship between age at onset of migraine and other background and clinical factors was evaluated by family history of migraine.
The search for the genetic basis of migraine has implicated specific genes only in familial hemiplegic migraine (FHM), a rare type of migraine with aura: CACNA1A located on chromosome19p13; FHM2, mapped to chromosome 1 q 21–23; and FHM3, linked to chromosome 2q24.
Past studies of the genetic basis of migraine have prompted genome-wide association studies that identified noncoding variants associated with migraine (8). The variants are concentrated in regulatory DNA marked by deoxyribonuclease I hypersensitive sites. A role has been suggested for the two-pore-domain potassium channel TRESK (TWIK-related spinal cord potassium channel). TRESK is involved in migraine by screening its encoded gene potassium channel, subfamily K member 18 (KCNK18). A frame sheet mutation has been reported that segregates perfectly with migraine without aura in a large pedigree. Prominent TRESK expression in migraine-salient brain areas such as the trigeminal ganglion has been reported as well (8). However, several twin studies found that genetic factors accounted for only up to 50% of the variation in migraine occurrence observed between dizygotic and monozygotic twins, with the remainder due to environmental factors (9,10), and complex segregation analyses also demonstrated a multifactorial hereditary model wherein multigenetic susceptibility factors interact with multiple environmental factors, rendering the individual susceptible to recurrent migraine attacks (4).
The present study suggests that having a genetic background of migraine makes a child more susceptible to the condition earlier in life than not having a genetic background. This finding is compatible with the results reported by Dzoljic et al. (11) who surveyed 245 migraineurs among 1943 female university-aged (18–28 years) individuals. Mean age at migraine onset was 14.5 ± 3.6 years in those with a positive family history of migraine compared to 16.9 ± 3.2 years in those with a negative history (p < 0.001). Reported headache frequency was one to two per month. However, this study is not directly comparable to ours because of differences in the characteristics of the population: older age, less severe migraine, lesser frequency of migraine, community-derived sample, and older mean age at migraine onset. Arruda et al. (12) found that the rate of headache frequency in children could be predicted by the presence of headache in the parents (p < 0.001) and was strongly associated with headache frequency in the mothers (p < 0.001). In the present study, we did not have access to data on the frequency of migraine in family members, although we showed that patients with any affected family member, including grandparents and aunts/uncles, had a longer duration of migraine attacks before presentation at the clinic than patients without a family history (Table 1, p = 0.026). No such difference was found on separate analysis of parental history specifically. It is possible that migraine is more severe in children with families that have harbored the disease for more than one generation.
The lack of association between age at onset of migraine and having a sibling with migraine might be explained by the small group size (n = 35) and lack of details on sibling age (that is, younger siblings may not yet have reached the age of migraine appearance (13)).
There are several potential biases in the study of migraine. One report suggested that children who have migraine in the family present earlier to the family physician than children who do not, and may be diagnosed earlier (13). However, in our study, children with a positive history actually presented significantly later (p = 0.004, Table 3). We speculate that in these cases, the parents may have been less anxious because they were familiar with migraine symptoms. The role of parental pain and the contribution to their children’s pain also has been reported. We did rule out some environmental factors that may have confounded results in other studies (13), such as overuse of medication, which was similar in patients with a positive or negative parental history of migraine (Table 3).
Our study has several limitations. The study group was restricted to patients attending a dedicated headache clinic in a tertiary medical center, who may have more severe disease than community-derived patients. Second, as mentioned, the subgroups of patients other than those with a parental history of migraine were quite small. Third, although the data on parental headache were derived by direct interview of at least one of the parents, data on headache in the grandparents, aunts, uncles, and other relatives were obtained second-hand. Ottman et al. (14) found that reports of adult patients regarding relatives with severe headache had a sensitivity of only 56% compared to interviews with the relative himself or herself. Russell et al. (15) interviewed 378 probands with and without migraine, 229 of their spouses, and 1109 of their first-degree relatives. The probands reported migraine in 288 family members whereas direct clinical interview yielded migraine in 472. The observed agreement rate was 78%, and the chance agreement (kappa) value was 0.47. Although the authors concluded that proband reports are unreliable for the assessment of migraine, recent accepted epidemiology literature define kappa values of 0.40 to 0.75 as representative of “intermediate to good” agreement (16). The kappa value in the present study falls within this range. The reliability of our findings is further supported by the similar results of the present study to the direct-interview study of Dzoljic et al. (11). Fourth, we did not have details of the severity of the parents’ headaches, their type of migraine or their complaints. Further studies addressing these issues are needed to corroborate our findings.
In conclusion, among patients being treated in a dedicated pediatric headache clinic associated with a tertiary medical center, migraine appears at a significantly younger age in those with a history of migraine in one or both parents than in those with a negative parental history. The findings suggest that having a genetic background of migraine makes a child more susceptible to migraine earlier in life than a child without a family history.
Clinical implications
Among children with migraine attending a headache clinic, those with a positive parental history of migraine may be more susceptible to acquiring the condition at a younger age than those with a negative parental history.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgments
The authors thank Pearl Lilos for the statistical analysis.
