Abstract
Background
During migraine, capsaicin-sensitive trigeminal sensory nerves release calcitonin gene-related peptide (CGRP), resulting in cranial vasodilatation and central nociception. Moreover, 5-HT is involved in the pathophysiology of migraine and depression. Interestingly, some limited lines of evidence suggest that fluoxetine may be effective in migraine prophylaxis, but the underlying mechanisms are uncertain. Hence, this study investigated the canine external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine before and after acute and chronic oral treatment with fluoxetine.
Methods
Forty-eight vagosympathectomised male mongrel dogs were prepared to measure blood pressure, heart rate and external carotid blood flow. The thyroid artery was cannulated for infusions of agonists. In 16 of these dogs, a spinal cannula was inserted (C1–C3) for infusions of 5-HT.
Results
The external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine remained unaffected after intracarotid or i.v. fluoxetine. In contrast, the vasodilator responses to capsaicin, but not those to α-CGRP or acetylcholine, were inhibited after chronic oral treatment with fluoxetine (300 µg/kg; for 90 days) or intrathecal 5-HT.
Conclusions
Chronic oral fluoxetine inhibited capsaicin-induced external carotid vasodilatation, and this inhibition could partly explain its potential prophylactic antimigraine action.
Introduction
Migraine is considered a neurovascular disorder characterised by an intense headache associated with several debilitating symptoms (1). Amongst other mechanisms, this disorder may involve: (i) activation of the trigeminovascular complex (2,3); (ii) high circulating plasma concentrations of calcitonin gene-related peptide (CGRP) (4,5); and (iii) vasodilatation of intracranial arteries and of the extracranial terminal branches of the external carotid artery, which are a significant source of pain in migraine (6,7), seemingly associated with CGRP release from capsaicin-sensitive trigeminal sensory nerves (8–10).
Migraine has also been described as a ‘low 5-hydroxytryptamine (5-HT, serotonin) syndrome’, suggesting that 5-HT plays a role in the pathophysiology of migraine (11,12). Interestingly, activation of central 5-HT1B receptors by triptans inhibits capsaicin-induced external carotid vasodilatation in vagosympathectomised dogs (13,14). Capsaicin is the pungent ingredient in chili peppers (15) and induces trigeminal release of CGRP (16,17) by acting on the transient receptor potential vanilloid type 1 (TRPV1) (18). Since intracarotid capsaicin increases jugular plasma levels of CGRP in pigs (16), it is reasonable to assume that capsaicin-induced external carotid vasodilatation in dogs could also be mediated by trigeminal release of CGRP, via TRPV1 activation (18), as previously discussed (13,14).
Furthermore, various types of drugs have been evaluated for migraine prophylaxis, including β-blockers, anticonvulsants and antidepressants (8). For example, fluoxetine (Prozac™), a selective 5-HT-reuptake inhibitor antidepressant (19), has been reported to be effective in migraine prophylaxis (20–23; sometimes inconclusively (20,21)), but the mechanisms involved are uncertain. In this context, the external carotid bed of vagosympathectomised dogs is a preclinical experimental model highly useful for the screening of compounds with potential antimigraine activity as well as for the identification of the mechanisms involved in their antimigraine action (24). On this basis, and considering the involvement of 5-HT and CGRP in migraine pathophysiology (9–12,24), the present study set out to investigate in anaesthetised vagosympathectomised dogs the external carotid vasodilator responses to capsaicin (presumably mediated by trigeminal release of CGRP), α-CGRP and acetylcholine before and after: (i) acute (intracarotid and intravenous (i.v.)) and chronic (oral) administration of fluoxetine; and (ii) acute intrathecal (i.t.) continuous infusions of 5-HT (the endogenous ligand) in an attempt to mimic the central effects of fluoxetine.
Materials and methods
Animals
A total of 48 male mongrel dogs (17–25 kg) were used in the present experiments. Only male dogs were used to avoid the confounding effects of hormonal fluctuations during the female oestrus cycle. The animals (housed individually on a 12:12 light/dark cycle with ad libitum access to food and water) were anaesthetised with an i.v. bolus injection of sodium pentobarbitone (30 mg/kg) and additional amounts (1 mg/kg, i.v.) were provided when required throughout the experiment, as previously reported (25). All experimental protocols of this investigation were approved by our institutional ethics committee (CICUAL-Cinvestav; permission protocol number 0353-06) and followed the regulations established by the Mexican Official Norm (NOM-062-ZOO-1999), in accordance with the United States National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals and the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines for reporting experiments in animals (26). All efforts were made to limit distress and to use only the number of animals necessary to produce reliable scientific data.
General methods
After induction of anaesthesia, all dogs were intubated with an endotracheal tube and artificially respired with room air, using a Palmer ventilation pump (rate: 20 strokes/min; stroke volume: 13–16 ml/kg) as previously established by Kleinman and Radford (27). Catheters were placed in: (i) the left femoral vein for the administration of vehicle or fluoxetine; and (ii) the left femoral artery, connected to a blood pressure transducer (P23XL, Grass Instrument Co., Quincy, MA, USA), for the measurement of arterial blood pressure. Moreover, in 16 of these dogs, a catheter was placed i.t. (28), through the subarachnoid space (spinal C1–C3 region; trigeminal nucleus caudalis) for the administration of 5-HT or its vehicle (saline). After administration of compounds, the i.v. and i.t. catheters were flushed with 3 ml and 0.3 ml of physiological saline, respectively. Mean arterial blood pressure (MAP) was calculated from the systolic (SAP) and diastolic (DAP) arterial pressures as follows: MAP = DAP + (SAP − DAP)/3. Heart rate was measured with a tachograph (7P4F, Grass Instrument Co., Quincy, MA, USA) triggered from the arterial blood pressure signal.
The right common carotid artery was dissected free and the corresponding internal carotid and occipital branches were ligated; under these experimental conditions, the blood flow through the right common carotid artery was considered to represent that of the external carotid artery (see Steel and Torrie (29) and Villalón et al. (30) for further considerations). Thereafter, an ultrasonic flow probe (4 mm R-Series) connected to an ultrasonic T206 flow meter (Transonic Systems Inc, Ithaca, NY, USA) was placed around the right common carotid artery. Bilateral cervical vagosympathectomy was systematically performed in order to prevent possible baroreceptor reflexes induced by the intracarotid continuous infusions of compounds. Subsequently, a catheter was introduced into the right cranial thyroid artery for the intracarotid continuous infusions of capsaicin, α-CGRP and acetylcholine. It is to be noted that the carotid arterioles are dilated under our experimental conditions (25,30). Since the main objective of our study is to investigate the effects of pretreatment with fluoxetine or 5-HT on the responses to vasodilator agents (such as capsaicin, α-CGRP and acetylcholine) on the external carotid circulation, we had to produce a selective carotid preconstriction with an intracarotid continuous infusion of phenylephrine (an α1-adrenoceptor agonist); this procedure allowed us to enhance the vasodilator responses to capsaicin, α-CGRP and acetylcholine, as previously reported (13,14,31). For this purpose, a 0.5 mm (external diameter) needle, connected to a suitable catheter, was inserted into the right common carotid artery for the continuous infusion of phenylephrine by another motor driven syringe. Then, the vasodilator responses to capsaicin, α-CGRP and acetylcholine during the intracarotid continuous infusion of phenylephrine were compared with those observed after intracarotid, i.v. or oral administration of fluoxetine, i.t. administration of 5-HT, or the corresponding equivalent volumes of vehicle. Thus, by using WPI model sp100i pumps (World Precision Instruments Inc, Sarasota, FL, USA): (i) capsaicin, α-CGRP, acetylcholine and fluoxetine as well as phenylephrine were infused into the carotid artery; and (ii) fluoxetine or equivalent volumes of physiological saline were infused into the femoral vein (for further details, see the experimental protocols described below). Arterial blood pressure, heart rate and external carotid blood flow were recorded simultaneously by a model 7D polygraph (Grass Instrument Co., Quincy, MA, USA). The body temperature of each dog was maintained between 37℃ and 38℃ by a lamp and monitored with a rectal thermometer.
Experimental protocols
After a stable haemodynamic condition for at least 60 minutes, baseline values of MAP, heart rate and external carotid blood flow were determined. Subsequently, all animals (n = 48) received an intracarotid continuous infusion of phenylephrine (1.5 µg/min) to obtain a sustained carotid vasoconstriction, as previously reported (13,14). Twenty minutes later, all the different treatments were given without interrupting the infusion of phenylephrine. Then, the animals were divided into four groups (n = 10, 9, 13 and 16).
The first group (n = 10) received consecutive intracarotid continuous infusions (1 ml/min, for one minute) of capsaicin (10, 18, 31 and 56 µg/min), α-CGRP (0.1, 0.3, 1 and 3 µg/min) and acetylcholine (0.01, 0.03, 0.1 and 0.3 µg/min). Then, this group was subdivided into two subgroups that received one-minute intracarotid continuous infusions of, respectively: (i) vehicle (physiological saline, 1 ml/min; six times given sequentially; n = 4); and (ii) fluoxetine (1, 3, 10, 30, 100 and 300 µg/min given sequentially in order to investigate whether fluoxetine can induce local effects on the external carotid arterial bed; n = 6). Ten minutes after having given the above doses of fluoxetine (total dose: 444 µg) or its equivalent volumes of saline, the responses to the above infusions of capsaicin, α-CGRP and acetylcholine were elicited again.
The second group (n = 9) received consecutive intracarotid continuous infusions (1 ml/min, for one minute) of the above doses of capsaicin, α-CGRP and acetylcholine. Then, this group was subdivided into two subgroups that received consecutive 10-minute i.v. infusions (0.5 ml/min) of, respectively: (i) vehicle (physiological saline; six times; n = 4); or (ii) fluoxetine (1, 3, 10, 30, 100 and 300 µg/kg given cumulatively to investigate if fluoxetine can produce systemic and carotid vascular effects; n = 5). Ten minutes after the highest dose of fluoxetine had been given (total dose: 300 µg/kg) or its equivalent volume of saline, the responses to the above infusions of capsaicin, α-CGRP and acetylcholine were elicited again.
The third group (n = 13) was subdivided into two subgroups that received a chronic oral daily treatment during 90 days of: (i) vehicle (meatball; n = 5); or (ii) fluoxetine (300 µg/kg; n = 8). Then, the animals were anaesthetised and prepared to receive consecutive intracarotid infusions of capsaicin, α-CGRP and acetylcholine as described above. It is noteworthy that this chronic oral daily schedule was based on the clinical observation that a significant reduction of migraine episodes was produced after 90 days of daily treatment with fluoxetine, as compared with placebo (20–22,32). For this reason, no pharmacodynamic/pharmacokinetic studies were conducted in our dogs to verify that a pharmacodynamically relevant plasma concentration of fluoxetine was achieved and maintained with the above dosing schedule.
The last group (n = 16) was subdivided into four subgroups that received intracarotid continuous infusions of the above doses of capsaicin, α-CGRP and acetylcholine before and 10 minutes after i.t. continuous infusions (during all the experiment) of, respectively: (i) vehicle (saline; 0.02 ml/min; n = 3); (ii) 5-HT (10 µg/min; n = 4); (iii) 5-HT (30 µg/min; n = 4); or (iv) 5-HT (100 µg/min; n = 5).
Other procedures applying to the experimental protocols
Each dose of capsaicin, α-CGRP and acetylcholine was in a solution administered at a rate of 1 ml/min during one minute. The dose-intervals between the different doses of these compounds (given sequentially as they produced transient responses) ranged between five (acetylcholine) and 20 (capsaicin and α-CGRP) minutes, as in each case we waited until the external carotid blood flow had returned completely to baseline values. The doses of these compounds were selected from results previously published (13,14,31), in which dose-dependent increases in external carotid blood flow were elicited with no changes in arterial blood pressure or heart rate.
Moreover, the acute administration of fluoxetine followed: (i) a sequential dosing schedule when given intracarotidly as no systemic or local carotid vascular responses were observed; and (ii) a cumulative dosing schedule when given i.v. as long-lasting external carotid vasoconstrictor responses were produced. In the case of i.v. administration of fluoxetine, each dose was given in 5 ml of solution during 10 minutes (at a rate of 0.5 ml/min).
In the last group (n = 16), the correctness of the i.t. administration was validated at the end of all experiments (after pentobarbitone overdose) by injecting 1% methylene blue (0.5 ml, i.t.) as previously reported (14). Then, the spinal cord was dissected by laminectomy (C1–C4 intervertebral disc) to look for the catheter tip and dye distribution. Dogs showing the catheter tip positioned at sites other than the C1–C3 spinal cord were not considered for data analysis.
Data analysis and statistical procedures
All data are presented as mean ± S.E.M. The peak changes in external carotid blood flow were expressed as percentage change from baseline. The difference between the variables within one group (or adifferent groups) of animals was compared by using a two-way repeated measures analysis of variance (or, correspondingly, aa two-way analysis of variance) (both randomised block design) followed by the Student-Newman-Keuls’ test (29). Statistical significance was accepted at p < 0.05.
Drugs
Values of mean arterial blood pressure (MAP), heart rate (HR) and external carotid blood flow (ECBF), before and after the different treatments.

p < 0.05 vs baseline value (before). 5-HT: 5-hydroxytryptamine; I.V.: intravenous; I.T.: intrathecal. The effects produced by fluoxetine given intracarotidly, intravenously or in chronic oral treatment on mean blood pressure and heart rate did not significantly differ from those produced by its corresponding vehicle.
Results
Systemic and carotid haemodynamic effects of the different treatments
Baseline values of MAP, heart rate and external carotid blood flow in the 48 anaesthetised dogs were 150 ± 9 mmHg, 174 ± 4 beats/min and 226 ± 12 ml/min, respectively. A significant (and sustained) decrease in external carotid blood flow (about 40%) was induced 20 minutes after the intracarotid continuous infusion of phenylephrine had been started, without significant changes in MAP or heart rate (see Table 1) (n = 48). Moreover, mean blood pressure and heart rate (Table 1) as well as external carotid blood flow (Figure 1) were not significantly modified in the animals that received: (i) intracarotid continuous infusions of physiological saline (1 ml/min; n = 3) or fluoxetine given sequentially (1, 3, 10, 30, 100 and 300 µg/min; n = 6); and (ii) 10-minute i.v. continuous infusions of physiological saline (0.5 ml/min; n = 3). Likewise, no significant effect was produced after i.t. infusions of saline (0.02 ml/min; n = 3), 10 µg/min 5-HT (n = 4), 30 µg/min 5-HT (n = 4) or 100 µg/min 5-HT (n = 5) (see Table 1). In contrast, Figure 1 shows that after i.v. administration of fluoxetine given cumulatively (1, 3, 10, 30, 100 and 300 µg/kg; n = 6) the external carotid blood flow was dose-dependently decreased (maximum effect: 27%), without significant changes in MAP or heart rate (see Table 1).
Effect of one-minute intracarotid infusions, given sequentially, of: (i) saline (1 ml/min, six times; n = 4) or fluoxetine (1, 3, 10, 30, 100 and 300 µg/min; n = 6); and (ii) an intravenous (i.v.) infusion during 10 minutes of saline (0.5 ml/min, six times; n = 4) or fluoxetine (1, 3, 10, 30, 100 and 300 µg/kg) (given cumulatively; n = 5) on the canine external carotid blood flow. Note that the X axis applies to the doses of fluoxetine given intracarotidly (µg/min) or i.v. (µg/kg). *, p < 0.05 vs. saline (i.v.).
Effect of intracarotid or i.v. continuous infusions of saline or fluoxetine on the canine external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine
One-minute intracarotid continuous infusions of capsaicin, α-CGRP and acetylcholine induced dose-dependent increases in the external carotid blood flow (see Figures 2 and 3; control responses) without significant changes in mean blood pressure or heart rate (not shown), as previously reported (13,14,31). These vasodilator responses to capsaicin and α-CGRP were longer lasting (between five and 20 minutes) than those to acetylcholine (between one and five minutes). The vasodilator responses to the three compounds remained unchanged after: (i) one-minute intracarotid continuous infusions, given sequentially, of either vehicle (1 ml/min of saline, given six times) (Figure 2, upper panel) or fluoxetine (1, 3, 10, 30, 100 and 300 µg/min) (Figure 2, lower panel); and (ii) 10-minute i.v. infusions of either vehicle (0.5 ml of saline) (Figure 3, upper panel) or fluoxetine (1, 3, 10, 30, 100 and 300 µg/kg; cumulatively) (Figure 3, lower panel).
Effect of one-minute intracarotid continuous infusions of capsaicin, α-calcitonin gene-related peptide (CGRP) and acetylcholine on the canine external carotid blood flow before (control responses) and after one-minute intracarotid continuous infusions, given sequentially, of: saline ((a), (b) and (c)) (1 ml/min; six times, n = 4); or fluoxetine ((d), (e) and (f)) (1, 3, 10, 30, 100 and 300 µg/min; total dose: 444 µg, n = 6). Effect of one-minute intracarotid continuous infusions of capsaicin, α-calcitonin gene-related peptide (CGRP) or acetylcholine on the canine external carotid blood flow before (control responses) and after 10-minute intravenous (i.v.) continuous infusions of: (i) saline ((a), (b) and (c)) (0.5 ml/min, six times; total volume: 30 ml, n = 4); or (ii) fluoxetine ((d), (e) and (f)) (1, 3, 10, 30, 100 and 300 µg/kg, given cumulatively; n = 5).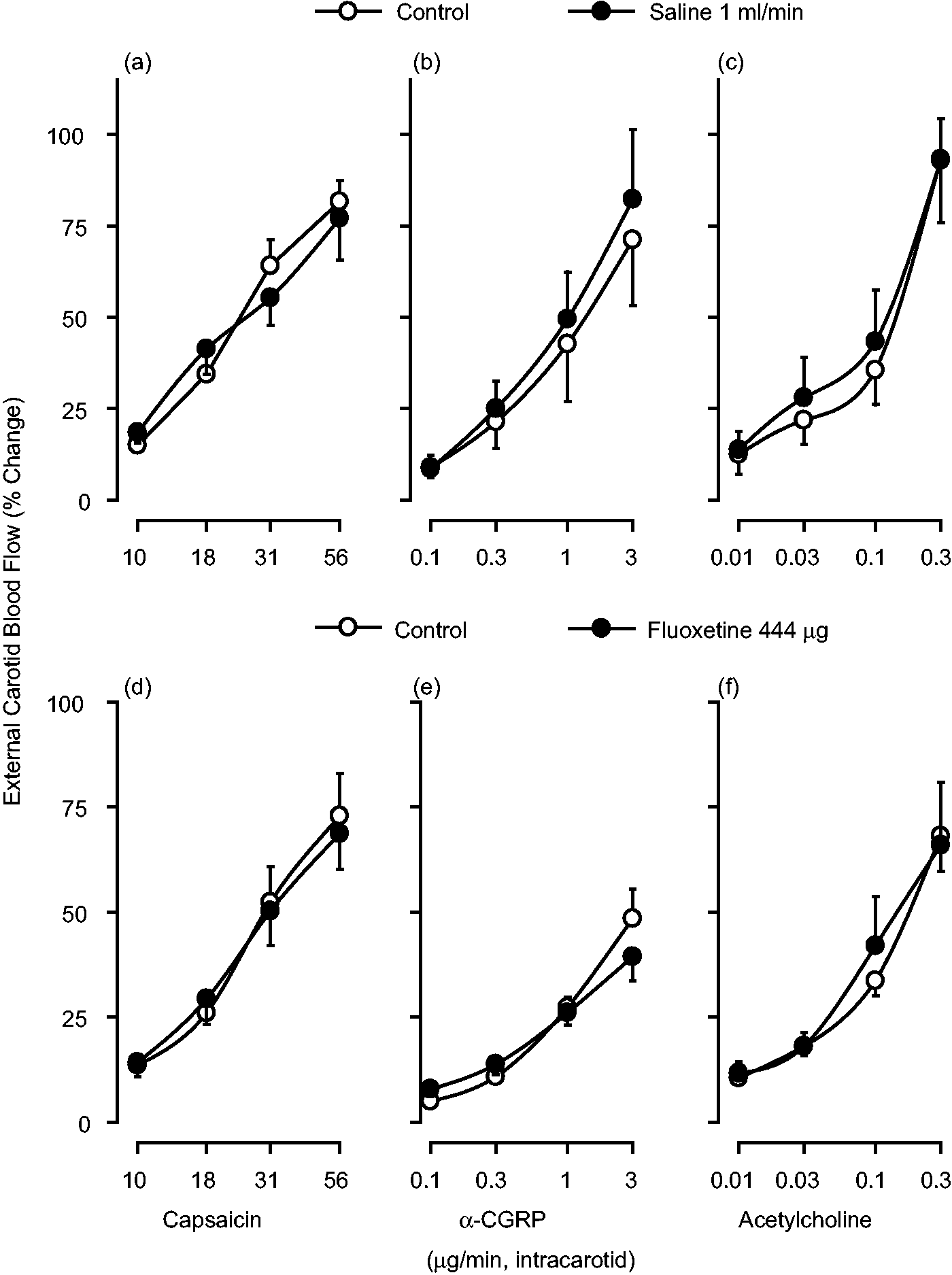
Effect of chronic oral treatment (daily; during 90 days) with vehicle or fluoxetine on the canine external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine
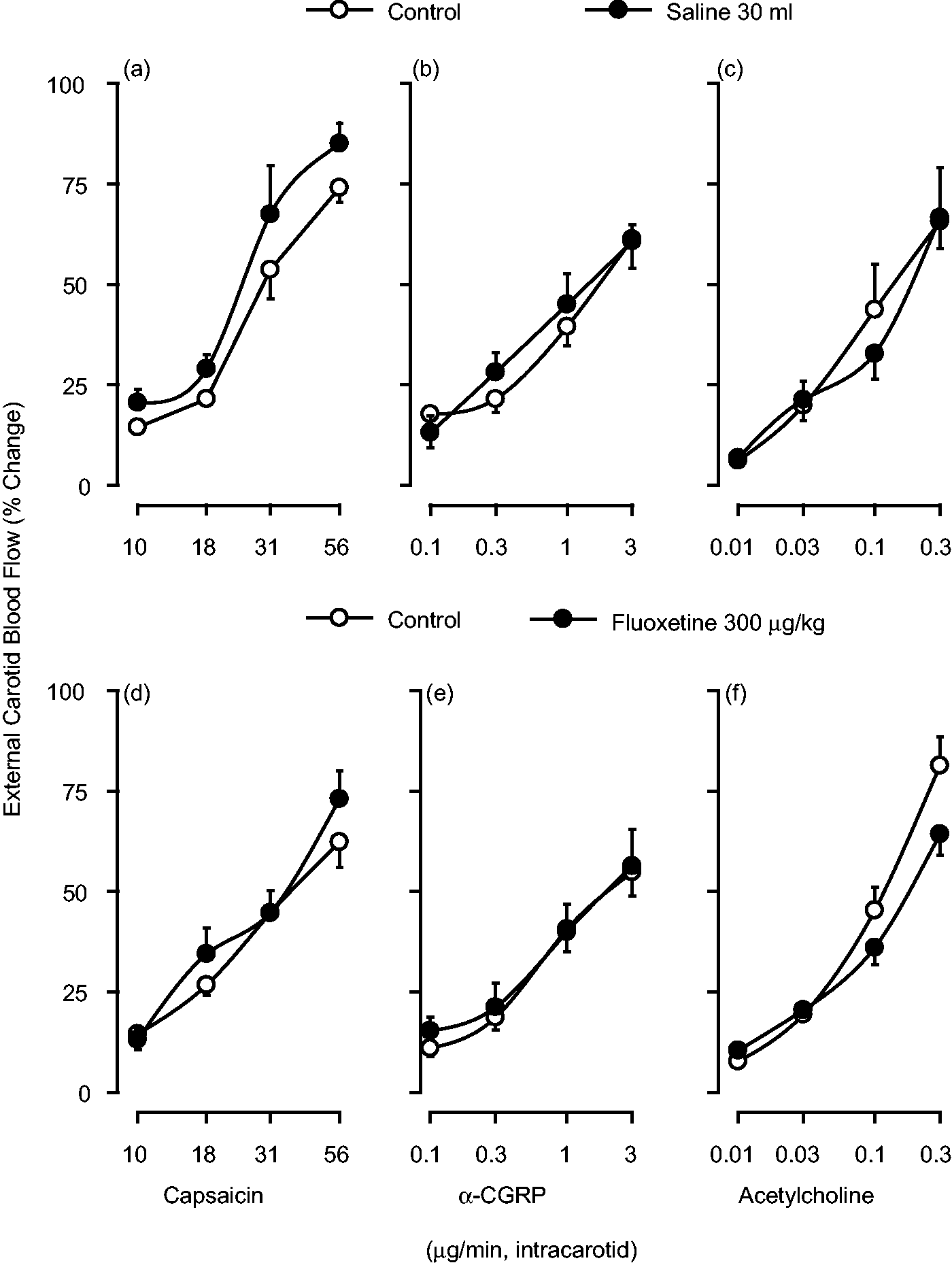
As compared to chronic oral treatment with vehicle (meatball), the vasodilator responses to capsaicin, particularly at 18, 31 and 56 µg/min, were significantly inhibited after chronic oral treatment with fluoxetine (300 µg/kg) (Figure 4(a)), whereas the responses to α-CGRP (Figure 4(b)) and acetylcholine (Figure 4(c)) remained unchanged.
Effect of one-minute intracarotid continuous infusions of capsaicin (a), α-calcitonin gene-related peptide (CGRP) (b) or acetylcholine (c) on the canine external carotid blood flow after chronic oral treatment (daily; during 90 days) with: (i) vehicle (meatball, control responses; n = 5); or (ii) fluoxetine (300 µg/kg, n = 8). *, p < 0.05 vs. the corresponding control response.
Effect of i.t. continuous infusions of physiological saline or 5-HT on the canine external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine
Since the inhibition induced by the chronic oral treatment with fluoxetine on capsaicin-induced external carotid vasodilatation may involve an increase in the central levels of 5-HT (although peripheral fluoxetine’s effects cannot be categorically excluded), we tried to mimic the central effects of fluoxetine by giving i.t. continuous infusions of 5-HT (10, 30 and 100 µg/min). Unlike i.t. administration of saline (Figures 5(a), (b) and (c)), i.t. administration of 5-HT (particularly at 30 and 100 µg/min) significantly inhibited the external carotid vasodilator responses to the highest doses of capsaicin (Figure 5(d)); but the responses to α-CGRP (Figure 5(e)) and acetylcholine (Figure 5(f)) remained unchanged.
Effect of one-minute intracarotid continuous infusions of capsaicin, α-calcitonin gene-related peptide (CGRP) and acetylcholine on the canine external carotid blood flow before (control responses) and after an intrathecal (i.t.) continuous infusion during all the experiments of: (i) saline ((a), (b) and (c)) (0.02 ml/min; n = 3); or (ii) 5-HT (10, 30 and 100 µg/min; n = 4, 4 and 5, respectively) ((d), (e) and (f)). The consecutive responses to capsaicin, α-CGRP and acetylcholine were started 10 minutes after the infusion of saline or 5-HT had commenced. *, p < 0.05 vs. the corresponding control response.
Discussion
General
Our findings show that capsaicin-induced external carotid vasodilatation was inhibited after chronic oral fluoxetine or i.t. 5-HT. This inhibition by fluoxetine may be mediated, at least in part, by central mechanisms; notwithstanding, the possible involvement of peripheral mechanisms (not explored in this study) cannot be categorically excluded. In contrast, the inhibition by i.t. 5-HT seems to involve exclusively central mechanisms (see below).
Systemic and carotid haemodynamic changes produced by the different treatments
Since intracarotid saline or fluoxetine, i.v. saline and i.t. saline or 5-HT failed to modify mean blood pressure, heart rate and external carotid blood flow (Table 1), no time-dependent changes occurred during our experimental protocols. Given that intracarotid phenylephrine decreased the external carotid blood flow (about 40%) without changing blood pressure or heart rate, a local vasoconstrictor effect mediated by α1-adrenoceptors is implied, as shown in dogs (33). A similar vasoconstrictor response (27%) was induced after the i.v. (but not the intracarotid) infusion of fluoxetine (Figure 1). This difference in fluoxetine’s carotid effects is probably due to the greater doses of fluoxetine given i.v. (total dose: 300 µg/kg) as compared to those given intracarotidly (total dose: 444 µg). Indeed, fluoxetine exerts complex acute effects on platelet function (34), including an increase in circulating plasma levels of 5-HT by inhibiting the platelet 5-HT reuptake transporter. This effect, in turn, would result in external carotid vasoconstriction to i.v. fluoxetine (Figure 1). If so, this effect most likely involves vascular 5-HT1B receptors, as previously shown for 5-HT and donitriptan (11,13,35).
Interestingly, the fact that chronic oral fluoxetine did not decrease the external carotid blood flow (Table 1) can be explained by other findings (34), in which an augmentation of platelet function (and higher 5-HT levels) after initiation of fluoxetine therapy may occur. However, this initial augmentation of platelet activation is likely offset by depletion of 5-HT from dense granules following chronic therapy (34).
Mechanisms involved in the vasodilator responses to capsaicin, α-CGRP and acetylcholine
Previous studies have suggested that the external carotid vasodilator responses to capsaicin, α-CGRP and acetylcholine in the dog may involve, respectively: (i) CGRP release, as shown in pigs (16); (ii) activation of CGRP receptors blocked by olcegepant, as shown in pigs (36); and (iii) muscarinic receptors on the carotid vascular endothelium in dogs (30).
Inhibition by chronic oral fluoxetine or i.t. 5-HT on the vasodilator responses to capsaicin
Some studies have reported that a chronic oral dosing with fluoxetine results in a prophylactic antimigraine effect (20–23,32; sometimes inconclusively (20,21)). Accordingly, our study shows for the first time that capsaicin-induced external carotid vasodilatation was inhibited after: (i) fluoxetine only when given chronically (Figure 4(a)), but not acutely (Figures 2 and 3); and (ii) i.t. 5-HT (Figure 5(d)). Hence, the inhibition by fluoxetine may probably involve increased central 5-HT levels although, as previously discussed, peripheral mechanisms can also play a role (34).
Admittedly, we did not measure central levels of 5-HT, which are reported to be increased after chronic fluoxetine (19), but we tried to mimic this central effect by a direct i.t. administration of 5-HT (spinal C1–C3 region; trigeminal nucleus caudalis). This inhibitory action by chronic fluoxetine or i.t. 5-HT may probably involve central stimulation of 5-HT1B receptors, as previously reported for the inhibitory action of donitriptan and sumatriptan on capsaicin-induced external carotid vasodilatation (13,14). Consistent with this speculation: (i) inhibition of neurotransmitter release is usually mediated by receptors coupled to Gi proteins, and in the case of 5-HT these are the 5-HT1 subtypes (37); and (ii) intracarotid capsaicin increases jugular plasma levels of CGRP in pigs (16). Hence, the inhibition by chronic oral fluoxetine and i.t. 5-HT in our experiments may involve inhibition of CGRP release although, admittedly, we did not measure jugular venous plasma levels of CGRP.
Moreover, the fact that chronic oral fluoxetine (Figure 4) or i.t. 5-HT (Figure 5) failed to modify the vasodilator responses to α-CGRP and acetylcholine suggests that the inhibition by fluoxetine and 5-HT does not involve interactions with vascular CGRP or muscarinic receptors. Although it could be argued that the inhibition by i.t. 5-HT was due to its leakage into the circulation, this can be ruled out as no changes in blood pressure, heart rate and external carotid blood flow were observed after i.t. 5-HT (Table 1). Thus, the inhibition by i.t. 5-HT is restricted to central interactions. In contrast, it is difficult to ascertain the extent to which central and peripheral mechanisms are involved in the inhibition by chronic oral fluoxetine, as this agent must firstly be absorbed to reach the systemic circulation before penetrating the blood-brain barrier.
Certainly, apart from 5-HT-reuptake inhibition, fluoxetine has other mechanisms of action including, amongst others: (i) blockade of Na+ channels (38); (ii) activation of K+ channels (39); (iii) an increase in gamma-aminobutyric acid (GABA)B receptor function (40); and (i.v.) a decrease in prostaglandin 2 (PGE2) production (41). Accordingly, the experiments of the present study do not allow us to discern the extent to which any of these additional mechanisms could be involved in fluoxetine-induced inhibition and, consequently, in its potential prophylactic antimigraine action.
Possible locus of the inhibitory action by fluoxetine and 5-HT
The inhibition of the capsaicin-induced external carotid vasodilatation by fluoxetine and 5-HT may involve the trigeminal nucleus caudalis and upper cervical cord because, amongst other findings: (i) spinal (C1–C3) administration of 5-HT inhibited the vasodilator responses to capsaicin (Figure 5(d)); (ii) spinal (C1–C3) administration of sumatriptan (a 5-HT1B/1D/1F receptor agonist) mimicked this response via 5-HT1B receptors (14); and (iii) sumatriptan activates a group of cells in the trigeminocervical complex (42). Thus, besides peripheral mechanisms, the central inhibition by chronic fluoxetine might be related to the trigeminocervical complex, whereas the inhibition by i.t. 5-HT is exclusively central.
Some limitations and ethical considerations of our study
Admittedly, four limitations of our investigation could be that:
A couple of studies report that there is little or controversial evidence that fluoxetine is effective in migraine prophylaxis (20,21). This is why fluoxetine is not suggested as a first-line treatment for migraine prophylaxis (European Federation of Neurological Societies (EFNS), American Academy of Neurology (AAN) practice guidelines). Nevertheless, the use of fluoxetine in migraine prophylaxis cannot be categorically excluded (23,43), and our study could, to some extent, explain its potential prophylactic action by shedding further light on the possible mechanism(s) involved. Our study is based on two seminal findings not specifically confirmed in this research, namely, that: (i) chronic fluoxetine increases central levels of 5-HT (44,45), although we tried somehow to mimic this effect by i.t. administration of 5-HT; and (ii) intracarotid infusions of capsaicin result in porcine carotid vasodilatation mediated by release of CGRP (16), a vasodilator neuropeptide released from capsaicin-sensitive trigeminal sensory nerves (10). The experiments involved in the above limitation comprise the use of dogs, the number of which is strictly controlled (and requires compelling ethical reasons) by our ethics committee (CICUAL). In any case, the objective of the present study is accomplished (see Introduction), and the results to be obtained from these experiments would only confirm previous findings. The right internal carotid artery was ligated in all dogs (see General methods section). This procedure: (i) may not have altered the total brain circulation; and (ii) could have possibly generated an intracranial ischaemic pain (not determined in our anaesthetised dogs).
Possible mechanisms by which spinal (C1–C3) administration of 5-HT or chronic oral fluoxetine result in a peripheral inhibition of capsaicin-induced carotid vasodilatation
Admittedly, additional studies that fall beyond the scope of this investigation will be required to ascertain the precise mechanisms by which an increase in central levels of 5-HT (after direct i.t. 5-HT) or after chronic oral fluoxetine result in a peripheral inhibition of capsaicin-induced carotid vasodilatation. Notwithstanding, amongst other possible mechanisms, these high 5-HT levels may have produced: (i) hyperpolarisation of the central ends of perivascular sensory nerves, which would prevent antidromic transmission with a resulting inhibition in the release of neuropeptides (presumably CGRP) at the neurovascular junction; and/or (ii) alterations in some reflex pathways passing through the central nervous system, as suggested for sumatriptan in the spinal control of peripheral inflammation (46). Consistent with these possibilities, activation of central 5-HT1B/1D receptors is associated with trigeminal/sensory inhibition, for example: (i) at spinal (C1–C2) levels, resulting in inhibition of capsaicin-induced external carotid vasodilatation (14,47); (ii) in the trigeminocervical complex, reducing the evoked nociceptive activity (48); and (iii) in the periaqueductal grey area, attenuating the evoked nociceptive neuronal firing in the trigeminocervical complex (49).
Furthermore, fluoxetine’s actions (particularly after chronic oral treatment) are much more complex and include additional pleiotropic effects that modulate neuronal excitability irrespective of the increase in central levels of 5-HT, which add up to its global effects, for example: (i) in depressed patients, fluoxetine racemate (0.27–1.5 mg/kg per day for eight weeks) increased the concentration of allopregnanolone (a positive allosteric modulator of GABAA receptors) in the cerebrospinal fluid (50); (ii) using the whole cell voltage clamp technique, fluoxetine (10–50 µM) blocked voltage-gated Ca2+ channels (51), ionotropic 5-HT3 receptors (52) and Na+ channels (53). In addition, norfluoxetine (the active metabolite of fluoxetine) potently inhibits the fast firing voltage gated potassium Kv3.1 channels (54). Within the context of our experimental model, these effects together could have favoured a (supra) spinal hyperpolarisation, making neurons more difficult to depolarise at this level when an antidromic potential from the periphery (evoked by intracarotid capsaicin) arrives.
Potential clinical implications
Since other prophylactic antimigraine agents have been reported to inhibit CGRP release from capsaicin-sensitive trigeminal sensory nerves (23,24), this mechanism could partly be involved in the potential prophylactic antimigraine action of fluoxetine. However, the pharmacological actions of fluoxetine are much more complex (see above) and, consequently, its potential prophylactic antimigraine action cannot be restricted to this mechanism only. In fact, other mechanisms could equally be involved. For example, 5-HT and sumatriptan inhibit GABAergic and glutamatergic synaptic transmission within the midbrain periaqueductal grey and this effect is mimicked by fluoxetine (55). Interestingly, our findings match well with those by Ayata et al. (56), who showed that prophylactic antimigraine drugs such as topiramate, valproate and amitriptyline inhibit cortical spreading depression (CSD) in rats when given chronically, but not acutely. Indeed, fluoxetine can similarly inhibit CSD in rats (57).
Conclusion
In conclusion
Article highlights
Using male mongrel dogs with bilateral vagosympathectomy, the external carotid vasodilator responses to capsaicin, α-calcitonin gene-related peptide (CGRP) and acetylcholine remained unaffected after intracarotid or intravenous administration of fluoxetine. In contrast, the external carotid vasodilator responses to capsaicin, but not those to α-CGRP or acetylcholine, were inhibited after chronic oral treatment with fluoxetine (300 microg/kg; for 90 days) or intrathecal administration of 5-hydroxytryptamine (5-HT). Therefore, chronic oral treatment with fluoxetine inhibited capsaicin-induced external carotid vasodilatation, and this inhibition could partly explain its potential prophylactic antimigraine action.
Footnotes
Funding
This work was supported by Consejo Nacional de Ciencia y Tecnología (CONACyT; projects 60789 and 219707; México D.F.).
Conflict of interest
None declared.
Acknowledgement
The authors would like to thank Mr Arturo Contreras for his skilful technical assistance.
a
Current address: Departamento de Biología Celular, Instituto Nacional de Perinatología, México
b
Current address: Instituto de Neurobiología, Universidad Nacional Autónoma de México, México
c
Current address: Unidad Académica Multidisciplinaria Reynosa Aztlán, Universidad Autónoma de Tamaulipas, México
