Abstract
Harold Wolff's theory of vasodilation in migraine is well-known. Less known is his search for a perivascular factor that would damage local tissues and increase pain sensitivity during migraine attacks. Serotonin was found to be among the candidate agents to be included. In the same period, serotonin was isolated (1948) and, because of its actions, an anti-serotonin drug was needed. Methysergide was synthesized from lysergic acid (LSD) by adding a methyl group and a butanolamid group. This resulted in a compound with selectivity and high potency as a serotonin (5-HT) inhibitor. Based on the possible involvement of serotonin in migraine attacks, it was introduced in 1959 by Sicuteri as a preventive drug for migraine. The clinical effect was often excellent, but 5 years later it was found to cause retroperitoneal fibrosis after chronic intake. Consequently, the use of the drug in migraine declined considerably, but it was still used as a 5-HT antagonist in experimental studies. In 1974 Saxena showed that methysergide had a selective vasoconstrictor effect in the carotid bed and in 1984 he found an atypical receptor. This finding provided an incentive for the development of sumatriptan. Bredberg et al. showed that methysergide is probably a prodrug for its active metabolite methylergometrine. Whereas methysergide is ‘a clean drug’, methylergometrine is ‘a relatively dirty drug’ with additional dopaminergic activity. The mechanism for the preventive effect of methysergide (methylergometrine) in migraine remains elusive. We describe the rise, fall and subsequent use as a third-choice drug of the first effective migraine prophylactic, methysergide.
Introduction
In previous papers, the history of drug treatment in migraine has been partly described, in particular the first use of ergot in acute treatment in 1868 (1, 2). Furthermore, the isolation in 1918 and subsequent research, spread, use and side-effects of ergotamine and dihydroergotamine have been studied (3). In the present paper we describe the origin and course of one of the first effective migraine and cluster headache prophylactics, based on research findings in migraine patients, i.e. methysergide. Prophylactic drugs had been used prior to the introduction of methysergide and included calcium salts, chrondroitin sulphuric acid, magnesium potassium thiocyanate, foreign protein injections, nicotinic acid, cannabis indica, sedatives, diuretics, pituitary and gonadal hormones, and prostigmine (4). Due to the similarities of migraine and epilepsy, anti-epileptic drugs have been used in migraine since the 19th century, e.g. bromides (1850s) and phenobarbital (1912). The latter drug was also applied in a combination drug (Bellargal: ergotamine, phenobarbitone, belladonna alkaloids).
Migraine and serotonin
An important factor in the history of methysergide was the isolation (5), identification and chemical synthesis (1948–1953) of serotonin, a serum (‘sero’) vasoconstrictor (‘tonin’) factor released from platelets during clotting of blood, hence its name (6). In 1953 serotonin was found in brain extracts (7). Because of its pressor effects, interest with respect to its relation to the study of hypertension arose among scientists. The finding of serotonin production by carcinoid tumours and the establishment of a central role of serotonin, due to the psychotomimetic effect of analogues and of lysergic acid (LSD), a serotonin antagonist on smooth muscle, caused further interest (6). In subsequent studies of carcinoid patients, however, no relation was found with hypertension, migraine or mental illness. Nevertheless, many drugs were tested in the era (1953–1970), including monoamine oxidase inhibitors, ergots (LSD and methysergide), methyldopa, carbidopa, tryptophan and tricyclic antidepressants. Peroutka and Snyder found multiple 5-HT receptors and made a classification (8), after which a new era of synthesis of new drugs started, including triptans and selective serotonin reuptake inhibitors.
Indications that 5-HT plays a role in migraine gradually accumulated in the 1950s and 1960s (9). Wolff was believed to have found evidence of tissue injury in the area of headache in patients. Lowered deep pain threshold around extracranial arteries and adjacent tissue was an invariable accompaniment of the headache. Although neither vasodilation nor lowering the pain threshold of itself could cause headache, interaction with a certain variability in the contractile state of cranial arteries was supposed to play a role in producing the headache. A phase of vasoconstriction was thought to be followed by dilation, intramural vascular oedema and lowered pain threshold (10). In 1957, Ostfeld, working in Wolff's group, studied the conjunctiva and skin of migraine patients and controls, applying several agents. In his experiments he included blister fluid, which has a pain threshold lowering effect, and serotonin that had recently been found to be ‘produced by nerve cells and that act on effectors’. Moreover, he applied local tissue fluid collected from subsurface tissues at the site of migraine headache. Chemical and microscopic analysis of sections of superficial temporal arteries removed from headache subjects and from non-headache subjects had shown that vasodilation was not the only cause of migraine, and that substances, possibly including serotonin, were present locally (11). Wolff et al. (12) injected serotonin perivascularly and were able to produce migraine-like symptoms. Furthermore, serotonin was suspected to play a role in allergic diseases, and migraine had been ascribed to allergic reactions (13). In a review of the physiology and biochemistry of migraine headache, Ostfeld concluded in 1960 that several candidates could be involved as the local substance in perivascular oedema, including acetylcholine, adenosine triphosphate, bradykinin, histamine and serotonin, the latter having ‘several attractive features’ as the more preferred candidate (14).
Subsequently, increased excretion of 5-HIAA in the urine during attacks was discovered in 1961 (15). Platelet serotonin was found to diminish (40%) at the onset of an attack (16). Anthony et al. (17) and Kimball et al. (18) found that attacks in migraine patients could be triggered by amine-releasing agents such as reserpine. In contrast to Wolff's previous findings, intravenous serotonin was found to alleviate migraine symptoms (17, 18).
Serotonin and methysergide. Successful prophylaxis in migraine and cluster headache
In the late 1950s, migraine as well as cluster headache were considered the two most important types of ‘vasodilating headaches’. With respect to prophylaxis, psychological and pharmacological methods were considered the most important treatments (4). In the late 1950s, LSD25, mentioned above, was found to be the most effective 5-HT antagonist (19), but it could not be used clinically because of its hallucinogenic effect (20). Several substances were tested, and it was found that the butolamide of lysergic acid had high potency as a 5-HT inhibitor. Furthermore, methylation at N1 in lysergic acid diminished the ergot-like action. The combination of these two characteristics was 1-methyl-D-lysergic acid-butanolamid (Fig. 1). Methysergide was found to possess more potency concerning 5-HT antagonism than LSD25 (20) and was usually without psychomimetic effects (20).

Chemical structure of L-methyl-lysergic acid butanolamide, methysergide.
Methysergide was then introduced in the clinic in 1959 as a highly specific 5-HT antagonist in the preventive treatment of migraine by the Italian neurologist Federigo Sicuteri (21), who continued on the path taken by Wolff and his co-workers. Like him, he stated that local release of substances, e.g. histamine, serotonin, bradykinin, etc., from mast cells could influence the vasa vasorum of the arteries and result in a decrease of the tonus in the arteries. Sicuteri published the results in an international journal on allergy, as he associated vasomotor activity with allergy following previous work on allergic factors playing a role in migraine. In this respect, we have to remember that in the 1950s cluster headache was often referred to as ‘histaminic cephalgia’ (21, 22). Sicuteri treated 18 migraine patients, 10 with particularly severe migraine, resistant to therapy, and two cluster headache patients. Eleven migraine patients were first treated with placebo. The migraine improved in all cases treated with methysergide, including nine patients who ‘have not had any real attack since the treatment began’ (15 patients were treated for 15–60 days) (21). This was also the case in cluster headache (21). Sicuteri concluded: ‘The remarkable prophylactic and therapeutic properties of this lysergic acid derivative, its powerful antiserotonin activity and absence of vasoconstrictor action, confirm the hypothesis that a disequilibrium of active tissue substance may play an important role in the pathogenesis of migraine’ (21). This finding led him to investigate the metabolism of serotonin and, as mentioned above, in 1961 he showed that excretion in the urine of 5-hydroxyindoleacetic acid, the metabolite of 5-HT, was increased during migraine attacks (15). In 1960, Heyck treated 38 migraine patients with serotonin antagonists (23). He reported that methysergide was effective in migraine, whereas the relatively weak serotonin inhibitor 2-bromine-LSD was not effective (23). In seven of eight cluster headache patients methysergide was effective (23).
Methysergide and trials
Subsequent trials were mainly done in the USA, where the drug was known by the name Sansert (4, 24–29) (see Table 1). In most studies, patients with severe, partly treatment-resistant migraine or cluster headache were included.
Early clinical studies of methysergide
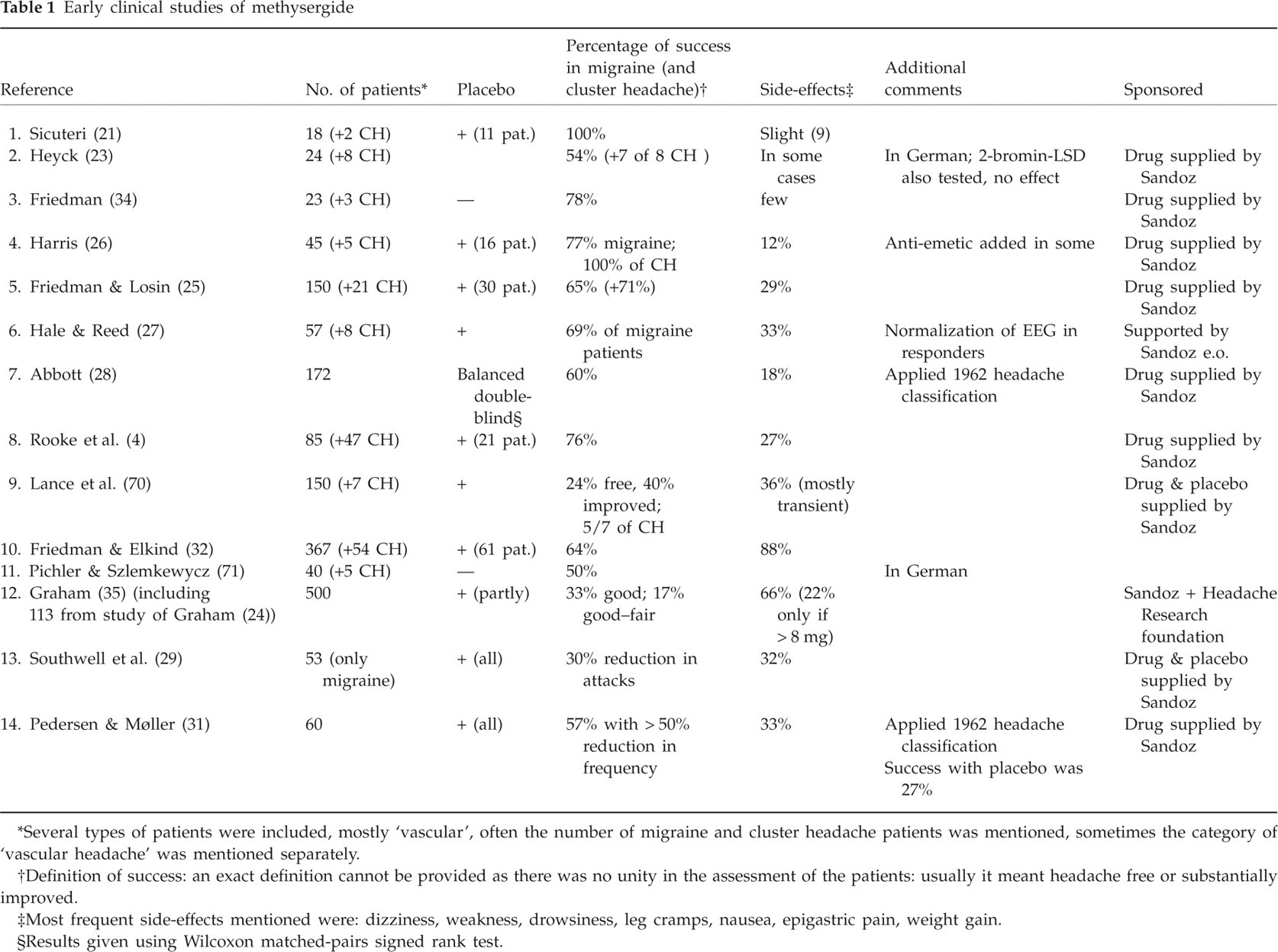
Several types of patients were included, mostly ‘vascular’, often the number of migraine and cluster headache patients was mentioned, sometimes the category of ‘vascular headache’ was mentioned separately.
Definition of success: an exact definition cannot be provided as there was no unity in the assessment of the patients: usually it meant headache free or substantially improved.
Most frequent side-effects mentioned were: dizziness, weakness, drowsiness, leg cramps, nausea, epigastric pain, weight gain.
Results given using Wilcoxon matched-pairs signed rank test.
In some studies, the authors referred to the new diagnostic criteria that were published in 1962 (30), including: (i) neuromuscular; (ii) vascular; (iii) cluster headache; (iv) true migraine (simple and cerebral type); (v) atypical facial pains; (vi) headache associated with known intracerebral aneurysms (unoperated and post surgical); and (vii) post-concussion headache (30). Possibly, the discovery of methysergide promoted the establishment of this classification. In a footnote to their study, Rooke et al. mention that ‘with the publication of the … classification … this suggestion [of nosological difficulties] comes a little late. However, the concept makes what we feel to be a useful distinction in classification …’ (4).
The first real European randomized clinical trial (RCT) was published by Southwell et al. in the Lancet in 1964. In modern terms, the latter study was a relatively well-conducted, double-blind, placebo-controlled trial, with carefully written results, in terms ‘though not so definite, do suggested that methysergide may reduce the number and severity of attacks'(29). In 1966, in another European RCT, methysergide was clearly superior to placebo for frequency of attacks (31).
Placebos were given in some of the studies (see Table 1) or at least in some of the patients, as ‘it was not practable to test the majority of our patients with placebo tablets’ (32). The number of side-effects was variable. The most frequent mentioned in most studies were dizziness, weakness, drowsiness, leg cramps, nausea, epigastric pain and weight gain. Fibrosis was not yet mentioned.
The first serious disappointments: retroperitoneal fibrosis
Following years of successful use of the drug, in Europe (23, 33) and the USA (34–36), in particular in cluster headache, but also in migraine patients, the first reports of more serious side-effects were published. The first paper appeared in 1965 (37). Although idiopathic retroperitoneal fibrosis had been known since the beginning of the 20th century, the condition was observed in three patients taking methysergide. Two of them were among the 500 patients of whom Graham described his experiences with the drug in 1964 (35), but at that time the relation had not yet been discovered. Utz et al. did not establish a direct causal relationship, but found that all other previously described associations were not present in the three (37). They concluded that the finding did not ‘constitute sufficient evidence to interdict its use’. They supposed it would be sensible to ‘discontinue medication for at least 3 months’ for any patient who has used it for a year. Moreover, urinalysis, renal function tests and excretory urography were advised. If the drug was continued beyond a year, the physician should be alert for evidence of fibrosis. Repetition of the tests every 3–6 months was also advised.
One year later, Graham published a larger series of patients with retroperitoneal fibrosis (38). The paper was based on 27 patients. In 14 the diagnosis was established by surgeon and pathologist. In the other 13, who were treated by discontinuance of the drug and from whom biopsy material was not obtained, it was less certain, but laboratory data and X-rays, consistent with retroperitoneal fibrosis, were available. Duration of treatment varied between 9 and 54 months, daily dosage between 2 and 28 mg. The incidence of this complication was estimated at about 1% (10 patients in a series of 1000 patients seen by Graham's group, quite different from the incidence that was mentioned later, i.e. one in 5000 (36)). Following discontinuance of the drug, partial or complete regression of the process was observed in all patients. Because of awareness of other fibrotic conditions in idiopathic patients, including cardiac and pleuropulmonary disease, some methysergide patients were also examined for this complication, but only a few doubtful cases were reported (38). The causal relationship between methysergide and retroperitoneal fibrosis was quite strong by the numbers presented and the regression after discontinuance. In a few patients, who had been operated on and who continued the drug, retroperitoneal fibrosis persisted. A year later, in 1967, Graham described cardiac and pulmonary fibrosis (39).
In the search for an explanation for the fibrotic disorders after methysergide, the pharmacokinetics of the drug was investigated with 14C-methysergide (40). The rate of elimination was the same in patients with retroperitoneal fibrosis and controls. There was therefore no indication that changes in metabolism resulted in the fibrotic disorder (40).
After the problems with fibrosis became known, the clinical use of methysergide declined considerably and its use thereafter has been reserved to a small minority of severely afflicted migraine patients (41).
Further studies on the action of methysergide; prelude to the discovery of sumatriptan
In 1978 it was stated in the Handbook of Experimental Pharmacology (volume 49) that small amide derivatives of lysergide acid, e.g. methysergide, ergometrine and methylergometrine, are among the most potent and selective 5-HT antagonists (42). Demethylation of methysergide during first passage in the liver results in the main active metabolite methylergometrine (42–44), which had a 10 times greater area under the curve than methysergide (Fig. 2) (44). Furthermore, it appeared that methylergometrine, as a 5-HT antagonist in the human temporal artery, is 40-fold more potent than methysergide (45). An interesting finding here is that there is a discrepancy between animal, where methysergide is the most potent (isolated rat uterus; see Table 2), and human studies (45).

Plasma concentrations of methysergide and methylergometrine after administration of oral methysergide. Note that the AUC is 10 times higher for methylergometrine than the AUC for methysergide. From Bredberg et al. (44).
Activity profiles for methysergide, methylergometrine and some ergot compounds

The relative activities on seven biological activities are listed. Modified from Berde & Schild (42).
Dopaminergic effects in the central nervous system.
The oral bioavailability of methysergide was 13% (44), so when given orally methysergide serves as a prodrug for methylergometrine. Given orally, methysergide was found to cause a decrease in prolactin in man, probably due to the dopaminergic effect of its metabolite methylergometrine (42). Chronic oral but not acute treatment with methysergide was reported to attenuate dural plasma extravasation following electrical stimulation of the trigeminal ganglion in the rat (46). The discrepancy between acute and chronic administration is probably due to of the presence of the metabolite methylergometrine during chronic administration (41, 44).
Whereas previous investigators had not found a peripheral vasoconstrictor effect of methysergide (47), it was found to cause selective carotid vasoconstriction in the carotid vascular bed (48). The possible relevance to the antimigraine action of methysergide was suggested (48). In 1976, based on the venoconstrictor test, Fanciullaci et al. suggested that methysergide acts as a partial agonist in migraine (49). In 1981 it was found that methysergide had an effect on the femoral circulation and on isolated saphenous vein of dogs (50, 51). In 1984, Saxena, investigating the effect of methysergide and 5-HT, found evidence for an ‘atypical’ 5-HT receptor in the carotid bed of pigs, (52) results that provided an incentive for the development of sumatriptan (41, 53).
It was around this period that Humphrey and Feniuk visited Saxena's laboratory in Rotterdam to learn the technique of measuring shunt flow with radioactive microspheres (52). Moreover, they tested a prototype analogue of sumatriptan, which acted on this ‘atypical 5-HT’ receptor. This receptor was later identified as a 5-HT1B receptor (53, 54). We now know that methysergide is a potent 5-HT2 and 5-HT7 blocker and a partial agonist at the 5-HT1B receptor (41). The preventive efficacy of methysergide has been ascribed to its 5-HT2 receptor antagonism, but this is unlikely, since the potent 5-HT2 antagonists mianserin, ketanserin and ICI 169 369, are ineffective in migraine (41, 55).
Recently acquired knowledge from studies with methysergide
It has been suggested that the effect of methysergide and its metabolite methylergometrine on the 5-HT1B receptor could be responsible for the antimigraine action (41), but how this could be the reason for the preventive effect remains unclear. Thus, chronic oral administration of methysergide in rats resulted in suppression of cortical spreading depression (CSD) and increased threshold for CSD (56), clearly a central nervous system (CNS) effect of the drug. This suppression of CSD could be relevant in migraine with aura, where spreading oligaemia of the regional cerebral blood flow (rCBF) has been observed (57). In migraine without aura, rCBF is unchanged in most studies (57), but also in this disorder a preventive drug must act on the CNS considering the premonitory symptoms, which occur before the headache and which are present in 87% of migraine patients (58).
As shown in Table 2, methysergide is a ‘rather clean drug’, whereas methylergometrine is a ‘somewhat dirty drug’ with central dopaminergic effects: emetic activity in the dog, effects on dopaminergic stereotyped behaviour in rats, and contralateral turning behaviour in 6-hydroxydopamine lesioned rats (42). In addition, methylergometrine is more potent than methysergide as a vasoconstrictor on bovine middle cerebral artery (59) and human coronary arteries (60), an effect on 5-HTB receptors. Furthermore, it appeared that when given intravenously acutely, methysergide caused a decrease in carotid arteriovenous blood flow in pigs, whereas subacute oral administration had no effect (52). Subacute oral administration would result in metabolism of methysergide to methylergometrine. This seemingly indicates that methylergometrine has no effect on arteriovenous anastomoses, but this could be a matter of doses. This vasoconstrictor effect, however, is unlikely to be responsible for methysergide's preventive effect in migraine. Thus, sumatriptan 100 mg tid was without any pre-emptive effect in a randomized controlled trial in cluster headache (61), and in clinical practice overuse of sumatriptan with daily or near-daily use of the drug causes medication-induced headache (62). Thus, constant 5-HT1B/1D stimulation is probably not a useful principle in migraine prophylaxis.
Could the dopaminergic effect of methylergometrine be responsible for the effect in migraine prophylaxis? Bergerot et al. found that dopamine can inhibit trigeminovascular transmission in rat (63), suggesting that dopamine agonism could be a new treatment modality in migraine, but in our opinion this is unlikely, since there are no reports of alleviating migraine with L-3,4-dihydroxyphenylalanine. In addition, migraineurs have been found to be hypersensitive to dopamine stimulation (64, 65). Methysergide has been used as a potent 5-HT antagonist for many years and is still used nowadays in both in vitro studies, e.g. (66), and animal in vivo studies, e.g. (67). It still has an important use as an experimental drug and as a potent 5-HT antagonist. It is well known that methysergide is a potent 5-HT2 receptor antagonist, but it does not distinguish between the 5-HT2A, 5-HT2B and 5-HT2C subtypes (36, 68). Thus, methysergide antagonizes the contractile effects of 5-HT on vascular and non-vascular smooth muscles with a pA2 (negative log of a constant for potency of an antagonist) of > 8 (55). Indeed, in the human isolated temporal artery, which contains predominantly 5-HT2 receptors (69), both methysergide and its active metabolite methylergometrine are potent antagonists; the latter compound is some 40 times more active than the parent drug (45).
Methysergide, not a synthesis in vain
Finally, one may ask: was methysergide a synthesis in vain? We have explained that in man, methysergide is most likely to act as a prodrug for methylergometrine (44). Methylergometrine is a potent 5-HT antagonist (see Table 2 and (45)) and in human temporal arteries it is 40-fold more potent than methysergide (45). Thus, methysergide ‘delivers’ a potent 5-HT antagonist in man. However, as mentioned above, methylergometrine is not such a ‘clean’ drug as methysergide, and it has, for example, a dopaminergic effect, as shown in Table 2 and by its prolactin-lowering effect in man (42). In contrast, methysergide has been used in experimental pharmacological studies as a potent and specific 5-HT2 antagonist. Another effect of methysergide, its effect on the ‘atypical’ 5-HT receptor in the carotid vascular bed (52), provided incentive for the development of sumatriptan (53).
Methysergide is still considered an effective migraine preventive medication (41) that, as shown in open studies (see above), is even more effective in resistant cases with a high attack frequency. All of the open and controlled studies that we have discussed attest to its efficacy in migraine and in cluster headache. Because of its side-effect profile, however, it should be reserved for severe cases in which other migraine preventive drugs are not effective (36). It should not be used continuously, but with a treatment-free interval of 1 or 2 months every half year in order to minimize the chronic side-effects of fibrosis. Obviously, this is not a problem for episodic cluster headache patients. The long-term side-effects of methysergide have thus limited its use considerably.
In conclusion, methysergide has served as a prodrug in clinical practice and has been used extensively as a potent, specific 5-HT antagonist in pharmacological studies. So overall, we believe, the synthesis of methysergide was not in vain.
