Abstract
In this randomized, double-blind, placebo-controlled, parallel-group study, patients received a single 50-mg oral dose of a 5-HT1D agonist, PNU-142633 (n = 34), or matching placebo (n = 35) during an acute migraine attack. No statistically significant treatment effects were observed at 1 and 2 h after dosing, even after stratifying by baseline headache intensity. At 1 and 2 h post-dose, 8.8% and 29.4% of the PNU-142633 group, respectively, and 8.6% and 40.0% of the placebo group, respectively, experienced headache relief; 2.9% and 8.8% of the PNU-142633 group and 0% and 5.7% of the placebo group were free of headache pain. Adverse events associated with PNU-142633 treatment included chest pain (two patients) and QTc prolongation (three patients). Results from this study suggest that anti-migraine efficacy is not mediated solely through the 5-HT1D receptor subtype, although this receptor may contribute, at least in part, to the adverse cardiovascular effects observed with 5-HT agonist medications.
Introduction
Migraine is a chronic neurological disorder characterized by repeated episodes of severe headache, often accompanied by nausea, photophobia and phonophobia. Migraine headaches are caused by dilation and inflammation of cranial blood vessels, resulting in stimulation of the trigeminal sensory system (1–3). Sumatriptan and other triptan compounds, such as zolmitriptan and rizatriptan, supposedly exert effects at both 5-HT1B and 5-HT1D receptors, reducing the migraine headache by constricting the dilated blood vessels and inhibiting the release of peptides from the trigeminal sensory fibres causing the inflammation, respectively (4–7). With regard to the cardiovascular adverse events associated with triptan therapy (8), they may be mediated through the 5-HT1B receptor, which is expressed in the human coronary artery, although at significantly lower levels than in the human cerebral artery (9, 10). Therefore, agents that are specific for the 5-HT1D receptor, and lack activity at the 5-HT1B receptor, may offer a safer strategy for the treatment of migraine.
PNU-142633 is a selective 5-HT1D receptor agonist, identified as such by radioligand binding assays with a binding affinity constant of [Ki] = 6 n
The intrinsic activity of PNU-142633F at the human 5-HT1D receptor was determined to be 70% that of 5-HT in the cytosensor cell-based assay. (Data on file. Pharmacia & Upjohn. Receptor binding methods: Radioligands and sources for receptors used to characterize the binding profile of PNU-142633 are summarized in Table 1. Rat cortical membranes were the source for adrenergic and cholinergic receptors. Rat striatal and bovine cerebellar membranes were the source for dopamine-D1 and histamine-H1 receptors, respectively. Other dopamine and serotonin receptors were harvested from CHO, HEK 293 or Hela cells following rinsing the cells with ice-cold CA++/Mg++ free phosphate buffered saline. Cells were pelleted (500
Proportion of patients with headache relief (combined and stratified by baseline headache intensity)

Fisher's exact test.
Cochran-Mantel-Haenszel test. Small number of responses.
In comparison with sumatriptan, PNU-142633 has similar efficacy but greater potency in the neurogenic inflammation model in guinea pigs, with an effective oral dose range of 0.1–1 mg/kg. PNU-142633, at intravenous doses of 0.1–1.0 mg/kg, and sumatriptan, at the intravenous dose of 0.3 mg/kg, were similarly efficacious in blocking stimulation-induced increases in blood flow in a trigeminal blood flow model in cats (Data on file. Pharmacia & Upjohn). This finding that the selective 5-HT1D receptor agonist, PNU-142633, is as efficacious as the non-selective 5-HT1D/5-HT1B agonist, sumatriptan, in this model suggested that the 5-HT1D receptor plays a role in the anti-migraine activity of sumatriptan and PNU-142633.
PNU-142633 was previously administered to 53 healthy volunteers in a phase I, single-dose, double-blind, dose-escalation study (11). Thirty-nine subjects received PNU-142633 in oral doses ranging from 1 to 100 mg; 14 received matching placebo. At this dose range, PNU-142633 was well tolerated; the most common adverse events were dizziness and headache. No serious adverse events were reported during the trial. Sporadic abnormalities in vital signs, ECG and clinical laboratories were observed, although no pattern in these abnormalities was apparent.
The objectives of the current study were to determine the efficacy, safety and tolerability of a single oral 50 mg dose of PNU-142633 vs. placebo in the treatment of acute migraine with or without aura.
Methods
Patients
Male and female migraineurs, aged 18–60 years, were eligible for participation in the study. Patients were required to have: a diagnosis of migraine with or without aura as defined by International Headache Society criteria; a history of migraine of at least 3 months; a typical occurrence of two to seven migraine attacks per month; the ability to distinguish between migraine attacks and other types of headaches; and a history of migraine responding to conventional methods of pharmacotherapy (i.e. non-steroidal anti-inflammatory drugs, ergotamine or sumatriptan). All patients provided written consent prior to their participation in the study. Female patients of childbearing potential were required to use an adequate method of birth control.
Patients were excluded if they had used: migraine prophylactic medication (anticonvulsants, beta-blockers, calcium channel blockers, antidepressants, or tranquilizers) within 2 weeks of treatment with study medication; sumatriptan, ergot derivatives or opiates within 24 h of treatment with study medication; or any other analgesic or antiemetic within 6 h of treatment with study medication. Patients were also excluded if they had any of the following: basilar, opthalmoplegic or hemiplegic migraine; a preponderance of mild migraine attacks; hypersensitivity or consistent non-response to triptan-like drugs; a prolonged QTc interval (> 450 ms for males or > 470 ms for females); a clinically significant history of medical or psychiatric illness; or any clinically significant abnormalities on physical examinations or laboratory tests.
Study design
In this double-blind, placebo-controlled, parallel-group trial, patients at six study centres were randomized to receive a single oral 50-mg dose of PNU-142633 or placebo during a moderate or severe migraine attack. The protocol was reviewed by a local Institutional Review Board at each centre, and was conducted under the World Medical Association Declaration of Helsinki.
Patients meeting entry criteria were instructed to call the centre upon onset of a migraine headache. If study staff assessed the headache to be moderate or severe in intensity, then the patient was instructed to report to the centre within a reasonable time-frame as judged by the investigator. Pre-dose evaluations included an abbreviated physical examination, 12-lead ECG (including assessment of QTc interval) and vital signs. If the patient continued to meet eligibility criteria, additional baseline measures including a pharmacokinetic blood sample, laboratory tests, headache assessments and adverse events, were conducted. Patients were then randomized to receive PNU-142633 50 mg or placebo. Following dosing with the blinded study medication, headache intensity was evaluated over a 12-h period. Rescue medication (e.g. non-steroidal anti-inflammatories or opiates) and anti-emetics were permitted if an adequate reduction in headache intensity was not achieved within 2 h of dosing. Efficacy assessments included the proportion of patients experiencing relief, as defined by a decrease in headache intensity from moderate or severe (score of 2 or 3) to mild or none (score of 0 or 1), and those who became pain free, both at 1 and 2 h after dosing.
Electrocardiographic measurements (including QTc interval) were assessed 2, 4, 8 and 12 h after dosing. If a prolonged QTc interval (an increase of > 30 ms compared with baseline; or > 450 ms for males and > 470 ms for females) was observed at the 12-h time-point, the patient was required to stay at the centre for an additional 12 h of cardiovascular monitoring. Laboratory tests were again conducted at 12 h post-dose. Vital signs and adverse events were monitored throughout the study. Symptoms related to the migraine attack were classified as ‘associated symptoms’ and were not reported as adverse events. Within 3–7 days after the treatment, patients returned to the centre for post-treatment safety assessments.
Serial blood samples for pharmacokinetic analysis were collected at pre-dose, and 0.5, 1, 2, 4, 8 and 12 h after dosing. PNU-142633 plasma concentrations were measured by a specific high performance liquid chromatographic method utilizing tandem mass spectrophotometric detection.
Statistical analysis
The primary efficacy variable was the proportion of patients experiencing headache relief at 2 h after dosing, which was defined as a decrease in headache intensity from moderate or severe to mild or none. The study was designed with a power of 90% (beta) to detect a difference as statistically significant that would occur with a change of 10% or less (alpha). The power calculation was based on the assumption of a 2-h headache response in the placebo group of 30% and in the PNU-142633 group of 65%. The study was conducted accordingly with 35 patients in each group. The proportion of patients who experienced headache relief at 1 h was also analysed, as well as the proportion of patients who were headache free (a reduction in pain from moderate or severe to none) at both time-points. The proportion of patients experiencing headache relief or who were headache free at 1 and 2 h in each treatment group was compared using Fisher's exact test. A P-value ≤ 0.10 was considered evidence of a significant difference between 50 mg PNU-142633 and placebo for these efficacy variables. The Cochran-Mantel-Haenszel test was used to compare headache relief and headache disappearance between groups, while controlling for headache intensity (moderate or severe) prior to treatment.
For each patient, the maximal plasma concentration (Cmax) of PNU-142633 and the time to reach maximal concentration (Tmax) were determined by inspection of the plasma concentration-time profile.
Results
Sixty-nine patients were randomized to receive PNU-142633 (n = 34) or placebo (n = 35). The two treatment groups were similar in age, gender and ethnic origin. The majority of patients in the PNU-142633 and placebo groups were female (62% and 83%, respectively) and Caucasian (78% and 88%, respectively). The mean age was 35.6 years in the PNU-142633 group (range 19–52 years) and 40.5 years in the placebo group (range 21–59 years).
Efficacy
The headache relief results are presented in Table 1. The proportion of patients experiencing relief at 2 h post-dose was not statistically significantly different (P = 0.45) in the PNU-142633 treatment group (29.4%) in comparison with the placebo group (40.0%). In addition, no significant differences (P = 1.00) in relief were observed between the two groups at the 1-h time-point. After stratifying by headache intensity at baseline, relief at 1 and 2 h after dosing was also not significantly different in the PNU-142633-treated and placebo-treated patients.
Table 2 presents the headache-free results. The proportion of patients who were headache pain free at 2 h post-dose was not statistically significantly different (P = 0.67) in patients who received PNU-142633 (8.8%) and in those who received placebo (5.7%). No significant differences (P = 0.49) for this measure were observed at the 1 h time-point, or at the 1 and 2 h time-points after stratification by headache intensity at baseline.
Proportion of patients who were headache free (combined and stratified by baseline headache intensity)

Fisher's exact test.
Cochran-Mantel-Haenszel test. Small number of responses.
Safety
The number of patients experiencing at least one adverse event was similar in the PNU-142633 treatment group (13 of 34; 38%) and the placebo group (12 of 35; 34%). The most common adverse event was dizziness, which was reported in three of 34 patients (8.8%) receiving PNU-142633 and two of 35 patients (5.7%) receiving placebo. Cardiovascular-related adverse events experienced by the PNU-142633 group and the placebo group are presented in Table 3. Chest pain was observed in two of 34 patients (5.9%) in the PNU-142633 group and in none of the patients in the placebo group. Mild QTc prolongation was observed in three of 34 patients (8.8%) in the PNU-142633 group and one of 35 patients (2.9%) in the placebo group. First degree AV block was observed in two of 34 patients (5.9%) in the PNU-142633 group and in none of the patients in the placebo group, while second degree AV block was observed in one of 34 patients (2.9%) in the PNU-142633 group and in none of the patients in the placebo group.
Cardiovascular-related adverse events
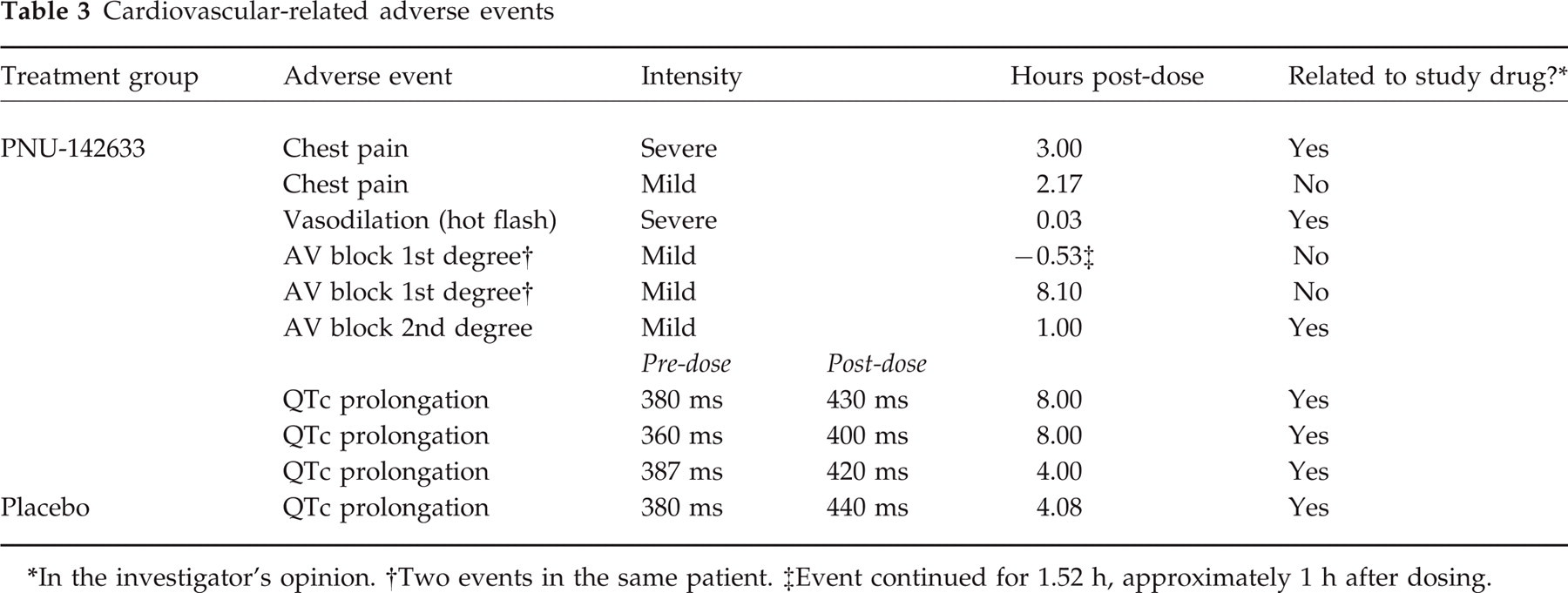
In the investigator's opinion.
Two events in the same patient.
Event continued for 1.52 h, approximately 1 h after dosing.
Pharmacokinetics
Plasma PNU-142633 concentration data were obtained from 32 individuals. The mean Cmax was 141 ± 69.9 ng/ml and the mean Tmax was 2.33 ± 2.17 h.
Discussion
In this double-blind, randomized, placebo-controlled study, the selective 5-HT1D agonist, PNU-142633, did not alleviate migraine headache in the population studied. The results suggest that activation of the 5-HT1D receptor alone may not be adequate for anti-migraine efficacy. Studies investigating the expression of 5-HT1 receptors have found that the 1B receptor is dominant in human cerebrovascular tissue while the 1D receptor is preferentially expressed in neuronal tissue (6, 9). Bonaventure et al. (12) found a high expression of 5-HT1D receptor mRNA and low expression of 5-HT1B receptor mRNA in guinea pig trigeminal ganglia. Collectively, these findings suggest that anti-migraine compounds such as sumatriptan reduce neurogenic inflammation through the stimulation of 5-HT1D receptor, while causing vasoconstriction through the stimulation of 5-HT1B receptors in the cerebral arteries. It is possible that both of these effects are necessary to induce headache relief, consistent with the efficacy of the triptans, which are agonists at both receptor subtypes.
Surprisingly, PNU-142633 was associated with adverse events including chest pain, mild AV block and mild QTc prolongation, indicating that activity at the 5-HT1D receptor contributes to some of the cardiovascular adverse events associated with triptan therapy (see below). This was unexpected, due to the predominance of the 5-HT1B receptor in the coronary arteries, as well as the implications of the involvement of the 5-HT1B receptor in the development of cardiovascular adverse events with sumatriptan therapy, particularly in patients with coronary disease (8, 9, 13). In contrast to these findings, however, the occurrence of cardiovascular adverse events with PNU-142633 suggests that the 5-HT1D receptor is also involved in the development of these events. These results suggest that selective 5-HT1D agonists may not be a safer, nor a more effective, strategy for anti-migraine compounds.
The mechanism of 5-HT1B/1D agonist-induced chest pain is unknown and may, in actual fact, not be cardiovascular, as ECG changes have not been found in patients reporting these symptoms (14, 15). ECG monitoring of 5388 patients receiving sumatriptan revealed a 5% incidence of chest pain/pressure and only a 1% incidence of ECG changes (16). Rare cardiac adverse events, including cardiac ischemia, coronary vasospasm, unstable angina pectoris, cardiac dysrhythmias, ventricular fibrillation and myocardial infarction, have also been reported in conjunction with sumatriptan use (17–22). In a study of alniditan, a 5-HT1B/1D receptor agonist unrelated to the triptan class, chest pain/pressure was reported in eight of 171 (5%) patients treated with one of four doses; however, no ECG abnormalities were found (23). Thus, while the incidence of chest pain/pressure was similar in patients treated with PNU-142633 compared with the triptans, the incidence of ECG abnormalities was higher. It is important to note, however, that the ECG abnormalities seen in PNU-142633 patients were mild in nature and have been observed in the normal population.
Pharmacokinetic results previously reported for healthy volunteers receiving an oral dose of PNU-142633 50 mg indicated a Cmax of 184 ± 74.9 ng/ml and a Tmax of 1.60 ± 1.39 h. Thus, the exposure of migraine patients to PNU-142633 in this study was similar to that observed in healthy volunteers. The data were also suggestive of somewhat delayed absorption during a migraine attack, as seen with other anti-migraine agents such as aspirin (24) and naproxen (25), but not with sumatriptan (26) or rizatriptan (27). Mean peak plasma concentrations in the present study (141 ng/ml or 334 n
There are several caveats to the conclusions of this study. The first is that the small sample size may have precluded the detection of differences between the placebo and PNU-142633 groups. This was a pilot study, powered at 90% to detect a difference based on an alpha level of 0.1, assuming a placebo response of 30% and a PNU-142633 response rate of 65%. However, the placebo response rate was 40% at 2 h post-dose, which is higher than expected. Placebo response rates in clinical trials of migraine patients have ranged from 15% to 26% at 2 h (28–31); however, in two recent studies, placebo responses as high as 37% (32) and 54% (33) were reported. The 2-h placebo response of 40% is, therefore, not out of the ordinary, especially considering the fact that the study was conducted in the clinic where the patients were allowed (and probably very often encouraged) to rest and relax.
In conclusion, PNU-142633 was not effective in reducing the pain of acute migraine in this study. This finding suggests that the efficacy of anti-migraine compounds is mediated through a 5-HT receptor other than, or in addition to, the 1D subtype. Additionally, cardiovascular adverse events, such as chest pain and mild QTc prolongation, were observed with PNU-142633, indicating that the 5-HT1D receptor or other unidentified serotonin receptors in cardiovascular tissue may contribute to the cardiovascular effects of current triptan therapies.
Footnotes
Acknowledgements
The Pharmacia & Upjohn Company sponsored this study. The authors wish to thank Fredrick S. Whaley PhD for statistical support, Richard Gibson MS for data management support, and Ann M Boldt MS for manuscript preparation.
