Abstract
Background and aims
Migraine headache is one of the most common primary headache disorders and is three times more prevalent in women than in men, especially during the reproductive ages. The neurobiological basis of the female dominance has been partly established. The present study aimed to investigate the effect of gender on the headache manifestations in migraine patients.
Methods
The study group consisted of 2082 adult patients from five different hospitals’ tertiary care-based headache clinics. The relationship between headache characteristics and gender was evaluated in migraine with aura (MwA) and migraine without aura (MwoA). The duration, severity, frequency of headache and associated symptoms were evaluated in both genders and age-dependent variations and analyzed in two subgroups.
Results
Women with migraine were prone to significantly longer duration and intensity of headache attacks. Nausea, phonophobia and photophobia were more prevalent in women. Median headache duration was also longer in women than in men in MwA (p = 0.013) and MwoA (p < 0.001). Median headache intensity was higher in women than in men in MwA (p = 0.010) and MwoA (p = 0.009). The frequency of nausea was significantly higher in women than in men in MwA (p = 0.049). Throbbing headache quality and associated features (nausea, photophobia, and phonophobia) were significantly more frequent in women than in men in MwoA. The gender impact varied across age groups and significant changes were seen in female migraineurs after age 30. No age-dependent variation was observed in male migraineurs.
Conclusion
Gender has an influence on the characteristics of the headache as well as on the associated symptoms in migraine patients, and this impact varies across the age groups, particularly in women.
Keywords
Introduction
Migraine is an important public health issue in all age groups as it causes disability and economic loss. Epidemiological studies have demonstrated a significant female preponderance of migraine worldwide with a ratio of 52% versus 37% (1–3). A recent population-based study in Turkey, which represents a population of both Asian and European origin, indicated that the prevalence of migraine headache is significantly higher in women (24%) than in men (12%) (4,5).
Migraine headache is three times more prevalent in women than in men, especially during the reproductive ages (6). The high prevalence of migraine in females is related to fluctuating levels of ovarian steroids to a great extent. Migraine frequency is high between puberty and menopause (7), and attacks are triggered by menstruation when the serum levels of estradiol and progesterone rapidly decline (8–10). Female sex steroids such as estrogen and progesterone also influence peripheral and central pain transmission via serotonergic, noradrenergic, glutamatergic, GABAergic and opioidergic neurotransmitter systems (11,12). However, persistence of the gender difference regarding the prevalence of migraine-type headache after menopause suggests that some other factors could also be involved. Pain susceptibility is also higher in females, even when healthy, independently of cycling hormone levels (13,14) and suggests an intrinsic gender difference. In this context, functional differences have been demonstrated in pain processing between the sexes even in healthy individuals (15–19). The sex difference affects functional brain circuits, with emotional circuitry apparently involved to a greater extent compared to sensory processing in migraine patients (20).
Studies are either focused on epidemiology or the description of pain characteristics in the literature. A few investigators have noted a gender influence on the duration, severity or frequency of migraine headache. Kelman demonstrated a gender effect on the duration of a migraine attack, while primarily studying the pain characteristics of acute migraine attacks (21). Some other studies have also noted a gender influence on the duration and intensity of headache (22–24). Nonetheless, the headache features and associated symptoms have not been studied in relation to gender as the main interest. It is still not clear whether the diagnostic criteria for migraine differ for females and males.
In this study, we aimed to test the hypothesis that gender has an impact on the clinical symptomatology of migraine. This broad-spectrum, case-controlled study conducted at our tertiary headache center consisting of multiple centers aimed to investigate whether the clinical diagnostic features of migraine such as duration, severity, frequency and the associated symptoms of nausea/vomiting, photophobia and phonophobia were different in females and males. To the best of our knowledge, this is the first study conducted with a detailed analysis of clinical variables according to gender with several age groups.
Methods
Data collection
Patients diagnosed by a neurologist after a face-to-face interview and proper neurological examination and investigations were entered in headache database. After obtaining written informed consent from center representatives, the data of 9300 patients entered into the Headache Database from 1999 to 2009 were screened for study inclusion. The study protocol was approved by the coordination center (MEU.0.01.00.06/265, 20.10.2008). Consent was also obtained from patients to use their data for academic purposes. Patients’ personal data were blinded except for gender and age and reviewed by two headache specialists. Patients with the following features were excluded from the study: i) diagnosis other than migraine (according to the International Classification of Headache Disorders, first edition (ICHD-I) (25) and second edition (ICHD-II) (26)); ii) missing or non-evaluable data for gender, age, headache characteristics and associated symptoms; iii) pregnant patient or using hormones; and iv) younger than 18 years.
Data evaluation
The data of 2082 patients pooled from five different university hospitals were included for the study and the database were analyzed for two migraine subtypes: The migraine with aura (MwA) group included patients with visual and sensory auras and the migraine without aura (MwoA) group included probable migraine without aura patients. The gender effect was further analyzed in both sexes using four different age groups: 18–29, 30–39, 40–49, and 50+ years.
Pain intensity was assessed using the visual analog scale (VAS). Descriptive analyses were performed as follows: The frequencies of MwA and MwoA by age and gender were determined. The pain properties for each type of pain (duration, intensity, frequency, laterality) and associated features of the patients were analyzed.
Statistics
The Shapiro-Wilk Test was used to examine the frequency distribution of headache duration, intensity and frequency data to determine whether they fit into the normal distribution in the gender, age and headache groups. For the descriptive statistics, means and standard deviations were calculated in normally distributed groups such as for age, whereas medians and quartiles were used in groups that did not have a normal distribution. Nominal and percentage values are provided for categorical variables. Kruskal-Wallis analysis was used to determine whether there was a difference between the age groups in terms of headache duration, intensity and frequency. The Dunn test was used to compare the group means in pairs. The z-test was used for proportional comparison of headache features in both genders. Chi-square analysis was applied to test the relationship between the age groups and headache characteristics. The Bonferroni correction (p/k) (k = 4) was applied for the prevention of inflation for type one error for the subgroup numbers that emerged during the sex-age interaction test. The error one ratio was accepted as 0.0125 for the sex-age interaction comparisons between the groups as a result. P values were reported without adjustment for multiplicity. The statistical evaluation of the data was performed using SPSS 11.5 and MedCalc@v11.0.1 software.
Results
The study covered a total of 2082 adult patients who were diagnosed with migraine. There was a nonsignificant difference between age according to gender in the two groups. A total of 312 MwA patients were included and the mean age was 46.43 ± 13.04 years for the 276 women and 40.08 ± 14.00 years for the 36 men. The MwoA group consisted of 1770 patients, of whom 1528 were female (mean age ± SD 45.34 ± 12.70 years) and 242 were male (mean age ± SD 42.74 ± 14.46 years).
MwA
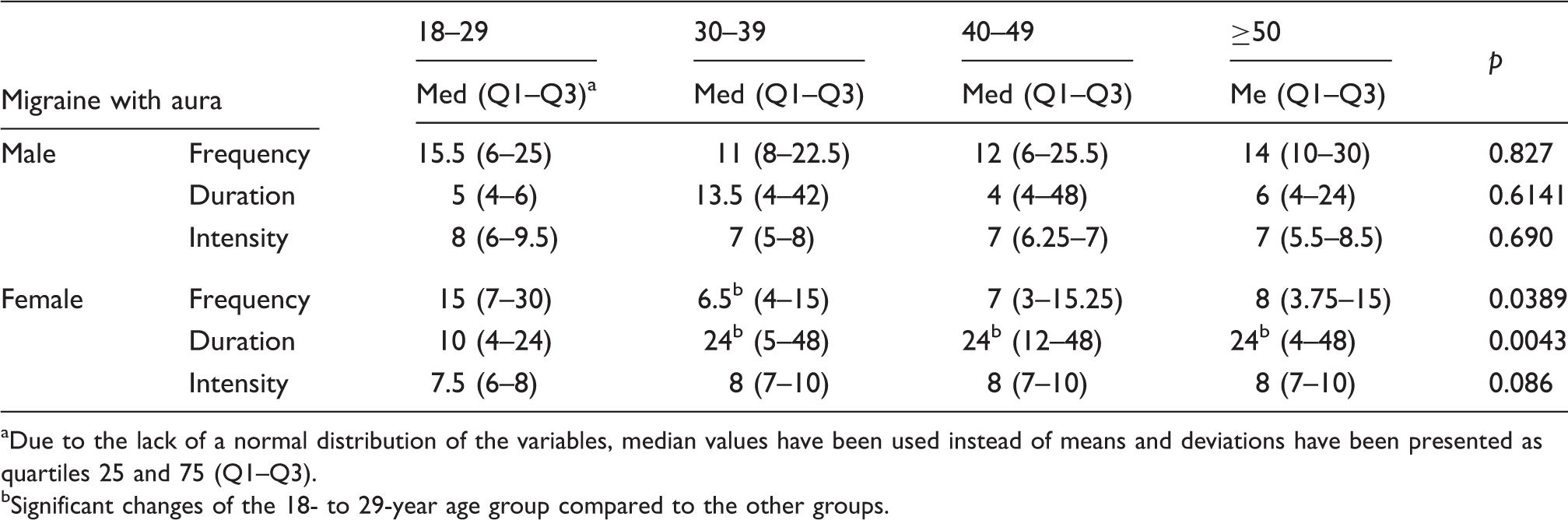
The median headache duration was longer in females, particularly after the age of 30 (p = 0.0043) (Table 1). The 18- to 29-years age group females had longer headache duration compared to the 30- to 39-years (p = 0.036), 40- to 49-years (p = 0.0025) and 50+ years (p = 0.0407) age groups (Figure 1(a)).
(a) The women in the 18- to 29-years age group had longer headache duration when compared with the 30- to 39- (p = 0.036), 40- to 49- (p = 0.0025) and 50+ years age groups (p = 0.0407). (b) There was no significant difference between the groups in terms of headache intensity. (c) Women in the 18- to 29-years age group reported higher headache frequency than the 30- to 39-years age group (p = 0.0308). The characteristics of headache attacks in migraine patients. Due to the lack of a normal distribution of the variables, median values have been used instead of means and deviations have been presented as quartiles 25 and 75 (Q1–Q3). Significant changes of the 18- to 29-year age group compared to the other groups.
Gender-dependent changes in clinical features.

MwA: migraine with aura; MwoA: migraine without aura.
MwoA
Median headache duration was longer and increased after the age of 30 years in women compared to younger ages. On the other hand, headache duration was significantly different between women in the 18- to 29-years age group and women aged 50+ years (p = 0.0407) (Table 2 and Figure 2(a)). Mean VAS and headache frequency were significantly different between women in the 18- to 29-years and 40- to 49-years age groups (p = 0.0355, p = 0.0000) (Table 2 and Figure 2(b)). There was a significant difference between women in the 18- to 29-years and 40- to 49-age groups (p = 0.0000) and the 50+ years age group (p = 0.0013) in headache frequency in the last month (Table 2 and Figure 2(c)).
(a) Two-group comparisons showed that women in the 18- to 29-years age group and the 30- to 39-years age group (p = 0.0062), 40-to 49-years age group (p = 0.0002) and 50+ years age group (p = 0.0000) had significant differences in headache duration. (b) There was a significant difference for headache intensity between women in the 18- to 29-years and 40- to 49-years age groups (p = 0.0355). (c) Two-group comparisons showed that there was a significant difference for headache frequency in the last month between women in the 18- to 29-years age group and the 40- to 49-years age group (p = 0.0000) and 50+ years age group (p = 0.0013).
The existence of gender impact and its variance with age in migraine patients.

Due to the lack of a normal distribution of the variables, median values have been used instead of means and deviations have been presented as quartiles 25 and 75 (Q1–Q3). bSignificant changes of the 18- to 29-year age group compared to the other groups.
Nausea was markedly more common in women (65.6%) then in men (48.6%) with MwA. In the same manner, phonophobia was more frequent in women (70.2%) then in men (51. 4%) in MwA. Photophobia was more prevalent after the age of 30 years in women with MwA.
Discussion
The predominance of the female gender and sex hormone influence is well established in migraine, yet it is not clear whether the characteristic features of migraine are the same in both genders. The present study aimed for the first time to investigate the impact of gender on characteristics of migraine. The results indicated that gender has an impact on the characteristics of migraine and its associated symptoms. In addition, age-dependent change in headache characteristics and associated symptoms were detected only in women migraineurs and not in male migraine patients.
Women diagnosed as having migraine experienced significantly longer and more intense headache attacks in all groups when compared with men. The longer duration of headache in migraine patients was also observed by other investigators (21,27). The higher intensity of headache attacks perceived in women migraineurs was demonstrated in previous studies (24,28). In addition to the higher prevalence of migraine headache, the longer and more severe migraine attacks in women suggest that the course of headache attacks can be different in women when compared with men. It has also been noted that unilaterality of MwA or MwoA attacks is not influenced by gender.
As regards the investigated migraine features, a gender impact was observed for nausea and phonophobia, and both symptoms were more prevalent in women migraineurs. A gender impact was not detected for unilaterality, vomiting and photophobia in the migraine with aura patients. On the other hand, existence of photophobia did not show any gender tendency in migraineurs with aura while it was more frequent in women in MwoA (Table 3). The latter finding indicates that sensitivity to light may be an intrinsic property of MwA in a gender-independent manner. Photophobia was found to be more prevalent after the age of 30 years in women with MwA. This finding was also noticed by Wöber-Bingöl (2004) (27). However, previous studies cannot be compared with our results on headache characteristics in migraine, as age-dependent variation was not investigated in relation to gender or aura (29). In addition, associated symptoms of photophobia and phonophobia were reported to be decreased or increased with age in migraineurs even when disregarding gender (29) or aura (27,30). Moreover, unilaterality of MwA attacks did not show any alteration between the age groups.
It is intriguing that the headache characteristics significantly vary with age in women with either MwA or MwoA, while headache features do not show any alteration in male migraineurs. We have no precise explanation for such a discrepancy between female and male genders and a simple explanation of sex hormonal influence does not seem to be sufficient.
Several characteristic features such as photophobia, phonophobia and nausea show significant alterations and become pronounced after 30 years of age. Avoidance and disengagement coping strategies to manage headache were recently found to be associated with nausea and vomiting (31). The increase in the above-mentioned features may be explained partly by a voluntary or implicit change in coping attitudes.
The significant gender difference in the intensity and duration of headache attacks needs further comment. The perception of pain severity is also subjective and depends on the first-person experience, while the duration of a migraine attack is a more objective measurement. However, the longer the headache duration, the more substantial impact on the perception of headache intensity can be expected. In addition, increased duration and severity of headache attacks associated with increased sensitivity to sensory stimuli are likely to induce more severe disability and reduce activity in women compared to men (1,32).
It is also well-established that women have lower pain tolerance and pain threshold when exposed to noxious stimuli (14,20). Moreover, gender role expectations should not be underestimated in pain research, i.e. men usually deny and/or tolerate pain. Gender roles are learned through interactions with the social environment. In addition, women were found to be more willing to report pain and to seek medical treatment more frequently than men (33,34), and use emotion-focused coping strategies (35–37).
Those differences could underlie a different course of MwA in women than in men for whom the disease manifests longer, more severely and with more prominent associated features such as nausea and phonophobia. In addition, this divergence is more prone to be enhanced between the fourth and fifth decades of life. The neurobiological mechanisms for the difference between the third versus the fourth and fifth decades are not exactly known. It is interesting that the age-dependent change of the gender impact was observed only in women. Although this fact is associated with the close relation between migraine and sex steroids, we found that this influence is common only at the age of 30 years. Nevertheless, 30 years of age is higher than the first menarche age, the first marriage age or the first motherhood age in general. Patients in our study were regularly menstruating women and were not using hormone therapy. However, increased weight gain at the age of 30 years in women may play a role at significant divergence, though we did not take body mass index (BMI) measurements into account because of inadequate patient data.
It is interesting that the gender impact on headache features apart from photophobia were not significantly different in the MwA and MwoA groups though experimental studies have demonstrated a notable impact of sex steroids on cortical excitability and pain in migraine models (38,39). In general, the impact of gender on MwoA was similar to that of MwA. Gender impact was detected in all eight features except for unilaterality and vomiting in the MwoA group. A notable difference from MwA patients was the photophobia that was subject to gender impact and more prevalent in women migraineurs in MwoA patients.
Women had longer and more severe headache attacks when compared with men. However, women demonstrated less frequent headaches within the preceding month after the age of 30 years in both the MwA and MwoA groups (Table 3). Among the associated symptoms, nausea and phonophobia exhibited an increase after the age of 30 years, but not photophobia. After 50 years of age, a significant decline in photophobia was observed in women in the MwoA group. One of the differences from MwA was in the unilaterality of MwoA attacks that was reduced after the age of 50 years in women. On the other hand, these features of migraine did not show any age-dependent change in men.
Migraine is three times more prevalent in women (40,41) and the prevalence declines with age for both genders although the ratio of women remains higher (42–44). Hormonal factors, onset of menstruation, the use of oral contraceptive drugs and pregnancy have a crucial impact on migraine attacks. However, the persistence of gender difference in the prevalence after menopause suggests that other factors are also involved (45).
The main limitations of the present study are as follows: i) Our database did not define the “probable” group separately as it included data from before 2004; ii) chronic migraine was not evaluated as a separate group in this study and these patients might have been overlooked within the large amount of data. These issues can be discussed specifically in future studies.
The strengths of the present study include the following: i) This is the first paper, to the best of our knowledge, to analyze age- and gender-based difference of headache characteristics of migraine patients in a broad tertiary center-based population; ii) the study included a dataset for which a headache diagnosis had been made by a headache specialist with a face-to-face evaluation of the patients rather than using a questionnaire; iii) the multicenter clinical survey was conducted within a long time period and included both headache classifications.
The present study indicates a gender-related discrepancy in the clinical features of the two migraine subgroups. A gender impact was detected for headache duration, headache intensity, nausea and phonophobia in all migraine patients but not for the unilaterality feature and vomiting symptom. Photophobia has a distinctive role in the two migraine subgroups; gender did not affect its prevalence in the MwA patients while the symptom was more prevalent in female MwoA patients. Headache properties and associated symptoms varied across age groups in female migraine patients while they were not subject to age-dependent changes in males.
Photophobia, phonophobia and nausea symptoms were more prevalent in women. Along with increased duration and severity of the headache attacks, the associated symptoms are likely to produce more severe disability and reduced activity in women. We suggest that patients should be informed of gender-based differences in the course of attacks and that gender impact on headache characteristics be integrated into the diagnostic criteria for migraine.
Clinical implications
Gender has an influence on the characteristics of migraine headache and its associated symptoms. Headache attacks in women migraineurs were significantly longer in duration and higher in intensity. Nausea and phonophobia were more prevalent in women with migraine headaches. Photophobia, more frequent in women with migraine without aura, was a gender-independent feature of migraine with aura. Gender impact varied across age groups, and significant changes occur in women migraineurs after the age of 30 years. No age-dependent variation was observed in male migraineurs.
Footnotes
Authors’ note
Part of this study was presented at the 15th International Headache Congress, 23–26 June 2011, Berlin, Germany.
Funding
This work was supported by the Turkish Neurological Society and the TUBA (Turkish Academy of Sciences, HB).
Conflict of interest
None declared.
