Abstract
Background
Fremanezumab, a fully humanized monoclonal antibody targeting calcitonin gene-related peptide, has demonstrated efficacy for the preventive treatment of migraine in adults.
Objective
To evaluate the effect of fremanezumab treatment on acute headache medication use and migraine-associated symptoms in patients with episodic migraine.
Methods
In the Phase 3 HALO trial, patients with episodic migraine were randomized to receive subcutaneous fremanezumab monthly (225 mg at baseline, weeks 4 and 8), fremanezumab quarterly (675 mg at baseline, placebo at weeks 4 and 8), or placebo over a 12-week period. The secondary endpoint was change from baseline in the monthly number of days with use of any acute headache mediation or migraine-specific acute headache medication; exploratory endpoints were change from baseline in the monthly number of days with nausea or vomiting, photophobia, or phonophobia.
Results
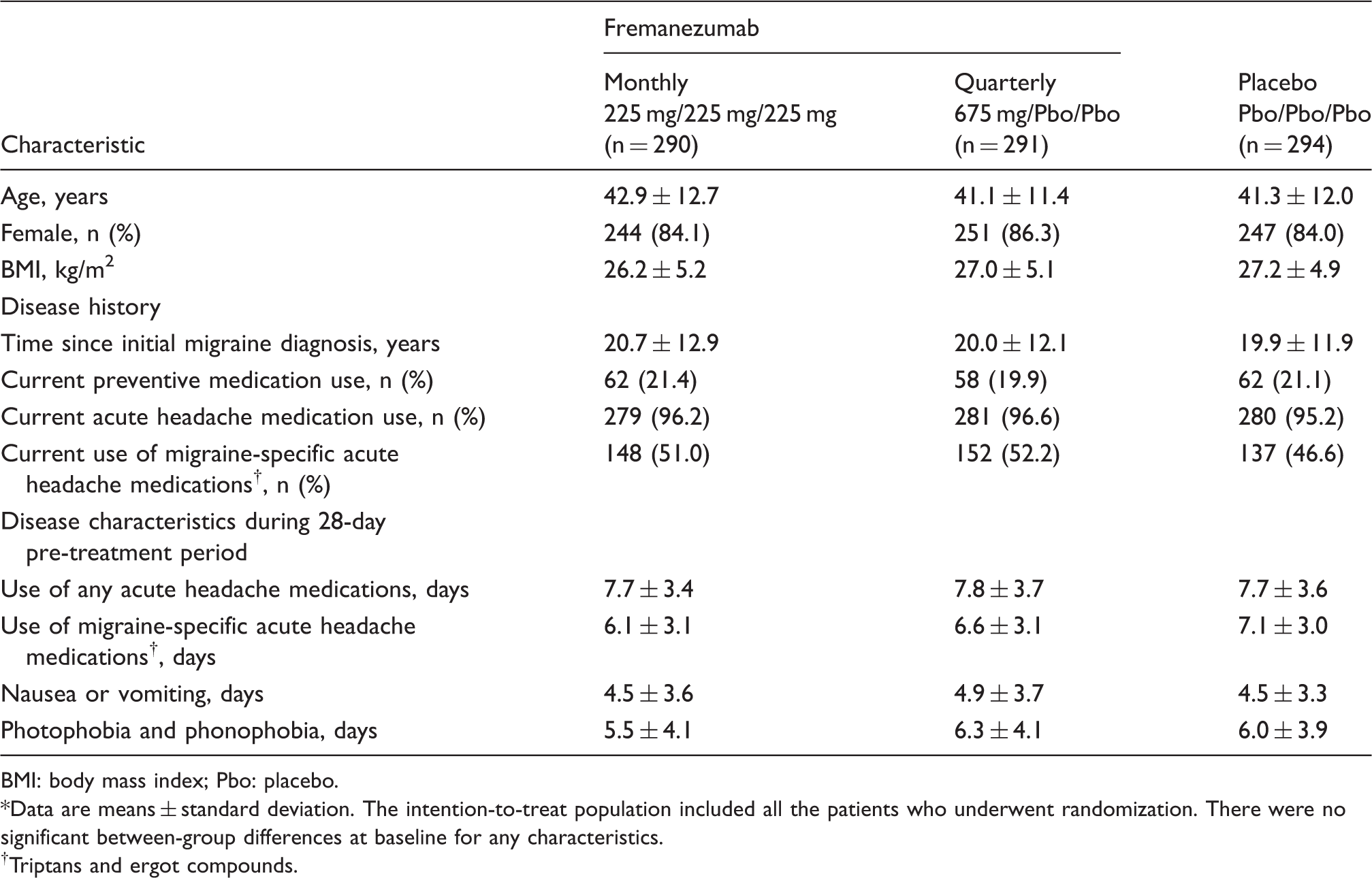
Of 875 patients randomized, 865 were included in the analysis (monthly, n = 287; quarterly, n = 288; placebo, n = 290). Baseline mean ± standard deviation days with: Any acute headache medication use (monthly: 7.7 ± 3.4; quarterly: 7.8 ± 3.7; placebo: 7.7 ± 3.6), migraine-specific acute headache medication use (6.1 ± 3.1; 6.6 ± 3.1; 7.1 ± 3.0), nausea or vomiting (4.5 ± 3.6; 4.9 ± 3.7; 4.5 ± 3.3) and photophobia and phonophobia (5.5 ± 4.1; 6.3 ± 4.1; 6.0 ± 3.9) were similar among treatment arms. Fremanezumab reduced the number of days of acute headache medication use ([least-squares mean change vs. placebo] monthly: −1.4 [95% confidence interval: −1.84, −0.89], p < 0.001; quarterly: −1.3 [−1.76, −0.82], p < 0.001) and migraine-specific acute headache medication use (monthly: −2.2 [−2.80, −1.56], p < 0.001; quarterly: −2.2 [−2.81, −1.58], p < 0.001) compared with placebo. Fremanezumab also reduced nausea or vomiting, photophobia, and phonophobia compared with placebo.
Conclusions
Fremanezumab reduced the need for acute headache medications, including migraine-specific medications, while treating migraine-associated symptoms in patients with episodic migraine.
Trial registration
Clinicaltrials.gov NCT02629861
Keywords
Introduction
The majority of individuals with migraine have episodic migraine, often defined as migraine occurring fewer than 15 days per month (1,2). Guidelines generally recommend preventive treatment when an individual has two or more disabling attacks per month that cannot be managed with acute therapy (3,4). Recognizing that treating the associated symptoms of migraine is also critical to migraine therapy is implied in the criteria for diagnosis of migraine; that is, the International Classification of Headache Disorders, third edition (beta version) (ICHD-3 beta) criteria for migraine require the presence of at least one of the following non-head pain symptoms: Nausea and/or vomiting, or photophobia and phonophobia (5).
Nausea is a common, debilitating migraine-related symptom that occurs in almost 50% of patients with episodic migraine and is associated with an increased risk of progression to chronic migraine (6). The presence of additional migraine-associated symptoms, such as nausea and/or vomiting and photophobia, has been shown to result in greater disease burden, higher healthcare resource utilization, and decreased quality of life (7,8). Data from a cross-sectional survey indicated that one-fifth of patients with migraine were prescribed triptans, but an estimated one-third discontinue use within 1 year either from lack of efficacy or side effects (9).
Acute headache medications, including simple and combination analgesics and migraine-specific agents (triptans and ergot derivatives), may be used to treat migraine (10). Many of these medications, however, are nonspecific and with limited efficacy, resulting in an increased need for their use. This increased use is associated with the development of medication overuse headache and progression from episodic to chronic migraine (11). Effective preventive treatment may reduce the use of acute headache medications, including migraine-specific acute medications.
Fremanezumab is a fully humanized monoclonal antibody (IgG2Δa) approved in the United States and the European Union for the preventive treatment of migraine in adults (12–14). Fremanezumab belongs to a new class of migraine preventive therapies that target calcitonin gene-related peptide (CGRP), a neuropeptide involved in migraine pathophysiology, or its receptor (13,15,16). Fremanezumab has a flexible dosing regimen with quarterly or monthly dosages and is administered by subcutaneous injection (12,14). In a Phase 2b study of patients with high-frequency episodic migraine, fremanezumab demonstrated significant reductions in the monthly average number of migraine days relative to placebo with no serious treatment-related adverse events (AEs) (17). A post hoc analysis of the Phase 2b study also found that fremanezumab reduced migraine-associated symptoms within the first few weeks of treatment (18).
While the efficacy, safety, and immunogenicity of fremanezumab monthly and quarterly for the preventive treatment of episodic migraine have been demonstrated in the pivotal randomized, double-blind, placebo-controlled, parallel-group, Phase 3 HALO EM trial (19), here we report the results from secondary and exploratory analyses evaluating the efficacy and safety of fremanezumab on acute headache medication use and migraine-associated symptoms in these patients.
Methods
Study setting and participants
The methodology of the HALO EM trial has been reported previously (19). Briefly, study participants were men and women aged 18–70 years with a history of migraine (ICHD-3 beta diagnostic criteria) for at least 12 months prior to screening and with onset prior to 50 years of age. During a 28-day pre-treatment baseline period, patients were confirmed (via an electronic headache diary device) to have episodic migraine: 6–14 headache days, with at least 4 days fulfilling ICHD-3 beta criteria for migraine with [1.2; B and C] or without [1.1; C and D] aura or probable migraine. Any day during which an acute migraine-specific medication (triptan or ergot derivative) was used was included as a migraine day. Patients had to demonstrate compliance with the electronic headache diary device by entering headache data on at least 24 days of the 28-day pre-treatment period (about 85% compliance). Concomitant use of one preventive migraine medication was allowed in a subset of patients, if the dosage was stable for at least 2 months prior to the pre-treatment period to the end of the study.
Use of onabotulinumtoxinA during the 4 months prior to screening, use of opioids or barbiturates on more than 4 days during the pre-treatment period, or previous failure of at least two medication clusters (divalproex sodium and sodium valproate; flunarizine and pizotifen; amitriptyline, nortriptyline, venlafaxine, and duloxetine; or atenolol, nadolol, metoprolol, propranolol, and timolol) after 3 or more months of treatment for migraine, was exclusionary. Patients were also excluded if they received an intervention or used a device for migraine during the last 2 months before screening.
Trial oversight
The protocol was approved by an independent ethics committee or institutional review board according to national or local regulations. This study was conducted in accordance with the International Conference on Harmonization Good Clinical Practice Consolidated Guideline and any applicable laws and regulations. Each patient provided written informed consent before any procedures or assessments were conducted.
Study design
This 16-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study consisted of a screening visit, 28-day pre-treatment period, 12-week treatment period, and final evaluation at Week 12. Based on screening and daily headache information captured during the pre-treatment period, individuals were enrolled in the current study for episodic migraine (Clinicaltrials.gov NCT02629861), or a concurrent study for chronic migraine (Clinicaltrials.gov NCT02621931), or excluded (19,20). Patients were randomized (1:1:1) to receive either fremanezumab monthly (225 mg at baseline and weeks 4 and 8), fremanezumab quarterly (675 mg at baseline and placebo at weeks 4 and 8), or placebo (at baseline, and weeks 4 and 8).
Study procedures and endpoints
Patients were evaluated at five scheduled site visits: Screening, baseline (dose 1), week 4 (dose 2), week 8 (dose 3), and end of treatment (week 12)/early withdrawal. Daily headache data, including the presence of migraine-associated symptoms (photophobia, phonophobia, nausea, or vomiting) and acute headache medication use, were captured via the electronic headache diary device.
The monthly average number of days of any acute headache medication use during the 12-week period, after the first dose of study drug relative to the baseline period, was evaluated as a secondary endpoint. Exploratory endpoints included: The mean change from baseline (28-day pre-treatment period) in the monthly average number of days of migraine-specific acute headache medication (triptan and ergot compounds) use during the 12-week period after the first injection; the mean change from baseline (28-day pre-treatment period) in the monthly average number of days with nausea or vomiting during the 12-week period after the first injection; the mean change from baseline (28-day pre-treatment period) in the monthly average number of days with photophobia and phonophobia during the 12-week period after the first injection.
Safety and tolerability assessments included reported AEs, vital signs, 12-lead electrocardiogram, clinical laboratory tests, and physical examinations.
Statistical analysis
Estimations from the primary analysis predicted that a sample of 675 patients (225 per treatment group) would provide 90% power to detect a 1.6-day (standard deviation, 5.2 days) difference in migraine days between an active and placebo group at α = 0.05 (19). With an expected dropout rate of 12%, 768 patients were planned for randomization.
Efficacy analyses were performed in the same full analysis set as the primary analysis: All randomized patients who received at least one dose of study drug and had 10 or more days of post-baseline efficacy assessments. The secondary and exploratory endpoints were analyzed via an analysis of covariance (ANCOVA) method (fixed effects: Treatment, sex, region, and baseline preventive medication; covariates: Corresponding baseline value and years since onset of migraine). Confidence intervals (95%) were determined for the least-squares mean differences between each fremanezumab group and the placebo group. An unconstructed covariance structure was used. If there was a deviation from normality assumption, as assessed by Shapiro–Wilk's test, then a Wilcoxon rank-sum test was performed as primary analysis. A mixed-effects repeated-measures (MRMM) model was used to estimate the mean change from baseline for the endpoints by month (fixed effects: Treatment, sex, region, baseline preventive medication use, month, and treatment*month; covariates: Baseline value and years since onset of migraine; random effect: Patient).
If a patient had missing electronic headache diary data in a month, these data were assumed to be missing at random: In patients with 10 or more days of data, the monthly number of that variable was prorated to 28 days for that month; in patients with less than 10 days of data, the monthly number of that variable was considered missing.
Safety data were summarized descriptively in all randomized patients who received at least one dose of study drug.
Results
Study participants
The primary efficacy and safety data for the HALO EM trial have been previously reported (19). Briefly, 875 patients randomized to one of the three treatment arms (fremanezumab monthly, n = 290; fremanezumab quarterly, n = 291; placebo, n = 294) (Figure 1), of which 791 patients (262 fremanezumab monthly; 264 fremanezumab quarterly; 265 placebo) completed the study. Baseline demographics and clinical characteristics were similar between all treatment arms (Table 1). A total of 21% of patients were allowed to continue treatment with one concomitant preventive migraine medication.
Study disposition. Among the 2995 patients who were assessed for eligibility, 875 were randomized to treatment in this study and 1036 were randomized in a parallel study in patients with chronic migraine (CM). Baseline characteristics of the patients in the intention-to-treat population according to study group*. BMI: body mass index; Pbo: placebo. Data are means ± standard deviation. The intention-to-treat population included all the patients who underwent randomization. There were no significant between-group differences at baseline for any characteristics. Triptans and ergot compounds.
Acute headache medication use
Reductions in the number of monthly average days of any acute headache medication use from baseline were significantly greater with fremanezumab monthly ([least-squares mean change ± standard error] −3.0 ± 0.22 days; difference vs. placebo: −1.4 [95% confidence interval: −1.84, −0.89], p < 0.001) and quarterly (−2.9 ± 0.22 days; difference vs. placebo: −1.3 [−1.76, −0.82], p < 0.001) dose regimens compared with placebo (−1.6 ± 0.21 days) during the 12-week treatment period (Figure 2(a)). In patients who used migraine-specific acute headache medications at baseline (fremanezumab monthly, n = 148; fremanezumab quarterly, n = 152; placebo, n = 136), significant reductions in the monthly average number of days of migraine-specific acute headache medication use from baseline period (fremanezumab monthly, 6.1 days; fremanezumab quarterly, 6.6 days; placebo, 7.1 days) were seen with fremanezumab monthly (−3.1 ± 0.26 days, reduction of 50.8%; difference vs. placebo: −2.2 [−2.80, −1.56], p < 0.001) and quarterly (−3.1 ± 0.26 days, reduction of 47.0%; difference vs. placebo: −2.2 [−2.81, −1.58], p < 0.001) dose regimens compared with placebo (−0.9 ± 0.27 days, reduction of 12.7%) during the 12-week treatment period (Figure 2(b)).
Use of any acute and migraine-specific acute headache medications. The effects of fremanezumab (monthly and quarterly) treatment on (a) the change from baseline in the monthly average number of days of any acute headache medication use from baseline and (b) the change from baseline in the monthly average number of days of migraine-specific acute headache medication use from baseline during the 12 weeks following first administration of the study drug in patients who used migraine-specific acute headache medication at baseline compared with placebo groups. Data are from the mixed-effects repeated-measures model, with treatment, gender, region, baseline preventive medication use, month, and treatment*month as fixed effects and baseline value and years since onset of migraine as covariates. Patient is a random effect. An unconstructed covariance structure was used. The primary analysis of covariance model includes treatment, gender, region, and baseline preventive medication use as fixed effects and baseline value and years since onset of migraine as covariates.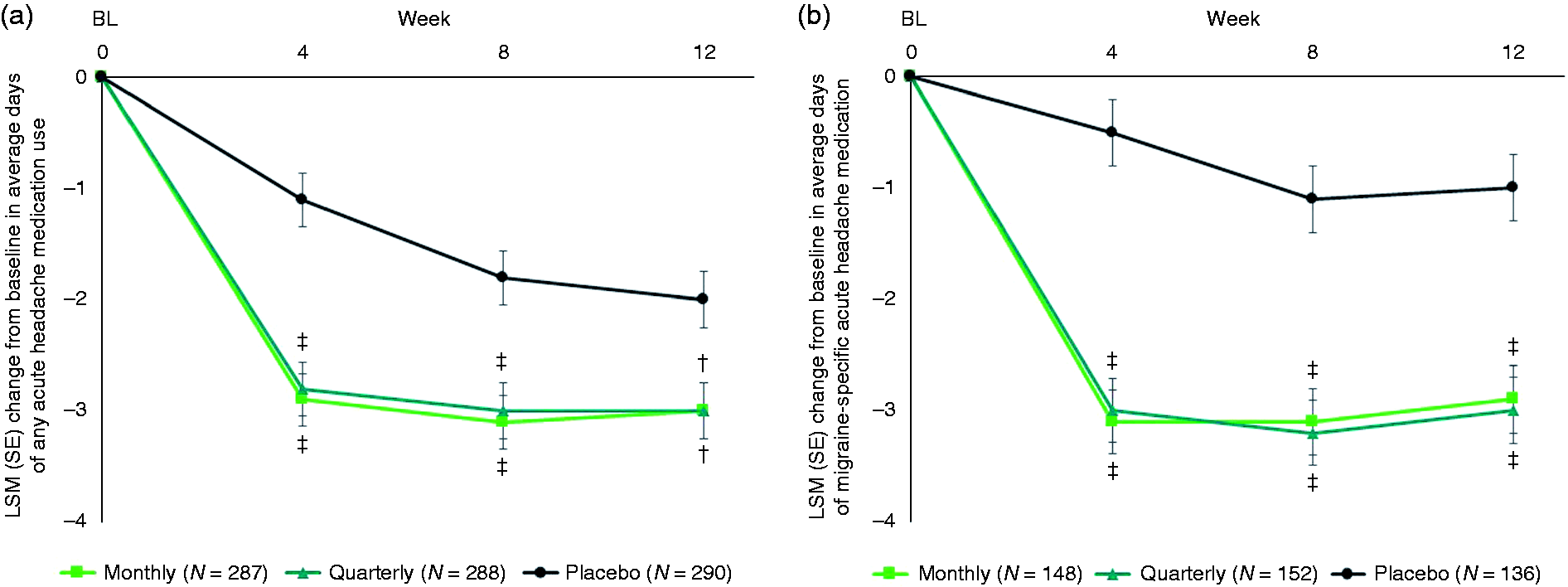
Migraine-associated symptoms
Fremanezumab significantly reduced the monthly average number of days with nausea or vomiting from baseline for monthly (−2.1 ± 0.19 days; difference vs. placebo: −0.7 [−1.12, −0.29], p < 0.001) and quarterly (−1.9 ± 0.19 days; difference vs. placebo: −0.5 [−0.87, −0.04], p = 0.031) dose regimens compared with placebo (−1.4 ± 0.19 days) during the 12-week treatment period (Figure 3(a)). Significant reductions from baseline were observed in the monthly average number of days with photophobia for fremanezumab monthly (−3.0 ± 0.23 days; difference vs. placebo: −0.9 [−1.43, −0.45], p < 0.001) and quarterly (−2.8 ± 0.23 days; difference vs. placebo: −0.8 [−1.27, −0.29], p = 0.002) dose regimens compared with placebo (−2.0 ± 0.22 days) during the 12-week treatment period (Figure 3(b)). Fremanezumab treatment significantly reduced the monthly average number of days with phonophobia from baseline for monthly (−3.0 ± 0.22 days; difference vs. placebo: −1.0 [−1.45, −0.49], p < 0.001) and quarterly (−2.7 ± 0.22 days; difference vs. placebo: −0.6 [−1.12, −0.16], p = 0.009) dose regimens compared with placebo (−2.1 ± 0.22 days) during the 12-week treatment period (Figure 3(c)).
Days experiencing non-head pain migraine symptoms. The effects of fremanezumab (monthly and quarterly) treatment on (a) the change from baseline in the monthly average number of days with nausea or vomiting from baseline, (b) the change from baseline in the monthly average number of days with photophobia, and (c) the change from baseline in monthly average number of days with phonophobia during the 12 weeks following first administration of the study drug, compared with placebo groups. Unless otherwise stated, data are from the mixed-effects repeated-measures model, with treatment, gender, region, baseline preventive medication use, month, and treatment*month as fixed effects, baseline value and years since onset of migraine as covariates, and patient as a random effect. An unconstructed covariance structure was used. The primary analysis of covariance model includes treatment, gender, region, and baseline preventive medication use as fixed effects and baseline value and years since onset of migraine as covariates.
Safety and tolerability
The overall safety results have been previously reported (19). At least one adverse event (AE) was reported in 66% of patients in both the fremanezumab monthly and quarterly groups, compared with 58% of patients who received placebo. Rates of severe AEs, serious AEs, and AEs leading to discontinuation were low and similar across active treatment and placebo groups ( ≤ 2%).
Injection-site reactions (pain, induration, and erythema) were the most common AEs reported in patients treated with fremanezumab, and these events occurred at slightly higher rates in the fremanezumab treatment groups than in the placebo group. No significant changes were observed in any patients in terms of vital signs, physical examinations, electrocardiogram findings, or laboratory parameters. Four patients developed antidrug antibodies against fremanezumab, one of whom developed neutralizing antibodies, without any significant safety issues.
One patient in the fremanezumab quarterly group died from diphenhydramine overdose (suicide) after withdrawing consent due to a family emergency and relocation. The patient was reported as deceased 109 days after receiving fremanezumab and had withdrawn from the study 38 days earlier. The death was determined by the investigator to be unrelated to treatment.
Discussion
In these pre-specified analyses from a Phase 3 study of fremanezumab for the preventive treatment of episodic migraine, treatment with fremanezumab quarterly and monthly improved a range of migraine-related symptoms and reduced acute medication use. The observed improvements in migraine-related symptoms complement the efficacy (reduction of migraine frequency) demonstrated in this clinical trial and a previously reported Phase 2b study in patients with high-frequency episodic migraine (17,19). Data are also consistent with a post hoc analysis of data from the Phase 2b study demonstrating a reduction in migraine-associated symptoms within the first few weeks of initiating fremanezumab treatment (18). The safety and tolerability profile of fremanezumab in this trial is consistent with previous clinical trials, with no clinically significant pattern of AEs or drug-related serious AEs (17,20,21).
Results here demonstrated that fremanezumab treatment decreased the need for acute headache medication use, particularly migraine-specific acute headache medication use. This may result in many potential benefits, as use of both acute headache medications and migraine-specific acute headache medications is associated with development of medication overuse resulting in progression to chronic migraine, systemic AEs, loss of effectiveness over time, anxiety related to limited availability, and high costs (9–11). Furthermore, the favorable side effect profile of fremanezumab is more likely to result in treatment adherence.
Additionally, fremanezumab treatment led to significant decreases in the most common non–head pain symptoms of migraine that are included in the diagnostic criteria for migraine: Nausea or vomiting, photophobia, and phonophobia. These reductions in associated features of migraine support the primary efficacy findings and further suggest that fremanezumab may improve patient outcomes, as both the headache and associated non-headache symptoms of migraine are reduced. Given that nausea is a common and debilitating symptom associated with an increased risk of progression from episodic to chronic migraine (6), the improvements in nausea or vomiting seen with fremanezumab treatment may be particularly consequential. Although our understanding of the pathophysiological correlates of the symptoms associated with migraine are not completely understood, the role of CGRP in photophobia has been established by investigations showing that peripherally administered CGRP can induce light aversion in mice as well as photophobia and nausea in humans (22,23).
Limitations of this study include that these were pre-specified secondary and exploratory analyses of the HALO EM trial. Exploratory endpoints were not adjusted for multiple comparisons. Further studies are needed to confirm or support these findings. With respect to the relationship between fremanezumab, medication overuse headache, and transformation from episodic to chronic migraine, the migraine-associated outcomes and symptoms assessed in this study (acute headache medication use, nausea) are associative rather than causative. Further studies will be critical to a more direct examination of these relationships.
Based on migraine-related outcomes, this study demonstrated that fremanezumab (both monthly and quarterly dose regimens) is an efficacious preventive treatment for patients with episodic migraine, with the potential to improve patient outcomes while reducing the need for acute headache medications, including migraine-specific ones.
Footnotes
Clinical implications
Fremanezumab reduced the need for acute headache medications (both non-migraine-specific and migraine-specific acute medications) in patients with episodic migraine.
Fremanezumab reduced migraine-associated symptoms, such as nausea or vomiting, photophobia, and phonophobia in patients with episodic migraine.
Fremanezumab may improve patient outcomes by means of these findings.
Acknowledgements
We thank the patients who participated in this study and their families; all investigators, site personnel, and the coordinating investigators; and Kristen Hokenson, PhD (Chameleon Communications International with funding from Teva Pharmaceutical Industries Ltd) for editorial assistance in the preparation of this report.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JLB declares research for Amgen, Eli Lilly and Company, CoLucid Pharmaceuticals, Teva Pharmaceuticals, Alder, Zosano, Allergan, and Biohaven Pharma and membership of advisory boards/speaker’s bureau for Amgen, Eli Lilly and Company, Teva Pharmaceuticals, Depomed, Promius Pharma, Supernus Pharmaceuticals, and Valeant. DK declares membership of advisory boards for Amgen, Eli Lilly and Company, and Alder BioPharmaceuticals, and research for Amgen, Alder BioPharmaceuticals, Eli Lilly and Company, Teva Pharmaceuticals, Biohaven Pharma, Allergan, CoLucid Pharmaceuticals, Dr Reddy’s Laboratories, Genentech-Roche, and UCB. PPY is a former employee of Teva Pharmaceutical Industries Ltd. FS declares membership of advisory boards for Amgen, Eli Lilly and Company, and Teva Pharmaceuticals. EA is a former employee of Teva Branded Pharmaceutical Products R&D, Inc. (USA). TB is a former employee of Teva Branded Pharmaceutical Products R&D, Inc. (USA). MG-W is an employee of Teva Branded Pharmaceutical Products R&D, Inc. (USA). RY is an employee of Pharmaceutical Industries Ltd. YM is an employee of Teva Branded Pharmaceutical Products R&D, Inc. (USA).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Teva Pharmaceutical Industries Ltd, Petach Tikva, Israel.
