Abstract
Background
Capsaicin induces the release of calcitonin gene-related peptide (CGRP) via the transient receptor potential channel V1 (TRPV1). The CGRP response after capsaicin application on the tongue might reflect the “activation state” of the trigeminal nerve, since trigeminal CGRP-containing vesicles are depleted on capsaicin application. We tested (i) the quantitative CGRP response after oral capsaicin application; (ii) the optimal concentration of red chili homogenate; and (iii) the day-to-day variability in this response.
Methods
Saliva was collected for two consecutive days after oral application of eight capsaicin dilutions (red chili homogenates) of increasing concentrations in 13 healthy individuals. Effects of homogenate concentration were assessed. Consecutively, saliva was sampled after application of vehicle and undiluted homogenates.
Results
CGRP secretion (pg/ml) increased dose-dependently with homogenate concentration (p < 0.001). CGRP levels were highest after application of nondiluted homogenate (vs. baseline: 13.3 (5.0) vs. 9.7 (2.9); p = 0.003, as was total CGRP secretion in five minutes (pg) with undiluted (vs. baseline): 89.2 (44.1) vs. 14.1 (2.8); p < 0.001. The dose-dependent response in CGRP was not affected by day (p = 0.14) or day*concentration (p = 0.60). Increase in CGRP (undiluted – baseline; pg/ml) did not differ between measurements on dose-finding (p = 0.67) and follow-up days (p = 0.46).
Conclusion
Oral application of red chili homogenate is well tolerated and causes a dose-dependent CGRP release in saliva, without day-to-day effects in this response. This model could be used to noninvasively study the activation state of the trigeminal nerve innervating salivary glands.
Introduction
Capsaicin is derived from hot peppers of the Capsicum genus and binds to transient receptor potential channels, such as the TRPV1 (formerly known as vanilloid receptor type 1) (1). Activation of the TRPV1 channels on trigeminal nerve terminals causes the release of calcitonin gene-related peptide (CGRP, a 37 amino-acid inflammatory neuropeptide present in the central, enteric and peripheral nervous system) as well as other inflammatory mediators (1,2), also from the major salivary glands and buccal mucosa (3–5). The salivary glands are integrated into the neuroendocrine system through complex regulatory pathways, and some salivary peptides are also found in cerebrospinal fluid (6,7). Clinically, nonstimulated elevated salivary CGRP levels were found during the premonitory or headache phase in two-thirds of attacks (8,9), or at baseline (10). The capsaicin-induced salivary CGRP response might thus reflect the “activation state” of the trigeminal nerve, innervating the salivary glands (10–13). Activation of the sensory C fibers in the ophthalmic branch of the trigeminal nerve induces this salivary release of CGRP and other neuropeptides, blood vessel dilation and increased glandular secretion (14,15). Subcutaneous and intradermal application of capsaicin to the facial skin causes local flaring and pain sensation (16–19), and topical application of capsaicin was recently described to induce reproducible increases in forehead dermal blood flow as a marker of trigeminal nerve-mediated vasodilation in humans (20). However, quantification of CGRP release after dermal capsaicin application is difficult (17–19). Application of capsaicin in the oral cavity also activates trigeminal nerve endings (16,21,22) and the subsequent release of CGRP in saliva can be quantified in animals (23). In humans there are no good reference studies yet. This, however, is useful since it enables the study of a variety of trigeminal conditions including headaches noninvasively and prospectively. The aim of this study is i) to quantify the CGRP response after capsaicin application; ii) to find the optimal concentration of red chili homogenate containing capsaicin for this response; and iii) to assess day-to-day variability.
Methods
Subjects
A total of 13 healthy controls (seven women; mean (SD) age 30.9 (5.9) years) were included in the dose-finding pilot study using a cumulative dose-response design, aiming to find the (dilution of) red chili homogenate with the highest salivary CGRP response that was still well tolerated. Eleven healthy controls (partly overlapping with the first group: six women; mean (SD) age 30.9 (5.9) years, all with Western food habits) were subsequently included in a follow-up study, aiming to confirm the previous findings, in a design in which a single dose was applied. All participants provided written informed consent and the study was approved by the local ethics committee.
Study design and collection methods
In the dose-finding study, saliva was collected for two consecutive days after oral application of eight capsaicin samples of inclining concentrations, from 106 times dilution to pure, undiluted red chili homogenate. Per concentration, saliva was collected during five minutes (see Figure 1), and homogenates of increasing concentration were applied consecutively. All individuals prepared the mouth by expectorating, rinsing with tepid tap water twice, and discarding to the sink prior to the first measurement each day. Individuals were given detailed verbal instructions (not to speak; not to move, not to swallow saliva) and were provided appropriate containers for saliva collection (50 ml conical tubes). Before the first sample, 2% citric acid solution was applied to the apex and sides of the tongue using a cotton-tipped applier, to stimulate salivary flow. On the second day, the whole procedure was repeated. Perceived heat sensation was measured during each homogenate concentration separately with a numeric rating scale (NRS) from 0 (no heat sensation at all) to 10 (highest heat sensation possible). The dose-finding study was nonrandomized and nonblinded deliberately for several reasons. First, the study was aimed at detecting the highest tolerable dose, enabling participants to indicate tolerability step by step. Second, adequate blinding of capsaicin application is very difficult, since oral administration directly affects heat sensation and saliva production. Third, with randomization, and thereby possible early exposure to the undiluted homogenate, (de)sensitization and depletion might largely affect the outcome parameters of subsequent measurements. In the follow-up study, we repeated salivary sampling in 11 participants after application of only vehicle and undiluted red chili homogenates using the same protocol.
Study design. Thirteen healthy volunteers were exposed on two consecutive days to eight increasing concentrations of Madame Jeannet chili homogenates containing capsaicin. Exposure lasted five minutes for each of the homogenate dilutions, which ranged from 106 times diluted to pure homogenate. Secreted saliva was collected during each of these periods of five minutes.
Preparation of red chili homogenate
Red chili homogenates containing capsaicin were obtained from fresh Madame Jeanette chili peppers (Capsicum chinense). The green peduncles as well as the seeds were removed from the chili peppers. The remaining part was homogenized, without addition of extra fluids, and immediately frozen in aliquots at −80℃ until use.
Saliva storage
Directly after sampling, saliva was placed on dry ice during the transfer to a freezer (−80℃).
CGRP analysis and determination
CGRP was determined by radioimmunoassay (Phoenix Pharmaceuticals Inc, Burlingame, CA, USA) according to the instructions of the manufacturer. Total protein was determined using the Pierve BCA Protein Assay Kit (Thermo Scientific, Rockford, IL, USA). Internal standards were used in every measurement. To assess potential cross-reactivity between the capsaicin in the red chili homogenates and the CGRP analysis kit, we also assayed homogenates themselves. Analysis of the undiluted homogenate indicated a mean (SD) CGRP concentration of 5.7 (0.4) pg/ml. These cross-reactivity effects were subtracted from outcome effects, and adjusted data were used for analysis.
Statistical analysis
CGRP responses after capsaicin application were quantified by using CGRP content (pg) and concentration (pg/ml) in saliva as outcomes. CGRP in baseline samples was compared to levels after maximum capsaicin concentration using a paired Student’s t test. The optimum for CGRP response was defined by the capsaicin concentration (ranging from 106 times diluted to pure, undiluted homogenate) leading to the highest CGRP content in salivary samples, while heat sensation was still acceptable. Day-to-day variability in CGRP response was assessed using a repeated measurement analysis of variance (ANOVA) with CGRP concentration and day as within-subject factors, and interaction between those variables. Correlation between heat sensation and capsaicin concentration (homogenate dilution) was assessed using linear regression. All data analyses were performed using SPSS 17.0 (SPSS Inc, IBM, USA), and p < 0.05 was considered to indicate significant differences.
Results
CGRP secretion at baseline and after capsaicin stimulation
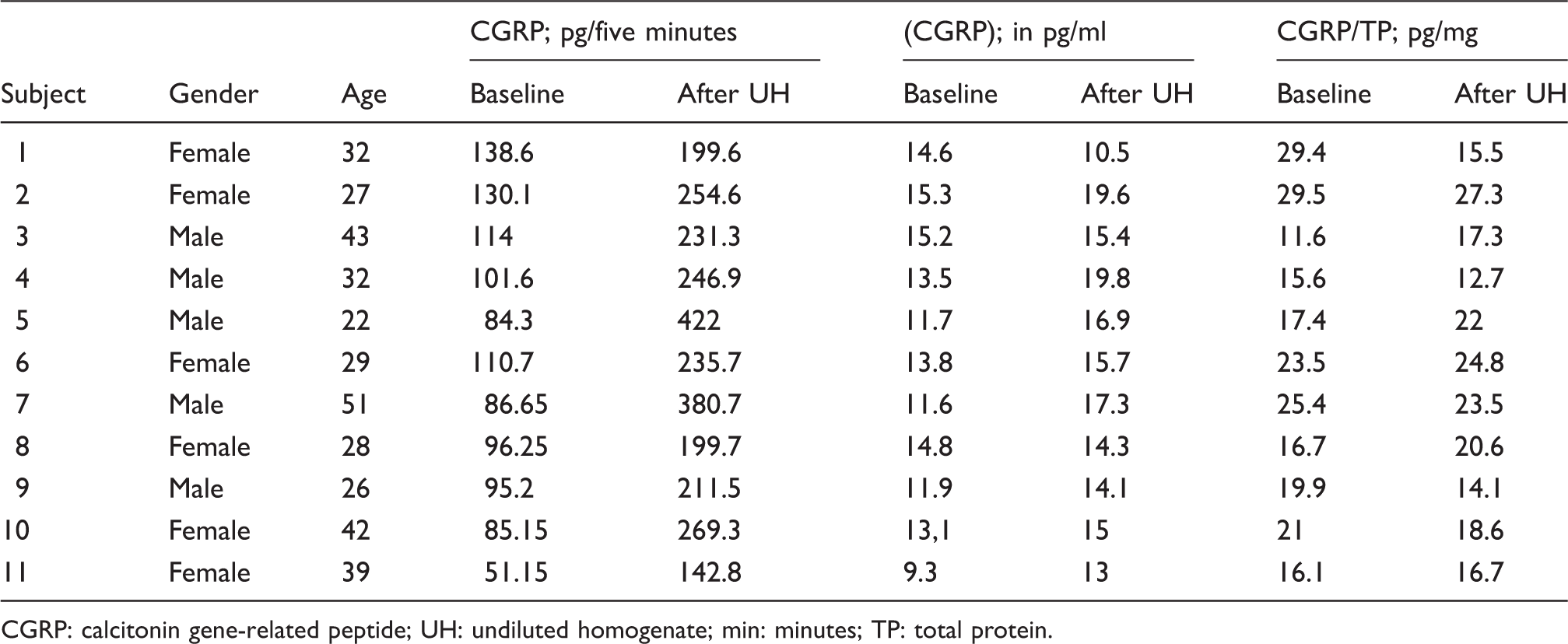
The concentration of the homogenate significantly affected CGRP secretion (p = 0.001). The highest CGRP levels were detected in samples after application of nondiluted chili homogenate in the dose-finding study (Table 1). Mean (SD) CGRP concentration (pg/ml) was higher after undiluted homogenate compared to baseline (13.3 (5.0) vs. 9.7 (2.9); p = 0.003; ratio 13.3/9.7 = 1.4/1), as was the total amount of CGRP (pg) secreted in five minutes (89.2 (44.1) vs. 14.1 (2.8); p < 0.001; ratio 89.2/14.1 = 6.3/1). These findings were confirmed by data from the follow-up study (Table 2): higher salivary CGRP concentration (pg/ml) using undiluted homogenate (15.6 (2.9) vs. 13.1 (2.0); p = 0.038), and higher totally secreted CGRP in five minutes (pg); 255.8 (85.1) vs. 98.3 (25.0); p < 0.001. Additionally, in the dose-finding study the ratio of salivary CGRP/total protein (pg/mg) was similar after stimulation of baseline vs. pure homogenates (19.3 (4.7) vs. 20.5 (5.8); p = 0.45), see Figure 2(c).
Total salivary volume and protein content with capsaicin stimulation. (a) Total secreted salivary volume (ml) depends on capsaicin concentration. (b) Secreted protein content (mg/ml saliva) depends on capsaicin concentration. (c) Ratio of calcitonin generelated peptide (CGRP)/total protein (pg/mg) does not depend on capsaicin concentration, after adjusting for cross-reactivity. In Figures 2(a) and 2(b), continuous lines depict day 1; dashed lines depict day 2. CGRP concentration (pg/ml saliva), amount of secreted CGRP (pg/five minutes), and CGRP/total protein ratio (pg/mg) in samples at baseline and after application of undiluted homogenate (highest concentration) on day 1 and 2 (n = 13) in dose-finding study. Data not from UH tube, but from UH:2 tube. CGRP: calcitonin gene-related peptide; UH: undiluted homogenate; min: minutes; TP: total protein. CGRP concentration (pg/ml saliva), amount of secreted CGRP (pg/five minutes), and CGRP/total protein ratio (pg/mg) in samples at baseline and after application of undiluted homogenate (highest concentration) in follow-up sample (n = 11). CGRP: calcitonin gene-related peptide; UH: undiluted homogenate; min: minutes; TP: total protein.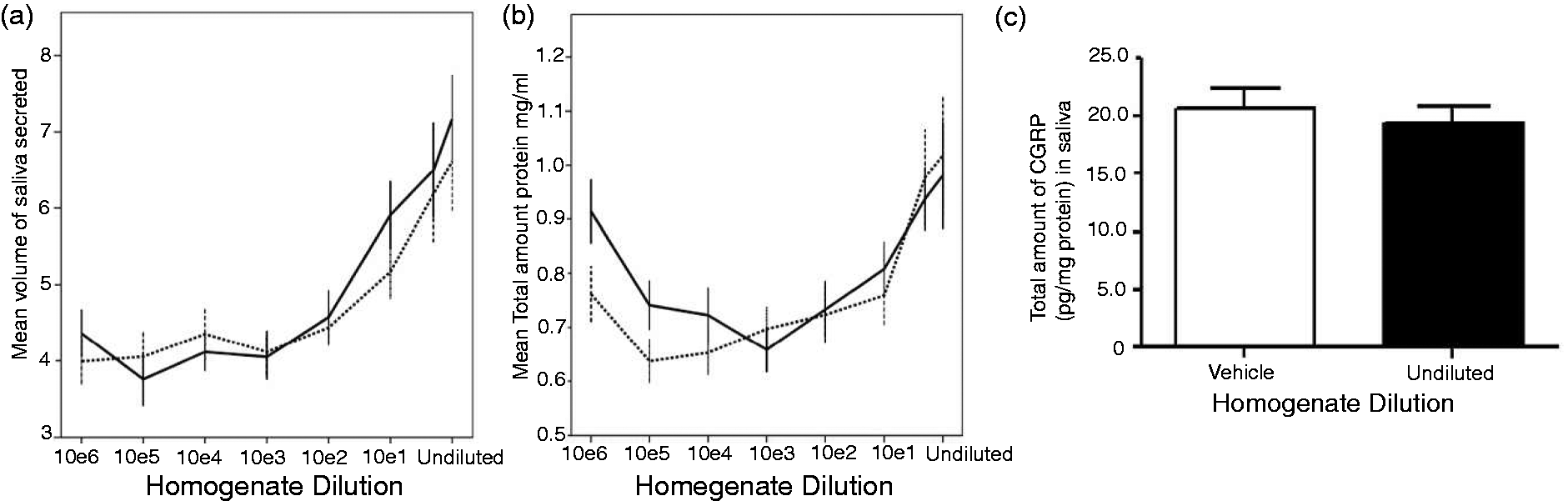

Day-to-day reproducibility
Salivary CGRP concentration (pg/ml) increased in a dose-dependent manner after stimulation with capsaicin on day 1 (p = 0.001) as well as on day 2 (p < 0.001). Combining data from these two days also results in an overall significant positive correlation (r = 0.39, n = 200, p < 0.001). Using analysis of covariance (ANCOVA), we found a significant effect of homogenate concentration (p = 0.001) on CGRP response, but not for the day and the interaction between concentration/day (p = 0.14 and p = 0.60, respectively).
The increase in salivary CGRP concentrations (pg/ml) between baseline and undiluted homogenate did not differ on the two consecutive days (mean (SD): day 1 = 3.0 (6.0) and day 2 = 4.1 (3.3); p = 0.67), nor was it different from the follow-up study (2.5 (1.05); ANOVA p = 0.46).
Total saliva volume and protein content
The total volume of saliva and the level of secreted protein increased with increasing capsaicin concentration, but the ratio of CGRP/total protein remained stable after adjusting for cross-reactivity (Figure 2).
Heat sensation
Red chili homogenates were well tolerated by study participants. Participants reported increasing sensation of heat from the 1:1000 dilution on both days (Figure 3), and mean (SD) NRS score on pure capsaicin homogenate was 8.1 (1.8) on both day 1 and day 2. There was a strong, positive correlation between heat sensation and capsaicin containing homogenate dilution (r = 0.87; p < 0.001). None of the participants terminated their participation because of the experienced heat sensation. Dilution of ≥104 times resulted in no heat sensation. Higher heat sensation was correlated with higher total amount of CGRP release (pg) in the sample (r = 0.50, p < 0.001).
Subjective heat sensation. Mean subjective heat sensation as reported by participants at increasing concentrations of orally applied capsaicin concentrations using a numeric rating scale (range 0–10). Participants perceived heat from the sample that wasdiluted 1000 (103/10e3) times. Error bars indicate ±1 standard error of the mean (SEM).
Discussion
This study shows that oral application of red chili homogenate is well tolerated and causes a dose-dependent salivary CGRP release, without day-to-day effects on this response. Capsaicin (-containing homogenates) have been applied in several ways before in previous studies, including subcutaneously (18), intradermally (17,19), topically (16,20) and orally (24), but this is the first study to quantify the salivary CGRP release. Our data show that there are inter-individual differences both in baseline salivary CGRP content as well as after capsaicin application, indicating that CGRP levels are variable and that salivary CGRP levels increase in response to compounds activating the TRPV1 channel.
Capsaicin, being an irritant for mammals, produces a sensation of burning in any tissue with which it has contact, including buccal mucosa. From previous studies, we know oral application of capsaicin on the tongue and other orofacial tissues elicits burning pain, and increases in blood flow and temperature, but no change in mechanical sensitivity in the glabrous lips or tongue (21). In our study, we show that application of capsaicin concentrations leads to a higher saliva secretion, which is in accordance with previous research that additionally reported higher secretion rates (22). This might be caused by the released CGRP itself, which is known to induce a delayed increase in rat salivary secretion (25). Furthermore, our data suggest that, although total protein content may increase after capsaicin application, the salivary protein composition might not be altered, since at least the proportion of CGRP does not change.
In the follow-up study (with use of single-dose homogenate), we found larger responses in CGRP secretion between vehicle and undiluted chili homogenates compared to the dose-finding study. The smaller CGRP response to the undiluted homogenate in the dose-finding study might be due to the cumulative application of increasing homogenate concentrations. This cumulative exposure to increasing concentration of chili homogenate is likely to have caused a partial depletion of CGRP-containing neurons innervating the salivary glands, and possibly to have induced early desensitization effects (25,26).
CGRP is expressed in subsets of peripheral and central nervous system neurons, including the trigeminal nerve branches that innervate the salivary glands. CGRP mediates the vasodilation part of neurogenic inflammation. Increased levels of CGRP reflect hyperactivation of the trigeminovascular system (TGVS) and subsequent signaling and processing of nociceptive stimuli in the brainstem (27). Since sublingual and submandibular glands are partly innervated by sensory CGRP-containing trigeminal nerves (3–5), levels of salivary CGRP can be used as a biomarker for trigeminal nerve activation (10). The trigeminal nerve is the major sensory nerve of the face with three major divisions (the ophthalmic, maxillary and mandibular) that join at the trigeminal ganglion. Although the three branches innervate different regions of the face, there is evidence in inflammatory disease that activation of one branch can cause activation of the other major branches through a process of sensitization, leading to elevated neuronal activity (28,29). It is, however, unknown, whether this is always the case, since a recent study showed that capsaicin-induced activation of the ophthalmic branch does not result in clinical headache (20). Furthermore, in migraine, referral pain to the lower branches of the trigeminal nerve is rarely reported. Overall, however, the CGRP present in saliva is most likely to originate from the trigeminal nerve endings (5). This mechanism enables us to monitor salivary CGRP changes that reflect pathophysiological changes in other trigeminal branches (10). Increases in salivary CGRP during trigeminal activation, such as is the case in migraine, have been reported before (8–10) and suggest a direct or indirect causal pathway. Detailed insight into the effects of intracranial dural stimulation on salivary CGRP release would be very interesting. Unfortunately, results from such studies are not available.
Limitations of this study include a relatively small sample size. Furthermore, the age homogeneity of our study sample does not allow generalizing results to a younger or elder population. Although the mean increase in CGRP secretion using undiluted homogenate might be small (3.6 CGRP pg/ml saliva; ratio 13.3/9.7 = 1.4), the range in CGRP responses varied between individuals with CGRP concentration changes from −27% to +775%. These data indicate large inter-individual variability in CGRP response, suggesting this secretion could be related to the (patho)physiological state of the individual.
Our study shows that it is possible to study trigeminal CGRP content and depletion with the use of the capsaicin model. Capsaicin stimulation is known to activate sensitive sensory neurons (30), leading to release of neuroactive biochemical substances including CGRP (1). We speculate that the mechanism behind this effect might be the depletion of CGRP-containing vesicles within trigeminal nerve endings. Increased salivary CGRP levels might thus reflect a high activity status of the trigeminal system, i.e. many intraneuronal CGRP-containing vesicles. Capsaicin might also induce CGRP production in the nerve cells, and subsequent release of CGRP in target organs, in this case the salivary glands, leading to higher levels within the saliva. We could speculate that stimulation with capsaicin in this condition would result in a higher salivary volume and a higher CGRP content. The capsaicin model could be used to assess the CGRP loading and functionality of CGRP-containing trigeminal nerve endings innervating the salivary glands, thereby reflecting the activation status of other branches of the trigeminal system (10). We hypothesize enhanced activity of the TGVS is reflected by enhanced CGRP depletion. Further development of analytic techniques may be of interest, especially for diseases in which dysfunction of the trigeminal system is known, for analysis of specific salivary compounds that are released into the saliva. In diseases such as (primary) headache syndromes including cluster headache (31) and migraine (10,31–36), but also in tension-type headache (36), and other diseases, analytical techniques that help qualifying and quantifying salivary (neuro)peptides including CGRP could be useful in elucidating pathophysiologic mechanisms. Especially when combined with measurements that are able to assess functionality of the other branches of the trigeminal nerve (ophthalmic and maxillary) synchronously, salivary CGRP measurements may help in understanding involvement of the trigeminovascular system in a variety of conditions.
In summary, we found that nondiluted chili homogenate can be used as a well-tolerated and effective capsaicin-provocation model to quantitatively measure salivary CGRP, with a good day-to-day reproducibility, which may be helpful in elucidating pathophysiologic mechanisms in diseases in which the trigeminal system is involved.
Clinical implications
Calcitonin gene-related peptide (CGRP) can be found in human saliva. Salivary CGRP is released by salivary glands, partly innervated by efferents of the trigeminal nerve. Oral application of undiluted red chili homogenate causes a dose-dependent CGRP release. The induced salivary CGRP response might reflect the activation state of the trigeminal nerve.
Footnotes
Funding
Dr Antoinette MaassenVanDenBrink was supported by the Netherlands Organization for Scientific Research (Vidi grant 917.11.349).
Conflict of interest
None declared.
Acknowledgement
The authors would like to thank Mrs K Ibrahimi for her help and support with regard to the medical ethics committee approval process.
