Abstract
Sumatriptan-induced changes in plasma calcitonin gene-related peptide (CGRP) concentration and headache intensity were investigated in 19 female migraineurs during nitroglycerin-induced migraine attack. Sumatriptan nasal spray was administered 120 min after the onset of the attack. Blood samples were obtained immediately before and 60 min after sumatriptan administration. In those subjects whose migraine attack improved considerably 60 min after the treatment the plasma CGRP concentration decreased significantly (P < 0.05). In contrast, plasma CGRP concentration failed to change in patients whose headache did not improve. In addition, plasma CGRP concentrations showed significant positive correlations with the headache scores both 60 and 120 min after sumatriptan administration (P < 0.05). According to our results plasma CGRP concentration decreases parallel to headache intensity during sumatriptan treatment and this decrease in CGRP predicts effectiveness of antimigraine drug therapy. This supports that one of the main effects of triptans is to decrease CGRP release.
Introduction
‘Triptans’, highly selective ligands for 5-HT1B/1D receptors are widely used and efficient antimigraine drugs. Sumatriptan, the prototype and first member of the family, causes marked contraction of cerebral arteries (1–4) and blocks neurogenic inflammation and the release of nociceptive neuropeptides (5–8). Based on animal studies, the effects of the triptans have been attributed, at least partially, to the inhibition of endogenous CGRP release (9, 10). Only one human study has monitored, however, the CGRP concentrations during acute migraine. The study monitored 8 patients prior to and post sumatriptan administration (6, 11, 12).
The aim of the present study was to evaluate the effects of sumatriptan on plasma CGRP concentration in peripheral blood circulation with relation to the drug's antimigraine effect. In this study the highly reproducible human migraine model of nitroglycerin-induced headache was used.
Materials and methods
Study subjects
Unrelated migraine patients without aura (mean age: 45 ± 1.4 years) were included in the study. A detailed medical history was taken from each subject, and they underwent complete physical, neurological and psychological examinations and laboratory tests before participating in the project. Patients taking oral contraceptives or antidepressants within 6 months of the study were excluded. The headache diagnoses were made according to the criteria of the International Headache Society (13). None of the subjects were suffering from psychiatric disorders of any kind according to the criteria of the DSM-IV (14). At the time of blood sampling all participants were medication-free for at least two weeks prior to the study and were not following a particular type of diet.
This study protocol was approved by the local ethics committee for experimentation on humans and every subject gave written informed consent form before participating in our research.
Procedure
The subjects received 0.5 mg sublingual nitroglycerin at 0800h. Headache intensity and characteristics were measured every 20 min thereafter for the duration of the observation period (until 1800h). Headache intensity was scored on a verbal scale that measured from 0 to 10, 1 representing a very mild headache including prepain (feeling of pressure), 5 a headache of medium severity and 10 a very severe headache. The subjects were asked to describe their headache after a standardized procedure and side-effects were recorded.
Nineteen patients who developed a typical migraine headache according to the criteria of the International Headache Society (13) were included in the study. A blood sample was obtained 120 min after the onset of the migraine, immediately before the application of sumatriptan nasal spray (Imigran, 20 mg/person, GlaxoSmithKline Ltd, Budapest, Hungary). Venous blood was drawn from an antecubital vein by the Vacutainer® system (Vacutainer is a vacuum test tube that sucks in blood once a needle has punctured a vein). The second blood sample was obtained 60 min after the administration of Sumatriptan nasal spray. Sumatriptan was administered 352 ± 13 min after the nitroglycerin administration.
Subjects were free to withdraw at any time during the study and treatment was offered if needed. None of the subjects whose data is presented here received any other drug treatment before the last blood sampling.
Determination of plasma CGRP concentration
125I-labelled Tyr-α-CGRP(23–37) was prepared in our laboratory (15). CGRP antiserum was provided by Dr T. Gorcs, Semmelweis Medical University, Budapest.
For the determination of plasma CGRP immunoreactivity venous blood samples (3 ml/patient) were taken into ice-cold tubes containing EDTA (7.5% 0.072 ml/3 ml blood) and aprotinin (2700 KIU/3 ml blood). Following centrifugation (1800 g for 15 min at 4°C) the plasma was aspirated and stored at −70°C until plasma CGRP determination. Plasma CGRP concentrations were measured by means of a specific and sensitive radioimmunoassay (RIA) method (16).
Statistical analysis
One-way, two way and repeated measure analyses of variance (
Results
The headache scores and plasma CGRP concentrations of the two patient groups are shown in Table 1. The first group (n = 6) consists of the patients whose headache improved at least 30%, 60 min after the sumatriptan nasal spray. In this group the decrease in headache intensity was significant (Friedman
Headache scores and plasma CGRP concentrations, in two patient groups, before and 60 min after the administration of sumatriptan. The first group (n = 6) contains the data of those patients whose headache scores improved by at least 30%, 60 min after the sumatriptan nasal spray. The second group (n = 13) contains the data of those patients whose headache scores did not improve by at least 30%, 60 min after the sumatriptan nasal spray
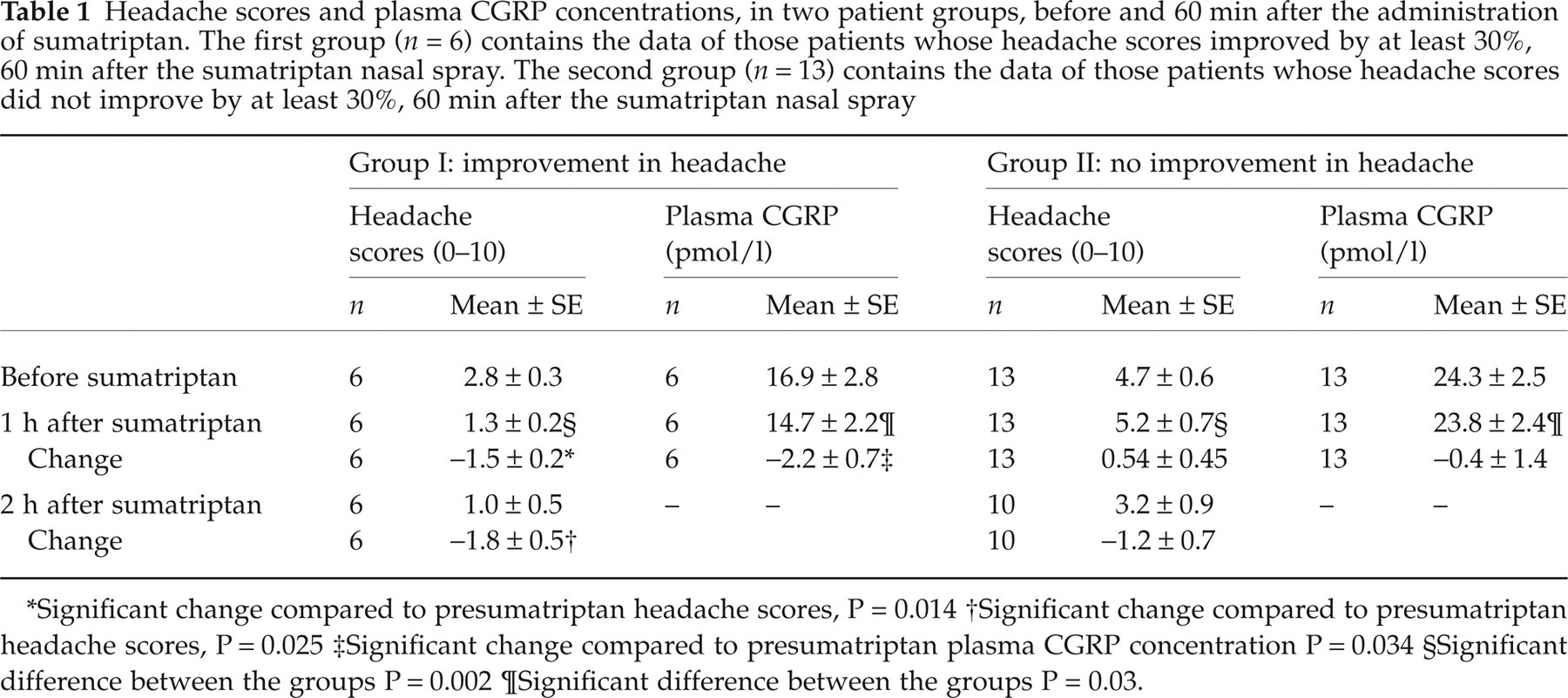
Significant change compared to presumatriptan headache scores, P = 0.014
Significant change compared to presumatriptan headache scores, P = 0.025
Significant change compared to presumatriptan plasma CGRP concentration P = 0.034
Significant difference between the groups P = 0.002
Significant difference between the groups P = 0.03.
Plasma CGRP concentration decreased significantly in those patients whose headache improved at least 30% (F = 8.39, d.f. = 1,5, P = 0.034) after sumatriptan administration. In contrast, there was no significant change in those patients whose headache did not improve at least 30% after sumatriptan administration (F = 0.09, d.f. = 1,12, P = 0.76).
When data from all of the patients were included in the analysis, plasma CGRP concentrations showed significant positive correlations with the headache scores, 60 min after sumatriptan administration (Spearman R = 0.580, n = 19, P < 0.01). Plasma CGRP concentrations obtained immediately before the administration of sumatriptan showed a positive trend with the headache scores (Spearman R = 0.415, n = 19, p = 0.077). Furthermore, plasma CGRP concentrations, obtained 60 min after sumatriptan, showed positive correlation with headache scores measured 60 min after the second blood sampling, that is 120 min after the administration of sumatriptan (Spearman R = 0.500, n = 16, P = 0.049).
Discussion
The nitric oxide (NO) donor nitroglycerin causes delayed headache in migraineurs that fulfils The International Headache Society criteria for migraine (17–21). NO induced migraine, such as spontaneous migraine, can be alleviated by antimigraine drugs (6, 22–24). Our study is the first to provide data about changes in plasma CGRP concentration during sumatriptan treatment of nitroglycerin-induced migraine attack.
Calcitonin gene-related peptide (CGRP) is a 37 amino-acid neuropeptide. It is a potent vasodilator and is also involved in pain sensations. The CGRP-containing nerve fibers of the trigeminal ganglia are activated in primary headaches. Several primary headaches, such as migraine, cluster and chronic paroxysmal hemicrania are associated with increased CGRP concentration in the jugular vein during pain attack (23). Furthermore, plasma CGRP concentration in the antecubital vein increases by 25–30% during nitroglycerin induced migraine attack and increase in the CGRP concentrations correlates markedly with the migraine headache intensity (21).
Our results demonstrate that sumatriptan decreases the peripheral plasma CGRP concentration during migraine attack. This decrease is present only in those patients whose headache shows parallel improvement. Furthermore, a more complete reduction of headache occurs in these patients 2 h after sumatriptan. These data mean that decrease in CGRP precede and thus, predict effectiveness of sumatriptan. In those patients whose headache does not improve, plasma CGRP concentrations fail to show a decrease after the administration of sumatriptan. Furthermore, plasma CGRP concentrations significantly correlate with headache scores after sumatriptan treatment. These results underline the causative relationship of CGRP release and migraine headache (25).
CGRP can be released in experimental models by NO-mediated mechanisms which cause morphological changes to the CGRP-immunoreactive peripheral dural afferents (26). It has been shown that 5-HT1B and 5-HT1D receptors are expressed in the human trigeminal ganglion and they are colocalized with CGRP and nitric oxide synthase (10). Activation of these receptors decreases CGRP release from the trigeminal perivascular axons (4, 5, 7, 8, 27–31). On the other hand, sumatriptan, which poorly passes through the blood–brain barrier, cannot prevent the activation of c-fos expression in interneurones of the caudal trigeminal nucleus (7) and thus, the effect of the drug is mediated mainly through peripheral mechanisms. This mechanism is probably independent of the vasoconstrictor effect, because cerebral artery blood flow velocity failed to show any correlation with headache scores (3). In another study, rizatriptan also abolished increasing level of CGRP during migraine attack (32). These data, together with our findings, suggest that sumatriptan’s, rizatriptan's and probably all triptan's main effect in alleviating migraine pain is mediated through the decrease in CGRP release from perivascular axons.
In conclusion, our results show that plasma CGRP concentration decreases parallel to headache intensity during antimigraine drug therapy and this decrease in CGRP predicts effectiveness of treatment. These results support the hypothesis that one of the main effects of triptans is to decrease CGRP release.
Footnotes
Acknowledgements
The authors would like to thank Sandorne Nagy and Ilona Ozorocyzne Szasz for assistance in blood sampling. The authors are indebted to Sophie Ryde for language corrections. The study was supported by the Hungarian Research Fund Grant T022256/1997, T032398/2000, T-039411, T-043467, TS-040753, NRDP: 1/047/2001, Ministry of Health, Social and Family Affairs Grant ETT 058/2003 and 03597/2003 and Postdoctoral Ph.D. Fellowship Program (G.J., 1999–2002) of the Semmelweis University, Ministry of Culture and Education, Hungary. Nemeth J was supported by the Istvan Szechenyi Fellowship. Sumatriptan nasal spray (Imigran) was graciously supplied by Glaxo Wellcome Ltd.
