Abstract
Aim
The aim of this article is to explore the progression of neurological, neuro-otological and cochlear features in benign paroxysmal vertigo (BPV) in children over time and its relation with migraine, neuro-otological and cochlear disorders in adulthood.
Methods
From January 2002 to December 2002, 15 children with BPV were prospectively recruited and then evaluated during a 10-year observational follow-up. All patients underwent detailed neurological, neuro-otological and cochlear examinations during interictal phases. Six children were also studied during ictal periods.
Results
At first assessment, four children reported migraine with aura (MwA) and six children reported migraine without aura (MwoA). Neuro-otological examinations were abnormal in two of 15 children. Cochlear examinations were normal in all patients. During the 10-year follow-up, recurrent vestibular symptoms and/or MwA and/or MwoA have been observed in the children. Neuro-otological examinations were abnormal in three of 15 individuals during the interictal period, and abnormal in four out of six patients who were studied during the ictal period. Two patients developed cochlear signs and/or symptoms.
Conclusions
During the 10-year follow-up, a phenotype variability in BPV patients has been observed. Specifically, de novo cochlear signs and/or symptoms developed in children with BPV, suggesting that cochlear symptoms should be properly investigated in these patients over time.
Introduction
Migraine-related symptoms in childhood are a group of heterogeneous periodic and paroxysmal neurologic disorders (1). Among these, cyclic vomiting, abdominal migraine and benign paroxysmal vertigo (BPV) have already been previously defined by the International Headache Society (IHS) criteria as childhood periodic syndromes that are usually precursors of migraine (2,3). Recently, the International Classification of Headache Disorders (ICHD-3 beta version) (4) has replaced the “periodic symptoms” terminology with “episodic symptoms which are associated with migraine” (1.6) and that will no longer be limited just to childhood.
BPV is the most frequent cause of paroxysmal vertigo in children and it is characterized by brief and recurrent attacks of incapacitating vertigo and postural imbalance occurring without warning and resolving spontaneously after minutes to hours in otherwise healthy children. These episodes are usually accompanied by vegetative symptoms such as paleness, sweating and nausea (5). However, BPV is currently underdiagnosed because of clinical manifestation heterogeneity and its pathophysiology is still not fully understood (5,6). Some authors have reported the presence of vestibular abnormalities in children with BPV (7,8), whereas others have not confirmed these findings (8,9). Furthermore, although considered to be “benign,” several studies have evidenced evolution of BPV to migraine and/or recurrent vestibular symptoms in long-time follow-up studies (10–12). In other terms, BPV in children may represent an early life disorder with a genetic predisposition to migraine that could be expressed later in life (1,13). However, there is still a paucity of data on the evolution of migraine or migraine-related symptoms starting in childhood, because there have been few prospective studies evaluating children with BPV.
To explore prospective progression of neurological, neuro-otological and cochlear features in children with BPV over time and its relation with migraine, neuro-otological or cochlear disorders in youth and adulthood, we have performed a 10-year observational follow-up study. Specifically, we hypothesized that: a) BPV is a migraine equivalent and/or precursor that could evolve into migraine and/or recurrent vestibular symptoms for a large number of patients; and b) cochlear dysfunction could be detected in some of these children.
Materials and methods
Eighteen consecutive children (11 female, seven male; aged 4–11 years, mean age ± SD: 7.12 ± 5 years) with BPV diagnosed according to ICHD-I criteria (2) as well as ICHD-II (3) and the more recent ICHD-III (beta version) (4) were prospectively recruited from the population referring to the Audiology and Vestibology Unit of the Department of Neuroscience of the University of Naples from January 2002 to December 2002. Fifteen children with BPV have been evaluated during the 10-year follow-up study while three performed only the first evaluation and were lost at follow-up because of family reasons.
An extensive neurological examination was performed at the first visit and once a year by a “headache expert” neurologist. In all children, neurological examinations were always negative. Headache diagnoses have been reached according to ICHD-I criteria (2).
Detailed neuro-otological and cochlear (pure-tone audiometry) evaluations were performed at the first visit and every six months. Otherwise, the children’s parents were instructed to ask for a clinical expert consultation at any moment, if necessary. Thus, some children were also evaluated during migraine headache or vertigo spells.
The neuro-otological test protocol, performed with infra-red videonystagmography (Ulmer VNG, Marseille, France), included: observation of spontaneous (patient in the upright position) and positional (patient in the supine, in head and body left side, in head and body right side, and in supine with head hanging in the center position) nystagmus without and with visual fixation; positioning nystagmus (Dix-Hallpike and Pagnini-McClure tests) (14,15); post-horizontal head-shaking nystagmus (16); head impulses in the horizontal plane (17); horizontal and vertical smooth pursuit; horizontal and vertical saccades; gaze-evoked nystagmus in eccentric positions in light. Spontaneous and/or positional nystagmus with a slow-phase eye velocity (SPEV) below three degrees/s was considered insignificant and was not further analyzed.
A peripheral vestibular dysfunction (PVD) was assumed when at least one of the following findings was present: 1) direction-fixed mixed horizontal torsional spontaneous nystagmus inhibited by visual fixation with abnormal head impulses in the horizontal plane; 2) direction-fixed mixed horizontal torsional positional nystagmus inhibited by visual fixation; 3) normal vestibulo-ocular reflex (VOR) suppression.
A central vestibular dysfunction (CVD) was assumed when at least one of the following findings was present: 1) purely horizontal, vertical or torsional spontaneous or positional nystagmus not inhibited by visual fixation; 2) direction-changing nystagmus; 3) down-beating nystagmus after horizontal head shaking (dbHS); 4) gaze-evoked nystagmus; 5) impaired VOR suppression; 6) slow or dysmetric saccades; 7) saccade pursuit.
Cochlear function was performed by pure tone audiometry with air (frequencies between 125 and 8000 Hz) and bone conduction (frequencies between 250 and 4000 Hz) (Amplaid 131).
Children’s clinical characteristics at first evaluation.
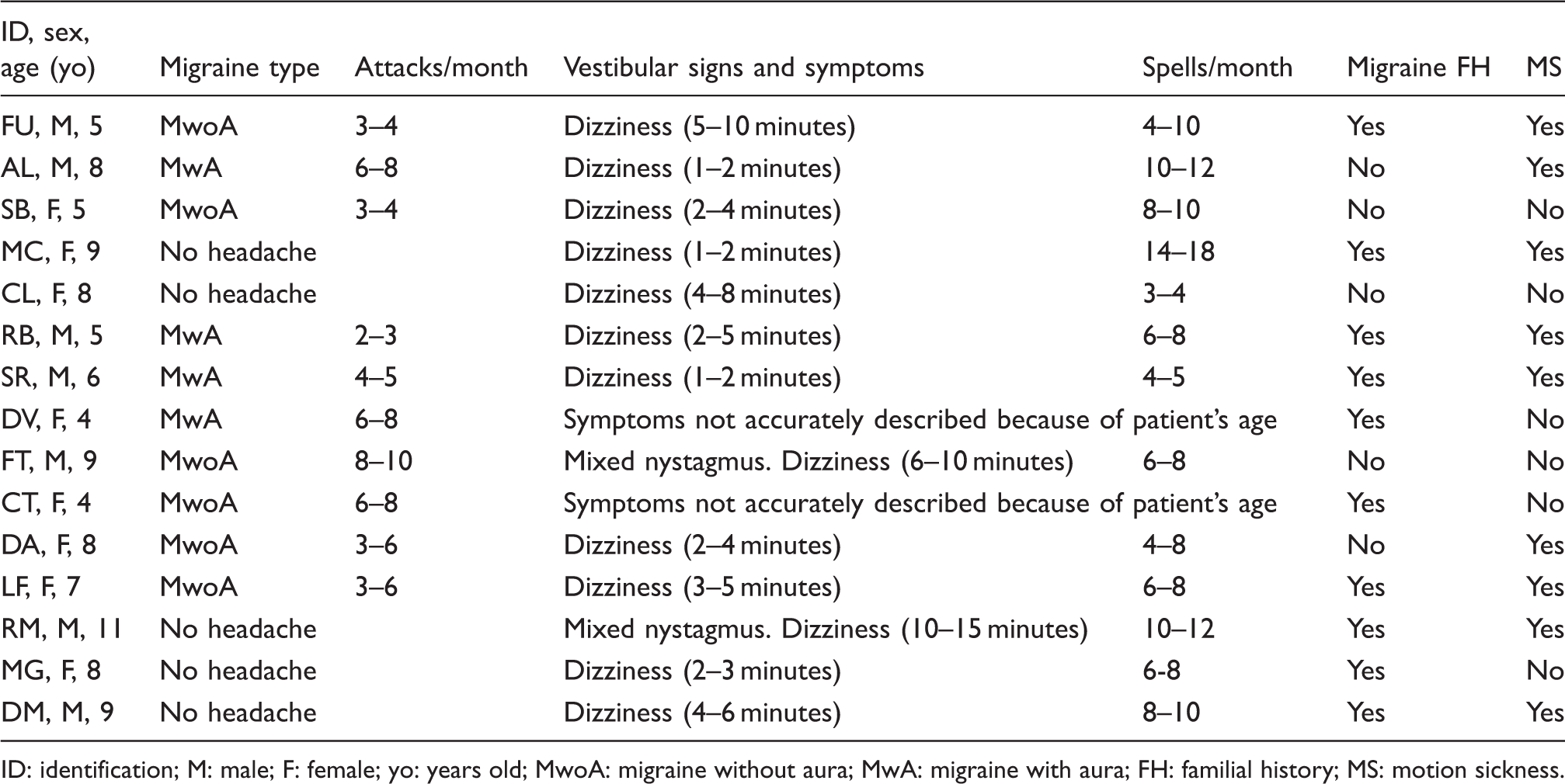
ID: identification; M: male; F: female; yo: years old; MwoA: migraine without aura; MwA: migraine with aura; FH: familial history; MS: motion sickness.
All children underwent a preliminary brain magnetic resonance imaging (MRI) scan before entering the present study to exclude any structural abnormalities.
Results
At the first clinical assessment, all children were diagnosed with BPV according to ICHD-I criteria (1). Among these, six children met ICHD-I criteria for migraine without aura (MwoA) and four children for migraine with aura (MwA). Five children had never experienced headache in their life. Only two of 15 children showed neuro-otological abnormalities in the form of positional nystagmus: One child showed a mixed horizontal torsional nystagmus, beating to the left and with the top pole beating toward the left ear in supine and in head and body right-side position (lbPN), inhibited by visual fixation (PVD related). Similarly, the other one showed nystagmus in supine, in head and body right side and in supine with head hanging in the center position (lbPN), inhibited by visual fixation (PVD related). Cochlear examinations were normal in all patients. During follow-up, in all children, neuro-otological and cochlear examinations were performed several times (from 12 to 19 evaluations) in the interictal periods. Neuro-otological evaluations were normal in 12 children (80%) and abnormal in three children (20%). In one child, in two of 12 examinations we found a mixed horizontal torsional nystagmus, beating to the right and with the top pole beating toward the right ear in supine and in head and body right-side position (rbPN), inhibited by visual fixation (PVD-related). In the second child a mixed horizontal torsional nystagmus, beating to the left and with the top pole beating toward the left ear in supine and in head and body right-side position (lbPN), inhibited by visual fixation (PVD-related) was detected in one of 18 examinations and a dbHS (CVD related) in two of 18 examinations. In the third child, a mixed horizontal torsional nystagmus, beating to the right and with the top pole beating toward the right ear in supine and in head and body left-side position (rbPN), inhibited by visual fixation (PVD related) was detected in one of 16 examinations and a down-beating nystagmus after horizontal head shaking (dbHS) (CVD related) in two of 16 examinations. In six children neuro-otological and cochlear examinations were also performed in the ictal phase (during headache or vestibular symptoms). In this context, neuro-otological evaluations were abnormal in four of six children (67%). Indeed, in one child we found a mixed horizontal torsional nystagmus, beating to the right and with the top pole beating toward the right ear in supine and in head and body right side position (rbPN), inhibited by visual fixation (PVD-related) and a dbHS (CVD related) in the one examination carried out during a vertigo spell. In two children, in one of two examinations performed during a dizziness spell, a mixed horizontal torsional nystagmus, beating to the left and with the top pole beating toward the left ear in head and body right-side position (lbPN), inhibited by visual fixation (PVD related) and a slow pursuit in the horizontal plane (CVD-related) were found. In one child, in two out of three different examinations performed during migraine headache attacks, a dbHS (CVD related) and a slow pursuit in the horizontal and vertical planes (CVD related) were detected. During both interictal and ictal periods, positional nystagmus (found in five children) and dbHS (found in three children) were the most frequent neuro-otological findings.
During the 10-year follow-up, 10 of 15 children (67% of the sample) reported the following symptoms: MwA (two of 15 patients), MwoA (one of 15 patients), recurrent vestibular symptoms (three of 15 patients), MwA and recurrent vestibular symptoms (two of 15 patients) or MwoA and recurrent vestibular symptoms (two of 15 patients).
Children’s clinical characteristics during follow-up evaluation.
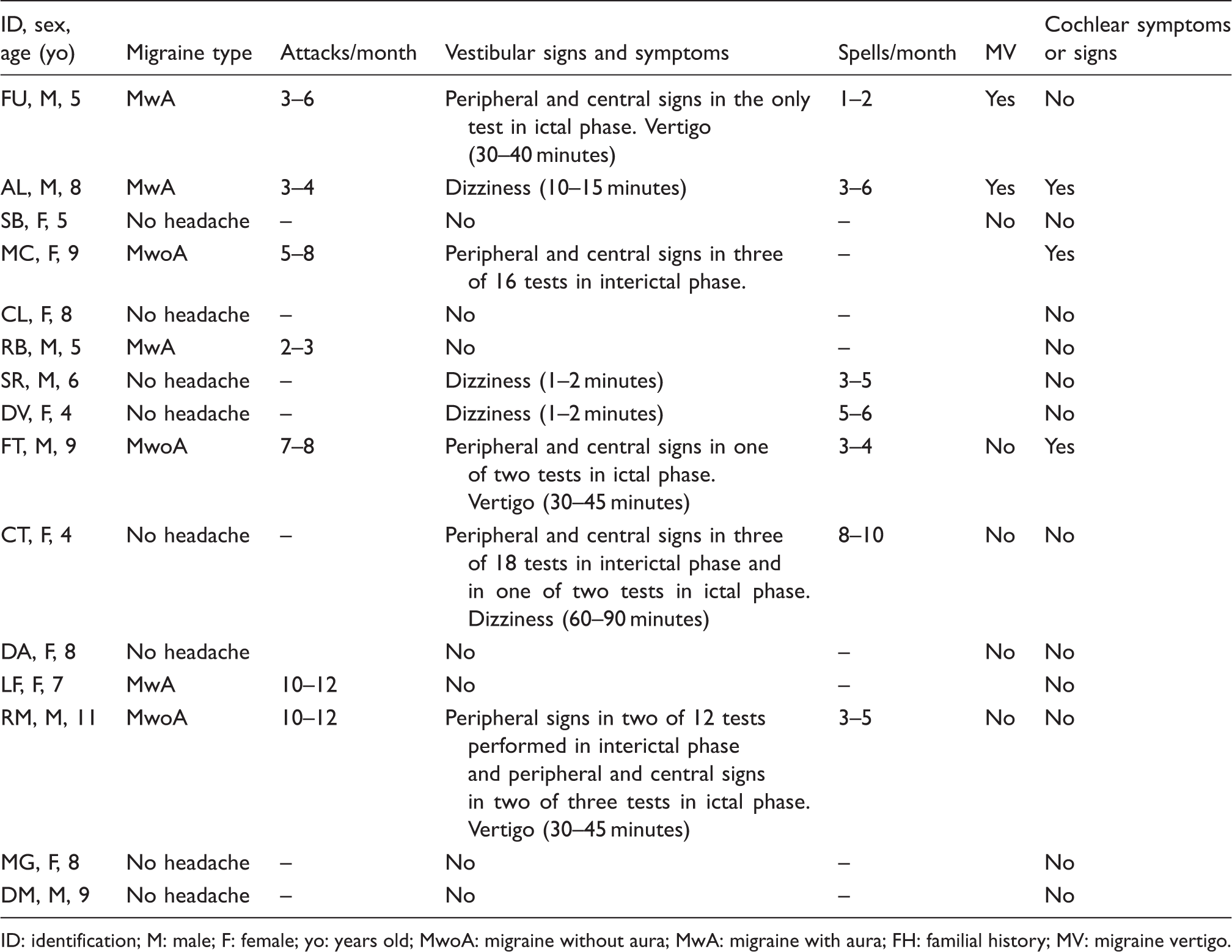
ID: identification; M: male; F: female; yo: years old; MwoA: migraine without aura; MwA: migraine with aura; FH: familial history; MV: migraine vertigo.
Regarding pharmacological therapy, seven patients used migraine-acute and -preventive medications, such as oral nonsteroidal anti-inflammatory drugs (NSAIDs) on an as-needed basis and oral flunarizine (5 mg/day; from three to six months, in most cases). The remaining patients used migraine-acute medications on an as-needed basis (oral NSAIDs).
Discussion
Herein, we report a prospective study exploring neurological, neuro-otological and cochlear signs and symptoms in children with BPV over a period of 10 years. During the follow-up period, we observed that two-thirds of patients with BPV reported onset or worsening migraine and/or recurrent vestibular symptoms. Neuro-otological examinations were normal during the interictal period for the majority of children, whereas they were abnormal in four of six children evaluated during migraine attacks, vestibular or dizziness spells, even without clinically evident balance issues. Positional nystagmus (e.g. suggesting peripheral vestibular system involvement) and dbHS (e.g. indicating central vestibular system involvement) were the most frequent neuro-otological signs. All children with MwA and two-thirds of those with MwoA continued to suffer from migraine and/or vestibular symptoms at the end of the study. Cochlear signs and symptoms were observed in two of seven children who developed migraine.
Since its first description (7), BPV has been considered as an early migraine manifestation or equivalent in childhood (8–10), and several studies have supported this hypothesis (18–20). In a retrospective study, it has been observed that in children with BPV migraine prevalence is higher than in the general population (21). However, only few studies have evaluated children with BPV for a sufficiently long time (22,23). In our follow-up study, an evolution or progression of symptoms has been observed in several children with BPV. Our findings are consistent with previous follow-up studies (24,25), showing a rate of headache remission from 22% to 34% of children with migraine.
Conversely, our results are not in line with other previous studies (26,27) showing a significantly higher BPV remission rate. The observed epidemiological discrepancies could be related to complexity of BPV diagnosis due to children’s difficulties in accurately describing the feelings of “vertigo” or “dizziness” (28,29). Furthermore, BPV might be under-recognized because of low sensitivity according to the IHS criteria. Indeed, these criteria include symptoms shared by other common childhood disorders (4,5). Finally, a problem further complicating BPV studies is the presence of different results depending on whether they originate from pediatric, otolaryngology or neurological departments. However, it is well known that patients with migraine (MwA or MwoA) are still most likely to suffer both from symptomatic and/or “silent” vestibular pathways involvements in the course of attacks over time (30). BPV pathophysiology is not completely understood and it is a matter of ongoing research. Nevertheless, our findings could be explained by means of different migraine mechanisms involving both the peripheral and central vestibulo-cochlear systems. These mechanisms include vestibular afferents modulation by neurotransmitters released during migraine attacks (31), transient ischemia of brainstem nuclei and vestibular and/or auditory pathways (31) as well as effects of “cortical spreading depression” or white matter microstructural abnormalities in the posterior circulation territory (31,32). A critical role of the brainstem and the migraine pathophysiological mechanisms might be argued observing that migraine is frequently preceded by a history of BPV as well as motion sickness, cyclic vomiting and recurrent abdominal pain (4,6,9).
To our knowledge, similar findings have rarely been reported in children with BPV. This is because of the difficulty in designing studies that consider the transition from childhood to adulthood through adolescence (22,23). However, the present study is not free from some limitations. First of all, the patient sample is too small to determine whether a specific BPV clinical feature could predict or affect symptom evolution. Secondly, neuro-otological and audiological assessments have not been conducted during the ictal phases in all children, limiting our knowledge about differences between ictal and interictal vestibular and audiological phenomena in only a few children with BPV, over time. Nevertheless, our data suggest that BPV in children could not exhibit a “self-limiting” evolution, nor as migraine. Indeed, BPV may evolve in different migraine or vestibular phenotypes. Furthermore, our findings allow us to assume that the cochlear system may be involved in children with BPV, though in a minority. Interestingly, a phenotype variability in the patients has been observed over time. For example, during the follow-up period, we have observed that among the children with migraine, some of the patients with MwA became patients with MwoA or vice versa. Similarly, among the children with vestibular symptoms, some experiencing dizziness switched to vertigo and vice versa. Also, some children without migraine at first observation, later developed migraine. Furthermore, a close correlation between migraine, neuro-otological signs and vestibular symptoms has not been observed: For example, children with or without migraine exhibited neuro-otological signs but not vestibular symptoms and vice versa.
Therefore, we suggest that BPV could be considered as a vestibulo-cochlear disorder with a migrainous etiology that may cause neuro-otological and cochlear signs and symptoms over time, as described for vestibular symptoms in vestibular migraine (33). Mechanisms underlying vestibular disorders such as BPV, vestibular migraine and motion sickness remain still widely unknown. We cannot exclude that hyper-sensitivity of the vestibular system due to the hereditary liability (similar to hypersensitivity to light or to sound in migraine) could represent a common functional pathway of different but correlated neuro-otological abnormalities. Moreover, sensitivity to estrogens could likely play a critical role in eliciting vestibular symptoms in migraine by common pathophysiological mechanisms underlying both migraine and vestibular multisensory control and compensation.
We speculate that hormonal changes from childhood to adolescence may lower the threshold of vestibular pathways activity both in children and in women with migraine. This may partially explain the increase of migraine and vestibular disorders predominance in female adult patients (31–36).
Based on the importance of diagnosing BPV in childhood in time for the limiting of unnecessary pain, the reducing of disability as well as avoiding inappropriate therapies or delaying the initiation of appropriate therapy, we suggest that BPV investigation should include a detailed history, clinical and neurological examination, and a complete neuro-otological and cochlear evaluation over time. Specifically, based on our findings, we suggest that ICHD-III criteria (4) of BPV (1.6.2) assuming a “normal neurological examination and audiometric and vestibular functions between attacks” could be sometimes, although rarely, confounding and, in turns, they should not be mandatory for the diagnosis. In our opinion, the above-mentioned criterion could be slightly modified as follows: “normal neurological examination between attacks” and “abnormal audiometric and vestibular functions between attacks do not exclude the diagnosis.” Therefore, in future studies, to better understand the correlation between migraine and vestibular pathophysiological mechanisms, it would be interesting to evaluate possible or preferential relationship between BPV in children and vestibular migraine in adulthood.
Conclusion
BPV is a common cause of vertigo in children, with a clinical phenotype characterized by non-specific or absent vestibular signs. Our data underline the importance of improving the understanding of BPV phenotypic evolution from childhood to adulthood through adolescence and its prospective prognostic factors. Indeed, it may be clinically relevant to establish whether a specific BPV phenotype could likely evolve into a different migraineous disorder or disappear. Finally, we suggest that the involvement of the cochlear as well as vestibular system in BPV in childhood can improve our knowledge on the pathophysiology of episodic symptoms that are associated with migraine, enlarging at the same time our view of different possible migraine phenotypes. Future studies testing a greater sample of patients are warranted to confirm our observations.
Clinical implications
Benign paroxysmal vertigo (BPV) is a migraine equivalent requiring a careful clinical follow-up because it could evolve into migraine (with or without aura), associated with vestibular disorders such as dizziness or vertigo in more than half of the patients. Interestingly, from both a pathophysiological and clinical point of view, cochlear symptoms should be properly investigated in these individuals. Of the 15 patients with BPV followed for 10 years, two-thirds reported onset or worsening migraine and/or recurrent vestibular symptoms. Neuro-otological examinations were abnormal in three of 15 individuals during the interictal period, and abnormal in four of six participants during the ictal period. Positional nystagmus and down-beating nystagmus after horizontal head shaking were the most frequent neuro-otological signs. Two patients developed cochlear signs and/or symptoms.
Ethics or institutional review board approval
The Regional Board of Medical Ethics at the University of Naples considered approval unnecessary as the observational study was limited only to the periodic evaluation of such patients as occurs in everyday clinical practice. All parents gave their informed consent to the study.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict or interest
None declared.
Acknowledgements
The authors thank Dr Antonio Giannone, Dr Gennaro Russo and Dr Enrico Marano for help with this paper.
