Abstract
Background and purpose
Headache may be a warning sign of subsequent stroke in patients with vertebral artery dissection (VAD). Even though the headache characteristics of VAD have been described predominantly in patients with extracranial VAD and neurological complications, headache semiology is not well known in patients with uncomplicated intracranial vertebral artery dissection (ICVAD). In the present study, we attempt to identify the headache semiology that characterizes ICVAD and validate the revised version of the International Classification of Headache Disorders (ICHD-3 beta) criteria for headache attributed to intracranial artery dissection.
Methods
Six patients with neurologically uncomplicated ICVAD presented at a participating medical center, and eight similar patients were reviewed in the literature. Combining these data, we analyzed headache characteristics of patients with uncomplicated ICVAD according to their pain onset and duration, nature, intensity, location, aggravating and relieving factors, associated symptoms, response to medication, and prognosis.
Results
Headache in uncomplicated ICVAD usually has an acute mode of onset (11/14) and persistent (10/14) temporal feature. Pain that has a throbbing quality (nine of 14) and severe intensity (13/14) on the ipsilesional (10/14) and occipitonuchal area (12/14) is a headache prototype in ICVAD. Additionally, headache was intensified by head flexion and rotation (three of six), and relieved by head extension and supine positioning (five of six). Headache of all patients in the present study fulfilled the ICHD-3 beta criteria.
Conclusion
Headache semiology of uncomplicated ICVAD is mostly homogenous in the present study. These characteristics may be helpful in the diagnosis of uncomplicated ICVAD.
Keywords
Introduction
Vertebral artery dissection (VAD) is one of the leading causes of stroke in young adults (1,2). Among the various neurological signs and symptoms of VAD, pain is known as the most common initial symptom (3). However, pain in VAD is not necessarily accompanied by cerebrovascular complications (4). Therefore, early recognition of pain attributed to VAD may be important, especially given the potential serious complications associated with VAD, such as infarction or subarachnoid hemorrhage (SAH). Previously, dissection has been reported as occurring frequently at extracranial (V2 and V3) segments (5), and pain has been described mostly in patients with extracranial VAD (ECVAD) (6–8).
An intracranial segment (V4) of the vertebral artery is also known to be vulnerable to dissection (9). However, characteristics of intracranial VAD (ICVAD)-induced pain, which may manifest primarily as headache, have been described in only a limited number of cases (10). This may be attributed to the following: 1) Most in-patients with ICVAD are neurologically complicated (3), which means that physicians are more likely to focus on serious neurological complications and ignore detailed headache semiology; and 2) several compounding effects such as mass effect in a large stroke, medical complications, and concomitant drugs used in treatment also may obscure headache characteristics in ICVAD. Therefore, in order to disclose the characteristics of headache attributable to ICVAD, it may be necessary to evaluate patients with ICVAD prior to the development of neurological complications.
Recently, the International Headache Society released a revised version of the International Classification of Headache Disorders (ICHD) criteria (11). According to the ICHD-3 beta criteria, headache attributed to intracranial artery dissection was defined through a causal relationship between intracranial artery dissection and headache (criterion C1) and headache semiology (criteria C2–4). Although uncomplicated ICVAD is not representative of intracranial dissection as a whole, it may be a potentially useful candidate to evaluate ICHD-3 beta criteria for headache attributed to intracranial artery dissection.
Based on the above, we describe patients with uncomplicated ICVAD who presented with headache alone. Through additional review of the literature, we try to identify the headache characteristics of ICVAD itself. Finally, we evaluate ICHD-3 beta diagnostic criteria for headache attributed to intracranial artery dissection.
Patients and methods
Patients
From January 2011 to December 2013, we recruited six patients diagnosed with uncomplicated ICVAD who presented at Korea University Ansan Hospital. Diagnosis of uncomplicated ICVAD was based on the following criteria: 1) lack of neurological signs, 2) lack of parenchymal lesions on magnetic resonance (MR) imaging, including diffusion-weighted imaging, and 3) the presence of either intimal flap or double lumen on vertebral artery angiography, or dynamic changes of angiographic findings with pearl and/or string sign, tapered occlusion, or intramural hematoma (10). The study protocol was approved and supervised by the institutional review board of Korea University Ansan Medical Center (AS13178).
Case reports
Patient 1
A 51-year-old female visited our outpatient clinic for headache for five days’ duration in October 2013.
Patient history
The patient had migraine without aura, mostly developed in the frontotemporal area, which responded well to acetaminophen.
Index headache
She had no preceding infection or trauma in relation to the current headache. The pain developed suddenly and reached maximum intensity over one day. It was located in the right occipital and high cervical regions, and radiated to the ipsilateral temporoparietal region. The intensity was severe (visual analog score, VAS = 7), and the nature was mainly pulsatile and intermittently stabbing. The pain was aggravated by physical activity and head flexion and rotation, and was relieved by head extension and supine positioning. The patient reported accompanying nausea, but not photophobia or phonophobia. The pain persisted all day despite use of talniflumate and muscle relaxants. Therefore, the index headache differed considerably from the prior migraine in location and response to medication.
Evaluation
There was focal tenderness on the right suboccipital area, which prevented head motion. MR imaging performed 10 days after the onset of the headache revealed a focal narrowing of the right intracranial vertebral artery and eccentric intramural hematoma. Follow-up imaging 20 days after headache onset showed complete occlusion of the vertebral artery (Figure 1 Serial angiographic features of intracranial vertebral artery dissection (ICVAD) in patients 1 and 2. (Patient 1, left) Initial MRA showed a flame-shaped stenosis with post-stenotic dilation in the right ICVA (arrow head, upper panel). Stenosis had progressed and the index artery was occluded, as demonstrated by sequential MRA (middle panel) and CTA (lower panel) performed five and 10 days after initial MRA. (Patient 2, right) Initial MRA showed a focal stenosis in the left ICVA (arrow head, upper panel). T1-weighted MRI revealed eccentric wall enhancement at the stenotic segment of the ICVA (arrow, middle panel). Follow-up CTA performed three months after initial MRA demonstrated an improved stenotic segment. MRA: magnetic resonance angiography; MRI: magnetic resonance imaging; CTA: computed tomographic angiography; ICVA: intracranial vertebral artery.
Treatment and prognosis
The patient received an antiplatelet agent (100 mg of acetyl salicylic acid per day) for vertebral artery dissection and propranolol (40 mg per day) and naproxen sodium (1 g per day) for pain control. Though there was some fluctuation, the intensity of headache slowly decreased and disappeared 50 days from onset. Follow-up until April 2014 demonstrated no neurological complications.
Patient 5
A healthy 36-year-old male visited our center for headache for five days’ duration during November 2013.
Patient history
The patient had a history of migrainous headache, characterized by unilateral, pulsating pain in the frontotemporal area, moderate intensity (VAS = 5–6), a duration of six to 12 hours, associated gastrointestinal symptoms, and augmentation upon physical activity. His previous headaches occurred spontaneously or with alcohol consumption and responded well to analgesic medication.
Index headache
He received a neck massage during a night of heavy drinking, and the headache developed at dawn the next day. Initially, the pain progressed over 12 hours. It was severe (VAS = 8) left occipitonuchal pain radiating to the ipsilateral hemicranium. The nature was mostly pulsating and intermittently stabbing. He denied augmentation of pain during physical activity and neck motion, but the pain was partially relieved by neck extension. There were no associated symptoms such as photophobia, phonophobia, or gastrointestinal discomfort. He took an over-the-counter (OTC) pain reliever before visiting our clinic, but the pain was not controlled. Therefore, the index headache was completely different from previous migraine in location, intensity, and response to medication.
Evaluation
Neurologic examination was normal at presentation. MR angiography performed seven days after headache onset demonstrated an intimal flap and unnaturally dilated left intracranial vertebral artery (Figure 2 Initial angiographic findings of intracranial vertebral artery dissection (ICVAD) in patients 5 and 6. (Patient 5, left) MRA showed aneurysmal change with intimal flap (inserted figure) in the left ICVA. (Patient 6, right) Pseudolumen was revealed by MRA source imaging (inserted figure) in the stenotic left ICVA. MRA: magnetic resonance angiography; CTA: computed tomographic angiography; ICVA: intracranial vertebral artery.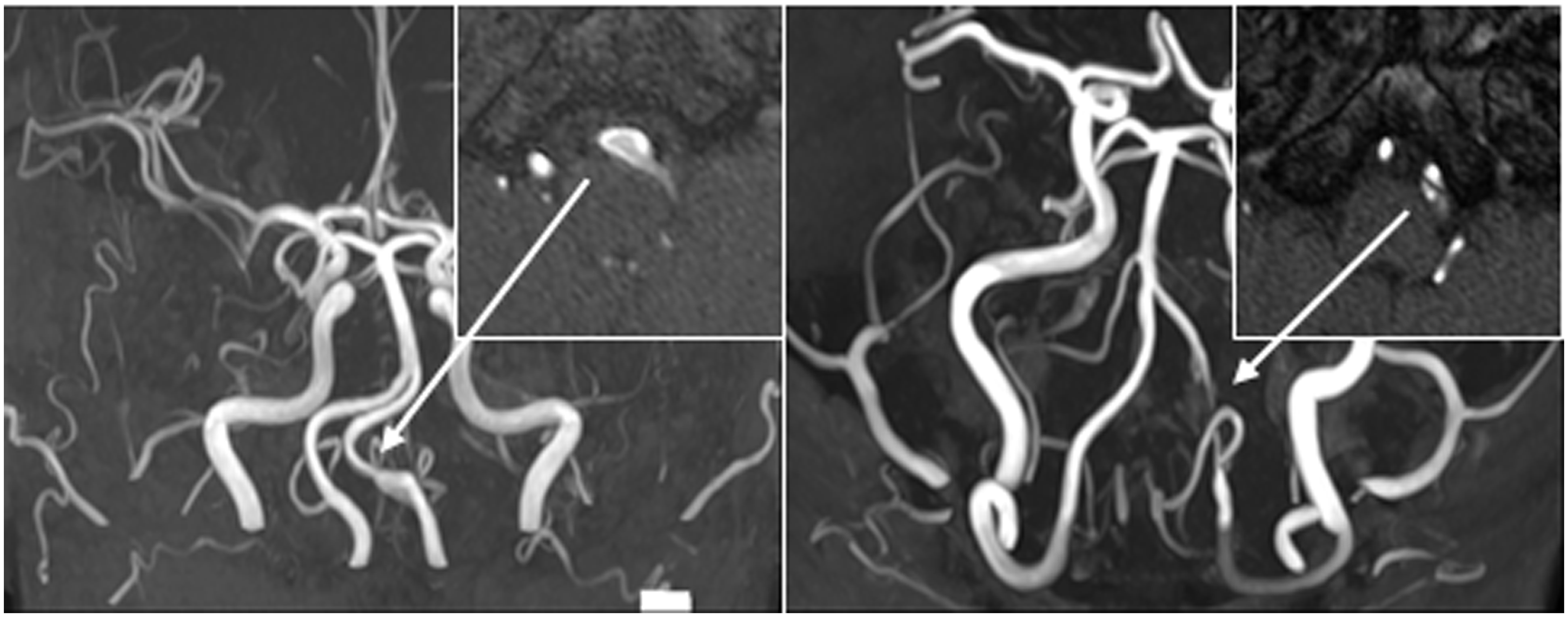
Treatment and prognosis
The patient was administered an antiplatelet agent. His headache was partially improved (VAS = 5) by 20 mg of propranolol and 500 mg of naproxen twice a day, and gradually disappeared over two months. He had no neurological complications during the follow-up period, up to April 2014.
Other patients
Case reports of the remaining four patients with uncomplicated ICVAD (patients 2–4, and 6) are described in the online Supplementary Material.
Serial angiographic findings of intracranial vertebral artery dissection (ICVAD) in patients 3 and 4 (patient 3, left; patient 4, right). Initial MRA (upper panel) revealed luminal narrowing in the left ICVA in both patients. MRA source imaging showed pseudolumen at the stenotic segment (inserted figure), which indicated arterial dissection. Follow-up MRA performed three months after initial evaluation (lower panel) indicated complete recovery of the dissected artery in both patients. MRA: magnetic resonance angiography; CTA: computed tomographic angiography; ICVA: intracranial vertebral artery.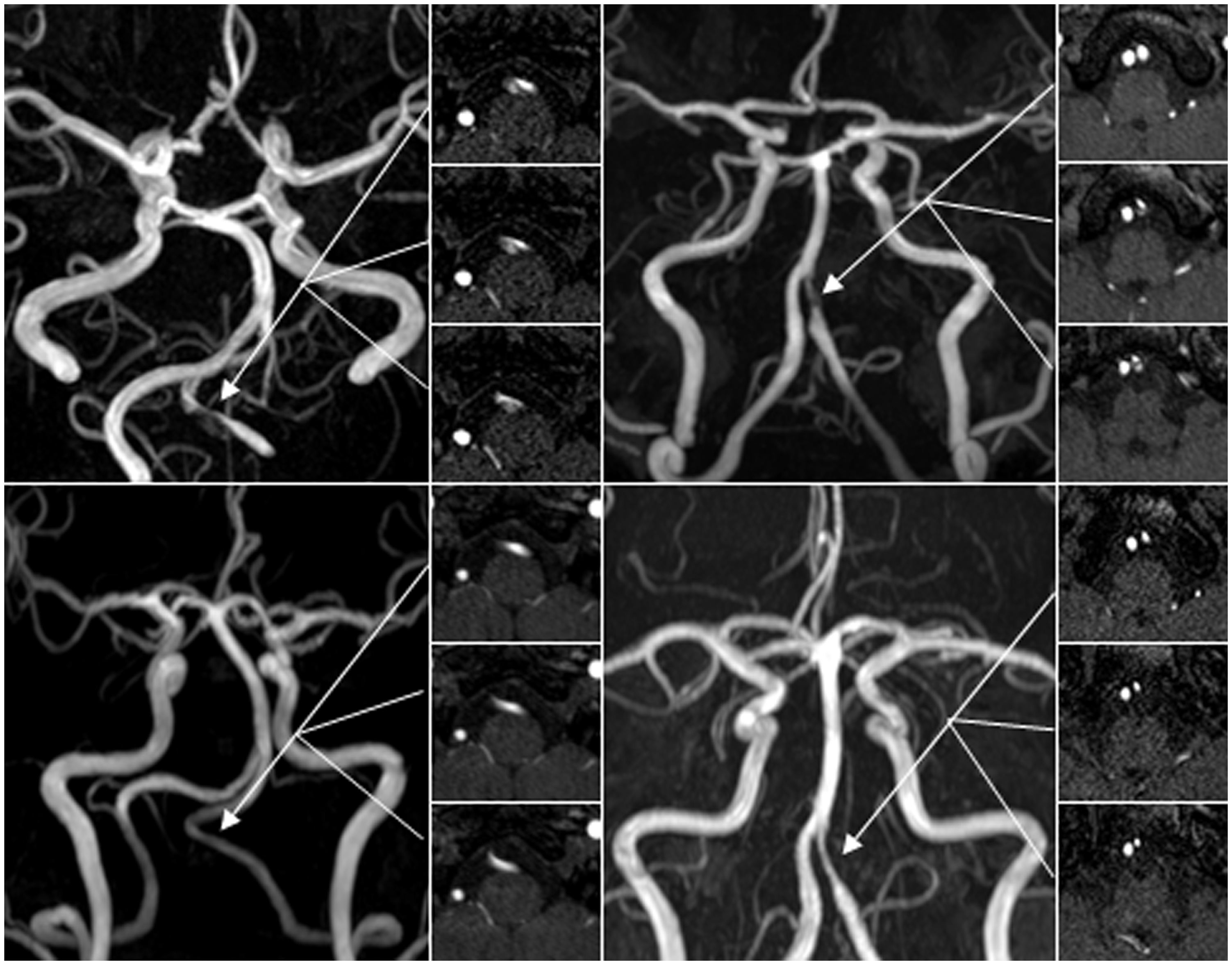
Literature review
We searched cases of ICVAD presenting with isolated headache published in English through the PubMed search engine. The terms “headache” with “cervical artery dissection” or “vertebral artery dissection” were used in our search. The initial review was conducted in December 2013, and was updated periodically through March 2014. A total of 103 articles were collected and reviewed. We included cases satisfying the following criteria: 1) headache should be faithfully described (mode of onset, quality, location, intensity, and duration were available); 2) dissection should involve an intracranial vertebral artery; and 3) neurological complications or signs (not symptoms) should be absent at presentation. Therefore, we excluded cases with ECVAD only, internal carotid artery dissection, and neurological complications, such as infarction and SAH, at diagnosis. Based on these criteria, we found eight cases of uncomplicated ICVAD presenting with headache alone.
Headache semiology
We systematically analyzed headache characteristics according to onset and duration, pain nature, intensity, location, aggravating and relieving factors, associated symptoms, response to the medication, and headache prognosis. The mode of onset was divided into thunderclap (sudden onset and reaching maximum intensity within one minute), acute (developing within 24 hours), and gradual (developing over greater than 24 hours) (10,12). Headache intensity was divided into moderate (VAS = 3–6) and severe (VAS = 7 or greater).
Validation of ICHD-3 beta diagnostic criteria
We evaluated the performance of each criterion of headache semiology as well as the total criteria for diagnosis of headache attributed to intracranial artery dissection in ICHD-3 beta, using data from our patients and selected cases from the literature.
Results
Frequency of uncomplicated ICVAD
From 2011 to 2013, 19 patients were radiologically confirmed to have cervicocranial artery dissection at our institute: Two had intracranial internal carotid artery dissection, three had middle cerebral artery dissection, one had basilar artery dissection, and 13 had VAD. In patients with VAD, 10 had ICVAD, one had ECVAD, and two had both extra- and intracranial VAD. At presentation, 13 patients had neurological complications, and the remaining six uncomplicated patients had only headache and were classified as ICVAD. Therefore, 32% patients with cervicocranial artery dissection (six of 19), 46% with VAD (six of 13), and 60% with ICVAD (six of 10) presented with headache without neurological complications in our institute.
Headache characteristics
Headache characteristics of intracranial vertebral artery dissection at presentation.

M: male; F: female; h: hour; R: right; L: left; B: bilateral; TCH: thunderclap; O: occipital; P: parietal; ON: occipitonuchal; FTP: frontotemporoparietal; TP: temporoparietal; VAS: visual analog scale; NA: not available.
Response to medication within first seven days.
Patients reported in this study.
Prognosis of headache and intracranial vertebral artery dissection.

M: male; F: female; h: hour; R: right; L: left; B: bilateral; TCH: thunderclap; O: occipital; P: parietal; ON: occipitonuchal; FTP: frontotemporoparietal; TP: temporoparietal; VAS: visual analog scale; NA: not available.
Patients reported in this study.
LMI: lateral medullary infarction.
Validation of ICHD-3 beta criteria
Validation of the International Classification of Headache Disorder – 3 beta criteria for 6.7.4 headache attributed to intracranial artery dissection.

Discussion
ICVAD is rare in the general population, given that the reported incidence of intracranial artery dissection was less than 10% of all cervicocranial artery dissection (13). The relatively high proportion of ICVAD in the present study may be surprising, but can be ascribed to ethnic differences in topography of cervicocranial artery dissection. Indeed, in contrast to a Caucasian population (5), the frequency of ICVAD was reported to be up to three- to 10-fold greater than that of ECVAD in Asian populations (14,15). Previously, approximately one-fifth of patients with cervicocranial artery dissection presented without neurological complications (10). In the present study, one-third of the patients with cervicocranial artery dissection presented with only headache, and the likelihood of an uncomplicated form increased in patients with VAD and ICVAD. Given that the diagnosis of cervicocranial artery dissection is not easy (16), our findings suggest that ICVAD may be underdiagnosed in clinical practice, particularly in uncomplicated cases. Even though technological advances in imaging have facilitated the diagnosis of ICVAD, a symptomatic approach using headache profiles may still be essential for its accurate diagnosis.
In the present study, the main characteristics of headache attributed to ICVAD can be summarized as follows: 1) The headache occurs acutely and persists more than two weeks; 2) the location of the headache is primarily in the ipsilesional occipitonuchal area; 3) the nature of the pain is mostly throbbing, suggesting a vascular origin; 4) the pain intensity is severe (VAS ≥ 7), and acute medical treatment is usually ineffective; 5) the headache is exacerbated by head flexion and rotation, and relieved by head extension or supine positioning; and 6) the headache is rarely associated with nausea, vomiting, photophobia, or phonophobia.
Some of these findings, such as location, duration, and intensity of pain, have been known, but the onset mode, nature, and intensifying factors of pain in ICVAD are still debated or less well known. Therefore, we will compare these components of headache in ICVAD to previous studies, including cases with focal neurological complications such as infarction and SAH. First, regarding mode of onset, a previous systematic study of 26 VAD patients (22 with focal neurological signs) described headache that occurred gradually in 72% of cases and suddenly in 22% (17). In another study of 14 ECVAD patients (all with either neurological signs or complications), it was reported that headache developed acutely in most cases (8). In those studies, however, the criterion for mode of onset was not specifically described. More recent systematic studies that enrolled patients with cervicocranial arterial dissection without neurological complications divided onset mode into thunderclap, acute, and gradual. As seen in previous studies, mode of onset was reported to vary in ECVAD (12), and, taken together, headache onset appears heterogeneous in patients with ECVAD. However, most patients with ICVAD had acute onset (10), as was shown in our cases. Moreover, this was reconfirmed when we selected only cases of ICVAD from the literature and evaluated these in conjunction with our cases (10,12,18). Given that neurological complications have been known to occur within a half-day of (median 14.5 hours) or simultaneously with VAD (17), an acute onset of headache may reflect temporal characteristics of ICVAD in evolution. Thunderclap headache has been reported frequently in patients with VAD (16,19), but has primarily been associated with SAH and reversible cerebral vasoconstriction syndrome (16,20). Therefore, the acute onset of headache in ICVAD seems to be influenced by associated intracranial neurologic complications.
Second, in our study throbbing pain is typical for headaches associated with ICVAD. Although the mechanism of pain in arterial dissection is not yet fully understood, it was proposed that dilation or distention of the arterial wall may result in mechanical stimulation of nociceptive receptors (21). Therefore, a throbbing quality may reflect an intrinsic nature of pain in VAD. Other types of pain have been described as well, ranging from sharp and dull (8), to constrictive (10,12), and stabbing and electrical pain (18,22). This variability may be ascribed to different study populations. Most previous studies enrolled patients with neurological complications at presentation, and ischemic or hemorrhagic stroke in the posterior circulation may be associated with diverse pain characteristics, which directly disrupts the ascending pain pathway or indirectly changes intracranial pressure through mass effect.
Finally, pain can be exacerbated or attenuated by head motion or a specific head position. Interestingly, this feature was described in an early study that investigated 14 ECVAD patients with neurological complications (8). In that study, 12 patients (85.7%) had dissection involving the V3 segments. Painful neck motion also was described in other cases of ECVAD (23). In general, though, this feature seems to be obscure because most studies since then have neglected this finding. Moreover, intensifying or relieving factors have not been described in patients with ICVAD. The pain mechanism exacerbation or attenuation in head motion or position could be ascribed to the anatomical features of the vertebral artery. The vertebral artery acutely bends nearly to a right angle at the level of the C1 (atlas) transverse foramen, and then runs transversely along the vertebral groove at the posterior atlas arch (V3 segment) (24). The C1 transverse foramen may serve as a relative fixing point, and specific head motion and position-induced elongation or shrinkage of a dissected vertebral artery distal to the C1 transverse foramen (V3 and V4 segments) may cause exacerbation or attenuation of pain. However, we did not determine whether the nature of exacerbated pain (i.e. throbbing pain) correlated with head motion or position, and so this point remains to be determined.
Diagnostic criteria for headache attributed to intracranial artery dissection in the ICHD-3 beta were applicable to patients in the present study. When we evaluated only headache semiology (criterion C2–4), intensity (C–3b) and location (C–4) criteria showed good performance, while the performance of duration (C–2) and onset mode (C–3a) appeared insufficient for patients in the present study. Elongation of duration and inclusion of “acute onset” should be considered to achieve better criteria performance. In addition, inclusion of throbbing or pulsating nature of headache as a sub-criterion may improve the diagnostic value of these criteria for intracranial artery dissection.
Differentiation of a primary headache, such as migraine or cluster headache, from a headache attributed to arterial dissection is important (11). Previous studies suggested that severe and unusual persistent occipitonuchal pain was suggestive of ICVAD (12,17). However, it still may be difficult to diagnose ICVAD in patients with headache and neck pain but without apparent neurological complications when presenting to an outpatient clinic or emergency department (10,17). It has been suggested that posterior headache in VAD without neurological signs is commonly misdiagnosed as tension headache (muscle contraction headache) (17). The long time to diagnosis (approximately 10 days) also may reflect difficulty in diagnosing VAD (16). The present study may help in the diagnosis of headache attributed to ICVAD. For example, unusual characteristics of headache compared with a previous primary headache in terms of location, intensity, duration, and response to medication may be strongly suggestive of secondary headache. Moreover, severe throbbing pain in the occipitonuchal area, while common in ICVAD pain, is atypical for migraine and trigeminal autonomic cephalalgias (TAC), which more commonly develop in the oculo-fronto-temporal area (25,26). Specific head motion- or position-induced pain exacerbation also does not correlate with migraine or TAC. Likewise, we can differentiate episodic tension-type headache by carefully asking about the quality (throbbing in ICVAD) and severity (severe in ICVAD) of pain (27,28).
Regarding prognosis, only one patient (patient 6) had a neurological complication after three weeks in the present study. However, it is not clear whether the natural outcome of initially uncomplicated ICVAD is good, since all patients with a favorable outcome in the present study were treated with antithrombotics and/or statins. In addition, patients with complicated ICVAD at presentation were excluded from this study (see Methods). Therefore, prognosis remains obscure and undetermined.
The present study has several limitations. First, the number of cases included was small. This may be attributed to the strict inclusion criteria (uncomplicated ICVAD cases with complete headache information). Second, some headache characteristics were obtained only in our limited number of cases. Finally, cerebrospinal fluid examination was not performed in all cases. Therefore the imaging-negative SAH cannot be excluded. Despite these limitations, headache semiology of ICVAD in the present study may help physicians in selecting which patients receive a neuro-imaging study, which may improve the diagnosis and treatment of ICVAD. Our findings and suggestions should be re-confirmed in a large-scale systematic study.
Clinical implications
The typical headache in uncomplicated intracranial vertebral artery dissection (ICVAD) is an acute-onset, severe, pulsating pain originating from the ipsilesional occipitonuchal area. Specific head motion or position may intensify or reduce the pain intensity in some patients. Given the serious potential neurological complications of ICVAD, early identification of uncomplicated ICVAD may be important. The headache semiology of ICVAD observed in the present study may help physicians to improve the diagnosis and treatment of ICVAD.
Footnotes
Authors’ contributions
J-G Kim analyzed the data and wrote the draft manuscript; J-Y Choi designed the research, analyzed the data, and made critical revisions of the manuscript; SU Kim, J-M Jung, D-Y Kwon and MH Park acquired the data and revised the manuscript; K Oh analyzed the data and made critical revisions of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
