Abstract
We report a case of secondary hypnic headache in a patient with a haemangioblastoma of the cerebellum. The number of secondary cases is steadily increasing in the medical literature and magnetic resonance imaging of the brain should be considered mandatory after arriving at a presumptive diagnosis.
Introduction
Hypnic headache was first described by Raskin (1) in 1988. Often referred to as ‘alarm-clock’ headache as the pain, which is usually bilateral but may be unilateral in location, will awaken the patient from sleep, with clockwork regularity, almost nightly. The mean age of onset is 63 years and women are more commonly affected. The pain is constant, moderately severe, often throbbing in character, lasting from minutes to 3 h. Autonomic symptoms are absent and nausea, phonophobia and photophobia are not prominent features (2,3). There have been more than 100 cases reported in the medical literature and while hypnic headache is usually a primary headache disorder, underlying pathology resulting in secondary hypnic headache is not uncommon. Posterior fossa meningioma, pituitary adenomas, sleep apnoea, hypertension, and pontine stroke have been reported to trigger typical episodes of hypnic headache (4–9). We describe a patient with seemingly classic hypnic headache who was found to have a cerebellar haemangioblastoma.
Case report
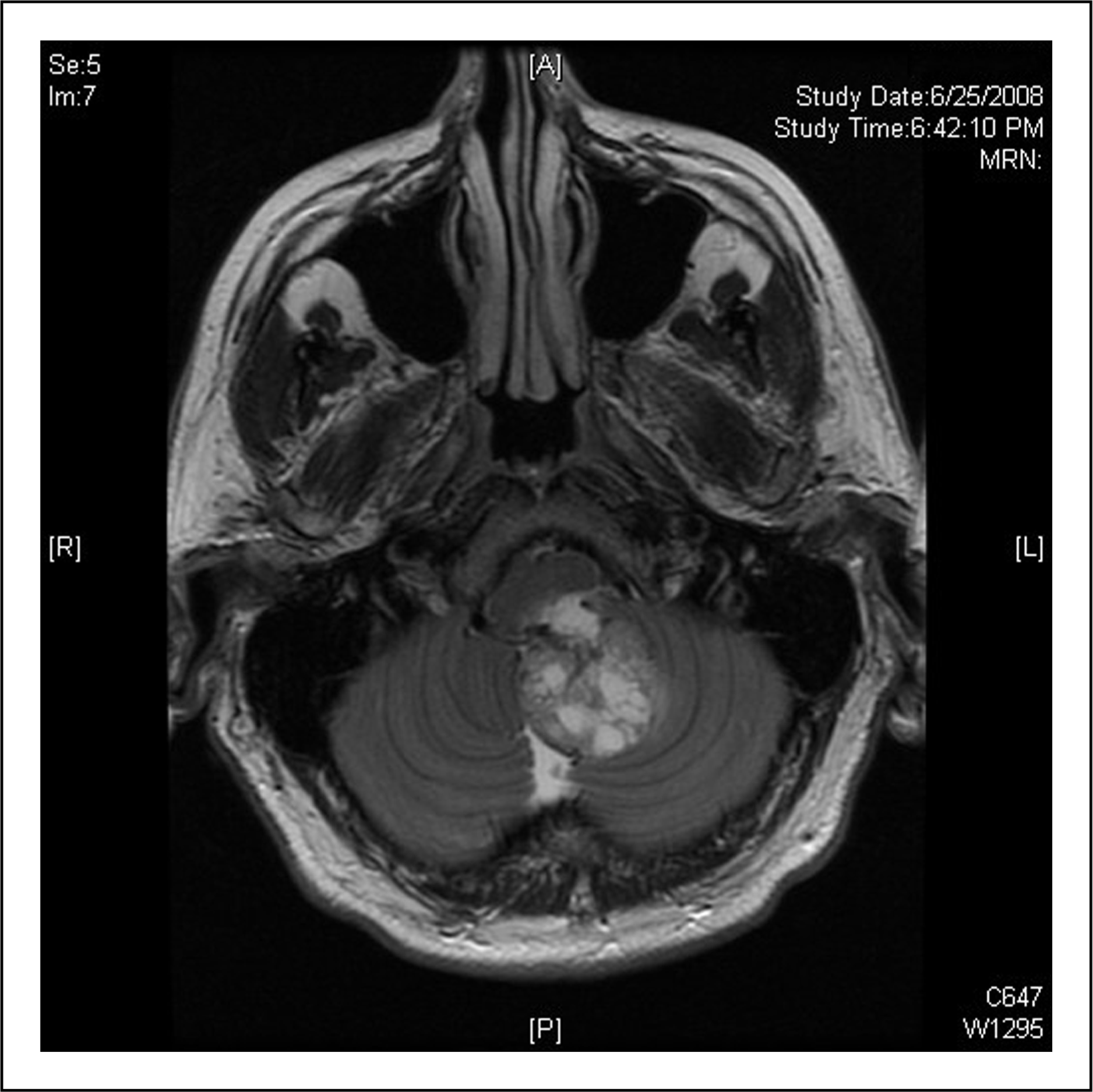
A 58-year-old man presented with a 2-month history of bi-occipital to bifrontal throbbing headaches awakening him from sleep 4–5 nights per week. The headaches occurred at the same time, several hours after falling asleep, and each episode lasted approximately an hour. When he was experiencing the pain, it was aggravated by lying down. The headaches did not occur during the day and there were no accompanying neurological symptoms, nausea or autonomic symptoms. Ibuprofen provided him with some relief, but cyclobenzaprine was not effective. He otherwise felt well and his past medical history and family history were negative. He did not have a history of a primary headache disorder. Neurological examination was normal and there was full mobility of the head and neck. Vital signs were normal with blood pressure of 130/80. While awaiting brain imaging, the patient tried lithium 300 mg and the headaches were reported to be less severe. A magnetic resonance imaging (MRI) scan revealed a heterogeneous mass in the fourth ventricle with compression of the medulla and pons with vasogenic oedema of the inferior cerebellum and obstructive hydrocephalus with transependymal oedema (Figures 1 and 2). The patient underwent a two-stage microsurgical resection of the tumour over a period of 14 days with suboccipital craniotomy, C1 laminectomy, right occipital ventriculostomy and duraplasty. Pathological examination was consistent with a haemangioblastoma. The patient experienced constant incisional head pain at the site of the craniotomy following the initial surgical procedure that gradually disappeared within 3–4 weeks following the second procedure. The patient subsequently received adjuvant radiation therapy. Imaging of the chest, abdomen, pelvis and spine was negative as was a neuro-ophthalmological examination. The nocturnal headaches resolved following the first craniotomy and he has remained free of headaches more than a year post surgery.
Axial T2 weighted image through the posterior fossa demonstrates heterogeneous, partially cystic mass with vasogenic edema, arising from the left cerebellar vermis. Coronal T2 weighted image through the posterior fossa demonstrates mass extending into the 4th ventricle and foramen Magnum, with resultant hydrocephalus and transependymal edema.
Discussion
We report a case of a 58-year-old man who presented with a headache fulfilling the 2004 International Headache Society criteria for hypnic headache who was found to have a haemangioblastoma of the cerebellum (10). The headache disappeared following recovery from surgery confirming that this was indeed a secondary headache.
It has been postulated that hypnic headache is the result of dysfunction of the brainstem neural pathways that regulate the sleep–wake cycle. Attacks tend to occur during REM sleep as documented by polysomnography studies when there is also diminished dorsal raphe nucleus activity. The regularity of the episodes during sleep suggests that the suprachiasmic nuclei of the hypothalamus are involved. There are neural connections between the pain modulating peri-aqueductal grey and the hypothalamus likely playing a role in the genesis of the headache. A decrease in melatonin secretion has been postulated possibly explaining a predilection for the elderly (2,11–13). In our patient, the headache appears to have been the result of direct brainstem compression and hydrocephalus.
Hypnic headache is a primary headache disorder that is relatively rare and only recognized as a distinct entity since 1988 (1). While initial reports suggested that this particular nocturnal headache was almost always benign, there have been an increasing number of reports of secondary hypnic headache, occurring as the result of a myriad of pathological processes (4–9). Our patient is the second case of seemingly typical hypnic headache that was found to be secondary to a posterior fossa tumour (5). Nocturnal headaches, while usually benign, should always raise a degree of concern about the possibility of underlying pathology.
Conclusions
We think that cranial MRI is mandatory in the evaluation of any patient who presents with a clinical history consistent with hypnic headache.
