Abstract
Background
Trigeminal neuralgia is a disorder of paroxysmal and severely disabling facial pain and continues to be a real therapeutic challenge. At present there are few effective drugs. Here the aim of this study was to investigate the role of BKCa channels in trigeminal neuropathic pain.
Methods
Rats were divided into two groups: a sham and a chronic constriction injury of infraorbital branch of trigeminal nerve (ION-CCI) group. Nociceptive behavior testing, immunohistochemistry, RT-PCR, Western blotting and whole-cell patch clamp recording were used.
Results
Relative to the sham group, rats with ION-CCI consistently displayed lower mechanical pain thresholds in the vibrissal pad region from day 6 to 42 after ION-CCI operation. ION-CCI induced a significant down-regulation of BKCa channels both in mRNA and protein levels in the ipsilateral trigeminal ganglion (TG), a lower threshold intensity of action potential, and decreased total BKCa currents in cultured TG neurons. TG target injection of NS1619 (20–100 µg), an opener of BKCa channels, dose-dependently increased the mechanical pain threshold, which was blocked by the BKCa channel inhibitor iberiotoxin (IbTX, 20 µg). NS1619 (10 µM) significantly increased the mean threshold intensities of action potentials in ION-CCI rats, while failing to affect those in the sham rats. The levels of phosphorylated extracellular signal-regulated kinase (ERK), p38 and c-Jun N-terminal kinases (JNK) in TG were significantly increased after ION-CCI operation. The ERK1/2 antagonist U0126, p38 antagonist SB203580 and JNK antagonist SP600125 significantly reversed the facial mechanical allodynia in ION-CCI rats. However, the ERK1/2 antagonist U0126, p38 antagonist SB203580 but not JNK antagonist SP600125 significantly increased BKCa currents in ION-CCI TG neurons.
Conclusions
Our results indicate the important involvement of mainly ERK and p38 MAPK pathways in modulating BKCa channels in ION-CCI TG neurons. BKCa channels represent a new therapeutic target for the clinical treatment of trigeminal neuropathic pain.
Keywords
Introduction
Trigeminal neuralgia (TN) is one of the classical pain disorders and constitutes a major therapeutic challenge to health care professionals (1–3). It is regarded as a neuropathic pain with abnormal pain hypersensitivity due to a lesion or dysfunction of the peripheral or central nervous system, probably caused by focal compression of the trigeminal nerve root at or near the dorsal root entry zone by an aberrant loop of artery or vein, or occasionally by tumors, cysts, anteriovenous malformations or aneurysms (4). However, the underlying mechanisms are unclear. One characteristic symptom of neuropathic pain is dynamic mechanical allodynia or pain in response to light, dynamic mechanical stimuli (e.g. gentle brush of the skin); that is, activation of the sensory fibers that normally detect touch elicits pain (5). Rats with a chronic constriction injury of the infraorbital nerve (ION-CCI) consistently display signs of spontaneous pain-related behavioral abnormalities (6), mechanical allodynia (6,7) and thermal hyperalgesia (8). The ectopic spontaneous discharges in TG neurons are closely related to the hyperalgesia and allodynia (9,10).
Large-conductance, calcium-activated potassium (BKCa) channels have a large, single-channel conductance (pS 100–300) and are activated by an increase in intracellular calcium and membrane depolarization (11,12). BKCa are expressed intrinsic membrane proteins and are involved in the regulation of neuronal excitability (13–17). Because of their high conductance, activation of BKCa channels shortens the action potential duration, enhances the rate of repolarization, reduces the depolarization and contributes to the rapid after-hyperpolarization. All these features make BKCa channels ideal feedback regulators of cell excitability (13–17). It has been reported BKCa channels are expressed abundantly in the trigeminal ganglion (TG) (18), dorsal root ganglia (DRG) (17) and superficial dorsal horn (16) and play a role in the anti-hyperalgesia effect in neuropathic pain (15,17). Expression of BKCa channels was significantly reduced in superficial dorsal horn after partial ligation of the sciatic nerve (16) and in DRG neurons after L4–L5 nerve-ligation injury (17). Activation of BKCa channels by NS1619 reversed allodynia and hyperalgesia caused by L4–L5 nerve injury. The effect was antagonized by selective BKCa channel antagonist iberiotoxin (IbTX) (17). In DRG neurons, opening BKCa channels by NS1619 leads to the reduction of depolarization-evoked action potential firing and 4-aminopyridine-induced neuronal hyperexcitability (19). IbTX itself presynaptically increased the frequency of spontaneous excitatory postsynaptic currents (sEPSCs) recorded in the superficial dorsal horn and thus modulated nociceptive transmission (16).
Mitogen-activated protein kinase (MAPK) is one of the most important molecules involved in the intracellular transduction cascade resulting in neuronal excitability changes (20–23). The MAPK family includes three major molecules: extracellular signal-regulated kinase (ERK), p38 and c-Jun N-terminal kinases (JNK). All of these molecules located in sensory neurons are known to have different functions in sensory processing (20–25). Inhibitors of ERK (26), p38 (27) and JNK (28) have been shown to effectively alleviate inflammatory and neuropathic pain in different animal models (20–23). Evidence has shown that ERK plays a role in the modulation of voltage-gated potassium channels, such as Kv4.2 and Kv4.3 (29–31). However, the functional significance of BKCa channels in TG neurons and the role of MAPK pathways are so far unknown. Therefore, the aim of the present study was to assess nociceptive behavior evoked by mechanical stimulation of the whisker pad skin, expression and function of both BKCa channels and the potential phosphorylation of MAPK pathway in ION-CCI rats.
Subjects and methods
Experimental animals
Animal care was consistent with the guidelines set by the Laboratory Animal Center of Second Military Medical University. All experimental procedures were approved by the Second Military Medical University Institutional Animal Care and Use Committee governing the participating laboratories. Adult male Sprague-Dawley rats weighing 200–250 g were used in these experiments. Rats were randomly divided into two groups: an ION-CCI group receiving a chronic constriction injury of the infraorbital branch of trigeminal nerve and a sham group receiving the same surgical procedure without constriction.
ION-CCI and sham operation
The unilateral ligation of the ION was made according to the method previously described (8). Briefly, rats were anesthetized with sodium pentobarbital (50 mg/kg intraperitoneally (i.p.)). The incision was made at the juncture between the zygomatic arch and nasal bone (near the back of the nose). Blunt dissection of the muscle was performed to expose the infraorbital branch of the trigeminal nerve until it was clearly visible through the incision. About 0.5 cm of the ION was dissected free of adhering tissue, and two chromic catgut ligations (4–0) were loosely tied (with about 2 mm spacing) around the nerve. To obtain the desired degree of constriction, a criterion formulated by Bennett and Xie (32) was applied: The ligatures reduced the diameter of the nerve by a noticeable amount and retarded but did not interrupt the circulation through the superficial vasculature. The scalp incision was closed using silk sutures (4–0). In the sham rats, the operation was performed in the same manner except the ION was not ligated. The contralateral sides were left intact in all rats. All operations were performed aseptically and no antibiotics were administered.
Behavior testing
The mechanical pain threshold was tested one day before and every three days after surgery for 42 days in the two groups. The rats were weighed and placed individually in a stainless steel cage (50 × 50 × 50 cm). They were allowed to acclimatize for at least one hour. The mechanical pain threshold was determined with a graded series of 15 von Frey filaments (Institute of Autonomic Neuroscience, London, UK). The filaments produced a bending force ranging from 0.01 to 14.9 g. The stimuli were applied within the ION territory, around the center of the vibrissal pad, on the hairy skin surrounding the mystacial vibrissae. Stimuli were applied in an ascending order of intensity. Each stimulus consisted of 10 consecutive placements (interval ∼2 seconds) of the stimulus filament. The response threshold was considered the lowest force of the filaments that produced a brisk head withdrawal, touching or scratching of the facial regions upon mechanical stimulation (6,7,33). Withdrawal threshold was expressed as the threshold level in grams. The mechanical response threshold was measured every 10 minutes after following the drug administration. The individuals conducting the behavioral measurements were blind to the treatments.
TG injections
We carried out the peripheral target injection to the TG via the infraorbital foramen as Neubert et al. described previously (34). Briefly, on postoperative day 15, rats were anesthetized with the appropriate diethyl ether. As soon as the animals revived within 20 seconds after injection, behavior testing was conducted. The sterile stainless steel needle was then inserted medially (1–2 mm) to the palpated portion of the zygomatic process through the infraorbital foramen. The needle was positioned at a ∼10 degree angle relative to the midline of the head. The tip of the needle was advanced approximately 20 mm along the infraorbital canal and subsequently through the foramen rotundum then the corresponding drugs were injected. The mechanical pain threshold was then determined every 10 minutes after injection.
Real-time quantitative reverse transcription-polymerase chain reaction (RT-PCR) analysis RNA isolation and complementary DNA synthesis
On postoperative day 15, total RNA from the TG was extracted using an RNeasy Mini Kit (QIAGEN, Clifton Hill, Australia). RNA purity was determined using a method of ultraviolet spectrophotometry at a wavelength of 260–280 nm. Two micrograms of total RNA was reversely transcribed to complementary DNA in a 20 µl reaction mixture containing 1 × reverse transcriptase buffer (15 mM MgCl2, 375 mM KCl, 50 mM dithiothreitol (DTT) and 250 mM Tris-HCl, pH 8.3), 10 mM deoxynucleoside triphosphates (dNTP), 20 U RNase inhibitor, 200 U Moloney murine leukemia virus (M-MLV) reverse transcriptase and 50 ng of oligo (deoxythymidine)15 primers. Reaction time was at least one hour at 42℃. The cDNA was stored at −20℃ until real-time PCR (RT-PCR). All reagents, with the exception of the RNeasy Mini Kit, were from Promega Corp. (Madison, WI, USA).
Quantitative (QT) real-time RT-PCR
Sequence of primers used for RT-PCR.

RT-PCR: real-time polymerase chain reaction; BKCa: large-conductance, calcium-activated potassium.
Western blotting analysis
On postoperative day 15, TG were harvested and homogenized in cold lysis buffer (20 mM Hepes buffer, pH 7.4, 10 mM KCl, 1.5 mM MgCl2, 1 mM ethylenediaminetetraacetic acid (EDTA), 1 mM ethyleneglycoltetraacetic acid (EGTA), 1 mM DTT, 0.1 mM phenylmethylsulfonyl fluoride (PMSF), 5 mg/ml pepstatin A, 10 mg/ml leupeptin and 10 mg/ml aprotinin) using a Dounce homogenizer. Protein concentration was determined with a bicinchoninic acid (BCA) assay kit using bovine serum albumin as a standard (Pierce Biotechnology Inc, Rockford, IL, USA) and then heated to 95℃. Proteins were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) on 12% TRIS-HCl gels (Bio-Rad, Hercules, CA, USA) and electrophoretically transferred to polyvinylidene difluoride membranes (Bio-Rad) at 120 V for one and a half hours in Towbin buffer, pH 8.3, to which 20% (V/V) methanol had been added. After transfer, the membranes were blocked with 5% (mass/vol) nonfat dried milk in Tris-buffered saline containing 0.05% Tween 20 (TBST) for one hour, then incubated with the primary antibodies: BKCa (rabbit anti-rat polyclonal, immunoglobulin (Ig)G 1:1000, Alomone Labs, Israel), ERK1/2 (rabbit mAb, IgG 1:1000, Cell Signaling Technology, USA), p-ERK1/2 (rabbit mAb, IgG 1:2000, Cell Signaling Technology, USA), p-p38 (rabbit mAb, IgG 1:500, Cell Signaling Technology, USA), p38 (rabbit mAb, IgG 1:1000, Cell Signaling Technology, USA), p-JNK (mouse mAb, IgG 1:1000, Cell Signaling Technology, USA), JNK (rabbit mAb, IgG 1:1000, Cell Signaling Technology, USA) and β-actin (mouse monoclonal, IgG 1:8000, Sigma, USA). After three washes with TBST, the membranes were incubated with the secondary antibody for BKCa (horseradish peroxidase (HRP)-goat anti-rabbit polyclonal, IgG 1:1000, Santa Cruz Biotechnology, Santa Cruz, CA), ERK1/2, p-ERK1/2, p-p38, p-38, JNK (HRP-goat anti-rabbit polyclonal, IgG 1:1000, Aogma, China), p-JNK (HRP-goat anti-mouse polyclonal, IgG 1:1000, Aogma, China) while for β-actin (HRP-goat anti-mouse polyclonal, IgG 1:800, Santa Cruz Biotechnology). Membranes were rinsed at room temperature in Tris-buffered saline containing Tween 20 (TBST) followed by TBS three times and visualized using an Odyssey Infrared Imaging System (LICOR, Lincoln, NE, USA). Densitometric quantification of the BKCa, ERK1/2, p-ERK1/2, and β-actin protein bands of the Western blot were determined using Odyssey software version 1.0 (LI-COR) and expressed as a relative ratio compared to that of β-actin.
Immunofluorescence histochemistry
Rats were transcardially perfused with 0.01 M phosphate-buffered solution (PBS), pH 7.4 and subsequently with fresh 4% paraformaldehyde on day 15 after the surgery. After the perfusion, the TG were harvested and fixed in 4% paraformaldehyde for four hours. They were then transferred into 20% sucrose for three days. Frozen transverse sections (10 µm thickness) were made through the TG, collected and then washed three × five minutes in cold PBS. The preparations were then preincubated in antiserum solution 1 (10% normal bovine serum, 0.2% Triton X-100, 0.4% sodium azide in 0.01 M PBS pH 7.2) for 30 minutes, followed by incubation with combinations of BKCa antibody diluted 1:400 (mouse mAb, Anti-Maxi Potassium channel alpha antibody (S6-60), abcam (Hong Kong) Ltd.) and the NeuN antibody (mouse anti-neuronal nuclei, Alexa floor 488 conjugated monoclonal antibody, Millipore, MA, USA) diluted 1:200 in antiserum solution 2 (1% normal bovine serum, 0.2% Triton X-100, 0.4% sodium azide in 0.01 M PBS pH 7.2) at room temperature. Subsequently, the preparations were incubated with Cy3-conjugated donkey anti-rabbit IgG diluted 1:400 for BKCa antibodies in antiserum solution 2 for two hours at room temperature. Cy3-conjugated donkey anti-rabbit IgG is specialized for multiple immunostaining from Jackson ImmunoResearch Lab (PA, USA). All the incubations and reactions were separated by three × 10 minute washes in PBS.
Whole-cell patch clamp recording
Dissociated neurons
On postoperative day 15, the sham and ION-CCI rats were used for patch-clamp recording. Rats were decapitated and ipsilateral TG were rapidly removed, de-sheathed, cut and incubated in 2 ml Ca2+- and Mg2+-free Hanks’ balanced salt solution with 10 mM HEPES buffer (pH 7.4) (HBSS; Life Technologies, NY, USA) containing 1.5 mg/ml collagenase (Class II, Worthington Biochemical Corporation, UK) and 6 mg/ml bovine serum albumin (Sigma Chemical Co., Poole, UK) at 37℃ in a shaking bath (170 rpm) for 30 minutes. This was followed by incubation in 2 ml HBSS containing 1 mg/ml trypsin (Sigma) at 37℃ in a shaking bath (170 rpm) for 10 minutes. The solution was replaced with 1 ml growth medium composed of L-15 medium supplemented with 10% bovine serum albumin, 50 ng/ml nerve growth factor, 0.2% NaHCO3, 5.5 mg/ml glucose, 200 i.u./ml penicillin and 200 µg/ml streptomycin. The ganglia were dissociated into single neurons by gentle mechanical trituration and plated onto 35 mm Petri dishes coated with 10 µg/ml laminin (Sigma) (36). Cells were maintained at 37℃ in a humidified atmosphere containing 5% CO2. All neurons were studied between three and 12 hours after removal from the animals.
Whole-cell patch-clamp recording was undertaken at room temperature with an Axopatch 200B amplifier (Axon Instruments, Foster City, CA, USA). Membrane potential was held at −60 mV, signals were filtered at 2 kHz (−3 dB frequency, Bessel filter, 80 dB per decade), then digitized at 10–50 kHz (Digidata 1320A interface, Axon Instruments). Patch electrodes had resistance of 2–5 MΩ. The pipette solution contained the following (in mM): K gluconate 120, KCl 10, NaCl 5, MgCl2 6H2O 2, CaCl2 2H2O 1, HEPES 10, EGTA 11, Mg-ATP 2, Li-GTP 1 (pH adjusted to 7.4 with KOH). The external solution contained (in mM): NaCl 145, KCl 3, CaCl2 2H2O 1, MgCl2 6H2O 2, HEPES 10, glucose 10, (pH adjusted to 7.4 with NaOH). Test solutions bathing the cytoplasmic face of the patch membrane contained (in mM): N-methyl-D-glucamine (NMDG) 145, KCl 3, MgCl2 6H2O 0.6, CaCl2 2H2O 2.5, HEPES 10, glucose 10 (pH adjusted to 7.4 with Tris-base and 300 mOsM). Apamin (200 nM) and 4-AP (10 mM) were routinely added to extracellular solutions to minimize contamination by small-conductance, calcium-activated K+ (SK) currents (37) and voltage-dependent K+ (KV) currents, respectively (38,39). A protocol was used as previously described (40), a pre-pulse (0 mV, 100 ms) was followed by test pulses (400 ms) from −80 to +80 mV in 10 mV increments, and only those cells that exhibited minimal outward currents during the pre-pulse were analyzed. The action potentials were recorded under current-clamp recording. During a 400-ms injection of a positive current (ranging from −40 to 450 pA), a single action potential could be evoked, depending on the type of the neuron (10). Only the cultured TG neurons with soma diameters ranging from 18 to 39 µm were used in this study.
Drugs
[1,3-dihydro-1-[2-hydroxy-5 -(trifluoromethyl)-phenyl]-5 -(trifluoromethyl)-2H-benzimidazol-2-one] (NS1619), a specific BKCa channel opener; IbTX, a BKCa channel blocker; 4-aminopyridine (4-AP, a selective blocker of members of the Kv1 (KCNA) family of voltage-activated K+ channels), apamin (an inhibitor of SKCa channels), NMDG (a selective blocker of Na+ channels) and U0126 (a highly selective inhibitor of MAPK/ERK kinase), SB203580 (an inhibitor of p38 MAPK), SP600125 (an inhibitor of JNK) were all purchased from Sigma. NS1619 was dissolved in dimethysulfoxide (DMSO), and then diluted in normal saline to the final concentration of 5 µg/µl in 0.1 ml. IbTX was dissolved in normal saline then diluted to a final concentration 10 µg/ml in 0.1 ml. Similarly, in the patch-clamp recording, 4-AP and IbTX were first dissolved in normal saline and then diluted in extracellular bathing solution to a final concentration of 10 mM and 100 nM, respectively. NS1619 (10 µM), U0126 (1 µM), SB203580 (10 µM and 30 µM) and SP600125 (10 µM) were all first dissolved in DMSO and then diluted in extracellular bathing solution to the final concentration, respectively.
Statistical analysis
All data are expressed as means ± SEM. The two-way repeated-measure analysis of variance (ANOVA) was used to compare the differences between groups in the pain behavior testing and to analyze BKCa currents in the sham and ION-CCI groups. Unpaired Student’s t test was used to analyze the expression of BKCa channels in the protein and mRNA levels, ERK expression, threshold intensities for activating action potential (AP) and peak currents induced by BKCa channels in the sham and ION-CCI groups. One-way ANOVA was used to evaluate the effect of NS1619 on the threshold intensities of AP in the sham and ION-CCI groups. Differences among groups were considered to be significantly different when the p value was lower than 0.05 (p < 0.05).
Results
Effects of NS1619 and IbTX on the mechanical pain threshold in the sham and ION-CCI rats
The mechanical pain threshold was determined one day before and every three days after ION-CCI surgery. The testing was conducted for 42 days post-operation. In accordance with previous results (6,7), the rats with ION-CCI showed marked increased responsiveness to mechanical stimulation of the territory of the ligated nerve (Figure 1(a)). ION-CCI rats developed a mechanical allodynia on day 6 (5.5 ± 0.5 g; n = 8; p < 0.05) and remained at least until day 42 (2.05 ± 0.01 g; n = 8; p < 0.05) after surgery. On postoperative day 15, the mechanical pain threshold on the ipsilateral side of ION-CCI rats reached the bottom (0.04 ± 0.01 g; n = 8), which was significantly lower than that observed in the sham rats (14.6 ± 0.3 g; n = 8; p < 0.05).
(a) Time course of the mechanical pain thresholds in the sham and ION-CCI rats after surgery. ION-CCI rats developed an allodynia from day 6 and remained at least until day 42 after surgery (n = 8 for each group). *p < 0.05 compared with those in the sham rats. (b) Effects of target injection of NS1619 (an agonist of BKCa channel) and iberiotoxin (IbTX, an antagonist of BKCa channel) on the ION-CCI rats at day 15 after surgery. Target injection of 20 (n = 6), 50 (n = 6) and 100 (n = 7) µg of NS1619 significantly reversed the facial mechanical pain threshold in the ION-CCI rats. Target injection of the 20 ng IbTX, 15 minutes before injection of NS1619 (100 µg) abolished the anti-allodynia effect of NS1619 (n = 6). *p < 0.05 compared with those in the vehicle group. (c) Quantification of the mRNA levels of BKCa channels in the TG between the sham and ION-CCI rats on postoperative day 15. Group data showed differential changes in α (511 bp), β2 (409 bp) and β4 (514 bp) subunit mRNA levels of BKCa channels in the TG between the sham and ION-CCI rats (n = 5). (d) Quantification of the protein levels of BKCa channels in the TG between the sham and ION-CCI rats on postoperative day 15. Representative gel images and summary data showed a differential change in protein levels of BKCa channels in the TG between the sham and ION-CCI rats (n = 6). *p < 0.05 compared with those in the sham group.
The BKCa channel agonist NS1619, a benzimidazolone analog (13,14), was selected to investigate the effect of activation of BKCa channels on mechanical allodynia in ION-CCI rats. Target injection of 20 µg (n = 6), 50 µg (n = 6) and 100 µg (n = 7) NS1619 dose-dependently increased the mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in the ION-CCI rats (Figure 1(b)). This effect started at 50 minutes (for 50 µg) and remained for more than 90 minutes (for 50 µg) after target injection. When NS1619 (100 µg) was administered, the mechanical pain threshold reached a maximum at 80 minutes (4.88 ± 0.7 g) and lasted more than 110 minutes (2.3 ± 0.8 g) after injection. Target injection of IbTX (20 ng), a highly selective blocker of BKCa channels (13–17) abolished the anti-allodynia effect of NS1619 (100 µg, n = 6, Figure 1(b)). Target administration of IbTX (20 ng) alone did not significantly change the facial mechanical pain threshold (n = 6, p > 0.05).The effect of NS1619 and IbTX were also tested in the sham rats on day 15 after surgery (n = 6 for each group). The results showed that neither NS1619 (100 µg) nor IbTX (20 ng) alone altered the facial mechanical pain threshold in sham rats.
Expression of constitutive α- and modulatory β-subunits of BKCa channels in TG from sham and ION-CCI rats
To investigate whether the expression of BKCa channels in TG was changed after ION-CCI, we used real-time RT-PCR and Western blotting to measure the mRNA and protein level of BKCa channels, respectively. RT-PCR results showed the existence of α, β2 and β4 subunits but not β1 or β3 subunits in TG, which was consistent with previous reports (15,18). The relative mRNA levels of α, β2 and β4 subunits of BKCa channels in TG from ION-CCI rats were significantly reduced compared with those in the sham rats. (Figure 1(c), n = 5, p < 0.05). Similarly, the expression of BKCa channels in protein levels in TG from ION-CCI rats was also reduced compared with that in the sham rats (Figure 1(d), n = 6, p < 0.05).
BKCa channel immunoreactivity in TG neurons between sham and ION-CCI rats
We determined whether the spatial distribution of BKCa channels in TG was altered after ION-CCI injury by double immunofluorescence labeling of NeuN and BKCa channels. We found that BKCa channel immunoreactivity was abundantly and densely present both in the cytoplasm and cell membrane of the ipsilateral TG neurons from the sham and ION-CCI rats (Figure 2(a)). Approximately 82.5 ± 2.8% (n = 786 neurons from three rats) of NeuN-positive TG neurons showed BKCa channel immunoreactivity in sham rats and 54.5 ± 2.3% (n = 863 neurons from three rats) in the ipsilateral TG from ION-CCI rats (Figure 2(b), p < 0.01). Most of the BKCa-immunoreactive neurons were concentrated in small and middle-sized (<30 µm and 30–40 µm) TG neurons both in the sham and ION-CCI groups. The BKCa-immunoreactive ratio from middle-sized (30–40 µm) neurons (15.1 ± 1.4%, n = 465 neurons from three rats) in the ION-CCI group were significantly decreased compared with that in the sham group (48.5 ± 6.4%, n = 634 neurons from three rats, Figure 2(c), p < 0.01). BKCa-immunoreactive neurons in small-sized (<30 µm) TG neurons in the ION-CCI group (35.9 ± 2.0%, n = 465 neurons from three rats) was not different compared with that in the sham group (30.2 ± 5.6%, n = 634 neurons from three rats, Figure 2(c), p > 0.05).
Changes in BKCa-immunoreactive and NeuN (a neuronal marker)-positive TG neurons in a sham and an ION-CCI rat on postoperative day 15. (a) Co-localization of the BKCa channel immunoreactivity (red) and NeuN (green) is indicated in yellow when the two images are digitally merged. All images are single optical sections. (b) Histograms showed the somatic size distribution of BKCa channel immunoreactive-neurons in the ipsilateral NeuN-positive TG neurons from the sham and ION-CCI rats (n = 3 rats for each group, *p < 0.01). A total of 786 and 863 neurons were counted from the sham and ION-CCI rats, respectively. Bar = 50 µm. (c) The BKCa-immunoreactive ratio from middle-sized (30–40 µm) neurons (15.1 ± 1.4%, 465 neurons) was significantly decreased in the ION-CCI group compared with that in the sham group (48.5 ± 6.4%, 634 neurons, *p < 0.01). BKCa-immunoreactive neurons in small-sized (<30 µm) or large-sized (>40 µm) TG neurons in the ION-CCI group were not significantly different from those in the sham group (*p > 0.05).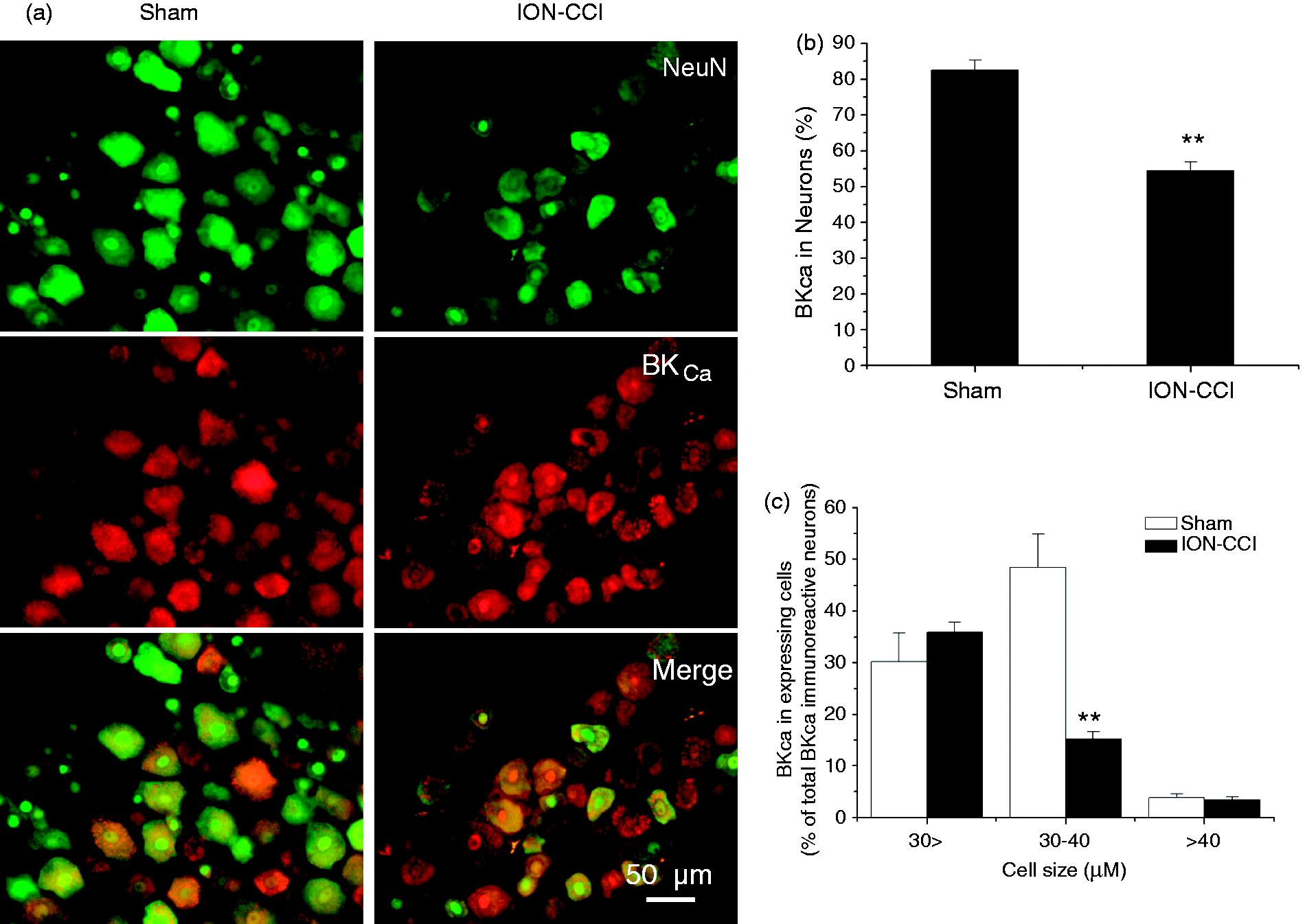
Effects of NS1619 on action potentials in TG neurons from sham and ION-CCI rats
Action potentials generated in TG neurons of sham and ION-CCI rats by square-pulse stimulation are illustrated in Figure 3(a). The mean threshold intensities for evoking the action potentials were 155.0 ± 25.9 pA in sham rats (n = 9) and 46.3 ± 16.3 pA in ION-CCI rats (n = 7). The threshold intensity was significantly decreased in the ION-CCI rats compared with that in the sham rats (Figure 3(b), p < 0.05). This result was consistent with a previous study (9). The passive electrical properties and action potentials in small-size TG neurons of the sham and ION-CCI rats are shown in Table 2 (Supplementary Data). There were no significant differences of rest membrane potential (RMP), action potential duration and action potential amplitude between the sham and ION-CCI groups. The amplitude of fast after-hyperpolarization (fAHP) was significantly decreased in the ION-CCI group (n = 7) compared with that in the sham group (n = 9, p < 0.05). To demonstrate the effect of NS1619 (10 µM) on the sham and ION-CCI rats, action potentials were generated in TG neurons in the presence or absence of NS1619. As illustrated in Figure 3(c), (d), in ION-CCI rats, NS1619 significantly increased the mean threshold intensities for evoking action potentials from 56.4 ± 12.5 pA to 294.3 ± 29.0 pA (n = 7, p < 0.01), while it failed to affect those in the sham rats (154.4 ± 18.0 pA to 173.3 ± 17.6 pA (n = 9, p > 0.05)). There was a significant difference of the NS1619 effect between the sham and ION-CCI rats (p < 0.05, Figure 3(d)).
Changes in action potential in TG neurons from the sham and ION-CCI rats on postoperative day 15. (a) Representative responses during current injection in two TG neurons from a sham rat and an ION-CCI rat, respectively. (b) Histograms show the mean threshold current intensities in the sham and ION-CCI rats (n = 9 for the sham group, n = 7 for the ION-CCI group). *p < 0.05 compared with that in the sham rats. (c) The responses during current injection in two TG neurons from the sham and ION-CCI rats in the presence of NS1619 (10 μM). (d) Histograms show the mean threshold current in the sham and ION-CCI rats in the presence and absence of NS1619 (10 μM). Application of NS1619 significantly enhanced the mean threshold intensities in the ION-CCI rats while having no effect in the sham rats (n = 9 for sham group, n = 7 for the ION-CCI group). In the presence of NS1619, the mean threshold intensities were significantly increased in the ION-CCI rats. *p < 0.05, **p < 0.01. Properties of TG Neurons. TG: trigeminal ganglia; RMP: rest membrane potential; ION-CCI: chronic constriction injury of the infraorbital nerve; fAHP: fast after-hyperpolarization. *There is a significant difference between the Sham and the ION-CCI group.

Functional changes in BKCa currents in sham and ION-CCI rats
As a selective antagonist of BKCa, IbTX is often used to confirm the BKCa current (19,41). In our study, in 33.3% (eight of 24) of small and medium TG neurons from the sham rats, application of IbTX (100 nM) reduced outward currents by 39.9 ± 9.0% (n = 8) at the membrane potential of+80 mV. In 18.5% (five of 27) of small and medium TG neurons from the ION-CCI rats, application of IbTX (100 nM) reduced outward currents by 47.0 ± 8.4% at the membrane potential of +80 mV (data not shown). We then found that IbTX-sensitive BKCa currents was significantly reduced in the ION-CCI rats (n = 11) compared with that in the sham rats (n = 10, Figure 4(a), (b), p < 0.05). The amplitude of currents at the membrane potential of +80 mV in the ION-CCI group was 74.5 ± 1.0% of that in the sham group (Figure 4(b), p < 0.05). I-V curve analysis also showed that BKCa currents were strongly depressed after ION-CCI (Figure 4(c)). The G-V curves were not significantly different between the two groups (Figure 4(d)).
Changes in BKCa currents in TG neurons from the sham and ION-CCI rats on postoperative day 15. ((a), (b)) Typical recordings of BKCa currents in neurons from sham and ION-CCI rats, respectively. In TG neurons from the sham (n = 10) and ION-CCI (n = 11) groups, compared with those in the sham group, ION-CCI neurons 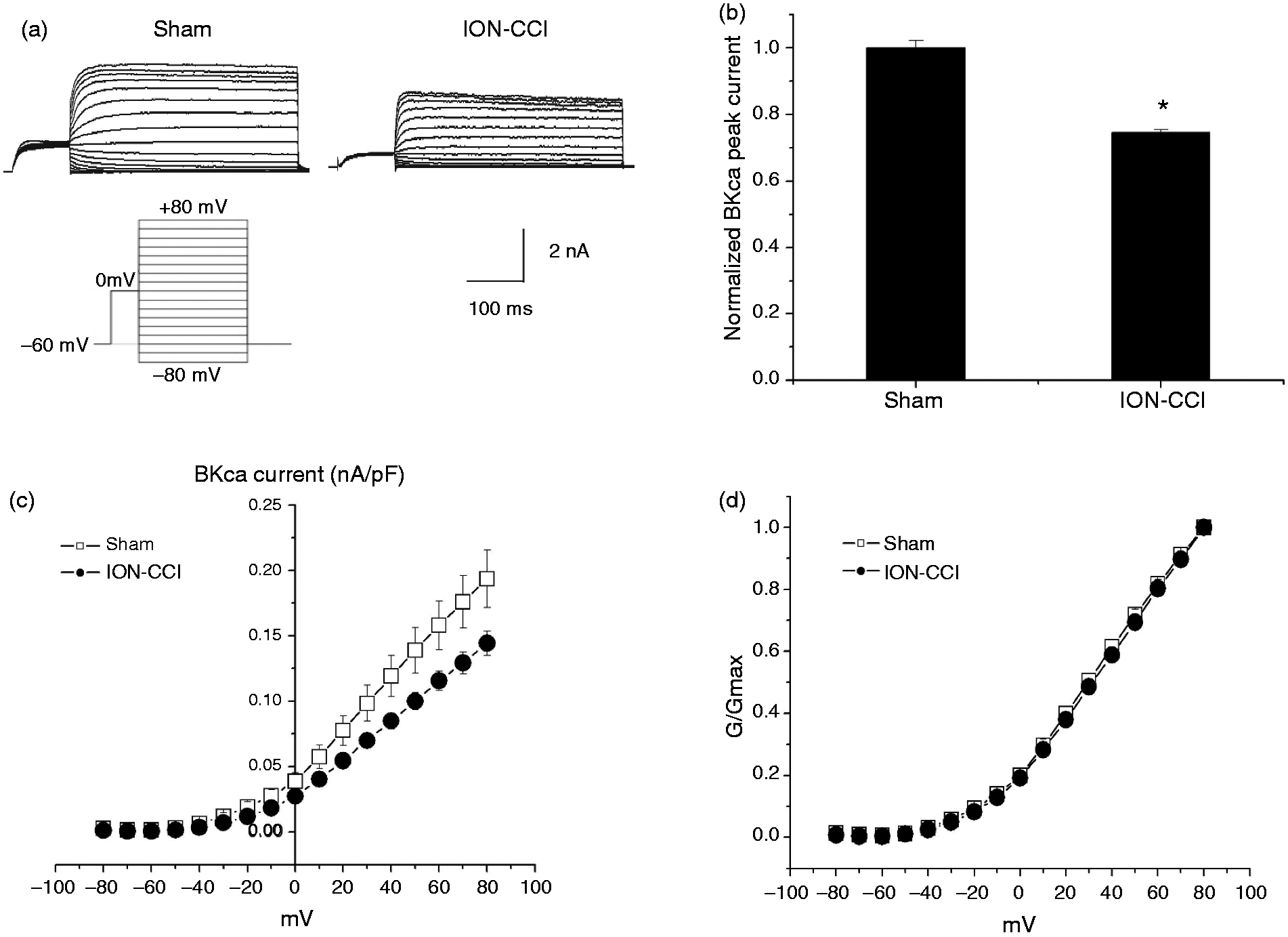
ERK1/2 was activated in ipsilateral TG following ION-CCI
Since ERK activation contributes to changes in membrane excitability as a result of direct or indirect phosphorylating kinases, key receptors, and ion channels (20,21,42), we then investigated whether ERK1/2 was activated in response to ION-CCI. The phosphorylated ERK1/2 in the ipsilateral TG from the sham rats (n = 6) and ION-CCI rats (n = 6) was determined by Western blotting. As shown in Figure 5(a), the amount of total ERK protein was not altered while the level of ERK1/2 phosphorylation was significantly increased in the ipsilateral TG on postoperative day 15 (1.45 ± 0.16) compared with that of the sham group (Figure 5(a), n = 6, p < 0.05).
(a) The active phosphorylated form of ERK in TG from the sham and ION-CCI rats and protein levels of total ERK in the same blot (left) on postoperative day 15. Representative gel images and summary data show that intensity of pERK/ERK is increased in ION-CCI rats compared with that in the sham rats (right). n = 6 for each group, *p < 0.05 compared with that in the sham group. (b) Target injection of U0126 (an ERK antagonist, 10 µg in 20 μl) alone had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in sham rats (n = 8, p > 0.05). (c) U0126 significantly increased the facial mechanical pain threshold in the ION-CCI rats (n = 8, **p < 0.01). When U0126 (10 µg, 20 μl) was administered, the increase of threshold started at 20 minutes and reached a maximum at 30 minutes (5.6 ± 1.2 g) after injection.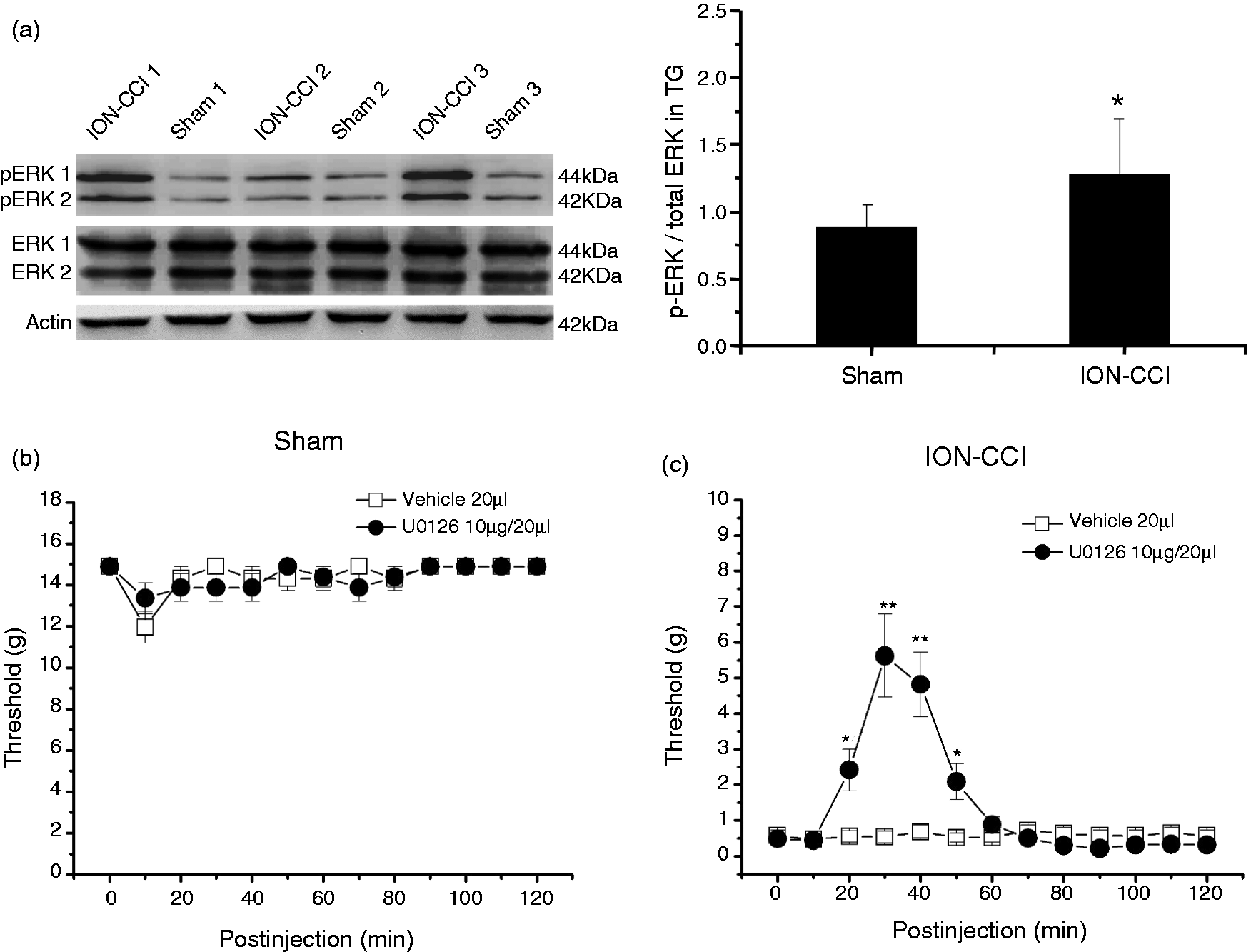
Target injection of U0126 (10 µg, 20 µl), a highly selective inhibitor of ERK kinase, had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in sham rats (Figure 5(b), n = 8, p > 0.05), while it significantly increased the threshold in the ION-CCI rats (Figure 5(c), n = 8, p < 0.01). When U0126 (10 µg, 20 µl) was administered, the increase of facial mechanical pain threshold started at 20 minutes and reached a maximum at 30 minutes (5.62 ± 1.16 g) after injection (Figure 5(c)).
ERK1/2 inhibitor U0126 enhanced BKCa currents in ION-CCI TG neurons
BKCa currents in ION-CCI TG neurons following application of U0126 (1 µM) are shown in Figure 6. After application of U0126 (1 µM) for two minutes, the amplitude of BKCa currents was significantly increased 27.2 ± 2.9% (Figure 6(a), (b), n = 9, p < 0.05). I-V curve analysis also showed that BKCa currents were strongly increased after application of U0126 (Figure 6(c), n = 9). The G-V curves were not changed after application of U0126 (Figure 6(d), n = 9).
Effect of U0126 on BKCa currents in TG neurons. (a) Representative recordings of BKCa currents in a neuron in the absence and presence of U0126 (1 µM), respectively. (b) The peak currents of BKCa showed the significant increase in the presence of U0126 (1 µM, n = 9,*p < 0.05). (c) The IA-voltage relationship curves showed the differences in the absence and presence of U0126 (1 µM, n = 9). U0126 exhibited a significant increase in the mean BKCa currents compared with those of the control. (d) The G-V curve of BKCa was not changed in the presence of U0126 (1 µM, n = 9).
p38 was activated in ipsilateral TG following ION-CCI
The phosphorylated p38 in the ipsilateral TG from the sham rats (n = 6) and ION-CCI rats (n = 6) was determined by Western blotting. As shown in Figure 7(a), the amount of total p38 protein was not altered while the level of p38 phosphorylation was significantly increased in the ipsilateral TG on postoperative day 15 (1.98 ± 0.38) compared with that of the sham group (Figure 7(a), n = 6, p < 0.05).
(a) The active phosphorylated form of p38 in TG from the sham and ION-CCI rats and protein levels of total p38 in the same blot (left) on postoperative day 15. Representative gel images and summary data show that intensity of p-p38/p38 is increased in ION-CCI rats compared with that in the sham rats (right). N = 6 for each group, *p < 0.05 compared with that in the sham group. (b) Target injection of SB203580 (a selective p38 antagonist, 10 µg in 20 μl) alone had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in sham rats (n = 8, p > 0.05). (c) SB203580 significantly increased the facial mechanical pain threshold in the ION-CCI rats (n = 6, **p < 0.01). When SB203580 (10 µg, 20 μl) was administered, the increase of threshold started at 30 minutes and reached a maximum at 60 minutes after injection.
Target injection of SB203580 (10 µg, 20 µl) had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in sham rats (n = 8, p > 0.05) (Figure 7(b)). However, SB203580 significantly increased the facial mechanical pain threshold in ION-CCI rats (n = 6) (from 1.14 ± 0.09 g at 30 minutes, and increased to the maximum at 60 minutes for 8.61 ± 1.35 g) compared with the vehicle group (0.58 ± 0.16 g at 30 minutes, at 60 minutes for 0.58 ± 0.16 g) (p < 0.01, n = 6, Figure 7(c)).
p38 inhibitor SB203580 enhanced BKCa currents in ION-CCI TG neurons
BKCa currents in ION-CCI TG neurons following application of SB203580, an inhibitor of p38 MAPK (10 µM), are shown in Figure 8. SB203580 10 µM (n = 11), 30 µM (n = 10) and 100 µM (n = 10) at two minutes, the amplitude of BKCa currents in the ION-CCI group TG neurons was significantly increased, SB203580 10 µM (from 0.16 ± 0.02 to 0.19 ± 0.02 nA/pF), SB203580 30 µM (from 0.17 ± 0.02 to 0.20 ± 0.02 nA/pF) (Figure 8(a), (b), p < 0.01). I-V curve analysis also showed that BKCa currents were strongly increased after application of SB203580 (Figure 8(c), n = 11 for the 10 µM group, n = 10 for the 30 µM group). The G-V curves were not changed after application of SB203580 (10 µM, 30 µM, Figure 8(d)).
Effect of SB203580, a p38 MAPK antagonist on the BKCa currents in TG neurons. (a) Representative recordings of BKCa currents in a neuron in the absence and presence of SB203580 (10 µM). (b) The peak currents of BKCa show a significant increase in the presence of SB203580 (n = 11 for 10 µM, n = 10 for 30 µM, **p < 0.01). (c) The IA-voltage relationship curves show the differences in the absence and presence of SB203580 (10 µM, n = 11). SB203580 exhibits a significant increase in the mean BKCa currents compared with those of control. (d) The G-V curve of BKCa was not changed in the presence of SB203580 (10 µM, n = 11).
JNK was activated in ipsilateral TG following ION-CCI
The phosphorylated JNK in the ipsilateral TG from the sham rats (n = 6) and ION-CCI rats (n = 6) was determined by Western blotting. As shown in Figure 9(a), the amount of total JNK protein was not altered while the level of JNK phosphorylation was significantly increased in the ipsilateral TG on postoperative day 15 (1.94 ± 0.34) compared with that in the sham group (Figure 9(a), n = 6, p < 0.05).
(a) The active phosphorylated form of JNK in TG from the sham and ION-CCI rats and protein levels of total JNK in the same blot (left) on postoperative day 15. Representative gel images and summary data show that intensity of pJNK/JNK was increased in ION-CCI rats compared with that in the sham rats (right). N = 6 for each group, *p < 0.05 compared with that in the sham group. (b) Target injection of SP600125 (a selective JNK antagonist, 10 µg in 20 μl) alone had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in sham rats (n = 8, p > 0.05). (c) SP600125 significantly increased the facial mechanical pain threshold in the ION-CCI rats (n = 6, **p < 0.01). When SP600125 (10 µg, 20 μl) was administered, the increase of threshold started at 30 minutes and reached a maximum at 60 minutes after injection.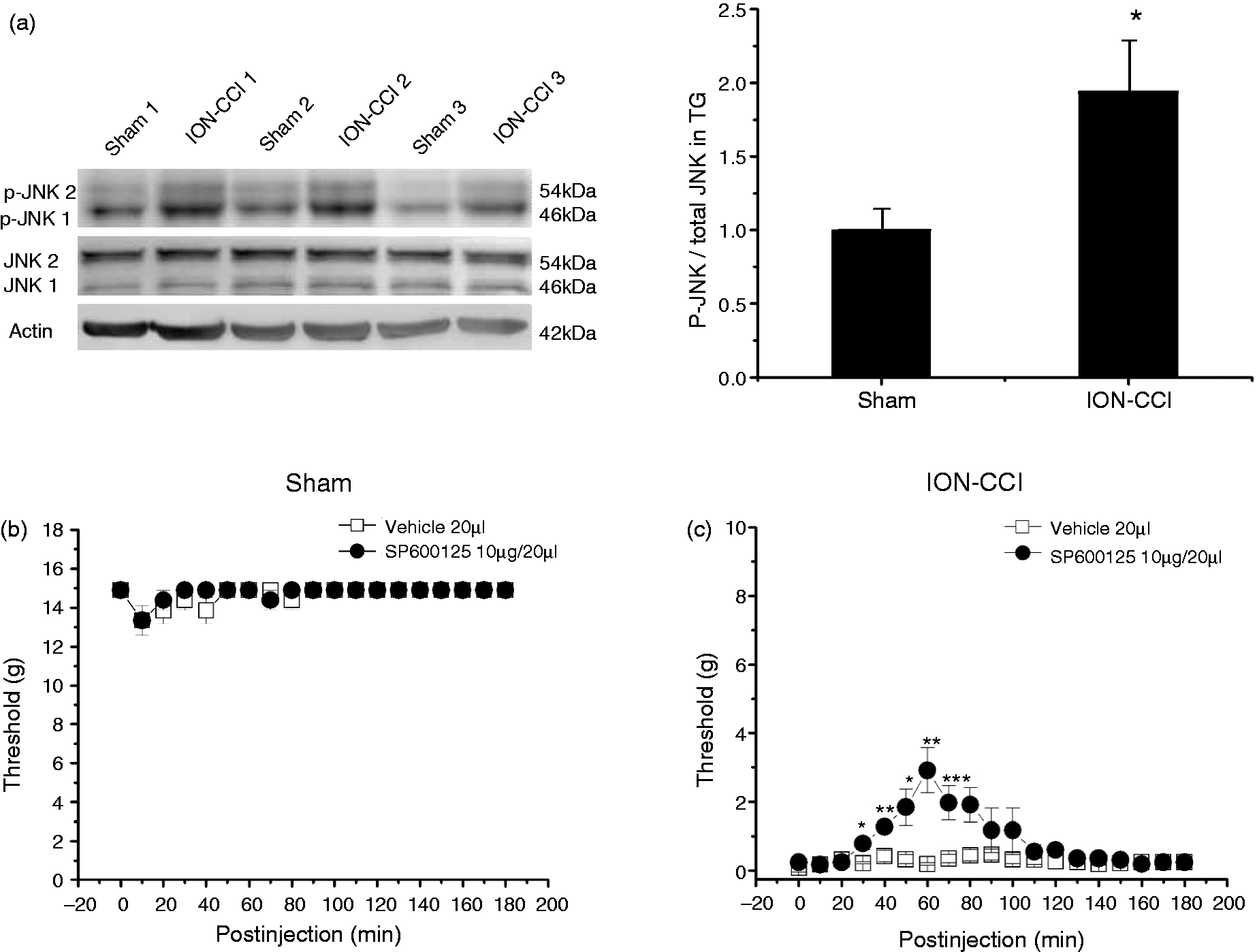
Target injection of the JNK antagonist SP600125 (10 µg, 20 µl) had no effect on the facial mechanical pain threshold in response to the stimulus of Von-Frey hair filaments in the sham rats (n = 8) (p > 0.05) (Figure 9(b)). However, SP600125 significantly increased the facial mechanical pain threshold in ION-CCI rats (n = 6) (from 0.78 ± 0.18 g at 30 minutes, to 80 minutes, increased to the maximum at 60 minutes for 2.90 ± 0.66 g) compared with the vehicle group (n = 6) (0.22 ± 0.03 g at 30 minutes, at 60 minutes for 0.19 ± 0.03 g) (Figure 9(c), p < 0.01).
JNK-inhibitor SP600125 did not enhance BKCa currents in ION-CCI TG neurons
BKCa currents in ION-CCI TG neurons following application of SP600125 (an inhibitor of JNK) (10 µM) are shown in Figure 10. With SP600125 10 µM (n = 9) and 100 µM (n = 9) at two minutes, the amplitude of BKCa currents in the ION-CCI group TG neurons was not significantly changed (Figure 10(a), (b), p > 0.05). I-V curves were not changed after application of SP600125 (Figure 10(c), n = 9). The G-V curves were not changed after application of SP600125 (Figure 10(d), n = 9).
Effect of SP600125, a JNK antagonist on the BKCa currents in TG neurons. (a) Representative recordings of BKCa currents in a neuron in the absence and presence of SP600125 (10 µM). (b) The peak currents of BKCa showed no significant effect in the presence of SP600125 (10 µM, n = 9, p > 0.05). (c) The IA-voltage relationship curves showed no differences in the absence and presence of SP600125 (10 µM, n = 9). SP600125 had no significant effect in the mean BKCa currents compared with those of controls. (d) The G-V curve of BKCa was not changed in the presence of SP600125 (10 µM, n = 9).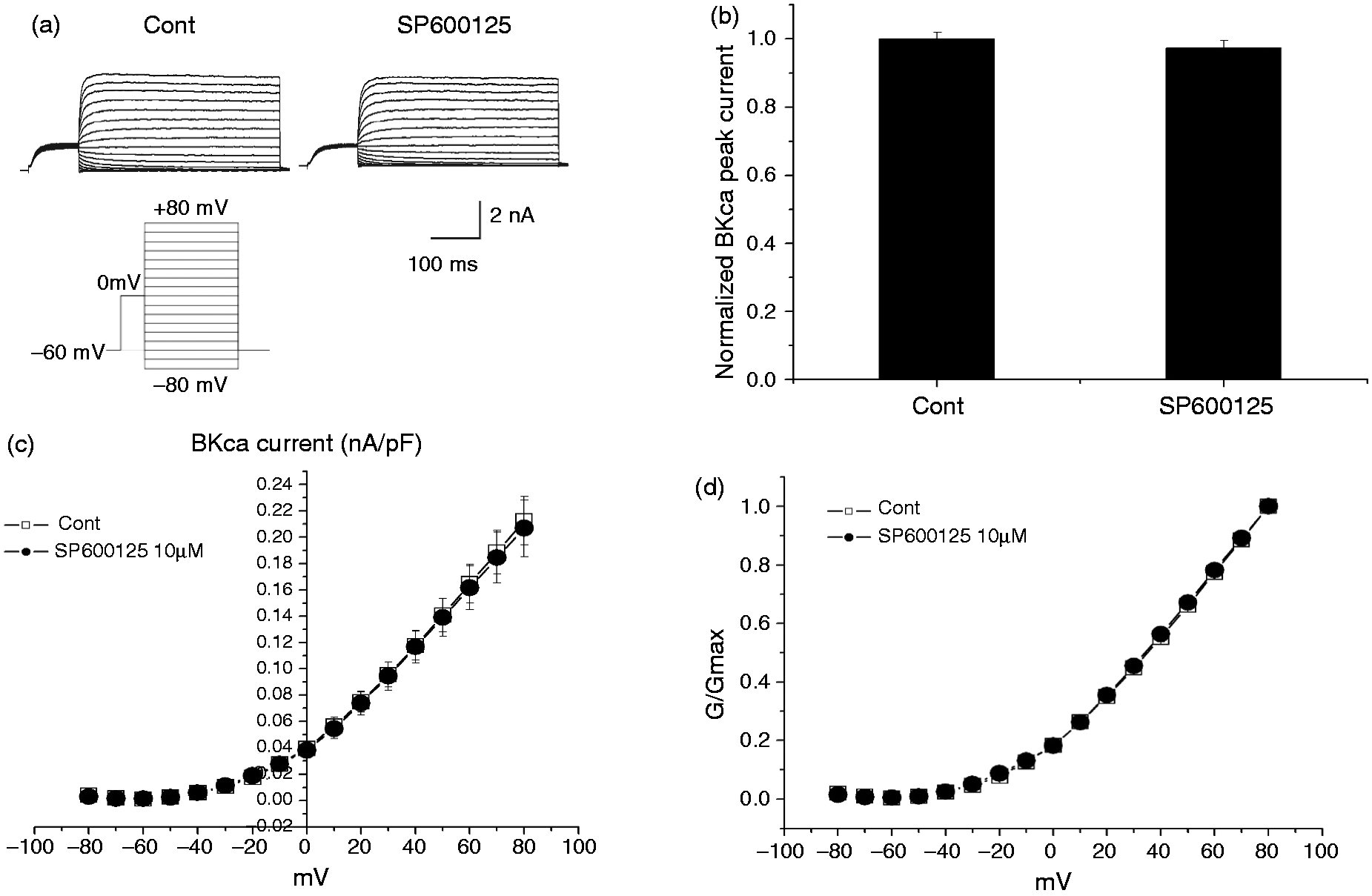
Discussion
In the present study, for the first time, the anti-allodynic effects of the BKCa channel-specific agonist NS1619 were found in the ION-CCI rats. The effect was blocked by the BKCa channel-selective antagonist IbTX. After ION-CCI surgery, BKCa channels in ipsilateral TG were significantly reduced both in the mRNA and protein levels. The ipsilateral TG neurons showed a higher excitability and significantly reduced BKCa currents. The levels of phosphorylated ERK1/2, p38 and JNK in TG were significantly increased. The ERK antagonist U0126 and the p-38 antagonist SB203580 significantly increased BKCa currents in TG neurons and facial mechanical pain threshold in ION-CCI rats, respectively. The JNK antagonist SP600125 demonstrated an anti-allodynic effect but did not affect BKCa currents in TG neurons. These results indicate that the important involvement of mainly ERK and p38 MAPK pathways in modulating BKCa channels in ION-CCI TG neurons. BKCa channels are probably novel therapeutic targets for the clinical treatment of trigeminal neuropathic pain.
BKCa channels are abundantly distributed in the central and peripheral nervous system (43–49), including primary sensory afferent pathways, such as DRG (17) and TG (15,18). Recent studies have shown the steady expression of the constitutive α subunit with varying expression of modulatory β subunits of BKCa channels in different tissues (15,18). β subunits of BKCa channels play a role in the regulation of membrane targeting as well as regulation of gating kinetics (11). For example, β1-subunits are restricted to smooth muscle tissue, β2, β4-subunits are mainly expressed in the human brain (50), and β3-subunits are mainly distributed in the pancreas, heart and testis (11,18). The β-subunits of BKCa channels could lead to different sensitivity and affinity to the calcium flux and thus modulate the function of BKCa channels (11,18). In the present study, we found that in both the mRNA and protein levels, BKCa channels from TG were significantly reduced after ION-CCI compared with those in sham rats. It has also been reported that the expression of BKCa channels was decreased significantly in L4–L5 superficial dorsal horn after ligation of the sciatic nerve (16) and in DRG after injury of the L5–L6 spinal nerve (17). These results indicate that the down-regulation of the BKCa channels could play important roles in trigeminal neuropathic pain.
Chen et al. (17) found a large reduction in IB4-positive DRG neurons after nerve injury with BKCa expression shifted to large DRG neurons, suggesting that decreased BKCa expression primarily occurred in small and medium-sized neurons that were mainly nociceptive afferent neurons (12–14,17,19). In this study, BKCa channel immunoreactivity was aggregated in the cytoplasm and cell membrane and most of the BKCa-immunoreactive neurons were concentrated in small and middle-sized (10–30 µm) TG neurons of both sham and ION-CCI groups. A large reduction of BKCa expression mostly occurred in the medium-sized TG neurons (30–40 µm) from ION-CCI rats compared with that from the sham group, which could contribute to the mechanical allodynia in the ION-CCI rats. Our findings suggested that the reduced expression, not the shifted distribution of BKCa, might contribute to the increased excitability of sensory neurons in TG. The discrepancy in the trigeminal afferent pathway could probably be explained by its unique characteristic that trigeminal afferents innervate the dura mater projecting to second-order neurons of the trigeminal nucleus caudalis, which is different from the spinal afferent pathway (13–15). In the present study we found the changes in BKCa observed in the histological studies were not only confined to the maxillary region (second division of the trigeminal nerve, V2) of the TG but found throughout the ganglia. As far as we understand, there could be several possible explanations for this phenomenon. Firstly, the trigeminal sensory neuron cell bodies with injured axons could alter the phenotypes of adjacent TG neurons. Indeed, the cross-talk between the primary afferent cell bodies occurred in DRG (51) or TG (52). Secondly, primary afferent axons could branch to the other territory through DRG or TG as variable branching patterns of primary afferent axons have been reviewed by Willis and Coggeshall (53). Thus, it is possible that some afferent axons from injured ION (maxillary, V2) may enter the mandibular (V3) or even ophthalmic (V1) regions. Therefore, we assume that down-regulation of BKCa channels both in injured and uninjured nociceptive TG neurons (medium-sized, 30–40 µm) played an important role in the mechanical allodynia after ION-CCI. The underlying mechanisms and specific reasons about how ION-CCI surgery changed BKCa expression throughout the ganglia require further investigation.
BKCa channels have been shown to contribute, in part, to repolarization of action potential and generation of after-hyperpolarization in sympathetic (17), dorsal vagal (54) and hippocampal neurons (55). Scholz et al. (56) demonstrated that the main functions of BKCa channels in small-diameter DRG neurons were to shorten action potential duration, enhance the speed of repolarization, and contribute to fAHP, which leads to the reduced repetitive activity. In our study, I-V curve analysis showed BKCa currents in ipsilateral TG neurons from ION-CCI rats were strongly depressed, which suggested the reduced function of BKCa channels following nerve ligation injury. The electrophysiological results indicated that ION-CCI could, in theory, produce a hyperexcitability of neurons probably through the decrease of amplitude of fAHP induced by the depression of BKCa currents both in injured and uninjured trigeminal sensory neurons throughout the ganglia. Considering the down-regulation of BKCa expression in medium-sized (30–40 µm) nociceptive TG neurons after ION-CCI in histological study, it is more likely that the decreased function of BKCa channels contributes at least in part, to the high-frequency spikes and neuronal hyperexcitability in the ION-CCI rats.
In the present study, the mean threshold intensity for evoking action potentials of TG neurons throughout the TG was significantly lower in the ION-CCI group than in the sham group. Tsuboi et al. (57) reported that changes in K+ current and Ih observed in the uninjured TG neurons reflected an increase in excitability of TG neurons innervated by the ION after inferior alveolar nerve (IAN, a branch of V3 division of the trigeminal nerve) transection, resulting in the development of mechano-allodynia in the area adjacent to the injured IAN innervated region (57). Tseng et al. (58) found that the transection of IAN produced allodynia in the whisker pad (V2 division) of rats; the hyperexcitability of injured IAN TG neurons may initiate the sensitization of uninjured TG neurons, which in turn generated high-frequency firing in thalamic neurons, contributing to neuroplastic changes in the thalamocortical circuits; and producing long-lasting neuropathic pain in the orofacial region (58). In addition, increasing the neurotransmitters/neuromodulators (e.g. substance P (SP)) released from the cell body of TG neurons through a paracrine or autocrine mechanism may also activate the neighboring TG neurons (59,60). Peripheral nerve injury could affect the somatosensory processing in the central nervous system such as the spinal/medullary dorsal horn (51), which then possibly modulates sensitivity of TG neurons. Therefore, we assume that the injured ION TG neurons after surgery could also possibly lead to sensitization of uninjured TG neurons from a neighboring region, mandibular (V3) or even ophthalmic (V1) branch of trigeminal nerve throughout the trigeminal ganglia.
It has been reported that intrathecal injection of NS1619 was able to reverse the mechanical allodynia after ligation of L5 and L6 spinal nerves (17). The anti-allodynic effect of NS1619 could be blocked by BKCa channel blocker IbTX (17). In this study, TG target injection of NS1619 showed the similar anti-allodynic effect that was blocked by IbTX, suggesting that BKCa channel activation might play a vital role in the trigeminal afferent pathway. BKCa channels might have no effect on the nociceptive signal transmission in the physiological condition while affecting an anti-allodynic role in trigeminal neuropathic pain state. The suppression of neuronal firing activity by NS1619 can be attributed to the increases of threshold for action potential firing, the prolonged duration of action potential (19), reduction in action potential frequency and the increases in amplitude of AHP (11,13,14,19). Our present studies demonstrate that NS1619 significantly enhanced the mean threshold intensities for evoking action potentials in TG neurons from the ION-CCI rats while failing to affect those from the sham rats. Also, in the presence of NS1619, the mean threshold intensity of TG neurons from the ION-CCI rats was significantly enhanced compared with that in the sham rats. A possible explanation is that the reduction in threshold caused by BKCa channel inhibition to evoke action potential was associated with the ectopic spontaneous discharges, and thus contributed to neuropathic pain from nerve injury (61,62). Therefore, BKCa channel agonist NS1619 could be a potential therapeutic approach for the clinical treatment of trigeminal neuropathic pain.
There are three major molecules in the MAPK family: ERK, p38 and JNK, that are known to have different functions in sensory processing (20–25). Alvarez et al. have found light and moving stroking of the infraorbital skin resulted in strong, bilateral up-regulation of pERK-1/2 in the insular cortex of ION-CCI animals but not in the sham rats (63). Similar studies showed that reduced Kv4.2-mediated A-type currents increased neuronal excitability in the spinal cord dorsal horn via ERK activation (64,65). In the present study, we found that ION-CCI significantly increased the level of ERK1/2 phosphorylation in TG. The ERK1/2 antagonist U0126 significantly enhanced BKCa currents in TG neurons and increased facial mechanical pain threshold after ION-CCI. Since activation of ERK contributes to changes in membrane excitability as a result of direct or indirect phosphorylating kinases, key receptors, and ion channels (20,21), these results suggest that the effect of ERK phosphorylation on BKCa channels might contribute to the mechanisms underlying abnormal pain after trigeminal nerve injury.
In addition, increasing evidence also suggests that p38 MAPK is a key factor for the development of neuropathic pain: P-p38 is elevated in DRG neurons following peripheral inflammation and nerve injury, and intrathecal inhibition of p38 reduces nerve injury-induced pain behavior (20,66–70). Hagenacker et al. (70) found that myricetin reduced voltage-activated potassium channel currents (IK(V)) in DRG neurons to increase neuronal excitability by p38-dependent mechanisms. BKCa channels both in principal cells (PCs) and intercalated cells (ICs) in the cortical collecting duct have also been shown to be stimulated by inhibition of ERK and p38 MAPKs (71). In the present study, the p38 MAPK antagonist SB203580 significantly enhanced BKCa currents in rat TG neurons and increased facial mechanical pain threshold after the ION-CCI. The possible explanation is that p38 is capable of direct modulation of BKCa channels.
Compared with ERK and p38, much less is known about how JNK regulates pain (20). Some evidence has shown that following nerve injury, JNK in DRGs and the spinal cord is rapidly activated by phosphorylation, and JNK expression remained elevated for weeks until the neuron dies or until its axon regenerated (72–74), which suggests that the JNK cascade might also be a critical signaling pathway for the onset and the maintenance of neuropathic pain. In the present study, JNK expression in TG was elevated after ION-CCI. However, no significant change in BKCa currents was found in TG neurons when using the selective JNK antagonist SP600125. Compared with SB203580 and U0126, The JNK antagonist SP600125 could slightly enhance the facial mechanical pain threshold after the ION-CCI operation. These results suggest that the inhibition of the JNK MAPK pathway induced analgesia in the ION-CCI rats, probably via modulating the other pain-related channels or proteins rather than BKCa channels.
Conclusions
Our results indicate the important involvement of mainly ERK and p38 MAPK pathways in modulating BKCa channels in ION-CCI TG neurons. BKCa channels represent a new therapeutic target for the clinical treatment of trigeminal neuropathic pain.
Clinical implications
Chronic constriction injury of the infraorbital nerve (ION-CCI) down-regulated large-conductance calcium-activated potassium (BKCa) channels, increased phosphorylation of extracellular signal-regulated kinase (ERK)1/2, p38 and c-Jun N-terminal kinases (JNK) in trigeminal ganglia (TG), and decreased BKCa currents in cultured TG neurons. The opener of BKCa channels NS1619 and the ERK1/2, p38 and JNK antagonists (U0126, SB203580 and SP600125) reversed the facial allodynia in ION-CCI rats. U0126 and SB203580 increased BKCa currents in ION-CCI TG neurons. Mainly ERK and p38 mitogen-activated protein kinase (MAPK) pathways are involved in modulating BKCa channels, which represents a new therapeutic target for clinical treatment of trigeminal neuropathic pain.
Footnotes
Funding
This work was supported by the Key Program of Shanghai Science and Technology Committee (No. 08JC1405400) and The Key Laboratory of Molecular Neurobiology of Ministry of Education.
Conflict of interest
None declared.
Acknowledgments
Thanks are due to Prof Arthur W. English and Dr Benjamin G. Huey from the School of Medicine at Emory University for their careful reading and commenting on the grammar and expression of English in the manuscript.
