Abstract
Background
The vascular effects of acute calcitonin gene-related peptide (CGRP) inhibition are well described, but the effects of sustained inhibition warrant further exploration in humans.
Objectives
The objective of this article is to assess the effects of sustained CGRP inhibition on blood pressure, heart rate, and ECGs in healthy women ≥40 years of age.
Methods
In this double-blind, placebo-controlled study, 31 women (mean age = 56) were randomized to receive placebo or an anti-CGRP monoclonal antibody at doses up to 2000 mg. Participants were confined for seven days and followed for 168 days. Cardiac telemetry was conducted for eight hours after infusion completion. Hemodynamic assessments and ECGs were conducted six times during Day 1 and periodically for three months.
Results
No clinically relevant changes in systolic or diastolic blood pressure, heart rate, or ECG parameters (RR, PR, QRS, or QTcF) were observed when comparing baseline vs. post-dose time-points or in-between groups. No significant changes were seen for adjusted QTcF (baseline subtracted and placebo and baseline subtracted). No significant differences or relevant abnormalities were seen when comparing parameters obtained at Tmax vs. any other time-point.
Conclusion
Sustained CGRP inhibition was not associated with hemodynamic or ECG changes in a population at an increased age risk for cardiovascular events.
Introduction
Calcitonin gene-related peptide (CGRP) is a well-studied neuropeptide of relevance for migraine pathophysiology (1,2). Jugular levels of CGRP are increased during migraine attacks, and intravenous (IV) CGRP administration induces migraine-like headache in most individuals with migraine (3,4). CGRP seems to modulate migraine processes peripherally (vasodilation, inflammation, and protein extravasation), at the trigeminal ganglion, and inside the brain (5). Inhibition of CGRP has demonstrated efficacy in the acute treatment of episodic migraine (6–8).
CGRP is a potent systemic vasodilator and its role in the control of cardiovascular homeostasis has received considerable attention (9–12). Preclinical and clinical studies have found that transient CGRP inhibition with receptor antagonists (CGRP-RA) is not associated with coronary vasoconstriction (13) and that under ischemic conditions, inhibition had no influence on heart infarct area (14). Olcegepant, a CGRP-RA, had no effect on systemic and cerebral hemodynamics (15) while telcagepant, another CGRP-RA, did not affect vasodilation induced by certain antihypertensive medications (16), nor did it negatively influence treadmill exercise time (TET) in patients with angina (17).
Most studies on the vascular consequences of inhibiting CGRP were conducted in short-duration exposure paradigms using medications that have short half-lives. A better understanding of the consequences of prolonged CGRP inhibition on cardiovascular parameters is necessary, since several monoclonal anti-CGRP antibodies (with relatively long half-lives) are currently under development (18).
LBR-101 is a fully humanized monoclonal antibody (mAb) that potently and selectively binds to both isoforms (α- and β) of CGRP, with a half-life of approximately 45 days. During Phase 1 development, the dose range tested was from 0.2 mg up to 2000 mg (19). Herein we report the cardiovascular safety results of a Phase 1 study (Study B0141008) which, relative to prior Phase 1 studies, enrolled an older female population and tested the highest doses of LBR-101. The aim of this study was to assess the effects of sustained CGRP inhibition on blood pressure, heart rate (HR), and electrocardiograms (ECGs) in healthy women ≥40 years of age.
Methods
Overview
The study was conducted at two sites in the United States under an Investigational New Drug (IND) application, and was registered in clinicaltrials.gov (NCT01511497). It was approved by the Independent IRB of Ft Lauderdale, FL (currently Shulman Associates IRB Inc, approval number 00003563). All subjects provided informed consent to participate.
In this double-blind, placebo-controlled, single-dose, dose-escalation study, 31 women (mean age = 56 years) were randomized to receive placebo or LBR-101 (T1/2 ∼ 45 days, Tmax ∼ 2 hours after the end of infusion) at doses of 300, 1000, 1500, or 2000 mg administered as a single one-hour IV infusion. Three of these doses are higher than the highest dose currently being tested in the Phase 2b program. Participants were confined and intensively monitored in a clinical research unit for seven days after the infusion. After being discharged, participants returned to the clinic for additional visits periodically through Study Day 168.
Participants were generally healthy, over 40 years of age, without known cardiovascular disease. Known cardiovascular risk factors (e.g. high cholesterol, diabetes) and significant history of cardiovascular disease or vascular ischemia were considered exclusion criteria; however, use of tobacco products up to the equivalent of five cigarettes per day was permitted. Participants were also excluded if their baseline ECG showed clinically significant abnormalities in the 12-lead surface ECG, including sinus pauses >2 seconds, second- or third-degree heart block or other abnormalities judged clinically significant by the investigator.
Assessments
All physical examinations, LBR-101 samples for pharmacokinetic analysis (PK), hemodynamic assessments, and ECGs were conducted six times during Day 1 (at pre-dose, at the end of infusion, and at three, five, eight, and 12 hours after the start of the infusion). On Days 2–7 the above were assessed once daily. After unit discharge, these assessments were repeated on Day 14, and at one, two, and three months after the LBR-101 infusion. PK samples were collected at the same times, with additional PK sampling at Weeks 6, 10, and 24.
Supine blood pressure was measured with the subject’s arm supported at the level of the heart, and recorded to the nearest mm Hg after five minutes of rest. The same arm (preference to the dominant arm) was used throughout the study. Likewise, properly calibrated automated devices and the same size blood pressure cuff were used to measure blood pressure at each assessment.
ECG and telemetry
Telemetry and pulse oximetry data were collected using a centralized system for storage and data analysis. To establish a baseline, continuous cardiac telemetry was initiated two hours before dosing on Day 1 and continued until eight hours after completion of infusion.
ECGs were collected as a single recording at screening and on Day 168. For all other assessments, triplicate recordings were made. All scheduled ECGs were performed after the subject had rested quietly for at least 10 minutes in a supine position and efforts were made to reduce noise. The triplicate 12-lead ECGs were obtained approximately two to four minutes apart, and are reported as the mean of the three ECG assessments. The primary ECG analysis was performed by a fully blinded ECG core laboratory assessment meeting International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) E14 criteria (20).
Analysis of pharmacokinetic parameters of LBR-101
Plasma samples were analyzed for LBR-101 concentrations using a validated chemiluminesence enzyme-linked immunosorbent assay (ELISA) method. Plasma concentration-time data were analyzed using WinNonlin (version 5.2, Pharsight Inc, Mountain View, CA). Non-compartmental analysis was performed to determine the PK parameters of LBR-101.
Data analysis
Clinical data, vital signs, and ECG parameters were summarized using descriptive tables and summary statistics. Summary tables are presented stratified by dose. Mean values and 95% confidence intervals (CI) are presented. Absolute values and changes from baseline for systolic (SBP) and diastolic blood pressure (DBP) and HR were summarized by treatment and time post-dose. Maximum absolute values and maximum changes from baseline for vital signs were also summarized. Absolute values and changes from baseline for the ECG parameters, heart rate, QT interval, PR interval, and QRS duration were summarized by cohort, treatment, and time post-dose. The QT interval was adjusted for the HR using the Fridericia’s (QTcF) heart rate correction formula and the primary analysis was the placebo-subtracted change from baseline at each time-point (delta-delta QTcF). The time-point with the largest delta-delta QTcF value was defined as the maximal mean QTcF (20). In addition, the change from baseline for the QTcF was also determined. The number of participants reaching specific categorical values (QTcF increase compared to baseline of 30–50 ms, or >50 ms) or absolute thresholds of QTcF >470 ms; or >500 ms was determined. In addition to the QTcF, the analysis plan examined the prevalence of subjects having a PR interval ≥ 200 msec or a QRS interval ≥ 110 msec.
Results
Sample disposition, adverse event rate, and pharmacokinetics
A total of 31 healthy women (80.6% white) participated in this study. Eight were randomized to receive placebo (mean age = 55, SD = 4.8), while six received 300 mg (mean age = 54, SD = 7.0), six received 1000 mg (mean age = 55, SD = 6.4), six received 1500 mg (mean age = 59, SD = 3.0), and five received 2000 mg (mean age = 57.4, SD = 5.0).
Adverse events during duration of the study (related to treatment or not) were reported by 63.3% of women receiving placebo and 60.9% of those receiving LBR-101. Serious or severe adverse events were not seen. Clinically relevant laboratory abnormalities were not observed.
The pharmacokinetic profile of LBR-101 was consistent with what is seen with mAbs (Figure 1). After a one-hour infusion of LBR-101, Tmax was achieved quickly (one to five hours). The group mean terminal half-life (T1/2) ranged from 42 to 53 days.
Plasma concentration of LBR-101 after a single intravenous administration as a function of dose and time after dose.
Heart rate
HR was similar comparing participants receiving placebo or LBR-101 (Figure 2). At baseline, both groups had resting HR of approximately 60 beats/minute. At Tmax (shortly after the end of the infusion) values were 64.0 (95% CI = 61.7–70.3) for those receiving placebo vs. 64.1 (61.0–67.1) for the LBR-101 group. HR remained similar across groups for the duration of the study. The same pattern was seen after stratifying LBR-101 subjects according to dose level.
Heart rate in individuals receiving LBR-101 or placebo. The figure displays pooled doses of LBR-101. Not all time-points are presented to maintain clarity. Data are presented as mean and 95% confidence intervals.
Blood pressure
SBP and DBP were also similar as a function of treatment group (Figure 3). Pooling all LBR-101 doses together, at the end of the infusion mean SBP was 117.4 mmHg (95% CI = 110.3–125.2) for placebo subjects and 118.2 mmHg (95% CI = 111.4–125.1) for LBR-101 subjects (the pre-dose baselines were almost identical). The SBP values remained at all later time-points to post-dose Day 168. A dose-response effect was not observed.
Systolic and diastolic blood pressure in individuals receiving LBR-101 or placebo.
The end of the infusion mean DBP values were 70.5 mmHg (95% CI = 62.9–78.9) for the placebo group vs. 68.9 mmHg (95% CI = 63.0–74.9) for the treated group (the pre-dose baselines were almost identical). Approximately three hours after the end of infusion, mean values were 70.7 mmHg (95% CI = 64.7–76.7) for placebo and 69.8 (95% CI = 65.5–74.1) for treated. Values remained similar between groups across the study period. Stratification for dose level showed similar findings.
ECG parameters
All ECG parameters were similar across all groups, including placebo, for all time-points. There were no relevant changes, specific trends, dose-response findings, or major individual changes seen.
Double-delta of the mean QTcF interval as a function of study dose and time since onset of infusion.

QTcF: QT interval after Fridericia’s correction. The delta-delta is defined as the difference of the QTcF interval for a time point relative to the baseline assessment, subtracted by the same difference in those receiving placebo. Measurements are in msec.
Assessments that are temporally closest to the CMax.
Doses higher than those being tested in the Phase 2 program.
Figure 4 describes the individual plots for the single delta QTc analysis (values at specific time-points subtracted by correspondent values at baseline). Increases above 50 ms or absolute values of 470 ms (changes that are considered to be clinically relevant) were not observed after dosing.
Baseline-subtracted (single-delta) QTcF in individuals receiving LBR-101 or placebo.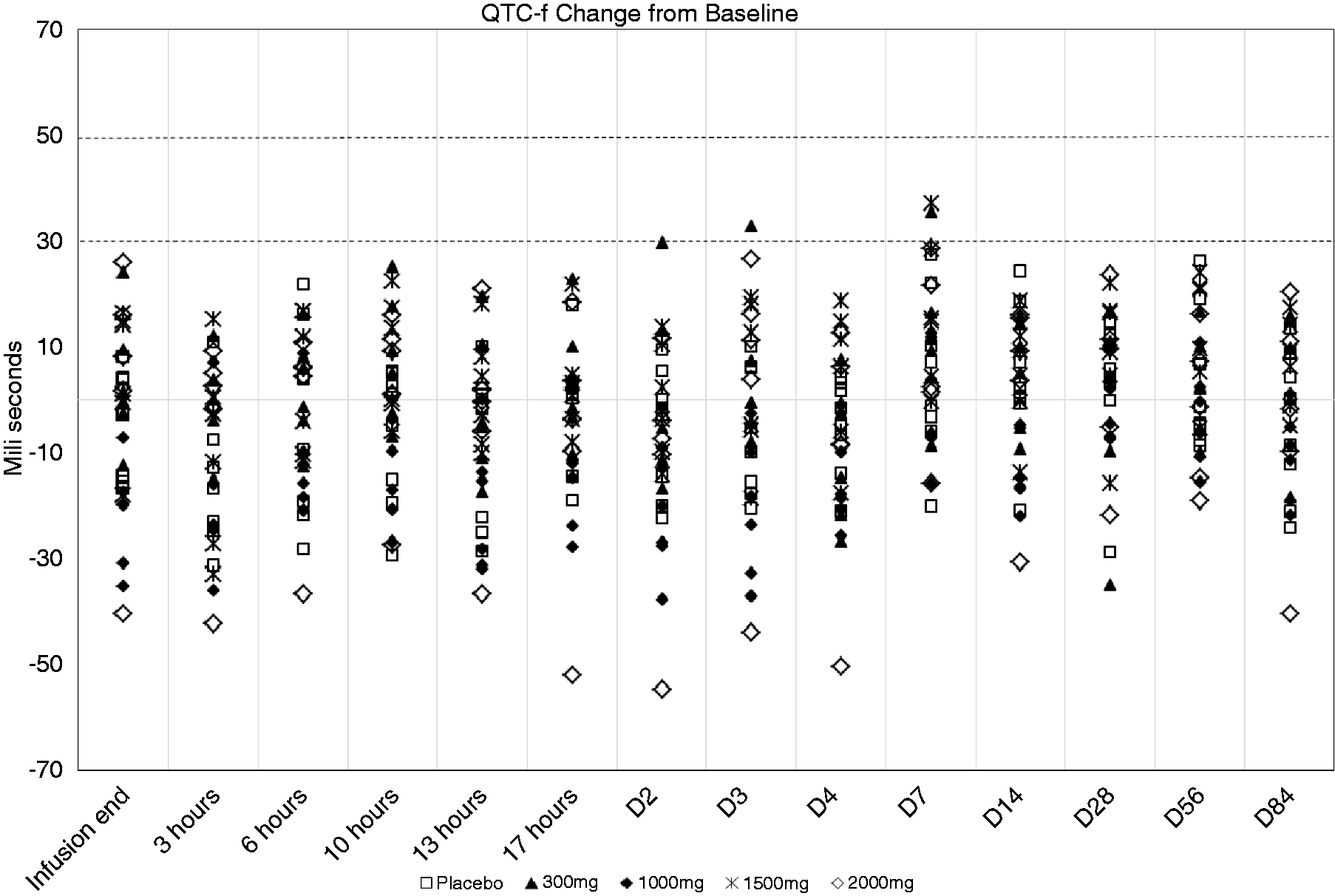
As for the PR intervals, around Cmax mean values were 158.3 ms for the placebo group relative to 167.4 ms in the 300 mg arm, 151.5 in the 1000 mg arm, 154.1 in the 1500 mg arm, and 169.1 in the 2000 arm. No clinically relevant measurements of PR interval, defined as ≥200 msec, were noted during the study. As for the QRS interval, mean values at three hours ranged from 80.8 ms (300 mg arm) to 87.1 (2000 mg arm). In those receiving placebo, values were 83.3 ms. Clinically relevant abnormalities (≥110 msec) were not observed.
Discussion
CGRP is distributed throughout the central and peripheral nervous systems and is often co-localized with other peptides in C fibers (21). Its relevance to migraine has been firmly established (22). Five distinct, small-molecule CGRP-RAs have demonstrated clinical proof-of-efficacy for the acute treatment of migraine (6, 23–26). Preclinical studies suggest that, as opposed to triptans, CGRP-RAs do not induce vasoconstriction (13, 14, 27). Unfortunately, their potential use as preventive medications has been limited by issues of liver toxicity associated with chronic administration. Anti-CGRP antibodies will hopefully avoid liver toxicity and reduce drug-drug interaction concerns since their elimination would follow physiologic processes for protein catabolism.
Both isoforms of CGRP are potent natural vasodilators. At the microvascular level, CGRP is around 10-fold more potent than the prostaglandins and 100–1000 times more potent than other classic vasodilators (28). In addition to its potency, CGRP also differs from other vasodilator substances in that it has a particularly long duration of action (29). The vasodilatory activity of CGRP extends to a wide variety of tissues and organs (30).
Animal studies showed that infusion of CGRP decreased the likelihood of onset of ischemia-reperfusion arrhythmias (31). In animal ischemia models, exogenously administered CGRP was found to improve the contractile function of the heart in dogs (32) and increased post-ischemic coronary artery flow in pigs (10). However, studies have failed to demonstrate that CGRP, when given during ischemia, had any protective effect as assessed by a reduction in infarct area (10). Further studies demonstrated that CGRP, given prior to ischemia, has a protective effect on infarct size and contractile function (14). This would suggest CGRP can play a role in preconditioning, a process by which tissue is protected from a future prolonged ischemic bout (33).
Many of the theoretical concerns that emerged from the in vitro and in vivo characterization of CGRP pathway have been investigated in human studies during the robust development of the CGRP-RAs. These molecules did not cause vasoconstriction of the human coronary arteries under normal conditions (13), of animal arteries under ischemic conditions (14), did not influence exercise-time in patients with angina (17), and did not counterbalance the vasodilation induced by certain antihypertensive medications (16). However, because CGRP-RAs inhibit the pathway for short periods of time, these findings cannot necessarily be extrapolated to the more prolonged inhibition that would occur with mAb administration.
LBR-101 is a fully humanized CGRP mAb with a long half-life in humans. Although a certain degree of accumulation occurs with monthly dosing until steady state is reached, the two highest doses tested in this study yielded higher LBR-101 plasma concentrations than the highest doses being tested in Phase 2, even after repeated dosing.
The pharmacological properties of LBR-101 have been well characterized and the safety of the product has been examined in the preclinical and clinical programs. A maximally tolerated dose has not been found (19). The safety characterization of the drug included two dedicated studies in monkey using very large doses (a single-dose safety pharmacology study at 100 mg/kg and a three-month repeat-dose study, 10 to 300 mg/kg, weekly IV dosing). In both studies, animals were intensively monitored. No biologically relevant changes were seen for any of the assessed cardiovascular parameters, including blood pressure and ECGs (34). In humans, LBR-101 has been administered to 118 subjects throughout the Phase 1 program. There have been no findings that caused concern about cardiovascular safety in individuals receiving the drug (19).
Because protein therapeutics are designed to be highly specific, adverse reactions are most commonly a result of exaggerated pharmacology (e.g., from prolonged CGRP inhibition) (35). CGRP does not seem to influence cardiac electrical conduction or repolarization, and its inhibition does not appear to cause any important ECG changes. In two monkey studies, relevant ECG changes were not seen at doses up to the highest dose of 300 mg/kg given weekly. Furthermore, during the LBR-101 Phase 1 program, no relevant differences were seen with any ECG parameters (including QTcF). The current study enrolled an older female population and administered the highest doses of LBR-101 tested to date, and also failed to identify any relevant ECG abnormalities. LBR-101 had no meaningful effect on ventricular repolarization as a function of administered dose or as a function of Cmax. A dose response was not seen among surrogate parameters (HR, BP, ECG, and laboratory tests), paralleling the clinical adverse event profile of the drug, where a maximally tolerated dose has yet to be identified (19).
Caution is advised when interpreting the results. Four major cardiovascular effects could be of concern with CGRP inhibition: medication-induced hypertension, counterbalancing the effect of antihypertensive drugs that have vasodilatory properties, inhibition of stress (or ischemia)-induced vasodilation, and impairment of cardioprotective mechanisms. We emphasize that these concerns are not addressed by the current study, which enrolled healthy volunteers. We can fairly say that examining both early changes (related to the maximal concentration of the drug) and also late changes (related to prolonged inhibition of CGRP) revealed no statistically significant or clinically significant changes in hemodynamic parameters in healthy individuals, but not necessarily in individuals at risk for cardiovascular disease. Since CGRP causes vasodilation, it is conceivable that inhibition of CGRP could translate into hypertension in patients with underlying hypertension though the lack of any hemodynamic effects in this study at the high doses studied reduces that likelihood. As mentioned, the current study was not designed to address other concerns of CGRP inhibition, such as impairment of preconditioning (or effects of CGRP inhibition in a population at risk for ischemic events). The most comprehensive human study to test this latter topic assessed the influence of supra-therapeutic doses of telcagepant (up to three times the highest dose being tested at the time) on TET in patients with reproducible exercise-induced stable angina with ischemic ST-segment depression. Patients performed treadmill exercise at Tmax (2.5 hours after the dose). The incidence of ischemic ST-segment depression ≥1 mm was 83.9% in those receiving placebo, 90.7% in those receiving telcagepant 600 mg, and 85.7% in those receiving telcagepant 900 mg. TET was not significantly different across groups and all other data were similar across groups. The authors suggested that the broad redundancy in vasodilatory mechanisms might preserve the compensatory vasodilatory response during myocardial ischemia, even in the presence of CGRP-receptor antagonism (17).
In summary, prolonged inhibition of CGRP with LBR-101 was not associated with hemodynamic or 12-lead ECG changes in a population at mild-moderately increased age risk for cardiovascular events. Thus, preclinical and clinical studies are encouraging and support further development of medications chronically inhibiting the CGRP pathway, as no other class of migraine medications, including those inducing vasoconstriction such as ergotamine and the triptans (36–38), has been so intensively tested in this regard.
Clinical implications
Monoclonal antibodies anti-calcitonin gene-related peptide (CGRP) have a long half-life and are being developed for the preventive treatment of episodic and chronic migraine. Prolonged inhibition of CGRP with LBR-101 is not associated with hemodynamic or 12-lead electrocardiogram (ECG) changes in a population at mild-moderately increased age risk for cardiovascular events.
Footnotes
Funding
This work was supported by Pfizer Inc.
Conflicts of interest
MEB, SW, MB, and RE are full-time employees of Labrys Biologics Inc.
