Abstract
Objectives
Migraine with aura is a common neurological disorder, and differentiation from transient ischemic attack or stroke based on clinical symptoms may be difficult.
Methods
From an MRI report database we identified 33 patients with migraine with aura and compared these to 33 age-matched ischemic stroke patients regarding perfusion patterns on perfusion-weighted imaging (PWI)-derived maps: time to peak (TTP), mean transit time (MTT), and cerebral blood flow and volume (CBF, CBV).
Results
In 18/33 (54.5%) patients with migraine with aura, TTP showed areas of hypoperfusion, most of these not limited to the territory of a specific artery but affecting two or more vascular territories. In patients with migraine with aura, TTP (1.09 ± 0.05 vs. 1.47 ± 0.40, p < 0.001) and MTT ratios (1.01 ± 0.10 vs. 1.19 ± 0.21, p = 0.003) were significantly lower compared to patients with ischemic stroke. In contrast to this, CBF and CBV ratios did not differ significantly between both groups.
Conclusions
Migraine aura is usually associated with a perfusion deficit not limited to a specific vascular territory, and only a moderate increase of TTP. Thus, hypoperfusion restricted to a single vascular territory in combination with a marked increase of TTP or MTT may be regarded as atypical for migraine aura and suggestive of acute ischemic stroke.
Introduction
Migraine with aura is a common neurological disorder with a prevalence of approximately 4% (1). The pathophysiological basis of migraine aura is cortical spreading depression, a state of spreading electrical discharge and depolarization of neurons and glia followed by complete recovery of cortical electrical activity. The phenomenon is usually accompanied by a short-lasting hyperperfusion for about one to two minutes and subsequent mild hypoperfusion for about one to two hours (2,3). The associated clinical symptoms include visual, sensory, perceptual, motor as well as behavioral alterations and are characterized by a progressive onset and slow evolution (4).
In assessing a patient with migraine with aura, taking the history is of particular importance and may help to make the distinction between migraine aura and acute ischemic stroke (4). Under certain circumstances, however, when taking a patient's history is hampered by aphasia or the patient reports a significant change in aura phenomenology, a differentiation of migraine aura, transient ischemic attack and stroke based on the reported clinical symptoms and the neurological examination may be difficult or even impossible and this may lead to acute therapy with intravenous thrombolysis (5,6).
More recently, perfusion-weighted magnetic resonance imaging (MRI) (PWI) has been used for a more precise characterization of hemodynamic changes in migraine aura in several case reports and smaller case series (7–21). In most cases, PWI demonstrated a mild uni- or bilateral hypoperfusion overlapping different vascular territories. Consequently, a comprehensive evaluation of patients with suspected migraine with aura using multimodal MRI might be useful in daily clinical routine to facilitate the diagnosis and support therapeutic decisions.
In this study we sought to investigate perfusion patterns on PWI in patients with migraine with aura and to compare these with the well-defined perfusion patterns in patients with acute ischemic stroke. Additionally, we aimed to evaluate diffusion-weighted MRI (DWI) for the presence of acute ischemic lesions and MR angiography (MRA) for vascular abnormalities in this particular patient group.
Methods
Patients
From a prospectively maintained MRI report database (2005--2013) we identified 698 patients with suspected acute ischemic stroke who underwent a standard stroke MRI protocol including PWI. For the present study we identified all those patients in whom clinical workup including multimodal MRI finally led to the diagnosis of migraine with aura according to the diagnostic criteria of the International Headache Society (22). For each patient with migraine with aura, we selected an age-matched patient with diagnosis of acute ischemic stroke. The demographic details and clinical presentation were abstracted from the case records. The study was approved by the local ethics committee, Medizinische Ethik-Kommission II, Medizinische Fakultät Mannheim (2013-815R-MA).
MRI studies
MRI was performed on a 1.5-T MR system (Magnetom Avanto or Sonata, Siemens Medical Systems, Erlangen, Germany) within six hours after onset of symptoms. A standardized protocol was used in all patients including (1) transverse, coronal and sagittal localizing sequences followed by transverse oblique contiguous images with a slice thickness of 5 mm aligned with the inferior borders of the corpus callosum (applied on sequences 2 to 6); (2) T1-weighted images; (3) T2-weighted images; (4) DWI; (5) fluid-attenuated inversion recovery (FLAIR) images; (6) PWIs following the first pass of contrast bolus through the brain; and (7) a three-dimensional (3D) time-of-flight MR angiography (TOF-MRA) with a slice thickness of 1 mm. PWI was acquired using a gradient-echo echo planar imaging sequence. The contrast agent gadoteric acid (Dotarem, Guerbet, Aulnay-sous-Bois, France) was bolus injected by a power injector (Spectris MR injection system, Medrad, Volkach, Germany) with a dose of 0.1 mmol/kg of body weight at a rate of 4 ml/sec. MRI was performed with informed consent of the patient.
Post-processing
The post-processing of the raw PWIs was performed by a specific software, Signal Processing In NMR (SPIN, The MRI Institute for Biomedial Research, Detroit, MI, USA) (23). Deconvolution with singular value decomposition (SVD) was used to create quantitative maps of mean transit time (MTT), cerebral blood flow (CBF) and cerebral blood volume (CBV). The position of the arterial input function (AIF) was automatically determined by using the maximum concentration (Cmax), time to peak (TTP) and first moment MTT (fMTT). The concentration-time curve for arteries has short fMTT, short TTP and high Cmax. Twenty voxels that best fit these properties were selected. Then the concentration-time curves of these voxels were averaged, smoothed and truncated to avoid the second pass of the tracer.
MRI analysis
MRIs were analyzed by two neuroradiologists in consensus according to the established criteria (24). First, PWI was analyzed visually, with the readers blinded to the clinical information and all other sequences. To obtain information on hemodynamic alterations, calculated TTP images demonstrating the delay of the contrast agent arrival in the brain parenchyma were used for visual analysis. TTP maps were classified as showing a normal perfusion pattern or areas of hypoperfusion. The topography and the corresponding dominant arterial territory of the hypoperfused areas were determined according to the maps by Tatu et al. (25,26) and categorized in (1) anterior cerebral artery (ACA), (2) middle cerebral artery (MCA), (3) posterior cerebral artery (PCA) territory, and (4) brain stem and cerebellum.
After one week, the remaining sequences including DWI and TOF-MRA were analyzed. DWIs were assessed for acute ischemic lesions. The topography and the corresponding dominant arterial territory of the identified ischemic lesions were determined and categorized corresponding to the hypoperfused areas on PWI as described above. Vascular pathologies were recorded on coronal and transverse views of 3D MRA reconstruction. Additionally, vessels of the circle of Willis were determined on the 3D TOF stack by use of OsiriX, a multidimensional image navigation and display software (27). MRA was classified as (1) normal, (2) dilated peripheral branches in comparison to the unaffected hemisphere, (3) decreased visualization of peripheral branches in comparison to the unaffected hemisphere, (4) stenosis, and (5) vessel occlusion.
Furthermore, the generated perfusion maps were qualitatively assessed by use of SPIN: A region of interest (ROI) was placed in an area of hypoperfusion on the generated maps (TTP, MTT, CBF, CBV) as well as mirrored to the contralateral unaffected hemisphere. Finally, ratios between the physiological estimates (TTP, MTT, CBF, CBV) of the area of hypoperfusion and of the contralateral mirror ROI were determined.
Statistical analysis
All statistical analyses were performed using Statistical Product and Service Solutions (SPSS) statistics for Windows (Release 17.0; SPSS, Chicago, IL, USA). Comparison of age was performed using the Student's t test. Patient demographics and clinical presentation were analyzed using chi2-based tests. Likewise, MRI and MRA findings were analyzed using chi2-based tests. Association of hypoperfusion in specific localizations and clinical symptoms was determined by use of chi2-based tests. Comparison of TTP, MTT, CBF and CBV ratios was performed using the Student's t test. All statistics was performed with a 0.05 level of significance.
Results
Baseline characteristics and clinical presentation
Demographic characteristics and clinical presentation in patients with migraine with aura and acute ischemic stroke.
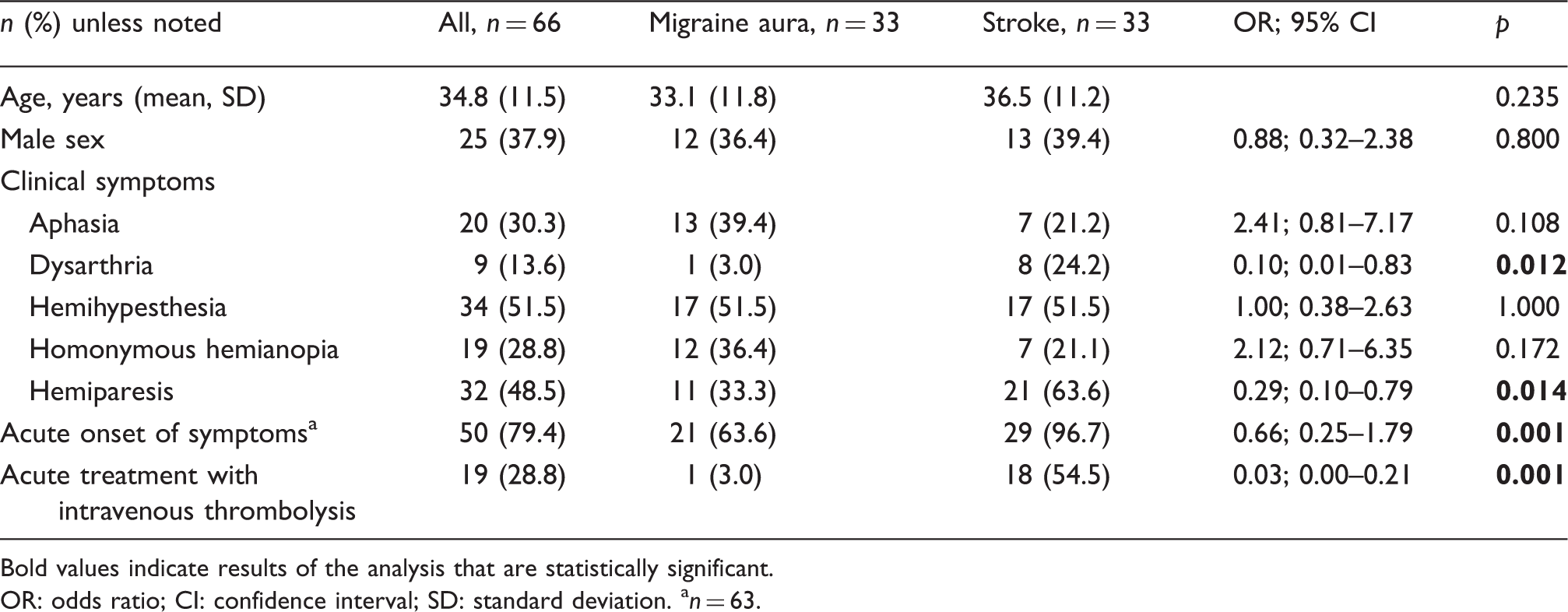
Bold values indicate results of the analysis that are statistically significant.
OR: odds ratio; CI: confidence interval; SD: standard deviation. an = 63.
MRI findings in patients with migraine with aura
MRI was performed within a mean time of 128.3 ± 45.5 minutes after onset of aura symptoms. Overall, PWI demonstrated an area of hypoperfusion in 18/33 (54.5%) patients with migraine with aura. In the remaining 15/33 (45.5%) patients, PWI was unremarkable. In only one of 33 patients (3.0%), PWI showed a perfusion deficit exclusively in the PCA territory while in the remainder the hypoperfused area involved more than one vascular territory. Quantitatively, the most pronounced changes were found in CBF (0.76 ± 0.16) and CBV (0.77 ± 0.16) followed by TTP (1.09 ± 0.05) and MTT (1.01 ± 0.10) signal ratios. In 11/18 (61.1%) patients an occipital predominance of hypoperfusion was observed, and in five of 18 patients (27.8%) hypoperfusion involved the occipital and parietal lobe (see Figure 1.1 (b)–(d) and 1.2 (b)–(d)), and in the remaining two (11.1%) patients hypoperfusion extended to the temporal and frontal lobe (see Figure 1.3 (b)–(d)). There was no clear association between clinical presentation and localization of perfusion abnormalities. For example, in patients with visual disturbances PWI demonstrated less often a hypoperfusion in the occipital lobe (four of 11 vs. seven of 11), occipital and parietal lobe (one of five vs. four of five) as well as in the occipital, parietal, temporal and frontal lobe (one of two vs. one of two) in comparison to patients with other clinical presentation (p = 0.706). Comparable findings were observed in patients with aphasia (p = 0.351) or hemiparesis (p = 0.516).
Three examples of multimodal MRI in migraine aura. In the first case, DWI is unremarkable (1 (a)), while perfusion maps (time to peak, cerebral blood flow, cerebral blood volume) show a mild hypoperfusion in the left parietal and occipital lobes (1 (b)–(d)). On MRA a decreased visualization of peripheral MCA branches on the left side can be observed (1 (e)). In the second case, DWI shows a small ischemic lesion in the head of the left caudate nucleus (2 (a)). The perfusion maps demonstrate a marked hypoperfusion in the left parietal and occipital lobes (2 (b)–(d)). MRA is unremarkable (2 (e)). In the third case, DWI is unremarkable (3 (a)) while perfusion maps (time to peak, cerebral blood flow, cerebral blood volume) demonstrate an extensive hypoperfusion in the left frontal, parietal and occipital lobes (3 (b)–(d)). MRA demonstrates subtle dilation of the left MCA branches (3 (e)).
DWI demonstrated a subacute ischemic infarction in the head of the caudate nucleus in only one of 33 (3.0%) patients with complex congenital heart disease probably indicating cardiac embolism (see Figure 1.2 (a)). The ischemic lesion was regarded as an incidental finding not related to the migraine aura.
MRA demonstrated side-to-side differences in peripheral branch visualization in seven of 33 (21.2%) patients. A mild dilation of the MCA/PCA branches in the affected hemisphere was observed in two (6.1%) patients, and a mildly decreased visualization of MCA/PCA branches in five (15.2%) patients. In the remaining 26 (78.8%) patients MRA was unremarkable. For an example of decreased visualization of MCA/PCA branches or mildly dilated vessels, see Figure 1.1 (e) and 1.3 (e), respectively.
MRI findings in patients with acute ischemic stroke
PWI demonstrated an area of hypoperfusion in 20/33 (60.6%) patients with acute ischemic stroke. In 17/33 patients (51.5%) PWI showed a perfusion deficit exclusively in the MCA or PCA territory while in only three of 33 (9.1%) the hypoperfused area involved more than one vascular territory. Quantitatively, the most pronounced changes were found in TTP (1.47 ± 0.40) and CBF (0.69 ± 0.29), followed by MTT (1.19 ± 0.21) and CBV (0.81 ± 0.32) signal ratios.
DWI demonstrated ischemic infarction in the MCA territory in 14/33 (42.4%), in the PCA territory in nine of 33 (27.3%), in the brain stem and/or cerebellum in four of 33 (12.1%), as well as in multiple territories in six of 33 (18.2%) patients.
MRA demonstrated vessel stenosis or occlusion in 15/33 (45.5%) patients, and side-to-side differences in peripheral branch visualization in only two of 33 (6.1%) patients. In the remaining 16/33 (48.5%) patients, MRA was unremarkable.
Comparison of MRI findings in patients with migraine with aura and acute ischemic stroke
MRI findings in patients with migraine with aura and acute ischemic stroke.
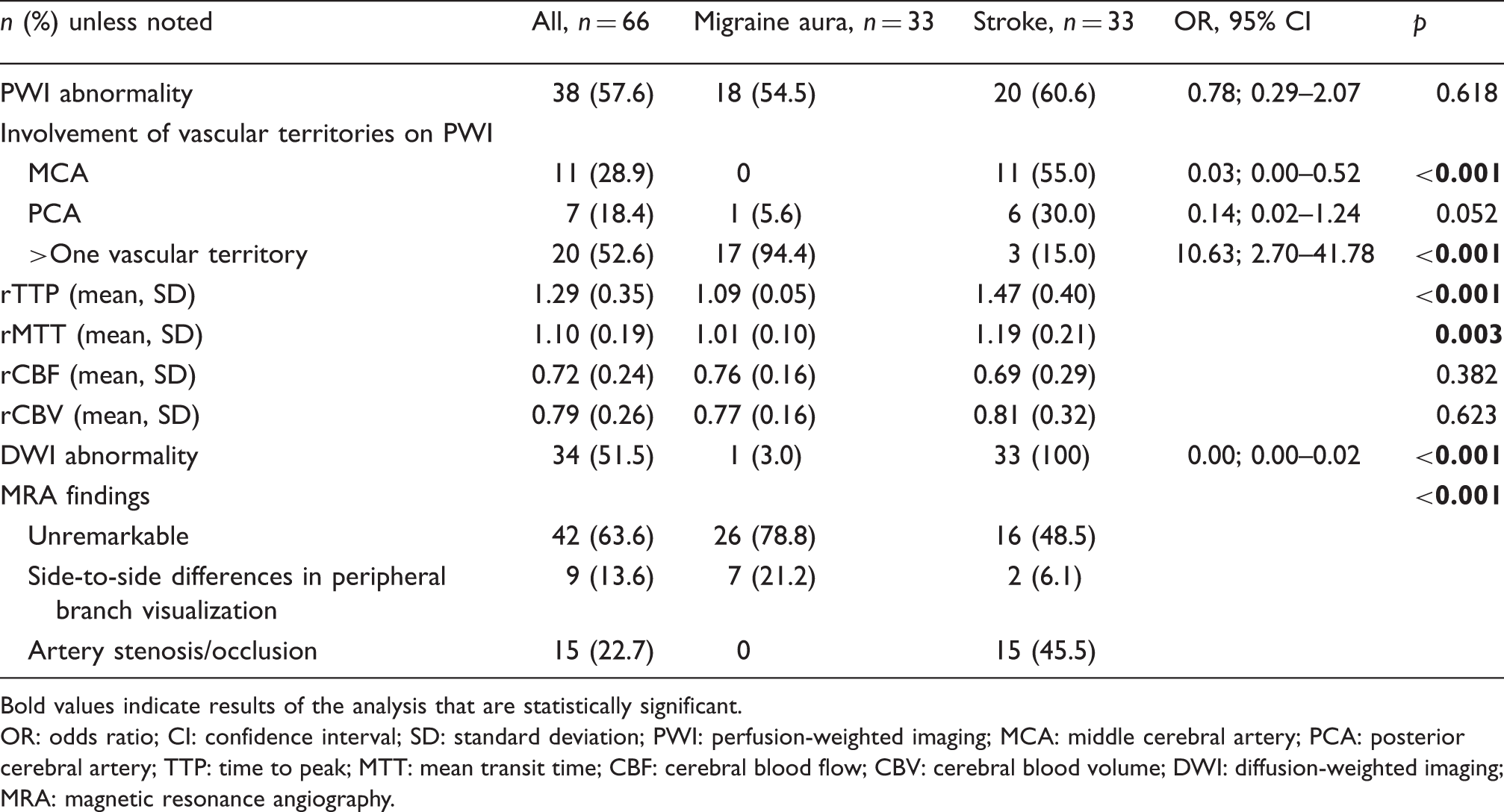
Bold values indicate results of the analysis that are statistically significant.
OR: odds ratio; CI: confidence interval; SD: standard deviation; PWI: perfusion-weighted imaging; MCA: middle cerebral artery; PCA: posterior cerebral artery; TTP: time to peak; MTT: mean transit time; CBF: cerebral blood flow; CBV: cerebral blood volume; DWI: diffusion-weighted imaging; MRA: magnetic resonance angiography.
Discussion
Perfusion changes in migraine with aura and acute ischemic stroke.

TTP: time to peak; MTT: mean transit time; CBF: cerebral blood flow; CBV: cerebral blood volume.
While DWI demonstrated one or more acute ischemic lesions in all stroke patients, DWI was unremarkable in all patients suffering from migraine with aura except in one patient with complex congenital heart disease. In this patient a small subacute ischemic lesion of the left caudate nucleus was observed most probably due to cardiac embolism. Acute migrainous infarction is a very rare subtype of ischemic stroke. With migraine aura in most cases not being linked pathophysiologically to hypoperfusion with rCBF and rCBV not falling below critical limits (31), in the above-mentioned patient the observed hypoperfusion was limited to the parietal and occipital lobe and did not reach the basal ganglia. Otherwise, there has been evidence that cardiac emboli might trigger migraine with aura (32) and possibly migraine aura was triggered by ongoing cardiac emboli in this patient. Consequently, we regarded the ischemic lesion as an incidental finding and did not make up the diagnosis of migrainous infarction.
On MRA none of the patients with migraine aura demonstrated an acute vessel occlusion typically observed in acute ischemic stroke (24). However, in a subset of patients with migraine with aura minor vascular abnormalities like dilated vessels or decreased visualization of peripheral branches was observed. Comparable findings were reported by Iizuka et al. in two patients with familial hemiplegic migraine (18). This observation may be of particular value for differentiation between patients with migraine aura and stroke patients with a so-called total mismatch. In the latter, a large area with hypoperfusion on PWI is found due to arterial occlusion while DWI is unremarkable (33,34).
The present study has some limitations. First, this is a retrospective clinical study of moderate size. However, to our knowledge this is the largest series of patients investigating perfusion abnormalities in migraine with aura in detail in comparison to acute ischemic stroke. Second, as a further consequence of the retrospective study design, a more comprehensive description of aura symptoms and evolution was not possible. Third, the hospital-based retrospective study design might cause several types of bias and statistical errors such as selection bias, sample bias, or image-based selection bias (35). Fourth, follow-up MRI was available in only a subset of patients and consequently no information about the evolution of perfusion abnormalities could be obtained.
In conclusion, migraine with aura is associated with moderate increase of TTP and a decrease of CBF and CBV not reaching critical levels nor limited to a specific vascular territory and normal MRA findings in the majority of patients. In a subset of patients subtle vascular abnormalities like dilated vessels or reduced visualization of peripheral branches can be observed. Knowledge of these MRI characteristics can help to distinguish migraine with aura from acute ischemic infarction and avoid unnecessary thrombolytic treatment in these patients.
Clinical implications
Migraine aura is usually associated with a perfusion deficit not limited to a specific vascular territory, and a moderate increase of time to peak (TTP). Hypoperfusion restricted to a single vascular territory with a marked increase of TTP and/or mean transit time (MTT) is atypical for migraine with aura and suggestive of acute ischemic stroke. Magnetic resonance angiography (MRA) may demonstrate subtle vascular abnormalities like dilated vessels or reduced visualization of peripheral branches in a subset of patients with migraine aura. Knowledge of these magnetic resonance imaging (MRI) characteristics can be useful to differentiate migraine with aura and ischemic infarction and thus avoid unnecessary thrombolytic treatment in these patients.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
