Abstract
Background
During migraine, trigeminal sensory nerve terminals release calcitonin gene-related peptide (CGRP), inducing nociception and vasodilation. Applied on the skin, capsaicin activates the transient receptor potential vanilloid type 1 (TRPV1) channel and releases CGRP from sensory nerve terminals, thus increasing dermal blood flow (DBF). Using capsaicin application and electrical stimulation of the forehead skin, a trigeminal nerve-innervated dermatome, we aimed to develop a model to measure trigeminal nerve-mediated vasodilation in humans.
Methods
Using laser Doppler imaging, forehead DBF responses to application of capsaicin (0.06 mg/ml and 6.0 mg/ml) and saline, with and without iontophoresis, were studied in healthy subjects. The within-subject coefficient of variation (WCV) of repeated DBF measurements was calculated to assess reproducibility.
Results
Maximal DBF responses to 6.0 mg/ml capsaicin with and without iontophoresis did not differ (Emax 459 ± 32 and 424 ± 32 arbitrary units (a.u.), WCV 6 ± 4%). In contrast, DBF responses to 0.06 mg/ml capsaicin were significantly larger with than without iontophoresis (Emax 307 ± 60 versus 187 ± 21 a.u., WCV 21 ± 13%). Saline with iontophoresis significantly increased DBF (Emax: 245 ± 26 a.u, WCV 11 ± 8%), while saline application without iontophoresis did not affect DBF.
Conclusion
Topical application of capsaicin and electrical stimulation induce reproducible forehead DBF increases and therefore are suitable to study trigeminal nerve-mediated vasodilation in humans.
Keywords
Introduction
Migraine is a neurovascular disorder (1) involving: (i) activation of the trigeminovascular system with the primary dysfunction located in brainstem centers regulating vascular tone and pain sensation (1); (ii) release of vasoactive neuropeptides from trigeminal sensory nerves including calcitonin gene-related peptide (CGRP) (2,3); and (iii) vasodilation of intracranial arteries and of the extracranial branches of the external carotid artery (4,5). Indeed, it has been reported that plasma concentrations of CGRP are elevated during migraine headache and they diminish to normal levels after treatment with triptans in parallel with alleviation of headache (6). In view of the key role of CGRP in the pathophysiology of migraine, CGRP and its receptors have now become a novel drug target for antimigraine therapy (7).
CGRP is predominantly located in sensory neurons and perivascular (including trigeminal sensory) nerves (2,3,8–10). Upon release in the cranial circulation, CGRP dilates cranial blood vessels and is involved in nociception (8). Release of endogenous CGRP from trigeminal nerves can experimentally be induced by electrical stimulation (6,11,12) or by chemical stimulation with capsaicin (13–15), the latter through activation of the transient receptor potential vanilloid type 1 (TRPV1) cation channel (16,17). Administration of capsaicin in the carotid artery has been reported to induce vasodilation of the extracranial circulation of pigs (18) and dogs (19). These capsaicin-induced vasodilator responses can be blocked by CGRP receptor antagonists (18,20). Similarly, both electrically and capsaicin-induced intracranial dural vasodilation in rats (21) and mice (22) are mediated by trigeminal CGRP release. Likewise, topically applied capsaicin on the human forearm skin increases dermal blood flow (DBF), and this response can be prevented by CGRP receptor antagonists (23) as well as by monoclonal antibodies preventing the binding of CGRP with its receptor (24,25). This “peripheral” human capsaicin model has been successful as a biomarker in early clinical drug development by providing proof of target engagement in phase I clinical trials and guiding dose selection for phase II clinical trials. Although the capsaicin-induced increase in DBF of the human forearm was shown to be reproducible and user friendly, its major limitation remains the uncertainty about the translation of peripheral versus cranial vascular responses induced by capsaicin.
Until now straightforward and reproducible human methods for the analysis of trigeminal nerve-mediated vasodilation with minimal demands of the test subject have been lacking. Since the dermatome of the forehead is innervated by the trigeminal nerve, the forehead seems well suited to investigate trigeminal mechanisms. Gazerani et al. have developed a capsaicin model for trigeminal sensitization and have applied the model to investigate for example gender-specific differences and the effect of botulinum toxin type A (26–29). However, this is an invasive model where human subjects receive intradermal capsaicin injections to the forehead, resulting in unilateral, throbbing, headache and symptoms of general trigeminal sensitization. Similarly, May et al. applied subcutaneous capsaicin injections in an experimental pain model to examine the neurovascular mechanisms in cluster headache (30). Participants of this study reported “terrible pain” after the subcutaneous capsaicin injections. We primarily aimed with our study to investigate whether measurement of local blood flow in the forehead region, using laser Doppler imaging, in response to either electrical stimulation or capsaicin application could be used as a novel model to assess local trigeminal nerve-mediated vasodilation in humans. Whereas application of capsaicin induces CGRP release via activation of TRPV1 channels (23), electrical stimulation induces neuropeptide release without direct activation of these channels (31). In recent years interest in transdermal drug delivery techniques that enhance permeation of drugs into the skin has increased (32). One such technique is iontophoresis, where a solution of a compound or drug is put in an electrode reservoir bearing the same charge as the compound or drug of interest. When an electric current is applied to the reservoir, ions set in motion toward the electrode of opposite charge and more easily penetrate into the skin (32). In this study, as a secondary aim, we wanted to investigate whether iontophoresis of capsaicin might improve its delivery on the human forehead, resulting in a higher response and/or a shorter response latency.
Materials and methods
Subjects
Demographics of the study population.

BMI: body mass index; BP: blood pressure; HR: heart rate. Mean (standard deviation (SD)).
Experimental setup
Experiments were performed in a quiet, temperature-controlled room. Participants were not allowed to use any drugs for 48 hours preceding the experiment and they fasted for at least three hours before the start of the experiment. To investigate short-term reproducibility, two research visits with three to four days in between were scheduled for the participants to study potential neuropeptide depletion and receptor desensitization in this period. Some of the subjects participated in multiple experiments; in these cases the time between two subsequent experiments was at least 14 days to exclude any potential interference of previous experiments. The forehead DBF responses induced by topical application of capsaicin, vehicle and physiological saline with and without iontophoresis were measured with a laser Doppler perfusion imaging device (LDPI; Periscan PIM 3 system, Perimed AB, Järfälla, Sweden). Subjects received topical applications of capsaicin (0.06 mg/ml and 6.0 mg/ml), vehicle (a mixture of ethanol 100%, Tween 20 and distilled water; 3:3:4) or physiological saline (0.9% NaCl, 0.5 ml in each case) on the forehead in electrodes with reservoirs specifically designed for this purpose (Perimed AB, Järfälla, Sweden). For electrical stimulation of the forehead skin, iontophoresis of physiological saline was applied.
Capsaicin was obtained from Sigma-Aldrich (Zwijndrecht, the Netherlands). We decided to use a methodology and dose similar to that previously reported by Van der Schueren et al. (33). In addition, since we intend to use our model also for studies aimed at observing trigeminal hyperreactivity, we furthermore decided to analyze the effect produced by a two log unit lower concentration in an attempt to be able to detect low responses around the activation threshold.
Trigeminal nerve-mediated vasodilation model
For the trigeminal nerve-mediated vasodilation model, we used two electrodes with 0.06 and 6.0 mg/ml capsaicin solution and a cumulative successive rate of currents applied to an electrode containing physiological saline (0.2, 0.4, 0.6, 0.8 and 1.0 mA, each lasting one minute, with an interval of six minutes between successive current intensities). To assess reproducibility these experiments were performed twice at an interval of three to 10 days. The trigeminal nerve-mediated vasodilation model was evaluated in 12 subjects.
In-depth characterization of the trigeminal nerve-mediated vasodilation model
Capsaicin iontophoresis was performed on the forehead by application of a small current (0.4 mA, lasting one minute) to two electrode chambers, filled with either 0.06 mg/ml or 6.0 mg/ml capsaicin in seven subjects. In these seven subjects, a third electrode was placed on the forehead for iontophoresis of vehicle (0.4 mA, lasting one minute).
To evaluate potential effects of electrode location, iontophoresis was performed on two electrodes placed on either the left or right side of the forehead during the same experiment.
To investigate iontophoresis-independent effects of the vehicle, vehicle was topically applied on the forehead without iontophoresis in four subjects.
Data presentation and statistical evaluation
The DBF values are presented in arbitrary units (a.u.) as means ± S.E.M. Student’s t test (paired or unpaired where appropriate) was used to compare maximal responses (Emax) to application and iontophoresis of different solutions for groups with n ≥ 7. Wilcoxon matched pairs test or Mann-Whitney test was applied where appropriate for groups with n < 7 to compare means. Emax was defined as the maximum DBF response in a.u. after subtraction of baseline DBF. Within-subject coefficient of variation (WCV) and intraclass correlation coefficient (ICC) were calculated as measures of reproducibility and agreement. We also performed correlation analysis using the Pearson correlation coefficient and Bland-Altman analysis. The between-subject coefficient of variation (BCV) was also calculated. Sample size calculations, given a type I error probability (α) of 0.05 and a power of 80%, were performed without assumptions on an eventual increase in statistical power because of a paired nature of measurements (unpaired model). Calculations were based on the application without iontophoresis of the 6.0 mg/ml capsaicin solution to detect a DBF response difference of 10%, 20% and 30%. A p value <0.05 was considered to indicate statistical significance.
Results
Forehead application of the capsaicin solutions (0.06 mg/ml and 6.0 mg/ml) with and without iontophoresis and forehead electrical stimulation were well tolerated and side effects other than local redness were not encountered. None of the subjects reported pain.
Trigeminal nerve-mediated vasodilation model
Application without iontophoresis of the 0.06 mg/ml and the 6.0 mg/ml capsaicin solutions resulted in fast increases in DBF (Figure 1) with an Emax of respectively 187 ± 21 and 459 ± 32 a.u. (Table 2). The DBF responses at 30 minutes, averaged for both visits, were 123 ± 10 a.u. for the low and 370 ± 28 a.u. for the high capsaicin concentration (Figure 2(a)). The time to reach Emax was 22 ± 3 and 11 ± 1 minutes for 0.06 and 6.0 mg/ml capsaicin, respectively. The DBF response to electrical stimulation differed from baseline for each applied current ranging from 0.2 to 1.0 mA (Figure 2(b)). The between-visit DBF responses for either of the two capsaicin solutions or electrical stimulation did not differ (Figure 2(a) and (b)).
(a) Illustration of electrode placement. From left to right: electrical stimulation, application without iontophoresis 0.06 mg/ml capsaicin and application without iontophoresis of 6.0 mg/ml capsaicin. (b) Color-coded perfusion images of forehead dermal blood flow measured at baseline and 10, 20 and 30 minutes. In the white circles from left to right: electrical stimulation, application without iontophoresis of 0.06 mg/ml capsaicin and application without iontophoresis of 6.0 mg/ml capsaicin. Perfusion is indicated in color code that ranges from black (no perfusion) to red (high perfusion). (a) Time course of forehead dermal blood flow responses to 0.06 mg/ml (○) and 6.0 mg/ml (⋄) application of capsaicin without iontophoresis during Visit 1 (open) and during Visit 2 (filled). N = 12. *Difference from baseline, t test: p < 0.05. (b) Response of forehead dermal blood flow to increasing currents of electrical stimulation of a physiological saline solution during Visit 1 (open) and Visit 2 (filled). N = 7. *Significantly different from baseline. Maximal forehead dermal blood flow responses to physiological saline, vehicle, 0.06 mg/ml capsaicin and 6.0 mg/ml capsaicin with and without iontophoresis. Iontophoresis with 0.4 mA during one minute.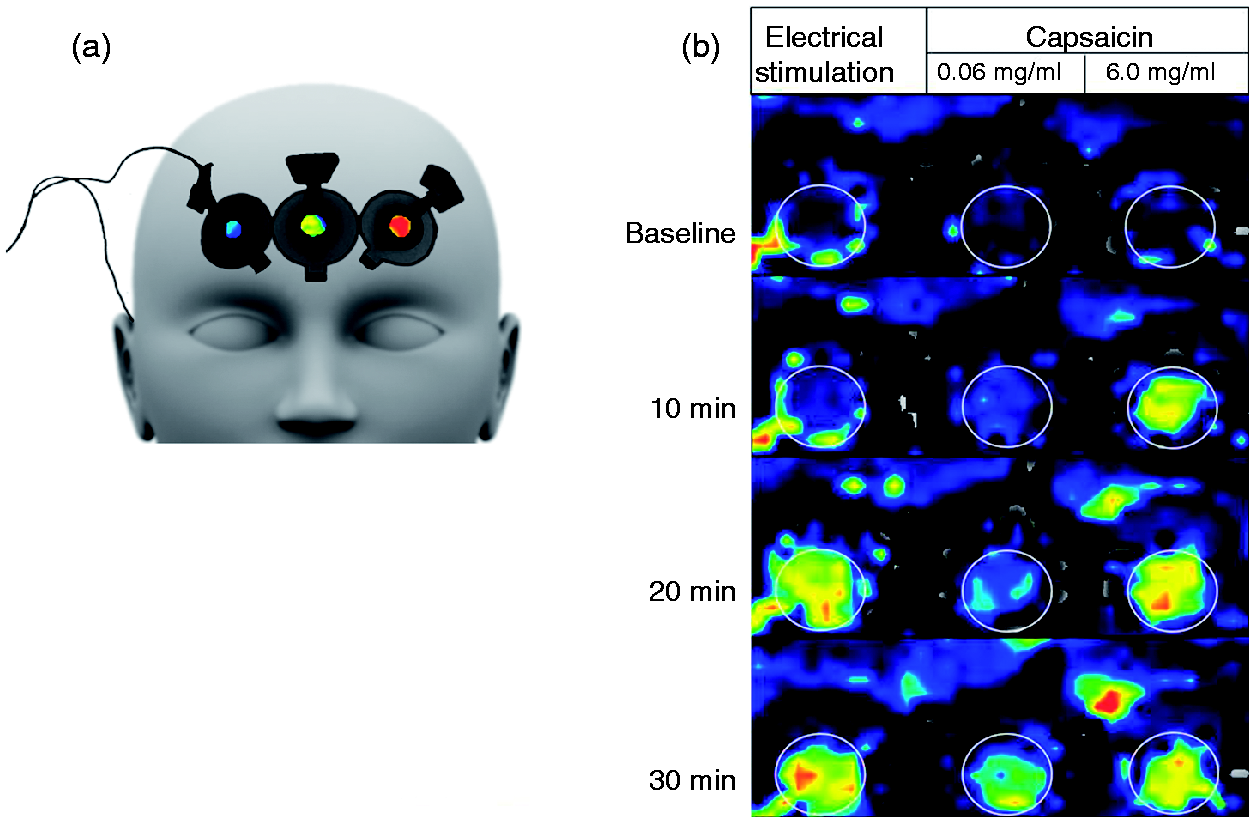

The application of 6.0 mg/ml capsaicin without iontophoresis and electrical stimulation with 1.0 mA at the end of the cumulative successive rate of currents were the best reproducible experimental approaches of the trigeminal nerve-mediated vasodilation model, whereas the reproducibility of the DBF response to 0.06 mg/ml capsaicin application without iontophoresis and electrical stimulation with currents lower than 1.0 mA was fair to good (Table 3 and Figure 3). The BCV was 38% for 0.06 mg/ml capsaicin, 24% for the 6.0 mg/ml capsaicin and 35% for electrical stimulation with 1.0 mA. We performed sample size calculations based on the application of 6.0 mg/ml capsaicin without iontophoresis, The required group sizes to detect differences in DBF response resulted in groups of n = 91, 23 and 11 subjects to detect a 10%, 20% or 30% shift, respectively.
Scatter plots (left-hand side) show correlation between Visit 1 and Visit 2 calculated for dermal blood flow (DBF) responses to 0.06 mg/ml capsaicin application without iontophoresis (upper panel), 6.0 mg/ml capsaicin application without iontophoresis (middle panel) and electrical stimulation (lower panel). 0.06 mg/ml capsaicin: r = 0.35, p = 0.26; 6.0 mg/ml capsaicin: r = 0.96, p < 0.0001; electrical stimulation: r = 0.93, p < 0.005. Bland-Altman plots (right-hand side) show bias (dotted lines) and limits of agreement (solid lines). N = 7–12. Reproducibility. ICC: intraclass correlation coefficient indicating strength of agreement; WCV: within-subject coefficient of variation; Emax: maximum dermal blood flow (DBF) response in arbitrary units (a.u.) after subtraction of baseline DBF; CI: confidence interval.

In-depth characterization of the trigeminal nerve-mediated vasodilation model
Compared to application only, iontophoresis (0.4 mA) of 0.06 mg/ml capsaicin resulted in a marked increase in forehead DBF (Table 3). In contrast, compared to application only, iontophoresis of the high dose of capsaicin did not cause a further increase in DBF (Table 3). In comparison to application without iontophoresis, the response latency was significantly shorter for application with iontophoresis of 0.06 mg/ml capsaicin (time to Emax of 13 ± 2 minutes), but not for application with iontophoresis of 6.0 mg/ml capsaicin (time to Emax of 10 ± 2 minutes).
Interestingly, application of vehicle, but not saline, without iontophoresis also caused a modest increase in DBF comparable to the DBF response to application without iontophoresis of 0.06 mg/ml capsaicin, whereas iontophoresis of vehicle and saline caused a further marked increase in DBF (Table 3).
Electrode placement site had no influence on the DBF response (Emax at 0.4 mA right side of the forehead: 430 ± 58. Emax at 0.4 mA left side of the forehead: 456 ± 91).
Discussion
We here present a novel human model for measuring trigeminal nerve-mediated vasodilation in the forehead skin. With our results we show reproducible DBF responses to topical capsaicin application and electrical stimulation. We have demonstrated that iontophoresis of capsaicin does not result in higher DBF responses or shorter response latency in comparison to application of capsaicin without iontophoresis. In addition to the chemical stimulation with capsaicin, electrical stimulation allows us to stimulate the trigeminal afferents without direct activation of the TRPV1 channel. Advantages of the model we developed are its noninvasiveness, its relative simplicity and its suitability to be used in humans. Furthermore, the topical application of a high and low concentration of capsaicin allows one to study not only high responses, but probably also to distinguish between subjects with a lower or higher threshold response to TRPV1 channel activation. Altogether, the developed model has the potential to be applied for studying trigeminal-nerve-mediated disorders as well as for the evaluation of (new) drugs for these conditions.
Analysis of our data revealed that the DBF responses to the highest intensity of electrical stimulation (1 mA) and the application without iontophoresis of the high concentration of capsaicin (6.0 mg/ml) were well reproducible, with an ICC that indicated an almost perfect strength of agreement for the maximal measured effects. These are also the two experimental approaches of our trigeminal nerve-mediated vasodilation model with the lowest WCV and the highest (maximally) observed values. The high maximal values, combined with the lower variability, suggest that in these experiments responses were approaching a physiological maximum response. In contrast, the application without iontophoresis of 0.06 mg/ml capsaicin had a low ICC and a high WCV. Electrical stimulation with current intensities lower than 1.0 mA also resulted in lower ICC values and higher WCV values. With the 0.06 mg/ml capsaicin the maximal DBF response was low, indicating an incomplete response. While acknowledging the higher variation of the low capsaicin concentration and electrical stimulation with currents lower than 1.0 mA, we included these parameters in the model as potential methods to detect individual response threshold differences. For example, capsaicin induces gender-specific sensory and vasomotor responses (26), indicating that individual threshold response differences may exist.
In our experiments we observed no significant difference in DBF responses between male and female subjects (results not shown). Obviously, the number of subjects in our experiment (three males and nine females) is too small to discern potentially small sex differences. Furthermore, we did not select female participants based on the phase of their menstrual cycle, pre- or postmenopausal status or use of hormonal contraceptives. Sex differences should be explored in future experiments as previous studies have shown that estradiol blood levels may influence vasodilatory responses of capillary blood flow in women (26,34).
The DBF response to vehicle application with and without iontophoresis and saline iontophoresis requires discussion. The vehicle consists of a mixture of ethanol 100%, Tween 20 and distilled water (3:3:4). Since distilled water can be considered harmless and Tween 20 has been shown to have a neutralizing effect on toxicity when mixed with skin irritants (35), the DBF response to application of vehicle without iontophoresis is most likely induced by ethanol. Indeed, ethanol is known to induce TRPV1 channel activation (36–38). Thus, it seems likely that a part of the blood flow responses to capsaicin in our study were caused by ethanol in its vehicle. This is especially relevant in the case of 0.06 mg/ml capsaicin, where the response was not significantly different from that to vehicle. Notwithstanding, we decided to include 0.06 mg/ml capsaicin solution in our model to have a mode of stimulation (ethanol plus capsaicin) of the TRPV1 channel with a low intensity, which, as described above, may be useful in detecting trigeminal hyperreactivity as observed in disease states (36). In this light, we have decided to express our responses relative to the baseline responses at the start of the experiment and not relative to the response to the vehicle, since this, as described above, also includes activation of the TRPV1 channel.
The application of vehicle with iontophoresis resulted in considerable increases in DBF. This could be explained by additional current-induced effects of iontophoresis that have been reported previously (31) on top of the DBF response to ethanol. Our exploration of the effects of iontophoresis on capsaicin revealed that DBF responses were higher and the response latency shorter only when applying 0.06 mg/ml capsaicin with iontophoresis to the forehead skin. Given that the DBF responses to 0.06 mg/ml capsaicin were not significantly different from the DBF responses to vehicle, the higher response and shorter latency could be attributed to the current induced vasodilation. Thus, we conclude that the addition of iontophoresis to the application of the capsaicin had no advantages in our model.
With iontophoresis of physiological saline the current-induced DBF effects were used as an electrical stimulation parameter in our model, complementary to the effects of chemical TRPV1-mediated trigeminal activation by capsaicin. Since the current-induced vasodilation can be inhibited by local anesthesia and is reduced by desensitization of C-nociceptive fibers, as demonstrated by Durand et al., we assume that these effects are largely mediated by CGRP and substance P (31). Contrary to the effects of capsaicin, CGRP release without direct activation of the TRPV1 channel may be studied with electrical stimulation. Thus, similar as described above for the two different concentrations of capsaicin, the application of several current intensities allows us to discern both differences in threshold for stimulation as well as in maximal response.
Admittedly, the involvement of CGRP in the responses to capsaicin, vehicle, as well as electrical stimulation could not unequivocally be proven in the current study since this would require the administration of a CGRP receptor antagonist, as was performed previously in the capsaicin-induced vasodilation model in the human forearm (23,33). Unfortunately, we do not have a CGRP receptor antagonist available that we can administer to human subjects. Similarly, an orally available TRPV1 antagonist would be required to unequivocally assess the involvement of this channel in DBF responses to capsaicin and electrical stimulation.
Comparison with capsaicin-induced dermal vasodilation in the human forearm
It is interesting to compare the properties of our model with those of the previously described model measuring capsaicin-induced dermal vasodilation in the human forearm (33). In the forearm model, maximal vasodilator responses to capsaicin (1000 µg, dose per mm2 of skin corresponding to our 6 mg/ml solution) are observed after 30–40 minutes. In contrast, in our model maximal responses to application without iontophoresis of this concentration were observed after an average of 11 minutes. Applying iontophoresis to the 6.0 mg/ml capsaicin solution did not result in a shorter response latency in our model, since Emax of 6.0 mg/ml with iontophoresis was reached after 10 minutes.
Intriguingly, in the forearm model, the vehicle of capsaicin did not induce a discernible increase in vasodilation, while in our model, as described above, vehicle-induced effects were considerable. These differences between forearm and forehead skin responses to capsaicin are probably related to the well-established variation in skin response to topical irritants that exists throughout the body (39). Furthermore, evidence exists that especially the face is more sensitive to capsaicin compared to other dermatomes of the body (40).
Conclusion
Using capsaicin topical application and electrical stimulation of the forehead skin, we have developed a noninvasive, reproducible model that can evaluate trigeminal-nerve mediated vasodilation. Future investigations should learn whether the model is suitable to differentiate between subjects with and without trigeminal nerve-mediated conditions including migraine and whether it is useful for the evaluation of novel potential antimigraine drugs.
Clinical implications
Until now straightforward and reproducible human methods for the analysis of trigeminal nerve-mediated vasodilation with minimal demands of the test subject have been lacking. We developed a model to study trigeminal nerve-mediated vasodilation in humans that is noninvasive, not painful and reproducible. In this model, trigeminal activation may be achieved via TRPV1 channel activation with capsaicin, or via electrical stimulation (neuronal depolarization without direct activation of the TRPV1 channel).
Footnotes
Funding
This work was supported by the Netherlands Organisation for Scientific Research (NWO) (grant number 91711349).
Acknowledgment
The authors would like to acknowledge the contribution of research assistant Evelien Jäger for her work during this study.
