Abstract
Background:
Pediatric migraine displays different clinical features from adult migraine. Because the trigemino-vascular system (TGVS) plays a pivotal role in migraine pathophysiology, this study compared TGVS responses in a migraine model induced by intracisternal (i.c.) instillation of capsaicin in adolescent and adult rats.
Methods:
TGVS responses measured included c-Fos-protein-expressing neurons in the trigeminal cervical complex (TCC), calcitonin gene-related peptide (CGRP) expression in the trigeminal ganglia (TG) and dura mater, and dural protein extravasation. The formulas for estimating total numbers of activated TCC neurons were established based on the c-Fos-positive neuronal numbers in three sample sections, +0.6, −1.2 and −9 mm and +0.6, −0.6 and −6 mm, from the obex in adult and adolescent rats, respectively.
Results:
After capsaicin instillation, adolescent rats had comparable TCC neurons activated as adult rats, but less TGVS peripheral responsiveness than adults, including CGRP immunoreactivity in the TG, and protein extravasation and CGRP depletion (inversely reflected by CGRP immunoreactivity) in the dura mater.
Conclusions:
Age-dependent differences in TGVS responsiveness in the i.c. capsaicin-induced migraine model of rats are reminiscent of less severe migraine in pediatric patients. This finding may provide new insight into the pathophysiology of migraine and guide the development of new anti-migraine drugs for children.
Introduction
Trigemino-vascular activation via both peripheral and central sensitizations is considered by many to be an essential neuropathogenic mechanism of migraine, a severe headache characterized by concurrent nausea, vomiting and autonomic instability. The trigemino-vascular system (TGVS), consisting of dural and superficial cortical blood vessels which are innervated by unmyelinated trigeminal afferent C fibers and myelinated Adelta fibers, is strongly implicated in the initiation of the headache pain (1). The TGVS transduces peripheral sensory signals, via trigeminal ganglia (TG), to the trigeminal nucleus caudalis (TNC) in the brainstem, which subsequently projects to higher-order pain centers (2,3). In addition to the TNC, the brainstem trigemino-cervical complex (TCC) also includes superficial laminar neurons of C1/2 spinal dorsal horn neurons which receive nociceptive inputs via dorsal root ganglia (DRG) from C1/2 dorsal roots (4). In addition, the TNC receives several modulatory inputs from other brainstem nuclei, including the periaqueductal gray (PAG), locus ceruleus (LC) and raphe nucleus (RN) (2).
Whether migraine is peripheral or central in origin is considerably debated. Peripheral sensitization of the TVGS via chemical stimulation can trigger migraine-like symptoms (5,6). The cardinal symptoms of migraine are similar to those resulting from meningeal irritation and inflammation (7). Recent genetic studies suggest the involvement of TWIK-related spinal cord potassium (TRESK) channels and transient receptor potential M8 (TRPM8) channels, which are enriched in peripheral sensory neurons, such as TG and DRG (8,9). Central sensitization of the TVGS by TNC activation, which is involved in both nociceptive processing and cerebrovascular regulation, also plays a pivotal role in migraine pathophysiology (10). Positron emission tomography showed early brainstem activation in a patient during spontaneous migraine attack but not in the headache-free interval (11).
In animal models, TGVS activation can be induced by intracisternal (i.c.) instillation of nociceptive substances like capsaicin (12) or autologous blood (7), resulting in both central and peripheral responses. The central response in the TNC, activated by glutamate (13) released from TG central terminals, can be measured by expressing c-Fos protein, a neuronal activation marker (14). Peripheral responses, including dural vasodilation and protein extravasation (15), are neurogenic inflammation phenomena mediated by neuropeptides, such as substance P or calcitonin gene-related peptide (CGRP), released from TG peripheral terminals, that is, perivascular nerve endings.
Capsaicin is an agonist of the vanilloid 1 type of transient receptor potential (TRPV1) channel and is co-expressed with substance P and/or CGRP in both TG peripheral and central terminals (3,16). Capsaicin activates the TRPV1 in TG peripheral terminals, the prominent population of meningeal nerve fibers, resulting in neuronal activation, as reflected by the c-Fos immunoreactivity (c-Fos-ir), in the TNC (12).
The clinical features and incidence of pediatric migraine are different from those of adult migraine. Pediatric migraine is characterized by more bilateral involvement, shorter duration, less severity (17) and lower prevalence than adult migraine (18,19). However, the mechanism(s) contributing to these differences remain unclear. Several animal models of migraine have been reported (15) but all were studied in adult animals. Thus, this study explored differences between adolescent and adult rats in terms of central and peripheral responsiveness of the TGVS in a rat migraine model induced by i.c. instillation of capsaicin.
Materials and methods
Animals
All animal care and experimental protocols were approved by the Institution of Care and Use of Laboratory Animals of the College of Medicine of National Taiwan University. Male Wistar rats at eight to nine weeks (280–310 g, adult group) and four to five weeks (120–140 g, adolescent group) were used. They were housed in an animal room with a 12-hour light/12-hour dark cycle and free access to food and water.
Intracisternal instillation of capsaicin
Under anesthesia with chloral hydrate (400 and 100 mg/kg intraperitoneially [i.p.], respectively, for inducing and maintaining), the rat received a midline skin incision from the occipital protuberance to the upper cervical area. After catheterization with a catheter (PE-10, SIMS Portex Ltd, Hythe, UK) inserted 3 mm deep into the cisterna magna, the rat was placed in a prone position for 6 hours. Then the capsaicin solution (10 nmol, 100 μl) was instilled through the catheter into the cisterna magna over one minute. The rat was then placed in a reverse Trendelenburg position (–30 degrees) for 30 minutes in order to facilitate capsaicin distribution within the subarachnoid space, followed by the prone position for another 90 minutes. Capsaicin (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in the vehicle solution containing 10% ethanol and 10% Tween 80, and sonicated for five minutes, and then further diluted (1:100) in normal saline as a stock solution stored at 4°C. For the control group, 100 μl of the vehicle was administered by i.c. instillation.
Two hours after capsaicin instillation, the rat was euthanized by an overdose of chloral hydrate (1.6 g/kg, i.p.) and then perfused via the ascending aorta with 150 ml 0.5% sodium nitrate, followed by a 500-ml fixative containing paraformaldehyde (4%) in 0.1 M phosphate buffer (PB, pH 7.4). For observing sustained CGRP expression in the TG and dura mater, the rat, with an interval of recovery from anesthetics, was euthanized at 20 and 44 hours after capsaicin instillation. The core temperature of the rat was maintained at 36–37°C, and its respiratory rate was monitored by observing the chest wall movement for one-minute blocks. Analgesics were used in the rats that showed autonomic changes during recovery.
TCC brain sections
The brainstem with attached cervical cord was dissected, stored overnight in the perfusion fixative and then dehydrated with 20% and 30% sucrose, in series. Brainstem and upper cervical spinal cord sections (50 µm) were serially sectioned using a cryostat (LEICA CM3050S, Nussloch, Germany) from 1 mm rostral to the obex to the C6 level of the spinal cord. Three rats in each age group were studied and 430 ± 10 and 330 ± 10 sections were obtained from each adult and adolescent rat, respectively. Every third section was collected and subjected to c-Fos immunohistochemical staining.
Dura mater preparations
The dura mater dissected from the cranial cavity of the rat was washed and shaken, in series, with 30% and 50% ethanol (10 minutes), 70% ethanol (15 minutes), 50% ethanol (10 minutes) and 0.1 M PB (3 minutes) twice. It was then kept in the fixative for CGRP immunohistochemical staining.
TG slice sections
The rat TG was dissected, stored in the fixative overnight, embedded by paraffin wax and then serially sectioned at 5 µm thickness using a microtome (LEICA RM2245, Nussloch, Germany). TG slices were collected and subjected to CGRP immunohistochemical staining.
Immunohistochemistry of c-Fos protein in TCC sections
Free-floating immunohistochemistry of c-Fos protein was conducted using the avidin-biotin method. Briefly, TCC sections were incubated in 1% NaBH4 in 0.1 M PB for 20 minutes, rinsed thrice with 0.1 M PB for 10 minutes and treated with 0.5% hydrogen peroxide in 0.1 M PB for 30 minutes. The sections were then washed thrice with 0.1 M PB for 10 minutes, followed by one-hour incubation with 0.1 M PB containing 5% goat serum and 0.2% triton X-100. The sections were incubated with an anti-c-Fos rabbit polyclonal antibody (Calbiochem, San Diego, CA, USA) in 1:7000 dilution with 0.1 M PB containing 5% goat serum and 0.2% triton X-100 at 4°C for 48 hours. After a 10-minute rinse with 0.1 M PB thrice, the sections were incubated with biotinylated anti-rabbit IgG (Vector Labs, Burlingame, CA, USA) in 1:200 dilution with 0.1 M PB containing 5% goat serum and 0.2% triton X-100 for two hours at room temperature. The sections were then washed thrice with 0.1 M PB for 10 minutes and incubated with horseradish peroxidase avidin D (Vector Labs, Burlingame, CA, USA) in 1:500 dilution with 0.1 M PB containing 5% goat serum and 0.2% triton X-100 for one hour in the dark at room temperature. Immunoreactions were visualized using the DAB reagent kit (KPL, Gaithersburg, MD, USA).
C-Fos-ir neurons, that is, neurons with stained nuclei, were counted under a microscope (Olympus BX51, Essex, UK) by an observer blinded to the age group. Data were confirmed in randomly selected sections by a second investigator who was also blinded to the age group.
Immunohistochemistry of CGRP in TG slices and dura mater
Additional rats (N = 4 in each group) were used for dura mater and TG preparations at 2, 22 and 44 hours after i.c. capsaicin. Immunohistochemistry of CGRP was performed with the avidin-biotin method with a protocol similar to that for c-Fos staining, except using a 30-minute blocking incubation, anti-CGRP rabbit polyclonal antibody (1:1000, Calbiochem, San Diego, CA, USA) and horseradish peroxidase avidin D (1:200 Vector Labs, Burlingame, CA, USA).
Protein extravasation in the dura mater
After anesthesia, additional rats (N = 4) were injected with Evans Blue (25 mg/kg) (Sigma-Aldrich, St. Louis, MO, USA) solution into the right femoral vein immediately before capsaicin instillation. Thirty minutes after capsaicin instillation, the rat was exsanguinated by infusing 60 ml saline, at 37°C, into the left cardiac ventricle. Then, the dura mater was dissected and incubated in formamide at 50°C for 24 hours (20). The fixed dura matter tissue with Evans Blue was examined by microscopy (Olympus BX51, Essex, UK).
Statistical analysis
Data were expressed as mean ± standard error (SE) unless otherwise specified. Formulas to estimate the total numbers of c-Fos-ir neurons in the TCC were established from the area under the curve (AUC) built by two linear regression lines, described in Figure 3. Differences between or among groups were compared using the Mann-Whitney U-test or analysis of variance (ANOVA) followed by Bonferroni post hoc test. Statistical significance was a p value < 0.05.
Results
Physiological monitoring after i.c. capsaicin
Rapid respiration occurred in rats five minutes after capsaicin instillation, with respiratory rates significantly increased from 94 ± 3.0 to 106 ± 2.9 per minute (p < 0.05) in adult rats (N = 13) and from 97 ± 4.4 to 116 ± 4.0 per minute (p < 0.05) in adolescent rats (N = 4). Changes in respiratory rates were not statistically different between the two groups (p > 0.05) and resolved within 30 minutes after capsaicin instillation in both groups (97 ± 2.3 and 97 ± 1.9 per minute, respectively). This suggests that i.c. capsaicin induced a respiratory-stimulating effect, implying an autonomic response which might be elicited by a painful stimulation, in both anesthetized adult and adolescent rats.
Spatial distribution of c-Fos-ir neurons in the TCC after i.c. capsaicin
Two hours after i.c. capsaicin, c-Fos-ir was demonstrated bilaterally and most intensely expressed in the superficial laminae of the TNC and the upper cervical spinal dorsal horn in both age groups (Figures 1A and B).
The expression of c-Fos-immunoreactive (c-Fos-ir) neurons in representative coronal sections in the trigemino-cervical complex (TCC) of adult and adolescent rats after intracisternal (i.c.) capsaicin. (A) The c-Fos-ir neurons induced two hours after capsaicin (10 nmol) i.c. instillation are depicted as dots in the TCC sections taken at three reference levels: the Vi/Vc transition zone (upper), rostral trigeminal nucleus caudalis (TNC) (middle), and C2 level of the cervical spinal cord (lower) in adult (left) and adolescent (right) rats. The distance (mm) rostral (positive) or caudal (minus) to the obex at each reference level was labeled. The findings are representative of the results obtained from five animals in each age group. Scale bar: 500 µm. (B) c-Fos-ir neurons were demonstrated by avidin-biotin immunochemical stain at the three reference levels in adult (upper) and adolescent (lower) rats. Scale bar: 100 µm.
The numbers of c-Fos-ir neurons after capsaicin were counted in TCC sections taken every 150 µm from +1.05 mm (rostral) to −15 mm (caudal) from the obex in five rats of each group. Figure 2 shows the distribution of c-Fos-ir cells along the TCC axis of adult and adolescent rats after i.c. instillation of capsaicin and vehicle. As compared with the vehicle control group, significantly more c-Fos-ir neurons in the TCC were observed in capsaicin-treated adult (Figure 2 A) and adolescent (Figure 2 B) rat. The sagittal distribution patterns of c-Fos-ir neurons were similar, but the total anterior-posterior distribution distances were different between the two age groups. In the adult group, the c-Fos-ir was maximal at −2.1 to −2.15 mm from the obex, followed by two smaller peaks at −5 to −6 mm and −8 mm and reaching a plateau at −9 mm; the total distribution distance was 10.05 mm (Figure 2A). In the adolescent group, the c-Fos-ir was maximal at −1.05 to −1.2 mm, followed by a smaller peak at −3.5 to −4 mm and a plateau at −6 mm from the obex; the total distance was 7.05 mm (Figure 2B). The first two peaks in the total anterior-posterior distance by percentage are 31% and 65% in the adult group, similar to 31% and 68% in the adolescent group.
Sagittal distribution of c-Fos-ir neurons in the trigemino-cervical complex (TCC) of adult and adolescent rats after intracisternal vehicle and capsaicin instillation. The c-Fos-ir neurons in the TCC were counted every third section (50 µm in each section) from a distance of +1.05 mm (rostral) to −15 mm (caudal) from the obex (total, 107 sections). Data are the mean ± standard error from five adult rats (A) and five adolescent rats (B) in vehicle- and capsaicin-treated groups, respectively. Method of establishing formulas to estimate the total number of c-Fos-ir neurons in the trigemino-cervical complex (TCC) by regression lines describing the data in Figure 2. The linear regression lines describing the data points in Figure 2, the means of c-Fos-ir neuronal numbers per section at each counted TCC section level, were obtained. The two regression lines cross at point C, which corresponds to the cell number at point D (−1.18 mm in adults and −0.6 mm in adolescents). The cell numbers at this points D (N2), A (+1.05 mm) (N1) and E (−9 mm for adults (A) and −4.5 mm for adolescents (B) (N3) can be used to estimate the total number of c-Fos-ir neurons by the formulas 16(N1 + N2)/2 + 53(N2 + N3)/2 in adults and 12(N1 + N2)/2 + 27(N2 + N3)/2 in adolescents, respectively, based on the formula for calculating the area under the curve, that is, two trapezoids. The coefficients, 16 and 53 in adults and 12 and 27 in adolescents, respectively, are the section numbers counted between points A and D and between points D and E. That is: ([1.05 + 1.18] mm/150 µm) + 1 and ([9–1.18] mm/150 µm) + 1 in adults and ([1.05 + 0.6] mm/150 µm) + 1 and ([4.5–0.6] mm/150 µm) + 1 in adolescents, respectively.

Establishing formulas to estimate total numbers of c-Fos-ir neurons in the TCC
To establish an adequate formula to estimate the total number of activated TCC neurons after capsaicin treatment from sampled sections, the regression lines for describing the distribution of c-Fos-ir neurons along the TCC axis were derived in each group. In the adult group, two regression lines were derived from +1.05 to −2.1 mm (y = −95x + 477) and from −2.25 to −9 mm (y = 68x + 670), which crossed at the point C (−1.18, 589.1) in Figure 3A. The total number of TCC neurons activated by capsaicin can be calculated by the AUC, that is, the areas of two trapezoids formed by two regression lines (Figure 3) with the formula: 16(N1 + N2)/2 + 53(N2 + N3)/2, where N1, N2 and N3 were the neuronal numbers measured at 1.05, −1.18 and −9 mm from the obex, respectively (points A, D and E in Figure 3A). The coefficients, 16 and 53, respectively, were the section numbers counted between points A and D and between points D and E in Figure 3A. In the adolescent group, the two regression lines were y = −169x + 475 and y = 91x + 632, which crossed at the point C (−0.6, 576.4) in Figure 3B. The formula to estimate total activated neuronal numbers was: 12 (N1 + N2)/2 + 27 (N2 + N3)/2, where N1, N2 and N3 were the neuronal numbers taken at 1.05, −0.6 and −4.5 mm from the obex (A, D and E in Figure 3B), respectively.
Deviation of the estimated number of c-Fos-ir neurons from the real counted number in three selected reference levels in the trigemino-cervical complex of adult rats after i.c. capsaicin instillation.

N1, N2 and N3 denote the numbers of c-Fos-ir neurons at the reference levels of +0.6, −1.2 and −9 mm rostral to the obex, respectively.
c-Fos-ir neurons were estimated by the formula: 16(N1 + N2)/2 + 53(N2 + N3)/2.
The error ratio was calculated by the difference of the estimated number from the real number, divided by the real counted number.
The adult mean is the average of nos. 1, 2, 3, 4 and 5 of adult rats.
i.c.: intracisternal.
Deviation of the estimated number of c-Fos-ir neuron from the real counted number in three selected reference levels in the trigemino-cervical complex of adolescent rats after i.c. capsaicin instillation.

N1, N2 and N3 denote the numbers of c-Fos-ir neurons at the reference levels of +0.6, −0.6 and −6 mm rostral to the obex, respectively.
c-Fos positive neurons were estimated by the formula: 12(N1 + N2)/2 + 27(N2 + N3)/2.
The error ratio was calculated by the difference of estimated number from real counted number, divided by real number.
The adolescent mean is the average of nos. 1, 2, 3, 4 and 5 of adolescent rats.
i.c.: intracisternal.
Comparison of the numbers of capsaicin-activated TCC neurons between adult and adolescent rats
The total numbers of c-Fos-ir neurons in the TCC in capsaicin- and vehicle-treated groups were estimated with the above formulas by taking into account of 65 sections from +0.6 mm to −9 mm from the obex in each adult rat and 45 sections taken from +0.6 mm to −6 mm in each adolescent rat. The results showed that in both age groups, capsaicin induced significantly more c-Fos-ir TCC neurons, compared to the vehicle control (Figure 4). The total number of c-Fos-ir neurons in adult rats tended to be higher than that in adolescents, but the difference is not statistically significant (p = 0.136). There is no significant difference between the ages in vehicle-treated groups. (Figure 4A). Adult rats have a longer TCC axis than adolescents. After correcting the TCC length, the numbers of c-Fos-ir neurons per section were not different between the two age groups (Figure 4B).
Comparison of c-Fos-ir neuronal numbers induced by intracisternal capsaicin or vehicle instillation in adult and adolescent rats. (A) Total numbers of c-Fos-ir neurons in capsaicin-treated rats in both age groups were significantly higher than in vehicle-treated rats (p = 0.000 for adults and 0.001 for adolescents). There is no significant difference between both ages in vehicle- (p = 1.0) or capsaicin-treated (p = 0.136) groups. (B) The numbers of c-Fos-ir neurons per section in both age groups in capsaicin-treated rats were significantly higher than in vehicle controls (p = 0.000 for either adults or adolescents). There is no significant difference between the two age groups in vehicle- (p = 1.0) or capsaicin-treated (p = 1.0) groups. Data are the mean ± standard error from five rats in each group. *p ≤ 0.05; N.S.: not significant; analysis of variance (ANOVA) (p = 0.000 in A and B), followed by Bonferroni post hoc test.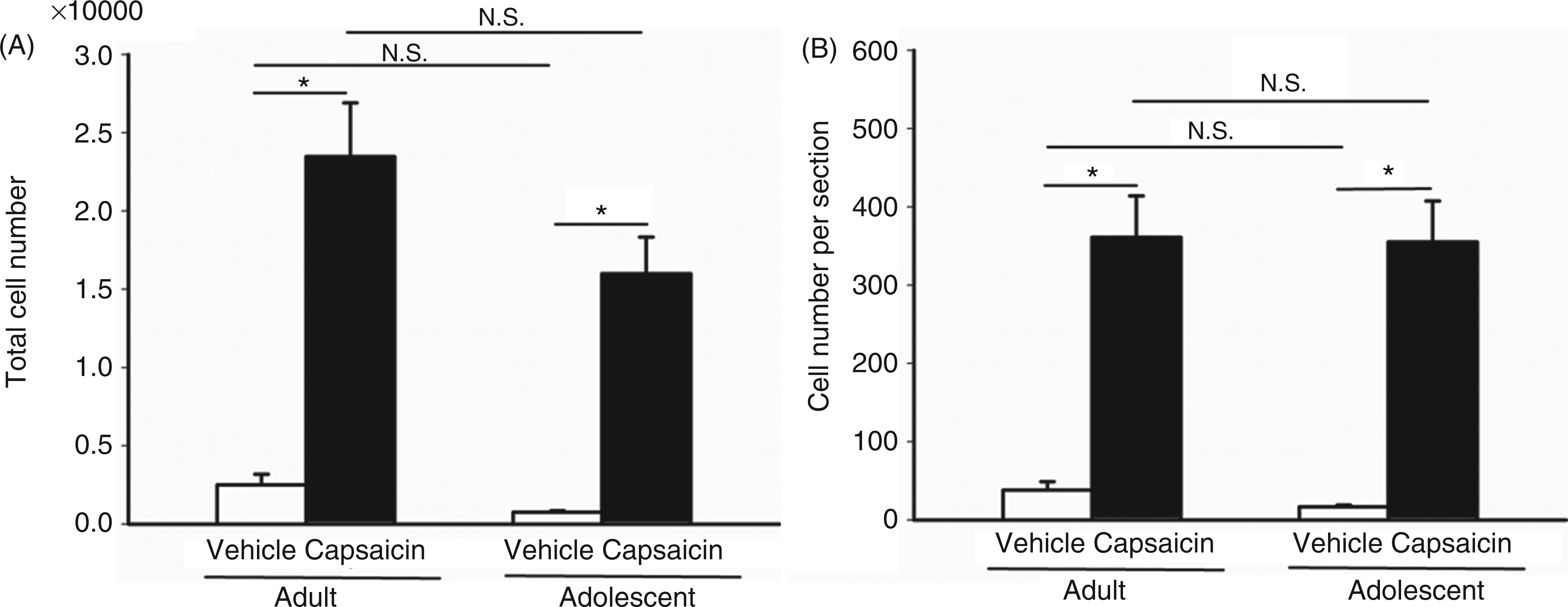
CGRP expression in the TG
In adult rats, sustained positive CGRP-ir in the TG was observed at 2, 20 and 44 hours after i.c. capsaicin instillation. However, in the adolescent group, only a weak CGRP-ir in the TG was observed at 2, but not at 20 or 44, hours after capsaicin instillation (Figure 5).
Calcitonin gene-related peptide (CGRP) immunoreactivity in the trigeminal ganglia (TG) of adult and adolescent rats after capsaicin instillation. CGRP immunoreactivity was photographied in TG sections isolated at 2, 20 and 44 hours after capsaicin or vehicle instillation in adult and adolescent rats. Negative control is the section shown from a naive rat. Note that capsaicin induces sustained CGRP immunoreactivity in the TG of adult rats, but less dense immunoreactivity in the adolescent group. Scale bar: 100 µm.
CGRP expression and protein leakage in the dura mater
The CGRP-ir in the dura mater of both age groups was measured at 2, 20 and 44 hours after i.c. capsaicin (Figure 6A). In the adult group, the CGRP-ir in dural nerve endings was depleted until 44 hours after capsaicin instillation. However, in the adolescent group, the CGRP-ir was highly preserved after capsaicin instillation, compared to the vehicle-treated group (Figure 6A). Similarly, significant protein leakage in the meningeal vessels of dura mater as shown by Evans Blue staining occurred in capsaicin-, but not vehicle-treated adult rats. However, there was little Evans Blue staining in capsaicin-treated adolescent rats, compared to vehicle-treated adolescents (Figure 6B).
Calcitonin gene-related peptide (CGRP) immunoreactivity and protein leakage in the dura mater of adult and adolescent rats after capsaicin instillation. (A) CGRP immunoreactivity was measured in dura mater preparations isolated at 2, 20 and 44 hours after capsaicin or vehicle instillation in adult and adolescent rats. Note that CGRP depletion in dural nerve terminals was triggered from 2 to 44 hours after capsaicin instillation in the adult group (upper two rows) but CGRP immunoreactivity is preserved in the adolescent group (lower two rows). Scale bar: 10 µm. (B) Protein leakage presented as granules distributed along the meningeal vessels (arrows) of the dura mater can be detected by Evans Blue staining after capsaicin instillation in the adult group, but not in the adolescent group. Scale bar: 100 µm.
Discussion
We demonstrated that in i.c. capsaicin-induced migraine model, adolescent rats showed weaker peripheral responsiveness than adult rats, but had comparable central responsiveness in the TGVS. Peripheral responses included the CGRP-ir in the TG, and protein extravasation and CGRP release (inversely reflected by the CGRP-ir) in the dura mater. The central response was measured by the number of c-Fos-ir TCC neurons. These age-dependent differences in TGVS responsiveness are consistent with the clinical observation that pediatric migraine is usually less severe than adult migraine.
TCC neuronal activation after i.c. capsaicin instillation
Capsaicin activated the TCC neurons starting from 0.6 mm to −9 mm rostral to the obex in adult rats (Figure 2A) and to −6 mm in adolescent rats (Figure 2B). The caudal coordinates (−9 and −6 mm) in both age groups correspond to their respective spinal C2 dorsal horns. Neurons in this spinal segment receive sensory inputs from supratentorial dura mater and superior sagittal sinus (21,22), both of which are likely to be the peripheral sites involved in migraine attacks. Therefore, in the formulas we established to quantify total c-Fos-ir neurons in the TCC in Wistar rats, the neurons in the spinal C2 dorsal horn were included in both adult and adolescent groups. These neurons were not included in a previous formula developed by Cutrer’s group for adult guinea pigs (12) and Sprague-Dawley rats (23). Interestingly, the distance between the spinal C2 level and obex is shorter in adolescent rats than in the adult group. Therefore, the formula used to estimate TCC c-Fos-ir neurons in this migraine model should be justified for a specific age group.
The adolescent rats used in this study were at the age of four to five weeks, which is a critical age in rodents for the development of nociceptive afferent activity in the TNC (24,25). Morphologic and functional studies demonstrate that adult-like nerve-fiber network structures are noted in rats at week 5, nociceptive reflexes mediated by the TNC are established at week 4 (25), the fast excitatory synaptic transmission in the trigeminal nociceptive afferent pathway matures after week 5 (24), and the mechanism for prolonged increase in central excitability first appears at week 6 (25). Interestingly, we found that the total number or neuron number per section of c-Fos-ir neurons in the TCC after capsaicin treatment was comparable between the two age groups. This suggests that the nociceptive reflex and the nerve-fiber network structures in adolescent rats at four to five weeks are relatively mature, having adult-like morphology and functions in the TCC, as compared to the adult age at eight to nine weeks.
Capsaicin given by i.c. instillation as a migraine model
The central response
Capsaicin (i.c.)–induced TCC neuronal activation can result from a direct activation of second order neurons in the TCC or indirectly from peripheral stimulation of primary sensory nerve endings through cerebrospinal fluid (CSF) circulation. To delineate the mechanism, we compared the TCC distribution pattern of c-Fos-ir neurons in the rat in a prone position (0 degrees) versus a reverse Trendelenburg position (−30 degrees), and found that the number of c-Fos-ir neurons in the rostral TNC is not higher in the rat in a reverse Trendelenburg position than in a prone position (supplemental figure 1). This suggests that in this model, instead of directly activating the TCC, capsaicin is mainly distributed widely throughout the subarachnoid space and activates primary sensory afferents supplying the meninges, thereby releasing CGRP as a consequence of the administration via the cisterna magna (15).
Peripheral responses
We also demonstrated that i.c. capsaicin upregulated the CGRP-ir in the TG and caused protein extravasation and CGRP depletion in the dura mater peripherally, mimicking neurogenic inflammation in migraine, in adult, but not adolescent, rats. Through stimulating small myelinated Aδ and unmyelinated C-fibers via TRPV1 receptors located in TG peripheral terminals (26), i.c. capsacin can indirectly activate second order neurons in the TNC via peripheral orthodromic activation of the TGVS (15). Alternatively, i.c. capsacin can also cause neurogenic release of vasoactive neuropeptides such as substance P or CGRP (27) via antidromic activation of neighboring peripheral terminals, resulting in an aseptic inflammation of the meninges, which induces protein extravasation, mimicking neurogenic inflammation in migraine. This explains our findings of CGRP depletion and protein extravasation in the dura mater after capsaicin treatment. In addition, TRPV1 receptors also exist in the TG (26) and direct chemical stimulation of the TG also causes protein extravasation in the dura mater and TNC (28,29). Therefore, it cannot be excluded that direct TG activation by capsaicin results in protein extravasation and central c-Fos expression in this model.
A limitation of this model is the inability to observe a lateralization difference within the TCC for studying adult migraines, which are clinically characterized by unilateral attacks. However, this disadvantage may be an advantage for studying pediatric migraines, which are mostly bilateral.
Age-dependent responsiveness in the capsaicin-induced migraine model
In the current model of migraine, adolescent rats exhibited comparable c-Fos-ir neurons in the TCC but less intense expression of CGRP in the TG, and no neurogenic inflammation in the dura mater after i.c. capsaicin instillation, as compared to adult rats. It is interesting that the central response, with neuronal activation in the TCC (second order neuron), was similar in both age groups, while the peripheral responses, with CGRP release in the TG (first order neuron) and dura, were different at the same amount of capsaicin stimulation. This suggests that peripheral responsiveness is more sensitive to capsaicin challenge than is central neuron activation in adults compared adolescents. The weaker peripheral responsiveness in the adolescent group may explain the clinical observation that migraine in children is less common and less severe than in adults. This age-related difference may be due to a lack of development of peripheral nociceptive fibers and the presence of neuropeptides. The possibility remains to be elucidated if this difference is attributed to an age-dependent difference in the density or sensitivity of TRPV1 in the TGVS periphery or TRESK or TRPM8 channels in the TG. This is the first study to demonstrate age-dependent vulnerabilities to i.c. noxious stimulation in peripheral neurogenic inflammation in the TGVS. The underlying mechanism for this age-dependent vulnerability, which is important for the development of anti-migraine drugs specific to pediatric patients, warrants further investigation.
Footnotes
Acknowledgments
This study was supported by the grants from the National Science Council of Taiwan (NSC93-2314-B002-179, NSC 94-2341-B-002-153, NSC 95-2314-B-002-297, NSC 99-2320-B-002-011-MY3 and NSC 99-2323-B-002-012), the National Health Research Institutes of Taiwan (NHRI-EX99-9506NI) and the National Taiwan University (Excellence Research Program 99R0066-51).
Conflicts of interest
The authors report no conflicts of interest.
