Abstract
It is suggested that during a migraine attack capsaicin-sensitive trigeminal sensory nerves release calcitonin gene related peptide (CGRP), resulting in cranial vasodilatation and central nociception. Hence, inhibition of trigeminal CGRP release may prevent the above vasodilatation and, accordingly, abort migraine headache. Therefore, this study investigated the effects of sumatriptan (100 and 300 μg/kg, i.v.) on capsaicin-induced carotid haemodynamic changes and on CGRP release. Intracarotid (i.c.) infusions of capsaicin (10 μg/kg/min, i.c.) increased total carotid, arteriovenous anastomotic and capillary conductances as well as carotid pulsations, but decreased the difference between arterial and jugular venous oxygen saturations. Except for some attenuation of arteriovenous anastomotic changes, the capsaicin-induced responses were not affected by sumatriptan. Moreover, i.c. infusions of capsaicin (0.3, 1, 3 and 10 μg/kg/min, i.c.) dose-dependently increased the jugular venous plasma concentrations of CGRP, which also remained unaffected by sumatriptan. The above results support the contention that the therapeutic action of sumatriptan is mainly due to cranial vasoconstriction rather than trigeminal (CGRP release) inhibition.
Introduction
Migraine is a neurovascular syndrome characterized by vasodilatation of cranial blood vessels with activation of perivascular trigeminal sensory nerves (1). Thus, activation of trigeminal sensory nerves results in the release of several neuropeptides, including neuropeptide Y, vasoactive intestinal peptide, substance P and calcitonin gene related peptide (CGRP). Interestingly, plasma concentrations of CGRP, but not of other neuropeptides, are elevated during the headache phase of migraine and these levels are normalized by triptans in parallel with alleviation of headache (2). CGRP, the most potent endogenous vasodilator described thus far, is predominantly located on sensory neurons and perivascular nerves surrounding blood vessels, where it is colocalized with other vasoactive neuropeptides, such as substance P and neurokinin A (3, 4). In view that an increase in circulating CGRP levels is considered as a biological marker for migraine headache, it is reasonable to assume that a substance capable of inhibiting the release of CGRP from trigeminal sensory nerves may be effective in migraine therapy (2).
CGRP can be released from the sensory nerves by electrical or chemical (capsaicin) stimuli (5, 6). In this respect, electrical stimulation of trigeminal sensory nerves evokes the release of CGRP in the cranial venous blood of rats and cats, which was attenuated by sumatriptan (7, 8). However, to the best of our knowledge, there is no comprehensive in vivo evidence to show that capsaicin-induced CGRP release is inhibited by triptans. Therefore, the main objective of this study in anaesthetized vagosympathectomised pigs was to investigate the effects of sumatriptan on capsaicin-induced: (i) carotid haemodynamic changes, and (ii) increase in plasma CGRP release.
Materials and methods
General
After an overnight fast, 28 domestic pigs (Yorkshire × landrace, female, 10–14 kg), divided into two groups (n = 15 and n = 13 for vehicle and sumatriptan, respectively), were sedated with azaperone (120 mg, i.m), midazolam hydrochloride (10 mg, i.m) and then anaesthetized with sodium pentobarbital (600 mg, i.v.). After tracheal intubation, the animals were connected to a respirator (BEAR 2E, BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH: 7.35–7.48, pCO2: 35–48 mmHg, pO2: 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of sodium pentobarbital (12–20 mg/kg/h). This anaesthetic regimen, together with bilateral vagosympathectomy (see below), increases heart rate and markedly dilates carotid arterioles and arteriovenous anastomoses. Consequently, carotid blood flow, particularly its arteriovenous anastomotic fraction, is considerably higher in these pigs than in conscious or thiopental-anaesthetized pigs (9).
Heart rate was measured with a tachograph (CRW, Erasmus University, Rotterdam, the Netherlands) triggered by electrocardiogram signals. Both common carotid arteries were dissected free and the accompanying vagosympathetic trunks were cut between two ligatures to prevent any possible influence via baroreceptor reflexes on the carotid vascular responses produced by capsaicin. Pulsatile and mean carotid blood flows were measured in the right common carotid artery with a flow probe (internal diameter: 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system, Skalar, Delft, the Netherlands). The amplitude of carotid blood flow signals provided an index of carotid flow pulse. Subsequently, three hub-less needles, connected to a polyethylene tube, were inserted into the right common carotid artery for the administration of capsaicin, radioactive microspheres and the α1-adrenoceptor agonist phenylephrine. The use of phenylephrine is necessitated by the fact that the carotid arterioles and arteriovenous anastomoses are in a dilated state under the present anaesthetic regimen (9); therefore, to study the effects of vasodilator agents (in the present case, capsaicin), one has to constrict these shunt vessels first. As described earlier (10), phenylephrine decreases the total carotid conductance exclusively by constricting carotid arteriovenous anastomoses, which results in an increase in the difference between arterial and jugular venous oxygen saturations (A-V SO2 difference) (11).
Lastly, catheters were placed in:
the right external jugular vein for the withdrawal of venous blood samples to measure blood gases (ABL-510; Radiometer, Copenhagen, Denmark) and plasma concentrations of CGRP (see below);
the inferior vena cava (via the left femoral vein) for the administration of vehicle or sumatriptan;
the aortic arch (via the left femoral artery) for the measurement of arterial blood pressure (Combitrans disposable pressure transducer; Braun, Melsungen, Germany) as well as withdrawal of arterial blood samples to measure blood gases.
Heart rate and systolic, diastolic and mean arterial blood pressures as well as mean and pulsatile carotid artery blood flows were continuously monitored on a polygraph (CRW, Erasmus University, Rotterdam, the Netherlands). Vascular conductances were calculated by dividing the respective blood flows (ml/min) by mean arterial blood pressure (mmHg), multiplied by one hundred and expressed as 10−2 ml/min/mmHg. During the experiment, body temperature was maintained at 37 ± 1°C by a heating pad and each animal was infused with physiological saline to compensate for fluid losses.
Distribution of carotid blood flow
The distribution of common carotid blood flow into tissue (capillary) and arteriovenous anastomotic fractions was determined in 14 pigs later receiving vehicle (n = 8) or sumatriptan (n = 6) with radioactive microspheres (diameter: 15.5 ± 0.1 µm; S.D), labelled with 141Ce, 103Ru, 95Nb or 46Sc (NEN Dupont, Boston, USA). For each measurement, a suspension of about 200 000 microspheres, labelled with one of these isotopes, was mixed and injected into the right carotid artery. At the end of the experiment, the animal was killed using an overdose of sodium pentobarbital and the heart, kidneys, lungs and different cranial tissues were dissected out, weighed and put in vials. The radioactivity in these vials was counted for 5 min in a γ-scintillation counter (Packard, Minaxi autogamma 5000 for 5 min), using suitable windows for discriminating the different isotopes (141Ce: 120–167 KeV, 103Ru: 450–548 KeV, 95Nb: 706–829 KeV and 46Sc: 830–965 KeV). All data were processed by a set of specially designed computer programs (12).
The distribution of total carotid blood flow to the different tissues (Qtis) was calculated by the formula:
Qtis = (Itis/Itotal) × Qcarotid
where Itis is tissue radioactivity, Itotal is the total radioactivity injected and Qcarotid is the total common carotid blood flow at the time of microsphere injection. Since little or no radioactivity was detected in the heart or kidneys, it can be assumed that all microspheres trapped in lungs reach this tissue from the venous side after escaping via carotid arteriovenous anastomoses. Therefore, the amount of radioactivity in the lungs can be used as an index of the arteriovenous anastomotic fraction of carotid blood flow (12, 13).
Determination of plasma levels of CGRP
Jugular venous blood samples were obtained from the 28 pigs, receiving either vehicle (n = 15) or sumatriptan (n = 13). Fourteen of these animals (8 and 6 animals from vehicle and sumatriptan groups, respectively) were used for carotid haemodynamic experiments, while the other 14 were separate experiments using the same protocol, except that the radioactive microspheres were not used. Blood was transferred immediately into a polypropylene tube containing ethylene dinitro-tetraacetic acid (1 mg/ml of blood) and aprotinin (500 KIU/ml of blood). Aprotinin was used to inhibit endogenous plasma proteases, since our unpublished studies have shown that CGRP is not detectable in biological samples without aprotinin. After centrifugation at 1600 g for 15 min, plasma samples were coded and stored at − 80°C until CGRP measurements were performed. The person measuring CGRP concentrations remained blind to the treatments, until all data had been collated.
CGRP was extracted from plasma using a C18 SEP-COLUMN, dried by lypholization, and measured by radioimmunoassay (14), as per protocol of the Peninsula Laboratories, Inc. (Belmont, CA, USA). The recovery of CGRP from the extraction procedure was ascertained by assaying control samples paired with the same sample spiked with known quantities of CGRP. The column recovery values were 85, 79, 81, 89 and 92% (mean, 85.2; standard deviation, 5.4; coefficient of variation, 6.3%). The CGRP concentrations measured in the actual samples were, however, not corrected for the loss in the extraction procedure.
Experimental protocol
Following surgery and after the haemodynamic condition of the animals (n = 28) had been stable for 15–20 min (heart rate: 109 ± 3 beats/min; mean arterial blood pressure: 101 ± 2 mmHg; mean carotid blood flow: 108 ± 5 ml/min; and A-V SO2 difference: 7.6 ± 1.3%) phenylephrine was infused into the right common carotid artery at a rate of 10 µg/kg/min for 10 min, followed by 3–6 µg/kg/min throughout the rest of the experiment to maintain carotid blood flow at a constant low level. The latter dose of phenylephrine was chosen so that the external jugular venous oxygen saturation was between 60 and 70% and mean carotid blood flow was about 40% of the original value. After a period during which the haemodynamic variables remained constant for at least 60 min (heart rate: 151 ± 4 beats/min; mean arterial blood pressure: 110 ± 2 mmHg; mean carotid blood flow: 63 ± 5 ml/min; and A-V SO2 difference: 23 ± 1.7%; n = 28), the animals received consecutive infusions (0.15, 0.45, 1.5 and 4.5 ml, i.c. for 3 min each) of capsaicin vehicle (see Compounds and kits section). It is important to mention that the vehicle of capsaicin was devoid of any systemic and carotid haemodynamic responses (see Results section).
Five to 10 min after the last infusion of capsaicin vehicle, blood samples were obtained for the measurements of blood gases and CGRP concentrations; moreover, the values of heart rate, mean arterial blood pressure and total carotid blood flow and conductance were collated (baseline values; 15 and 13 pigs for vehicle and sumatriptan, respectively). In 14 out of the 28 pigs (8 for vehicle and 6 for sumatriptan) the first batch of radioactive microspheres was injected for determining the baseline distribution of carotid blood flow. The animals then received 4 consecutive infusions of capsaicin (0.3, 1, 3 and 10 µg/kg/min, i.c. for 3 min each) and heart rate, arterial blood pressure and total carotid blood flow were determined at the end of each infusion. In addition, after the last infusion of capsaicin (10 µg/kg/min), blood gases, plasma CGRP concentration and carotid blood flow distribution were measured as described above (control values). Subsequently, a recovery period of 20 min was allowed until all haemodynamic parameters had returned to baseline levels. At this point, the animals were divided into two groups, which were treated with i.v. infusions (rate: 0.5 ml/min for 10 min) of either vehicle (two times 5 ml of acidified distilled water; n = 15) or sumatriptan (100 and subsequently 300 µg/kg; n = 13). Ten min after each infusion, capsaicin was given and the haemodynamic and biochemical variables were measured again, as described above.
In the remaining 14 animals, subdivided into two subgroups (n = 7 each; for vehicle and sumatriptan, respectively), apart from determining the variables described above, venous blood samples (for plasma CGRP concentration) were withdrawn after each dose of capsaicin (0.3, 1 and 3 and 10 µg/kg/min) given before and after treatment with vehicle (n = 7) or sumatriptan(300 µg/kg; n = 7).
Data presentation and statistical analysis
All data are presented as mean ± s.e.mean. The statistical analysis was performed using the SPSS package for windows (version 10.0; SPSS Inc., Chicago, IL, USA). The significance of changes within one group (vehicle or sumatriptan) was analysed with repeated-measures
Ethical approval
The Ethics Committee of the Erasmus MC, Rotterdam, dealing with the use of animals in scientific experiments, approved the protocols for this investigation.
Compounds and kits
The following compounds were used: aprotinin (5850 KIU/mg; Roth, Karlsruhe, Germany), azaperone (Stresnil®; Janssen Pharmaceuticals, Beerse, Belgium), sumatriptan succinate (gift from Dr H.E. Connor, Glaxo Group Research, Stevenage, Hertfordshire, UK), capsaicin, tween 80, ethanol and phenylephrine hydrochloride (all from Sigma-Aldrich Chemie b.v., Zwijndrecht, the Netherlands), ethylene dinitro-tetraacetic acid (Merck, Darmstadt, Germany), heparin sodium (to prevent blood clotting in catheters; Leo Pharmaceutical Products, Weesp, the Netherlands), midazolam hydrochloride (Dormicum®; Hoffmann La Roche b.v., Mijdrecht, the Netherlands) and sodium pentobarbital (Sanofi Sante b.v., Maasluis, the Netherlands). The radioimmunoassay kit for CGRP was purchased from Peninsula Laboratories, Inc. (Belmont, CA, USA).
Capsaicin was initially dissolved in tween 80, ethanol and physiological saline in the ratio of 0.5 : 1 : 8.5 ml, respectively. Phenylephrine was dissolved in distilled water, while sumatriptan was dissolved in physiological saline.
Results
Baseline values
Baseline values (i.e. after capsaicin vehicle infusion) in the 28 pigs used were: heart rate, 133 ± 3 beats/min; mean arterial blood pressure, 108 ± 2 mmHg; total carotid blood flow, 47 ± 4 ml/min; total carotid conductance, 44 ± 3 10−2 ml/min/mmHg; A-V SO2 difference, 36 ± 2%; and plasma CGRP concentration, 13 ± 1 pg/ml. No significant difference in the baseline values were found between the two groups that subsequently received vehicle (n = 15; data not shown) or sumatriptan (n = 13).
Effect of different doses of capsaicin on heart rate, blood pressure and total carotid blood flow and conductance
Figure 1 depicts heart rate, mean arterial blood pressure and total carotid blood flow and conductance values at baseline and following different doses of capsaicin (0.3, 1, 3 and 10 µg/kg/min, i.c.) given before (control response) and after treatment with sumatriptan (100 and 300 µg/kg, i.v.) or corresponding volumes of vehicle (data withheld in this and subsequent figures for the sake of clarity, but these will be included in the PhD thesis of U. Arulmani). In both groups, capsaicin infusion dose-dependently increased the heart rate (last two doses), mean arterial blood pressure and total carotid blood flow and conductance. These changes to capsaicin remained essentially unmodified following vehicle or sumatriptan treatment. However, a small, but significant, attenuation in capsaicin-induced increase in total carotid conductance was observed following the highest dose of sumatriptan treatment (300 µg/kg, i.v.; P < 0.05; Fig. 1).
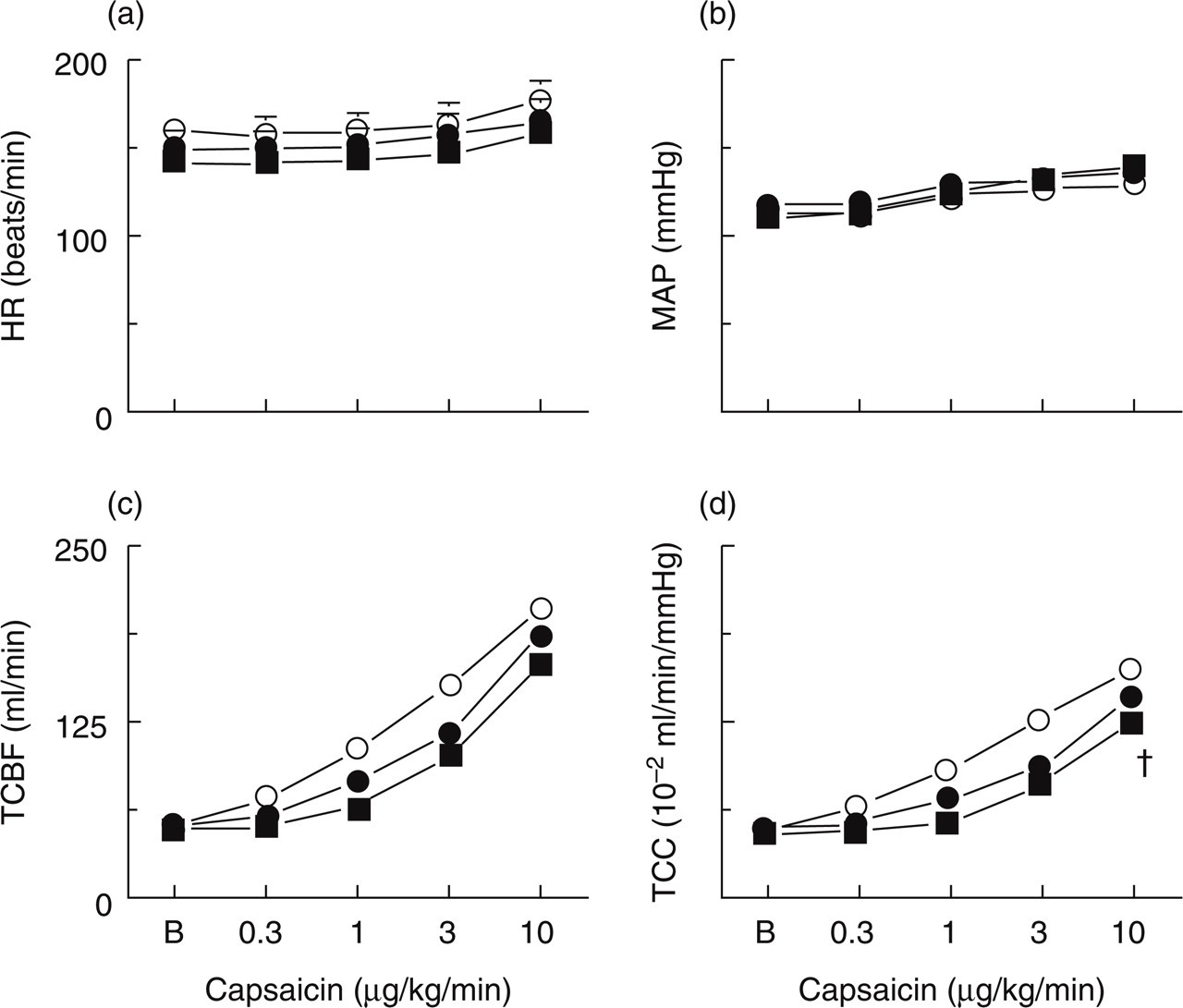
(a) Heart rate (HR), (b) mean arterial blood pressure (MAP), (c) total carotid blood flow (TCBF) and total carotid vascular conductance (TCC) values at baseline (B) and following infusions of capsaicin (0.3, 1, 3, 10 µg/kg/min, i.c.) in anaesthetized pigs before (○ Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (• 100 and ▪ 300 µg/kg; n = 13 each). All values are expressed as mean ± s.e.mean. Capsaicin increased MAP as well as TCBF and TCC (significance not shown for the sake of clarity). Apart from a small decrease in TCC to the highest dose of sumatriptan, capsaicin-induced changes were not altered by vehicle or sumatriptan treatment. †P < 0.05 vs response after the corresponding volume of vehicle.
Capsaicin-induced carotid haemodynamic changes
The effects of capsaicin (10 µg/kg/min, i.c.) on carotid haemodynamics were investigated in detail in animals receiving vehicle or sumatriptan.
Carotid blood flow, conductance and pulsations
The effects of capsaicin (10 µg/kg/min, i.c.) on total carotid blood flow and conductance (depicted as maximum absolute changes) as well as pulsations (represented as arbitrary units; a.u.), before (control response) and after treatment with sumatriptan (100 and 300 µg/kg/min, i.v.) are shown in Fig. 2. In both treatment groups (vehicle group data not shown) the control capsaicin infusion significantly increased the carotid blood flow and conductance as well as pulsations. While vehicle and sumatriptan (100 µg/kg, i.v.) were devoid of any significant effect on capsaicin-induced increases in carotid haemodynamics, a small, but significant, decrease in the carotid vascular conductance was observed in the animals treated with the highest dose of sumatriptan (300 µg/kg, i.v.; P < 0.05; Fig. 2).

Maximum changes in (a) carotid blood flow, (b) vascular conductance and (c) pulsations measured at baseline (□) and following infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before ( Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (
Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan ( 100 and ▪ 300 µg/kg; n = 13 each). All values are expressed as mean ± s.e.mean. a.u., Arbitrary units. ∗P < 0.05 vs baseline values; †P < 0.05 vs response after the corresponding volume of vehicle.
100 and ▪ 300 µg/kg; n = 13 each). All values are expressed as mean ± s.e.mean. a.u., Arbitrary units. ∗P < 0.05 vs baseline values; †P < 0.05 vs response after the corresponding volume of vehicle.
Fractionation of carotid vascular conductance
As shown in Fig. 3, capsaicin infusion significantly increased total carotid, arteriovenous anastomotic and capillary conductances. The capsaicin-induced increases in conductances from baseline values (maximal percent changes) were, respectively: total carotid, 329 ± 39; arteriovenous anastomoses, 554 ± 406; and capillary fraction, 340 ± 30. While vehicle (data not shown) as well as sumatriptan treatment did not affect capsaicin-induced changes in total carotid and capillary fractions, the increase in arteriovenous anastomotic conductance was markedly attenuated by sumatriptan. Moreover, following the highest dose of sumatriptan (300 µg/kg, i.v.), a small decrease in capsaicin-induced increase in total carotid conductance was observed (P < 0.05; see Figs 1 and 3).

(a) Total carotid, (b) arteriovenous anastomotic (AVA) and (c) capillary vascular conductances measured at baseline and following infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before ( Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (
Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan ( 100 and ▪ 300 µg/kg; n = 6 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values; †P < 0.05 vs response after the corresponding volume of vehicle.
100 and ▪ 300 µg/kg; n = 6 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values; †P < 0.05 vs response after the corresponding volume of vehicle.
Furthermore, capsaicin infusion significantly increased the vascular conductance to the different cranial tissues, including skin, ear, skeletal muscles, fat, bone, salivary glands, eye, tongue and dura mater, but not that to brain. These vasodilator responses to capsaicin remained unaltered after sumatriptan (100 and 300 µg/kg, i.v.; Fig. 4) or the corresponding volumes of vehicle (data not shown).

Distribution of carotid vascular conductances to head tissues measured at baseline (□) and following infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before ( , control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (
, control) and after i.v. administrations of vehicle (data not shown) or sumatriptan ( S100 and ▪ S300, 100 and 300 µg/kg, respectively; n = 6). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values.
S100 and ▪ S300, 100 and 300 µg/kg, respectively; n = 6). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values.
Difference between arterial and jugular venous oxygen saturations (A-V SO2 difference)
Consistent with the increase in arteriovenous anastomotic conductance, capsaicin infusion (10 µg/kg/min, i.c.) significantly decreased the A-V SO2 difference from baseline values from 39 ± 4% to 4 ± 0.6% (control response; n = 28). This response to capsaicin remained unaffected after treatment with vehicle (data not shown) or sumatriptan (Fig. 5).

Differences between arterial and jugular venous oxygen saturations (AV SO2 difference) measured at baseline (□) and after infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before ( , Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (
, Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan ( , 100 and ▪ 300 µg/kg; n = 13 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values.
, 100 and ▪ 300 µg/kg; n = 13 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values.
Capsaicin-induced jugular venous plasma concentration of CGRP
Figure 6 depicts the plasma CGRP concentrations at baseline (i.e. after capsaicin vehicle infusion) and following capsaicin infusion (10 µg/kg/min, i.c.) before (control response) and after sumatriptan (100 and 300 µg/kg, i.v.) or the corresponding volumes of vehicle (data not shown). The capsaicin infusion significantly increased plasma CGRP concentrations from a baseline value of 11 ± 1 pg/ml to 28 ± 4 pg/ml (maximal percent change from baseline: 155 ± 38). These responses to capsaicin were not attenuated by either vehicle or sumatriptan.

Jugular venous plasma CGRP concentrations measured at baseline (□) and after infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before ( ,Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (
,Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan ( , 100 and ▪ 300 µg/kg; n = 6 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline value.
, 100 and ▪ 300 µg/kg; n = 6 each). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline value.
Furthermore, capsaicin infusions (0.3, 1,3 and 10 µg/kg/min, i.c.) dose-dependently increased plasma CGRP concentrations (a significant increase was observed during the last two doses of capsaicin; Fig. 7). These responses remained unaffected after treatment with either vehicle (data not shown) or sumatriptan (300 µg/kg, i.v.).

Jugular venous plasma CGRP concentrations measured at baseline (B) and after different doses of capsaicin infusion (0.3, 1, 3 and 10 µg/kg/min, i.c.) given in anaesthetized pigs before (○, Control) and after i.v. administrations of vehicle (data not shown) or sumatriptan (•, 300 µg/kg; n = 7). All values are expressed as mean ± s.e.mean. ∗P < 0.05 vs baseline values.
Finally, it is noteworthy that even with further increasing the dose of sumatriptan (i.e. 1000 µg/kg/min, i.v.), the above responses to capsaicin (carotid haemodynamic changes as well as increased plasma CGRP concentrations) did not significantly differ from the vehicle-treated animals (data not shown).
Discussion
General
Migraine headache is a neurovascular syndrome in which the neural events seem to involve stimulation of the trigeminal system and an ensuing release of CGRP from perivascular trigeminal nerves (1, 2).
The introduction of triptans(5-HT1B/1D/1F receptor agonists) in migraine therapy has focused on the role of serotonin receptors in migraine (19, 20). Triptans seem to abort migraine attacks by two main mechanisms: firstly, constriction of dilated cranial blood vessels and carotid arteriovenous anastomoses via the stimulation of 5-HT1B receptors (21, 22) and, secondly, inhibition of CGRP release as well as of nociceptive transmission on peripheral and central trigeminal sensory nerves via 5-HT1B/1D receptors (2, 23). Since trigeminal inhibition of CGRP release may reduce cranial vasodilatation and nociception, the present study set out to investigate the effects of sumatriptan on capsaicin-induced porcine carotid haemodynamic changes and the associated increase on plasma CGRP concentrations. Our results in anaesthetized pigs show that i.c. administration of capsaicin increased total carotid (including arteriovenous anastomotic and capillary) blood flows and conductances, carotid pulsations as well as jugular venous plasma CGRP concentrations and narrowed the A-V SO2 difference. Interestingly, sumatriptan failed to modify capsaicin-induced carotid haemodynamic changes, except those in arteriovenous anastomoses and capsaicin-induced increase in plasma CGRP concentrations.
Systemic haemodynamic effects of capsaicin
The systemic haemodynamic effects of capsaicin have been investigated extensively (24, 25). Indeed, the significant increases in heart rate and mean arterial blood pressure observed in our study with capsaicin are in accordance with these findings, which are due to a central activation of the sympathetic outflow.
Carotid haemodynamic changes by capsaicin
Several studies have shown that sensory nerves innervating the cerebral vasculature contain substance P and CGRP (6, 26). However, capsaicin-induced relaxation of guinea-pig isolated basilar artery is mediated by CGRP rather than by substance P (27, 28). Moreover, BIBN4096BS, a potent CGRP receptor antagonist, abolished capsaicin-induced porcine carotid haemodynamic responses (24), a finding which shows the involvement of CGRP in the responses to capsaicin.
Apart from some attenuation of arteriovenous anastomotic changes, the capsaicin-induced responses were not affected by sumatriptan (Figs 1–4). These results are in keeping with other findings showing that sumatriptan failed to block capsaicin-induced relaxation of guinea pig isolated basilar artery (28) as well as carotid vasodilatation induced by trigeminal ganglion stimulation (29–31).
It is noteworthy that the decrease in total carotid conductance by sumatriptan is predominantly due to the decrease in carotid arteriovenous anastomotic conductance, even when all triptans slightly increase the nutrient conductance (32, 33). Considering this, it is most likely that the above apparent inhibition by sumatriptan on capsaicin-induced responses (Fig. 3) is due to physiological antagonism (i.e. vasoconstriction by sumatriptan) rather than inhibition of CGRP release, since sumatriptan failed to modify this variable (see Figs 6 and 7). In contrast, the apparent failure of sumatriptan to attenuate capsaicin-induced increase in carotid conductance may have been due to massive bursts of CGRP release produced by capsaicin and/or an insufficient dose of sumatriptan. However, the latter possibility can be excluded as treatment with an even higher dose of sumatriptan (1000 µg/kg/min, i.v.) did not modify capsaicin-induced carotid haemodynamic changes (unpublished observation).
A-V SO2 difference
The dilatation of carotid arteriovenous anastomoses with an associated decrease in the A-V SO2 difference is a characteristic feature observed during migraine headache (34). Consistent with the above finding, our study shows that capsaicin decreased the A-V SO2 difference, which is presumably due to a CGRP-mediated dilatation of carotid arteriovenous anastomoses (24). As expected, sumatriptan decreased the carotid arteriovenous anastomotic conductance in our study (Fig. 3); however, its failure to normalize the capsaicin-induced A-V SO2 difference may be explained in terms that capsaicin-induced increase in capillary conductance may lead to oxygen saturation in the cranial tissues. Therefore, these tissues cannot extract oxygen further, thereby shunting the oxygenated blood via capillaries rather than via carotid arteriovenous anastomoses.
Plasma levels of CGRP
Several lines of evidence have shown that stimulation of the trigeminal system with electrical or chemical (capsaicin) stimuli releases the endogenously stored CGRP (7, 24, 28, 35–38), which produces cranial vasodilatation (7, 24, 39). Interestingly, triptans have been reported to attenuate CGRP release elicited by both electrical (7, 35–37,40) and chemical (capsaicin) (38) stimulation; the latter effect was barely significant and observed with sumatriptan in a concentration (50 µ
Notwithstanding, other lines of evidence seem to show just the opposite, namely, that triptans do not modify the cranial vasodilatation produced by either trigeminal stimulation (30, 31) or capsaicin (28). Admittedly, in these studies CGRP concentrations were not measured in parallel and, consequently, no categorical conclusion can be drawn with regards to the role of inhibition of CGRP release in the antimigraine action of triptans. The present in vivo study shows that sumatriptan failed to modify the capsaicin-induced increases in plasma CGRP levels and the associated carotid vasodilatation and the carotid arteriovenous anastomotic constriction by sumatriptan is not associated with a corresponding change in CGRP levels.
Admittedly, there is no clear-cut explanation why sumatriptan seems to attenuate CGRP release upon electrical stimulation but not by capsaicin. It is known that capsaicin activates a subset of small sensory fibres that cover a major proportion of C and some Aδ fibres (39, 42–44), while low intensity electrical stimulation recruits Aδ fibres alone (39). Thus, one explanation for the differential effect of sumatriptan may be that, compared to electrical stimulation, capsaicin releases high quantities of CGRP that can be potently antagonized by selective vanilloid VR1 antagonists (45), but not by a presynaptic mechanism involving sumatriptan. Another possibility is that, in contrast to the capsaicin-sensitive C and δ fibres, the δ fibres recruited by electrical stimulation possess 5-HT1B/1D/1F receptors stimulated by sumatriptan (2, 20). We have indeed shown that the mRNAs for 5-HT1B and 5-ht1F receptors are expressed in the porcine trigeminal ganglia (46, 47). Finally, sumatriptan does not easily penetrate the blood brain barrier (20) and, should CGRP be mainly released from intracerebral sources, it will inhibit such a release only if the blood brain barrier is disrupted.
Possible clinical implications
Finally, the possible clinical implications of our results with sumatriptan within the context of antimigraine therapy must be considered. Therefore, based on our findings, the blockade of the postjunctional effects of CGRP (with BIBN4096BS) (24) would seem to be a better therapeutic strategy to prevent neurogenic vasodilatation rather than trigeminal inhibition of CGRP release (via the activation of prejunctional 5-HT1B/1D receptors by sumatriptan). Moreover, in view of the putative pathophysiological role of arteriovenous anastomotic dilatation in migraine (34, 48), the constriction of these non-nutrient vessels by sumatriptan in our study may be responsible for the therapeutic action of this drug in migraine.
In conclusion, our results imply that prejunctional 5-HT1B/1D receptors (activated by sumatriptan) do not inhibit capsaicin-induced vasodilatation in the porcine carotid circulation and increase in plasma CGRP concentrations. Therefore, the primary mechanism behind the clinical efficacy of sumatriptan in migraine may be due to constriction of cranial blood vessels rather than neurogenic inhibition of CGRP release.
Footnotes
Acknowledgements
The authors would like to thank Dr H.E. Connor, Glaxo Group Research, Stevenage, Hertfordshire, UK for providing sumatriptan and Erasmus Pharma b.v. for the financial support.
