Abstract
Objective
The current study evaluated the consistency of eletriptan response.
Methods
Using a within-patient crossover design, patients with migraine completed a three-attack, open-label, lead-in period, before being treated, double-blind for four attacks, with either eletriptan 40 mg (ELE-40; N = 539) or eletriptan 80 mg (ELE-80; N = 432); placebo was randomly substituted for the treatment of one attack.
Results
On an a priori analysis of within-patient consistency, double-blind treatment was associated with similar 2 hour headache response rates using a ≥2/3 response criterion for ELE-40 (77%) and ELE-80 (73%), and using a 3/3 response criterion for ELE-40 (46%) and ELE-80 (47%). Within-patient consistency in achieving pain-free status at 2 hours using a ≥2/3 criterion was slightly higher on ELE-40 (42%) compared with ELE-80 (38%), and was similar using the 3/3 criterion (18% on ELE-40, 17% on ELE-80). On a repeated measures logistic regression analysis across all treated attacks, the probability of achieving a headache response at 2 hours ranged from 71% to 74% on ELE-40 vs. 17% to 28% on placebo (p < 0.0001), and from 66% to 74% on ELE-80 vs. 21% to 27% on placebo (p < 0.0001). The incidence, per attack, of adverse events was low for both ELE-40 and ELE-80. Few adverse events occurred with incidence ≥10% on ELE-40 (asthenia, 5.0%) or ELE-80 (asthenia, 10%; nausea, 5.8%). Discontinuations because of adverse events were 0.2% on ELE-40, and 1.6% on ELE-80
Conclusion
In this multiple attack study, eletriptan was well-tolerated and demonstrated consistent and significant efficacy in the treatment of migraine.
Clinicaltrials.Gov Identifier: NCT01859481
Introduction
Community surveys indicate that more than 40% of individuals with a diagnosis of migraine have an average headache frequency of two or more attacks per month, while approximately one out of four report experiencing one or more attacks per week (1). Consistency of response is rated by patients as being one of the key characteristics of any medicine used to treat migraine (2). Underscoring its importance, lack of consistency of response has been cited in patient surveys as one of the top three reasons for dissatisfaction with current migraine treatments (3).
Among important clinical outcomes in migraine treatment studies, consistency of response is the least often reported, and even fewer studies are designed to provide an a priori evaluation of this parameter. The recommendation of the Clinical Trials Subcommittee of the International Headache Society (IHS) is that consistency of response should be assessed using a randomized, double-blind, crossover design in which placebo is substituted for one of five treated attacks (4). IHS guidelines also recommend that consistency of response be evaluated utilizing a within-patient analysis in addition to the more commonly employed group average response.
Typically, studies report consistency data based on results from long-term extension phase studies (5,6). These studies may overestimate consistency as they lack a placebo control, are subject to selection bias (only acute phase responders enter the extension phase), are biased toward super-responders, as these are the subjects most likely to stay in an open-label study across a year, and consistency of response is calculated as a group mean across all attacks, instead of calculating a within-patient (intra-patient) mean. In contrast, only a few published studies that we are aware of meet IHS design guidelines for consistency studies (7–9). In a study of the 10 mg dose of rizatriptan, the within-patient consistency rate for headache response was 73% on three out of four attacks, and 37% on four out of four attacks (7,8) with a response rate when placebo was substituted ranging from 28% to 54%. In a second report (9), data were presented from two identical randomized, double-blind, crossover studies in which four attacks were treated within 1 hour after pain onset, with sumatriptan/naproxen sodium, and placebo was substituted for the treatment of a fifth attack. Pain-free response at 2 hours in two out of three attacks was achieved by 55% of patients in Study 1 and 52% of patients in Study 2. The strength of this study is the use of remission endpoints such as sustained pain-free at 24 hours.
Eletriptan has demonstrated within-patient consistency of response based on three previous studies using a randomized, double-blind, placebo-controlled, parallel-group design (10–12). For eletriptan 40 mg (ELE-40), consistency of response on two out of three attacks ranged from 63% to 77%, and for eletriptan 80 mg (ELE-80), two out of three consistency ranged from 72% to 82%.
We now report the results of a randomized clinical trial using IHS-recommended double-blind, placebo-controlled, crossover methodology, and designed to provide a rigorous test of within-patient consistency of response for eletriptan versus placebo.
Patients and methods
Patients
Male or female adults, aged 18–65 years were eligible for study entry if they met IHS criteria for migraine with or without aura (13) and reported a minimum of one migraine attack every 6 weeks, but not more than six attacks per month, or had failed to respond to previous adequate medication therapy. Patients were excluded from the current trial if they reported a history of coronary artery disease, clinically significant arrhythmia, heart failure or uncontrolled hypertension; active, clinically significant renal, hepatic, gastrointestinal, neurological, endocrine, metabolic or psychiatric disease as determined by medical history and/or physical examination; clinically significant electrocardiogram abnormalities at screening; severe limitation of gastrointestinal absorption; or clinically significant allergic reaction to eletriptan, sumatriptan or any other 5-HT1 agonist. Women who were pregnant or lactating were excluded.
The study was conducted between March 2000 and March 2002 at 180 outpatient headache centers and private practice sites in 14 European countries according to the standards set forth in the Declaration of Helsinki (1996 revision) and consistent with all International Conference on Harmonization good clinical practice guidelines. The protocol was approved by an ethics review committee at each site. Study procedures were explained to prospective patients, and written informed consent was obtained prior to study entry. The screening medical evaluation consisted of a physical examination, vital signs, urine pregnancy testing and 12-lead ECG; routine laboratory assessments were not performed.
Study design
This was a double-blind, four-attack consistency of response study in which three attacks were treated with ELE-40 or ELE-80, and one randomly chosen attack was treated with placebo. At randomization, each patient was allocated to a treatment sequence covering treatment for all three attacks. This randomization was created by a computer-generated pseudo-random code using the method of random permuted blocks. Prior to randomization, all patients first completed an open-label lead-in period during which they treated three migraine attacks with ELE-40 within a 3 month time-period. Based on the efficacy and tolerability exhibited during the three-attack lead-in period, the investigator, in consultation with the patient, made a decision regarding whether the patient should continue on ELE-40 mg during the double-blind phase, or escalate to the 80 mg dose. Once this decision was made, patients were not permitted to change dosage levels during the next 24-week, four-attack experimental phase of the study. Thus, patients who elected the 80 mg dose of eletriptan were a heterogeneous mixture of patients who did not achieve optimal headache or pain-free response on each of the three attacks during the lead-in period with 40 mg, and those who did achieve a good response, but elected to increase nonetheless for better efficacy, as subsequent up-titration was not permitted.
During both open-label and double-blind treatment, patients were instructed to take study medication when they experienced a typical migraine attack, after the aura phase (if one occurred) had ended, but not until the pain was of at least moderate severity.
Patients recorded migraine-related symptoms in a diary at baseline (immediately pre-dose), and at 30 minutes, 1 hour, 2 hours, 4 hours and 24 hours after dosing. Use of rescue medications was also recorded in the diary.
Patients were permitted to take rescue medication if they failed to achieve a headache response (defined below) by 2 hours post dose. They were not permitted to take any other triptan, ergotamine or ergotamine-like substance for 24 hours post dose. Patients who achieved a 2 hour headache response, but experienced a recurrence of moderate to severe headache were permitted to take a second dose of study medication (40 mg or 80 mg), provided that 2 hours had elapsed since the initial dose. The time of recurrence and the second dose, and information on rescue medication, were noted in the diary card. Rescue medication was permitted 2 hours after this second dosing if needed.
Evaluation of efficacy
The primary efficacy endpoint used to assess intra-individual consistency in the current study was the proportion of patients with headache response at 2 hours after the first dose of study medication in three out of three attacks during the double-blind phase. Headache pain was rated by the patient on a four-point scale as 0 = none, 1 = mild pain, 2 = moderate pain, and 3 = severe pain. Headache response was defined as improvement in headache intensity (on this four-point global scale), from a pre-treatment level of moderate or severe to a 2 hour post-dose intensity of none (pain-free) or mild in the three attacks treated with active drug.
Secondary efficacy endpoints consisted of the following: 1. individual headache and pain-free response at 2 hours; 2. headache recurrence between 2 and 24 hours after ingestion of study medication; 3. use of rescue medication; 4. sustained headache and pain-free response within 24 hours after the first dose of study medication; 5. acceptability of study treatment, defined by the response at 24 hours to the question: ‘Given the choice between this and any other previous medication you have used to treat a migraine attack, would you take this again?’; and 6. satisfaction with study medication, defined by self-rating on a seven-point Likert scale ranging from ‘7’ (completely dissatisfied, couldn’t be worse) to ‘1’ (completely satisfied, couldn’t be better).
Statistical analyses
The primary objective of this study was to assess the consistency of headache response in subjects on optimum treatment with eletriptan 40 mg or 80 mg. The primary, a priori, outcome of the study was the proportion of subjects who achieved a headache response at 2 hours after the first dose of study medication in three out of three attacks during the double-blind phase (Phase 2). All efficacy analyses were performed on the sample of patients who took at least one dose of study medication and had a valid baseline and at least one post-baseline headache evaluation. The safety sample consisted of all patients who took study medication, regardless of whether the treated migraine attack met study severity criteria and therefore was a valid attack. Sample size determination was based on a measure of consistency of headache response, the proportion of subjects who responded to treatment at 2 hours post dose for all three eletriptan treated attacks in initial sample of subjects in Phase 2 of the current study. A confidence interval approach was used for determining the sample size requirement.
The power analysis determined that a sample size of 271 subjects per group in the double-blind phase was needed to provide 95% confidence for detecting a difference in consistency of response between eletriptan and placebo. As approximately 35% and 30% dropout rates were expected in Phases 1 and 2 of the study, respectively, it was expected that approximately 1200 subjects would need to be enrolled at study entry.
The efficacy of eletriptan versus placebo was evaluated using a logistic regression model, with repeated measurements, performed on patients with available data on all four attacks in the double-blind phase. Generalized estimating equations (GEE) were used to account for within-patient correlation among observations (using PROC GENMOD in SAS with the REPEATED option). The model included terms for treatment, attack and baseline headache severity for each attack. All statistical tests of significance were performed at the 5% level of significance, and were two-sided tests unless otherwise specified. Analyses were not adjusted for multiplicity.
Results
Patient sample and disposition
A total of 1191 patients entered, and 971 (82%) completed the three-attack lead-in period (Figure 1), and continued into the double-blind, experimental phase of the study, 539 on ELE-40, and 432 on ELE-80. The three most common reasons for discontinuation during the lead-in period were other/administrative reasons (5.5%), protocol violation (3.3%) and adverse events (2.9%; Figure 1). During the double-blind phase, 45 patients (8.3%) on ELE-40 and 62 patients (14%) on ELE-80 discontinued prematurely (Figure 1). Only two patients (<1%) on the 40 mg dose and nine patients (2%) on the 80 mg dose discontinued because of insufficient response.
Flow diagram.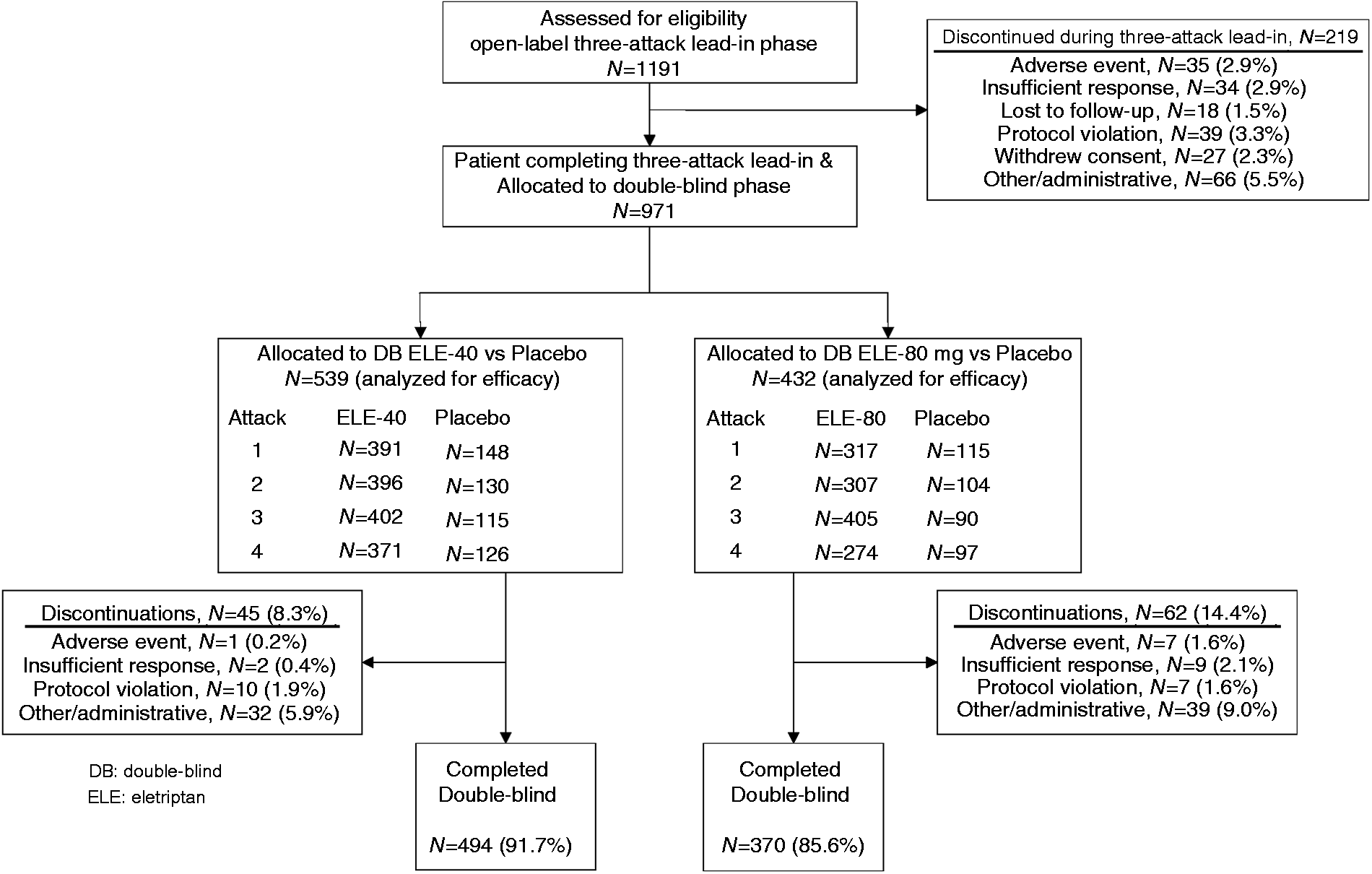
A total of 1156 subjects out of 1191 had at least one valid attack in Phase 1 and comprised the intent-to-treat population. Entering double-blind phase with at least one valid attack in Phase 1, 422 (36.5) out of 1156 elected to escalate to 80 mg dose of eletriptan and 531 (46%) patients continued on the 40 mg of eletriptan; 203 (17.5%) did not continue to Phase 2. Of the 422 subjects who elected to escalate to 80 mg, 215 (51%) had achieved a headache response on the 40 mg dose of eletriptan in at least two out of three migraines during the three-attack lead-in period. Of the 531 patients who continued on the 40 mg dose of eletriptan, 436 (82%) had achieved a headache response in at least two out of three migraines during the three-attack lead-in period, while 18% were non-responders using these criteria.
Characteristics of the patient sample.

*Time since first diagnosis.
Headache response and pain-free rates: consistency data
On an a priori analysis of within-patient consistency (Figure 2(a)), treatment with ELE-40 (n = 381) and ELE-80 (n = 279), respectively, were associated with similar 2 hour headache response rates using an at least two out of three response criteria (77% and 73%), and a three out of three response criteria (46% and 47%). Headache response at 2 hours was achieved in at least one out of three attacks by 92% of patients on ELE-40 and 87% of patients on ELE-80. Within-patient consistency in achieving pain-free status at 2 hours was proportionately lower, whether based on the at least two out of three criterion (42% on ELE-40, 38% on ELE-80), or the three out of three criterion (18% on ELE-40, 17% on ELE-80; Figure 2(a)). The current analysis did not include subjects with lower attack frequency. For example, 37/531 (7%) of patients treated with ELE-40 and 40/422 (9%) treated with ELE-80 treated only one migraine attack during the double-blind phase of the study. Among these patients with low frequency attacks, pain-free rates at 2 hours were higher for both ELE-40 (41%) and ELE-80 (40%).
Within-patient consistency of headache response and pain-free. (a) 2 hour headache response and pain-free rates in ≥2/3 and 3/3 treated attacks, %. (b) Sustained headache response and pain-free rates at 24 hours in ≥2/3 and 3/3 treated attacks, %.
Sustained headache response and pain-free rates: consistency data
Using the IHS within-patient consistency metric, the proportion of patients achieving a 24 hours sustained headache response on two out of three attacks was 26.5% for ELE-40 and 29.0% for ELE-80; and on three out of three attacks was 23.4% for ELE-40 and 18.6% for ELE-80 (Figure 2(b)). Using the same IHS metric, the proportion of patients achieving a 24 hour sustained pain-free outcome on two out of three attacks was 18% for ELE-40 and 19% for ELE-80; and on three out of three attacks was 11% for ELE-40 and 8% for ELE-80 (Figure 2(b)).
Headache response and pain-free rates: individual attack data
On a repeated measures logistic regression analysis across all four treated attacks (Figure 3), the probability of achieving a headache response at 2 hours ranged from 71% to 74% on ELE-40 versus 17% to 28% on placebo (p < 0.0001), and from 66% to 74% on ELE-80 versus 21% to 27% on placebo (p < 0.0001). The probability of achieving a pain-free outcome at 2 hours ranged from 40% to 44% on ELE-40 versus 6% to 12% on placebo (p < 0.0001), and from 38% to 39% on ELE-80 versus 6% to 10% on placebo (p < 0.0001).
Headache response and pain-free rates at 2 hours: individual attack data based on a logistic regression analysis with repeated measures.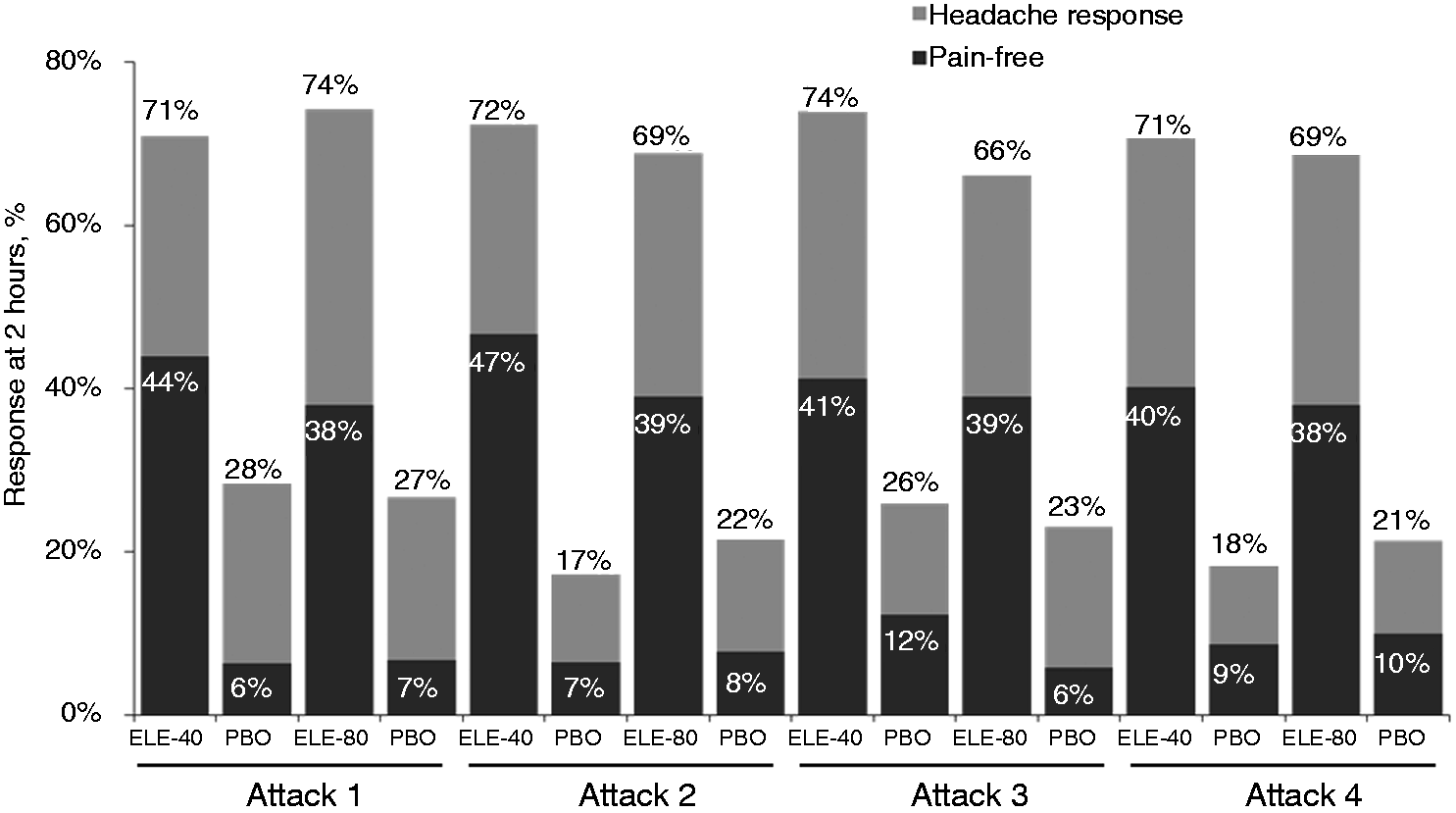
Sustained headache response and pain-free rates: individual attack data
On a repeated measures logistic regression analysis across all four treated attacks (Figure 4), the probability of achieving a sustained headache response ranged from 50% to 52% on ELE-40 versus 0% on placebo (p < 0.0001), and from 44% to 47 on ELE-80 versus 0% on placebo (p < 0.0001). On a repeated measures logistic regression analysis across all four treated attacks, the probability of achieving a sustained pain-free outcome ranged from 31% to 34% on ELE-40 versus 0% on all four attacks on placebo (p < 0.0001), and from 27% to 28% on ELE-80 versus 0% on all four attacks on placebo (p < 0.0001).
Sustained headache response and sustained pain-free rates at 24 hours: individual attack data.
Treatment satisfaction and acceptability versus previous treatments
Patient satisfaction with study medication, rated on a seven-point scale, was calculated using the logistic regression method. The proportion of patients reporting ‘somewhat’, ‘very’ or ‘complete’ medication satisfaction across the four treated attacks ranged from 75% to 77% for ELE-40 and 65% to 68% for ELE-80, compared with 24% to 36% for placebo. Similarly, the proportion of patients rating their current study medication as preferable to previous migraine treatments across the four treated attacks ranged from 79% to 80% for ELE-40 and 64% to 69% for ELE-80, compared with 29% to 44% for placebo. Previous treatment with a triptan was reported by 72.5% of patients.
Effectiveness of ELE-80 mg in patients with persistent non-response to ELE-40 mg
During the three-attack lead-in period, a subgroup of patients treated with the 40 mg dose of eletriptan responded to zero out of three attacks (N = 60) or one out of three attacks (N = 120), and were titrated to the 80 mg dose of eletriptan during the double-blind, experimental phase. Titration to an 80 mg dose of eletriptan resulted in conversion to responder status on the first attack in more than half of patients. Among patients who had responded to zero out of three attacks on ELE-40 during the lead-in period, headache response rates at 2 hours ranged from 42.5% to 60% on ELE-80 versus 7% to 31% on placebo, and pain-free rates at 2 hours ranged from 23% to 27% on ELE-80 versus 3% to 6% on placebo. Among patients who had responded to one out of three attacks on ELE-40 during the lead-in period, headache response rates at 2 hours ranged from 50% to 58% on ELE-80 versus 18.5% to 25% on placebo and pain-free rates at 2 hours ranged from 42.5% to 56% on ELE-80 versus 0% to 12.5%% on placebo.
Tolerability and safety
Treatment-emergent adverse events. a

Data shown are average per attack incidence across four treated attacks in the double-blind phase; sample sizes for eletriptan 40 mg and placebo for attack 1 (n = 391; n = 148), attack 2 (n = 396; n = 130), attack 3 (n = 402; n = 115), attack 4 (n = 371; n = 126); and for eletriptan 80 mg and placebo for attack 1 (n = 317; n = 115), attack 2 (n = 307; n = 104), attack 3 (n = 305; n = 90), attack 4 (n = 274; n = 97).
NNH: number needed to harm, calculated as the inverse of the eletriptan vs. placebo difference of the incidence of each adverse event.
Note that NNH cannot be calculated where the adverse event rate is higher on placebo.
Discussion
The results of the current consistency study, utilizing the IHS-recommended within-patient design, indicate that 77% of patients treated with a 40 mg dose of eletriptan demonstrated consistency of response based on the criterion of headache response at 2 hours in two out of three attacks, and 46% achieved headache response on three out of three attacks. The consistency rates for the 80 mg dose of eletriptan were similar (2/3 = 73%; 3/3 = 47%).
In the past decade, sustained headache and pain-free response have emerged as important efficacy criteria in migraine treatment studies, especially in clinical trials where triptans are taken early, when headache pain is mild. The current study did not use the early (mild pain) dosing strategy, and thus is not comparable with more recent trials that used this dosing strategy (9). The only previously reported study that utilized a within-patient design that was similar to the current study (except that it was a four-attack study) and a similar time of administration (when headache pain was moderate-or-greater) was the study of the 10 mg dose of rizatriptan (7,8). In that study, treatment with rizatriptan was associated with a consistency rate for headache response of 73% on three out of four attacks, and 37% on four out of four attacks. However, the placebo response rate in the rizatriptan study ranged from 28% to 54%, which was much higher than the placebo response rate observed in the current trial (17–28%). The reason for the widely variable placebo response rate in the previous trial is not known.
Cross-study comparisons must be made with caution because of differences in the clinical characteristics of patients in each study. Notable in the current study is the unusually high proportion of treated attacks in which headache pain was reported as ‘severe’. In single-attack migraine treatment studies utilizing the conventional dosing strategy, the proportion of patients reporting headache pain as ‘severe’ is typically in the range of 35–40% (5,14). In the current multiple attack study, approximately 60.5% of patients overall reported at least two out of four treated attacks as severe. By contrast, in the similarly designed rizatriptan consistency study (7), only 17% of patients reported having a severe headache in at least two out of four treated attacks. The low severity of the migraine patient sample in the rizatriptan study, and the resulting high placebo response rate (20% higher than in the current study) means that it is impossible to make useful cross-study comparisons of response consistency between the two studies, as the populations studied appear quite different.
Multiple attack studies provide a more stringent test than single attack studies of the acceptability and satisfaction with an acute migraine therapy. This study found high rates of satisfaction on both the 40 mg dose of eletriptan (75–77%) and the 80 mg dose (65–68%), with the majority of the satisfaction ratings contributed by patients who were ‘very’ or ‘completely’ satisfied). Similarly, on the basis of the overall efficacy and tolerability across multiple attacks, eletriptan was rated as the preferred treatment choice by a high proportion of patients on both the 40 mg dose (79–80%) and the 80 mg dose (64–69%).
Several limitations of the current study should be noted. First, as noted above, patients were instructed not to take study medication until their headache pain was moderate-to-severe in intensity. More recent research has demonstrated higher pain-free rates if a triptan is taken early, when headache pain is still mild. The extent to which the delayed treatment strategy used in the current study reduced headache response and pain-free rates is uncertain. A second study limitation is the duration of the trial. Although a four-attack design is consistent with IHS recommendations, nonetheless, treatment of multiple attacks over a 6–12 month period would have provided invaluable information on patterns of response to long-term treatment. A third study limitation is the absence of an active comparator drug. We are unaware of any study, based on an IHS-recommended design that compares consistency of response for two triptans, or for a triptan and non-triptan therapy. A fourth study limitation is the potential for a selection bias resulting from study entry criteria that excluded patients with high frequency migraine attacks (at least seven per month), and subjects who had failed to respond to previous adequate medication therapy.
A final point is that patients on the 80 mg dose of eletriptan in the current study were a more treatment-resistant subgroup, many of whom (42%) had not achieved a consistent (at least two out of three) response rate on the 40 mg dose of eletriptan administered during the open-label lead-in phase of the study. A secondary finding of the current study is that titration to the 80 mg dose of eletriptan is an effective treatment strategy for the subgroup of patients who have demonstrated non-response to the 40 mg dose (zero out of three attacks). Among the 60 patients who reported a zero out of three headache response rate when treating three migraine attacks with the 40 mg dose of eletriptan during the open-label lead-in period, titration to the 80 mg dose was associated with a 2 hour headache response rate in the range of 42.5% to 60% during the three subsequent attacks. Triptans, with the possible exception of rizatriptan, typically have not been reported to exhibit a significant dose-response curve for efficacy when treating headache of moderate-to-severe pain (15). However, few migraine studies are designed to evaluate the efficacy of various treatment options (e.g. dose escalation versus switching) in patients who have failed to respond to initial treatment. The design of the current study appears to suggest that increasing to an 80 mg dose of eletriptan may be an effective treatment option for patients who have failed to respond to the 40 mg dose. It is important to note that the 80 mg dose of eletriptan is not an approved dose in the USA.
It is also important to note that despite various treatment strategies (e.g. early dosing, dose-escalation or switching to an alternative triptan), only a minority of migraine patients across all trials of oral triptans achieve true remission as judged by the IHS recommended outcome of pain-free at 2 hours in three out of three attacks (5).
In conclusion, the results of the current study confirm that treatment with eletriptan resulted in within-patient consistency of headache response in two out of three attacks in 77% of patients, and with sustained response and pain-free rates across multiple attacks that are relatively high for a conventional dosing strategy where treatment is taken when pain is moderate-to-severe. Both doses of eletriptan were well tolerated, with adverse events that were typically transient and mild. As a result, satisfaction was high, with three out of four patients preferring eletriptan to all previous migraine treatments.
Clinical implications
Given the frequency and chronicity of migraine attacks, studies evaluating consistency of response, especially utilizing the recommended within-patient design, provide evidence for efficacy that is important, but continues to be under-reported. The results of the current trial indicate that eletriptan demonstrates consistency of response in three out of four patients. Overall, there is no evidence of a dose-response effect when treating migraine attacks of moderate to severe in intensity. However, a subgroup of patients was identified that were non-responders to the 40 mg dose of eletriptan, but who achieved a response when the dose was titrated up to 80 mg.
Footnotes
Acknowledgments
We would also like to express our appreciation to the Investigators who participated:
M De Tourtchaninoff, P Louis, T Piette, J Schoenen, MEH Van Zandijcke
P Docekal, V Martina, I Rektor, G Waberzinek
A Korsgaard, P Staehr, P Tfelt-Hansen, F Winther-Bach, A Ziebel
A Autret, M Bouchacourt, G Chazot, P Cormier, B Delfiner, G Geraud, P Henry, P Houdart, C Kubler, M Lanteri-Minet, B Laurent, P LeMarquis, C Lucas, J Marcombes, H Massiou, G Mick, B Mihout, A Pradalier, J Siboni, T Solsson, D Valade, J Vaunaize, M Veyrie-Courtade, J Visy
H Beikuefner, U Dickmann, R Frieling, R Kreyer, B Kukowski, E Mauch, G Schumann, B Steinberg, S Strache, T Wallasch
S Harmoussis, K Karageorgiou, I Mylonas, E Nikolakaki
J Bank, L Csiba, G Gacs, P Harcos, G Jakab, I Jelencsik, S Komoly
G Bussone, MG Buzzi, A Carolei, M Del Zompo, M Diomedi, R Di Perri, V Gallai, GC Manzoni, P Montagna, B Panascia, F Perini, L Pinessi, L Provinciali, G Relija, G Sandrini, L Savi, E Sternieri, D Volta, G Zanchin
J Carpay, P van Erven, R Groen, R van Leeuwen, W Linssen, L Mulder, W Mulleners, J Niewold, P Pop, P Schiphof, MG Smits, T Tacke, J Ter Bruggen
K Amthor, I Monstad, K Nestvold, S Strandquist
L Bigotte de Almeida, M Palmeira, J Palmeiro
E Alias Linares, Angeles del Real, J Burguera Hernandez, O Fábregues, JC García Moncó, E Governa Ortiz, P Granés Ibáñez, C Hernández Lahoz, F Iglesias Díez, D Jiménez, A Jiménez-Escrig, JF Martí Massó, JA Pareja Grande, JAH Pérez, J Quesada Córcoles, G Rubio Esteban, C Targa Benet, F Titus Albareda, E Varela de Seijas Slocker, T Villanueva Lorente
L Åkerman, S Boes-Hansen, B Borre, L Brattström, R Brinkeborn, C Dahlöf, L Edvinsson, I Eilenberg, M Elm, U Ericsson, S Eriksson, G Fogdegård, A Granvik, R Hagströmer, Y Hallström, G Hedman, T Hermansson, B Jacobson, B Jansson, G Malmqvist, T Moberg, O Olsson, P Östgård, Rollsjö, M Rosengren, E Stockenwall, E Strandman, B Tengmark, E Waldenlind
M Özmenoğlu, Y Sarica
Funding
Funded by Pfizer Inc.
Conflict of interest
M. Almas and E. Ramos are full-time employees of Pfizer, Inc.
Dr. Tepper has served as a speaker, consultant, or scientific advisory board member for Allergan, Inc.; Autonomic Technologies, Inc.; GlaxoSmithKline; Helsinn Group; MAP Pharmaceuticals, Inc.; Merck & Co., Inc.; Nautilus Neurosciences, Inc.; NuPathe, Inc.; and Zogenix, Inc.; and has received research support from Autonomic Technologies, Inc.; Bristol-Myers Squibb; Depomed, Inc.; GlaxoSmithKline; MAP Pharmaceuticals, Inc.; Merck & Co., Inc.; NuPathe, Inc.; and Zogenix, Inc. Dr Tepper has stock options from Autonomic Technologies, Inc.
Dr. Landy has no conflicts to declare.
E. Schweizer is the owner of Paladin Consulting Group, Inc., which was a paid consultant to Pfizer Inc in connection with the development of this manuscript; and owns stock in Pfizer Inc and Novartis.
