Abstract
This double-blind, placebo-controlled study was designed to evaluate the efficacy and tolerability of early treatment of a single migraine attack, when headache pain was mild, with two doses (20 mg and 40 mg) of eletriptan. Patients (N = 613; female 79%; mean age 39 years) meeting International Headache Society criteria for migraine were encouraged, but not required, to utilize early treatment, thus providing an opportunity to assess the relative contribution to efficacy of pain severity and timing of dose. For the total patient sample (mild-to-severe headaches), 2-h pain-free rates were significantly higher than placebo (22%) on both eletriptan 20 mg (35%; P < 0.01) and eletriptan 40 mg (47%; P < 0.0001). For the cohort of patients who treated their headache when the pain intensity was mild, the 2-h pain-free rate on eletriptan 40 mg was 68% compared with 25% on placebo (P < 0.0001). Pain intensity at the time of taking eletriptan appeared to influence outcome more than the timing of the dose relative to headache onset. Eletriptan was well-tolerated, with adverse event rates similar to placebo when mild headaches were treated.
Recent post hoc analyses (1–5) of patients in standard double-blind, placebo-controlled clinical trials who took study treatment early have consistently shown that treatment of headache when pain is mild is associated with higher pain-free rates at 2 h than when treatment is administered once pain is moderate to severe in intensity.
It has been suggested that the benefit of early treatment may derive, at least in part, from the fact that it occurs prior to the hypothesized development of central trigeminovascular sensitization (6). Patients who treat with a triptan after central sensitization has developed appear to be less likely to achieve a pain-free state (6).
Several prospective, double-blind, placebo-controlled trials have now been reported that confirm the efficacy advantage conferred by early treatment intervention. Winner and colleagues (7) reported pain-free rates at 2 h from two studies, combined, that were significantly higher for sumatriptan 100 mg (57%) compared with placebo (29%; P < 0.001). Most of the patients (>95%) took sumatriptan within 30 min of the onset of their headache, when pain intensity was mild. The efficacy advantage of early treatment includes higher migraine-free rates at 2 h, where migraine-free is defined as no headache pain and no associated symptoms at 2 h. Combined study data (7) found significantly higher migraine-free rates at 2 h for sumatriptan 100 mg (49%) compared with placebo (24%; P < 0.001). More recently, data on the efficacy of a new formulation of sumatriptan used to treat mild migraine has been reported (8). Migraine-free rates at 2 h were also significantly higher on the new 100-mg formulation of sumatriptan (51%) compared with placebo (17%; P < 0.001).
Eletriptan is a potent 5HT1B/1D receptor agonist that is rapidly absorbed with relatively high (50%) bioavailability, a short time to maximal concentration (T max = 1–1.5 h), and a relatively long half-life among triptans (4.0 h). The current study was designed to evaluate the efficacy and tolerability of two doses of eletriptan (20 mg and 40 mg) in the early treatment of migraine, when headache pain was mild. As described in Patients and methods, patients were encouraged but not required to utilize early treatment, when headache pain was mild.
Patients and methods
Patients
Eligible patients consisted of women or men, aged 18–65 years, who met the International Headache Society criteria for migraine with or without aura (9), with a duration of illness of at least 1 year, and who reported an average frequency of one to four attacks per month over the past 3 months. Unlike previously reported early treatment studies (7), patients were not required to have migraine headaches in which a mild pain phase preceded the moderate-to-severe phase.
Patients were excluded for the following reasons: (i) presence of frequent non-migrainous headache, chronic daily headache, or atypical or migraine variant presentations (e.g. familial hemiplegic, basilar migraine, migraine with prolonged aura, etc.); (ii) any history of cerebrovascular or coronary artery disease, heart failure, uncontrolled hypertension or abnormal electrocardiogram (ECG); (iii) any clinically significant medical illness or laboratory abnormalities; (iv) severe reduction in gastrointestinal absorption; (v) current use of prophylactic medication for the treatment of migraine; (vi) hypersensitivity or known contraindication to treatment with eletriptan; (vii) concomitant use of potent CYP3A4 inhibitors; (viii) misuse or abuse of alcohol or other substances, including analgesics or ergotamine; (ix) use of any experimental drug within the past month; and (x) women who were pregnant, breast-feeding, or sexually active without use of medically acceptable contraception.
At screening, the study was explained to prospective patients, and written informed consent was obtained prior to study entry. All patients had a physical and neurological examination, including blood pressure, ECG, and urine pregnancy testing (for women). Study conduct was consistent with the Declaration of Helsinki. The study protocol was approved by Institutional Review Boards (Ethics Committees) at each site.
Study design
This randomized, double-blind, parallel-group, out-patient study was conducted at 33 centres in the USA and seven centres in Canada. Patients who met all eligibility criteria were randomly assigned in a 1 : 1 : 1 ratio to treat one attack with either 20 mg or 40 mg of eletriptan, or placebo.
Patients were instructed to take study medication as soon as they were sure they were experiencing a typical migraine headache, and after the aura phase (if present) had ended. Patients were encouraged, but not required, to take study medication when headache pain was mild. Furthermore, patients were instructed not to take study medication if they had already taken an analgesic or antiemetic during the current attack, or in the previous 6 h, or if another triptan or ergotamine-containing or ergot-type medication (e.g. dihydroergotamine) had been taken in the previous 48 h.
Patients recorded migraine-related symptoms in a diary at baseline (immediately predose), and at 30 min, 1 h, 1.5 h, 2 h, 4 h, and 24 h after dosing. Use of rescue medications was also recorded in the diary.
If patients failed to achieve a headache response by 2 h, they were permitted to take rescue medication, but were not permitted to take another dose of eletriptan or any other triptan, ergotamine or ergotamine-like substance for 24 h postdose. Patients who achieved a 2-h headache response but experienced a recurrence of headache were permitted to take a second dose of study medication (the second dose was the same as the first dose).
Evaluation of efficacy
Severity of headache pain was measured using a 4-point pain intensity scale, with scores ranging from 3 = severe pain to 0 = no pain. The primary efficacy endpoint was the per cent pain-free (scored 0 = no pain) at 2 h postdose.
Secondary endpoints consisted of the following: (i) percentage of patients who were pain free at other assessment time points (30 min, 1.0 h, 1.5 h, 4 h, and 24 h); (ii) headache response (in the subgroup of patients with moderate-to-severe headache pain at treatment baseline); (iii) relief of the associated symptoms of nausea, vomiting, photophobia, and phonophobia; (iv) improvement from pretreatment baseline in a 4-point functional impairment scale; [improvement was analysed in terms of both functional response (per cent of patients whose functional status improved from 2 or 3 to 0 or 1) or normal functioning (per cent who improved from 1, 2, or 3–0)]; (v) headache recurrence within 24 h; (vi) use of rescue medication; and (vii) sustained pain free, defined as achieving a pain-free state within 2 h postdose, and remaining pain free up to 24 h with no additional use of rescue or study medication.
Statistical analysis
The primary efficacy comparison, pain free at 2 h, was between the 20-mg and the 40-mg doses of eletriptan and placebo. The study was powered at 85% to detect as significant (α level = 0.05, two-tailed) an 8-point difference between eletriptan 40 mg and placebo on the 2-h pain-free endpoint. The 8-point difference was considered to represent a clinically meaningful difference between the two medications.
Baseline characteristics of the sample were compared for homogeneity across treatment groups. All efficacy analyses were performed on the intent-to-treat (ITT) sample, defined as all subjects who took at least one dose of study medication and had a valid baseline and at least one post-baseline evaluation. The primary and secondary analyses were conducted using a categorical linear model based on the SAS procedure CATMOD, which included terms for treatment and baseline severity. The response function was the logit of the responder rate. The primary comparisons were between the eletriptan groups and the placebo group. The first comparison was eletriptan 40 mg vs. placebo. If this result was significant, eletriptan 20 mg was to be compared with placebo. This step-down procedure preserved the experimentwise error rate at 5% level on the parameter. All treatment comparisons were performed at the 5% significance level and were two-sided.
Results
Study sample
A total of 799 patients were screened, of whom 613 met all eligibility criteria and were randomized to one of the three study treatments. Twenty patients failed to meet day-of-attack criteria, and 28 patients had missing headache pain intensity assessments at 2 h; thus, the primary a priori analysis, pain free at 2 h, was based on total sample of 565 patients.
The demographic and clinical characteristics of the patients were similar across the three treatment groups, eletriptan 20 mg, eletriptan 40 mg, and placebo, respectively, in terms of gender (female, 79% vs. 83% vs. 85%), mean (+ SD) age (39.1 ± 10.8 vs. 38.7 ± 11.5 vs. 39.1 ± 11.7 years), mean duration of illness (13.4 ± 11.3 vs. 14.0 ± 11.2 vs. 13.6 ± 11.5 years), proportion of patients without aura (73% vs. 68% vs. 67%), and mean (+ SD) monthly attack frequency (8.3 ± 3.2 vs. 8.6 ± 3.0 vs. 8.0 ± 3.1).
Despite encouragement from the investigators to treat attacks early when headache pain intensity was mild (but when they were sure it was a migraine), only 21–28% of patients took study medication when pain was mild and only 16–18% took study medication within 1 h of headache onset, when pain was mild (Table 1). Almost half (44–47%) of patients reported that their headache pain was moderate to severe despite dosing less than 1 h after headache onset. In fact, of the subgroup of 234 patients who took study drug within 30 min of headache onset, 70% reported that their pain was already moderate to severe in intensity.
Clinical characteristics of the treated attack categorized by time from headache onset to intake of study medication (intent-to-treat sample)

Relative to headache onset.
Regardless of the time of dosing, moderate to severe levels of headache pain (vs. mild) were associated with notably higher rates of associated symptoms such as nausea (51% vs. 31%, P = 0.001), photophobia (79% vs. 66%, P = 0.001), and phonophobia (66% vs. 47%, P = 0.001). This was especially true of functional impairment, where rating of ‘severe’ or ‘bed rest’ occurred almost exclusively in patients whose pain was moderate to severe (75% vs. 7%, P = 0.001).
Efficacy: pain-free response
For the total sample, pain-free rates at 2 h postdose were significantly higher on both eletriptan 20 mg (35%; P < 0.01) and eletriptan 40 mg (47%; P < 0.0001) compared with placebo (22%). As summarized in Table 2, achieving a pain-free response at 2 h appeared to be more strongly associated with the intensity of headache pain at the time of drug administration than with the time of administration relative to headache onset. This is shown graphically in Fig. 1, and is especially notable for the 40-mg dose of eletriptan, which showed similar pain-free rates at 2 h when pain intensity was mild at the time of taking eletriptan, regardless of whether the time of dosing was less than 1 h after headache onset (69%), or more than 1 h (67%). For the baseline-mild subgroup (Fig. 2), the slope of the pain-free response over the first 4 h postdose was notably steeper for eletriptan 40 mg compared with eletriptan 20 mg.
Pain-free response at 2 h after administration of eletriptan or placebo: comparison of outcome based on time from headache onset to dosing, and pain severity at time of dosing
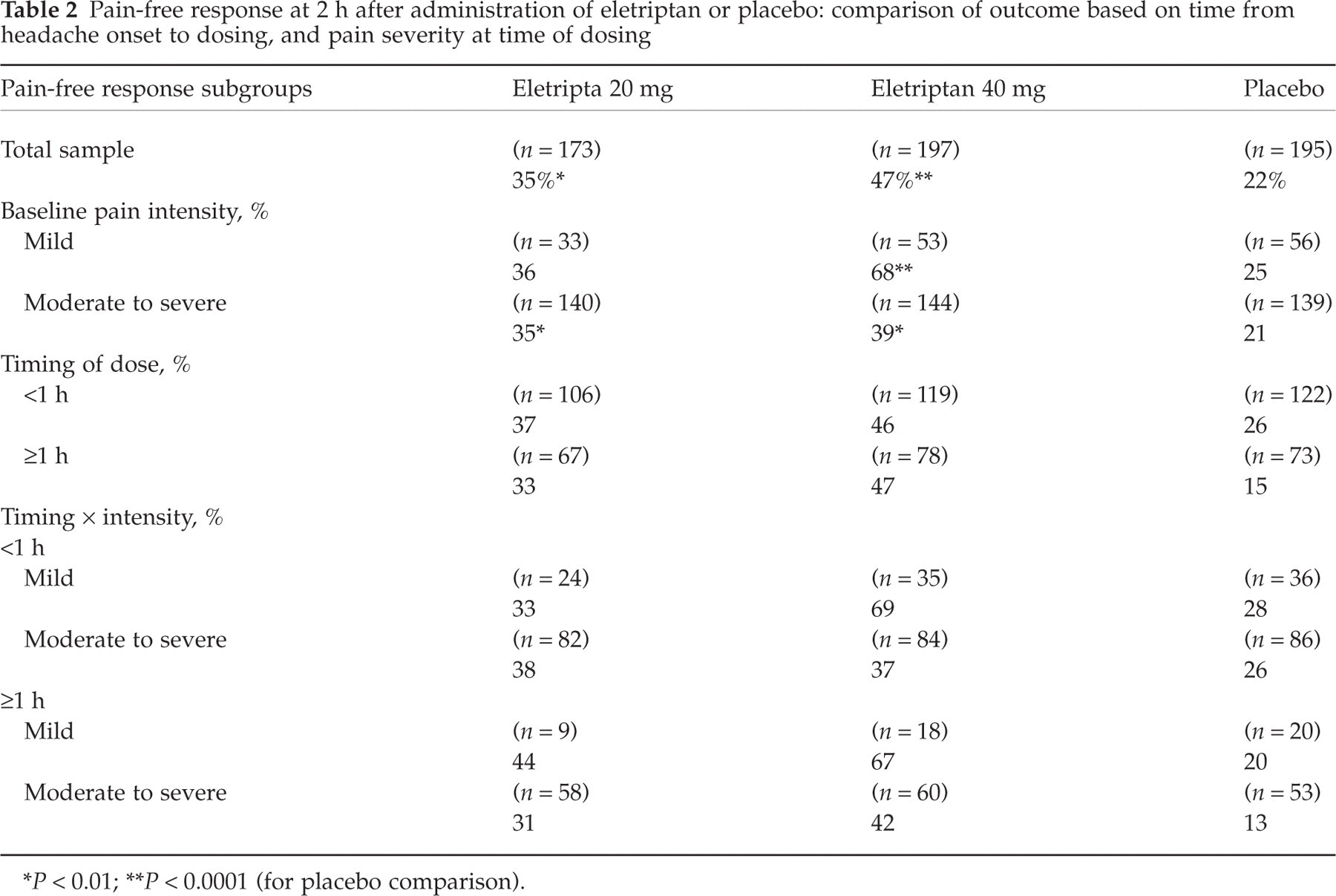
P < 0.01;
P < 0.0001 (for placebo comparison).

The interaction of timing of dose and baseline pain intensity of pain-free response at 2 h. ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.001.

Pain-free and sustained pain-free rates in the subgroup of patients with mild headache pain at the time of taking study drug. ∗P < 0.05; ∗∗P < 0.0001. •, Eletriptan 20 mg (N = 38); ▪, eletriptan 40 mg (N = 55); ▵, placebo (N = 57).
The relative contribution of baseline pain intensity and timing of dose (after headache onset) was examined in a regression analysis, using pain-free status at 2 h as the dependent variable. A preliminary analysis showed Spearman correlations of 0.109 for baseline pain intensity, and 0.043 for time of dose. The regression model showed that only baseline pain intensity was a significant independent predictor of 2 h pain free, with an odds ratio of 1.8 (95% confidence interval 1.2, 2.7) for mild vs. moderate-to-severe pain (P < 0.0039).
When pain-free status at 2 h was analysed in the smaller subgroup of patients (n = 113) who treated mild pain within 30 min after headache onset vs. > 1 h, respectively, rates were found to be similar for eletriptan 20 mg (40% vs. 44%), eletriptan 40 mg (71% vs. 67%), and placebo (23% vs. 20%). Furthermore, early treatment of migraine (<30 min after headache onset vs. > 1 h after onset) did not increase pain-free rates at 2 h for the subgroup of patients whose headache pain was moderate-to-severe level of intensity: for eletriptan 20 mg, 39% vs. 31%; for eletriptan 40 mg, 32% vs. 42%; and for placebo, 30% vs. 13%.
Efficacy: treatment-emergent headache pain
Treatment of mild headache pain may be considered an acute treatment that preempts the progression of pain. Thus, prevention of emergent moderate-to-severe pain in the 4 h postdose was analysed as a secondary efficacy outcome. Among the subgroup (25%) who took study drug when their headache pain was mild, 30% on placebo experienced worsening of headache pain, which was significantly higher than the incidence of worsening headache pain at 4 h on eletriptan 20 mg (2.9%; P < 0.05) and eletriptan 40 mg (3.7%; P < 0.01).
Efficacy: associated symptoms
Treatment with the 20-mg dose of eletriptan was associated with a significantly lower incidence (vs. placebo) at 2 h of nausea (83% absent vs. 75%; P < 0.05), photophobia (66% absent vs. 51%; P < 0.001), and phonophobia (74% absent vs. 55%; P < 0.0001). Treatment with the 40-mg dose was associated with a significantly lower incidence of photophobia (74%; P < 0.001) and phonophobia (81%; P < 0.0001) but not nausea (76%).
Efficacy: improvement in functioning
A significantly higher proportion of patients reported fully normal functioning at 2 h on both the 20-mg (46%) and 40-mg (51%) doses of eletriptan compared with placebo (31%; P < 0.001 for both comparisons). For the subgroup of patients (58% of the total sample) with moderate-to-severe functional impairment at baseline, rates of functional response at 2 h were significantly higher for eletriptan 20 mg (70%) and 40 mg (75%) compared with placebo (44%; P < 0.001 for both comparisons).
Complete (migraine-free) response at 2 h
The most stringent acute outcome occurred in patients who were pain-free, reported no associated symptoms, and had normal functioning. This complete or ‘migraine-free’ outcome was analysed in the subgroup of patients who reported mild headache pain at the time of drug administration. For this mild subgroup (Fig. 3), a complete response was achieved at 2 h by 33% of patients on eletriptan 20 mg, 68% of patients on eletriptan 40 mg, and 23% of patients on placebo (P < 0.001 for the 40-mg dose of eletriptan vs. placebo). For the total ITT sample, complete relief at 2 h was achieved by 32% of patients on the 20-mg dose of eletriptan, 43% of patients on the 40-mg dose, and 20% of patients on placebo (P < 0.01 for the eletriptan 20 mg vs. placebo comparison, and P < 0.0001 for the eletriptan 40 mg vs. placebo comparison).

Complete migraine relief rates in the subgroup of patients with mild headache pain at time of taking study drug. ∗P < 0.01; ∗∗P < 0.001; †P = 0.06. •, Eletriptan 20 mg (N = 38); ▪, eletriptan 40 mg (N = 55); ▵, placebo (N = 57).
Use of rescue medication, recurrence, and sustained pain-free at 24 h
There was a significantly lower use of rescue medication among patients treated with both doses of eletriptan regardless of the level of headache severity at the time of drug administration (Table 3). Using the stringent criterion, return of mild or greater pain, recurrence rates were lower on both doses of eletriptan, but were only significant for the 40-mg dose vs. placebo for the total sample (15% vs. 30%; P < 0.05) and for the mild pain subgroup (6% vs. 41%; P < 0.01; Table 3). Among patients in the total sample who were pain free at 2 h, significantly more showed sustained pain-free rates at 24 h on eletriptan 20 mg (29%) and 40 mg (37%) compared with placebo (15%; P < 0.001 for both comparisons). Lower baseline pain severity was associated with an increase in sustained pain-free rates (Table 3; Fig. 2). It should be noted that the definition of sustained pain free was absence of any headache pain at 2 h and no return of any pain within 24 h.
Outcomes at 24 h: comparison based on pain severity at time of initial administration of eletriptan or placebo
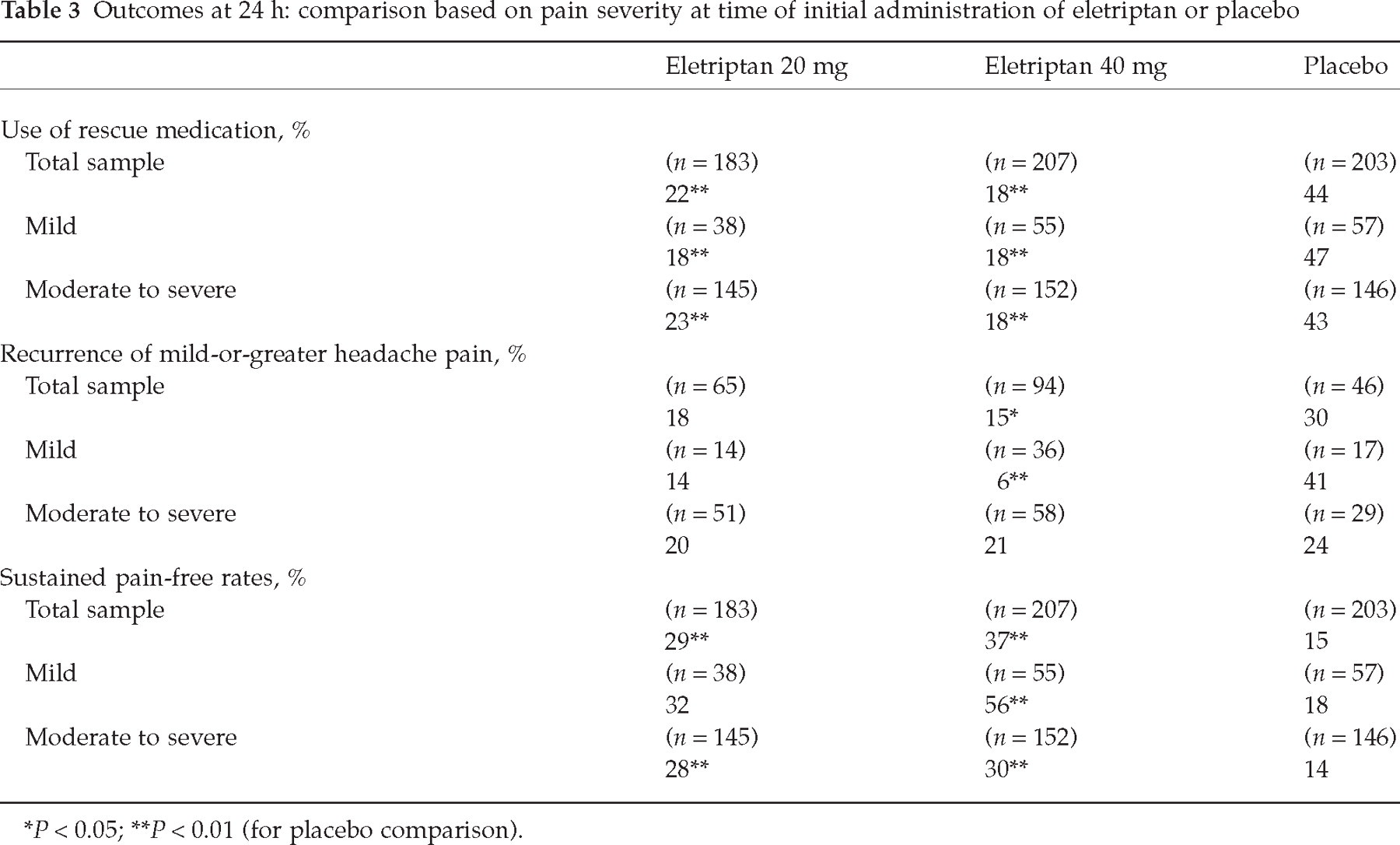
P < 0.05;
P < 0.01 (for placebo comparison).
Tolerability and safety
The following all-causality adverse events were reported at a rate of ≥ 2% (and more frequently than placebo) for eletriptan 20 mg, eletriptan 40 mg, and placebo, respectively: vomiting (4.7% vs. 3.8% vs. 3.8%), dizziness (2.6% vs. 1.4% vs. 1.9%), and asthenia (2.1% vs. 1.9% vs. 0.5%). No serious treatment-related adverse events occurred on eletriptan. Overall, the incidence of any adverse event, regardless of causality, was lower on eletriptan 20 mg (28%) and 40 mg (23%) compared with placebo (32%).
Discussion
Treatment of mild headache pain with eletriptan was associated with high 2-h pain-free rates on both eletriptan 20 mg (36%) and 40 mg (68%) compared with placebo (25%). The high pain-free rates resulted in almost identical complete migraine relief rates (no pain, normal functioning, and no associated symptoms), 33% and 68%, respectively, on the 20-mg and 40-mg doses compared with 23% on placebo. The benefit of treating mild headache pain was maintained over the 24 h postdose, with sustained pain-free rates of 32% on eletriptan 20 mg and 56% on eletriptan 40 mg compared with 17% on placebo.
The design of the current study encouraged, but did not require, patients to take study medication early, when headache pain was mild. As a result, patients were heterogeneous in terms of both time from headache onset to dosing (early vs. late) and in terms of pain intensity at the time of dosing (mild vs. moderate to severe). The study therefore provides a unique opportunity for evaluating the relative contribution of the two variables, headache severity and time of dosing, on triptan efficacy. The results suggest that the efficacy advantage of early treatment is more strongly correlated with pain intensity at the time of dosing than it is with the time of dosing itself. This was confirmed by the results of a regression analysis, which indicated that treatment of mild headache results in a significant 80% increase in the odds of achieving a pain-free state at 2 h postdose. In contrast, early treatment (within 30–60 min of headache onset) was not significantly associated with a pain-free outcome. The benefit of administering treatment when headache pain was still mild was especially notable for the 40-mg dose of eletriptan, where treatment of mild pain was associated with pain-free rates at 2 h that were 25–30% higher, regardless of time of administration relative to headache onset.
The benefit of taking eletriptan when headache pain is still mild is clear when the results of the current study are placed in the context of results from a pooled analysis of data from three standard eletriptan clinical trials in which patients were treated when headache pain was moderate to severe in intensity (10). Pain-free rates at 2 h for the mild subgroup of patients in the current study were 68% on eletriptan vs. 25% on placebo. This represents an 11% efficacy advantage over patients receiving standard treatment (10), where pain-free rates were 57% on eletriptan (vs. 26% on placebo). The efficacy advantage of early treatment is amplified when sustained pain-free rates are compared for early treatment (eletriptan 40 mg, 56% vs. placebo, 18%) and standard treatment (eletriptan 40 mg, 22% vs. placebo, 3%). It is important to note that caution should be exercised in comparing any results across studies.
The results of the current study, taken together with the three previously reported sumatriptan early treatment studies (7, 8), all provide data supporting the importance of rapid treatment intervention when headache pain is still at mild levels. Such early treatment intervention appears to be effective in preventing the emergence of moderate-to-severe pain. When treatment-emergent worsening of pain is examined as an outcome in its own right, the 40-mg dose of eletriptan was found to be highly effective compared with placebo in preventing the emergence of headache, with worsening rates at 2 h of 3.8% on eletriptan 40 mg compared with 20% on placebo, and 3.7% at 4 h compared with 30% on placebo.
In a recent editorial, Ferrari was appropriately critical of studies that claimed early treatment benefit, while constructively articulating ideal clinical trial designs of variable complexity and practicality to address the question rigorously and conclusively (11). This was not the primary purpose or design of our present study, but, more than prior studies, it does contain elements that approach the ideal parallel group design of stratified and randomized groups of early or late treatment of mild or severe headache called for in the editorial (11). In our study, these four subgroups evolved out of baseline characteristics generated by loose compliance with medication instructions. Although groups were of small number and possibly not appropriately balanced for baseline headache characteristics, the results were sufficiently encouraging that such a clinical trial design conducted prospectively might resolve the issue. Further, we were able to establish benefit of treating mild pain using the most rigorous outcomes suggested by Ferrari, namely sustained relief of headache and associated symptoms without rescue medication (11). Our study is also distinguished from others by assessing prevention of progression from mild headache, an essential criterion according to Ferrari.
Both doses of eletriptan were well tolerated in the current study. Adverse events typically reported with triptans occurred at a low rate with eletriptan, raising the possibility that central sensitization mechanisms might be associated with a more generalized sensitivity to somatic symptoms. Thus, early treatment of mild headache pain might increase the tolerability of therapy with eletriptan, as well as other triptans. Future research is needed to confirm this hypothesis. Long-term studies are also needed to evaluate whether early intervention is associated with an increased risk of over-treating headaches with triptans. The benefit of early intervention (when headache pain is mild, but before the headache is confirmed as a migraine) must be balanced against the risk of medication overuse and the potential for increasing migraine frequency.
In conclusion, early treatment with eletriptan, when headache pain was mild, was associated with higher pain-free and sustained pain-free rates than previously observed in clinical trials with moderate-to-severe headache pain. The results shown here also demonstrate that the timing of drug administration had less influence on outcome than the level of pain intensity at the time of treatment. The tolerability of eletriptan was similar to placebo when administered when headache pain was mild. These results confirm and extend the findings of previous studies demonstrating the clinical benefits of early treatment of migraine headaches.
Footnotes
Acknowledgements
We would like to express our appreciation to the investigators who participated in this study: Rudolf Arts, Jan L. Brandes, Roger Cady, Bruce Corser, James Couch, Merle Diamond, George Dooneief, Amin R. Elashker, John Ervin, Grace Forde, W. Thomas Garland, Jr, Larry I. Gilderman, Jerome Goldstein, Jon Heiser, David King, Rebecca Knight, David Kudrow, Michel Lebel, Elizabeth Loder, Ninan Mathew, Alexander Mauskop, Michael Norman, William Patterson, Charles Powers, Gary Ruoff, Joel Saper, Gary Shapero, Fred Sheftell, Stephen Silberstein, Denis Simard, Timothy Smith, Ward Smith, J. Christopher Stringer, Abraham Sunshine, Duncan Sze-Tu, Harvey Tilker, Randal Von Seggern, Geoffrey E. Starr. Supported by funding from Pfizer Inc.
