Abstract
Objective
The purpose of our study was to evaluate the effect of atrial septal defect (ASD) closure on migraine headache attacks (MHA).
Methods
A total of 247 patients who underwent percutaneous ASD closure at our facilities were sent a structured questionnaire. We diagnosed MHA according to the criteria of the International Headache Society.
Results
A total of 207 patients were included in the study. New-onset MHA occurred in 23 patients and persisted in 15 at a mean follow-up of 45 months. Of the 29 patients who had MHA prior to ASD closure, 11 reported exacerbation of MHA, 11 reported no change and seven reported improvement within three months after ASD closure. Compared with the patients who had no MHA, patients with de novo MHA were younger and patients with MHA improvement tended to be older. Switching from aspirin to ticlopidine or clopidogrel rapidly aborted frequent MHA in nine patients who had severe symptoms after ASD closure.
Conclusions
These results indicate that Amplatzer device implantation can act as a permanent trigger of MHA in not a few patients, and that age may be an important predictive factor of the influence of ASD closure on MHA.
Introduction
Recently, a relatively high prevalence of migraine, up to 30%, was found in patients with unclosed secundum type atrial septal defect (ASD) (1–4). Several studies have demonstrated a significant reduction or even an abolition of migraine headache attacks (MHA) after transcatheter closure of atrial septal defect (ASD) or patent foramen ovale (PFO) (5,6). However, it has also been reported that ASD closure can lead to de novo MHA or exacerbation of existing MHA (2,3,5,7–17). Most of these studies lack pediatric patients, and do not provide detailed and systematic descriptions of the MHA related to ASD closure. Therefore, the effect of ASD closure on MHA is not well understood. The purpose of our study was to evaluate MHA after ASD closure in a large cohort of consecutive patients including pediatric patients.
Methods
Patient selection
A total of 247 consecutive patients underwent successful transcatheter ASD closure at the Saitama Medical University Hospital between August 2005 and March 2007 (n = 45), and Saitama International Medical Center between April 2007 and March 2012 (n = 202). Both institutions are affiliated facilities of Saitama Medical University, Japan. All procedures were performed by femoral approach, under general anesthesia, and with transesophageal echocardiography guidance. The Amplatzer septal occluder device (AGA Medical Corporation, Golden Valley, MN, USA) was used in all cases. Heparin was administered in all cases. All patients received antithrombotic therapy for the six months after ASD closure. Antithrombotic therapy at discharge was confirmed from medical records. Most patients received echocardiography within one month of ASD closure, where residual shunt, device thrombosis, and cardiac abnormalities were assessed.
A structured questionnaire was composed in such a way that the information provided would enable a neurologist to diagnose migraine with or without aura (MA or MO, respectively), according to the International Classification of Headache Disorders, second edition (18). The questionnaire was mailed to all 247 patients from May 2012 to March 2013. The patients were asked to complete the questionnaire and return it in a prepaid envelope. The study was performed in accordance with the institutional ethics committee, and all subjects and/or their relatives gave informed consent for the study. The prevalence of MHA in patients aged ≥12 years was compared to the prevalence previously reported in a Japanese population (19–21), after adjustment for gender and age.
Evaluation of MHA
The structured questionnaire asked about the presence, frequency, intensity, duration, type, and site of headache, the occurrence of scotoma, paresthesis, paresis, aphasia, aggravating factors, accompanying symptoms, vomiting, sono- and photo-phobia, and whether the headache interrupted regular daily activities. All patients who reported daily headache before or within the three months after ASD closure underwent an additional interview referring to the medical record of those days about headaches. The neurologist determined if patients had a history of MHA prior to ASD closure, and if they had a history of MHA after ASD closure. Patients who had no history of MHA prior to ASD closure but had MHA within the three months after ASD closure were classed as de novo MHA. Patients who reported repeated MHA after ASD closure with little or no intervening periods free from MHA were classed as “status migrainosus.” The final follow-up time equals the time the questionnaire was sent. We analyzed our patients with a follow-up of at least one year. At final follow-up, patients who suffered from MHA after ASD closure were asked to grade the frequency of MHA as exacerbated, unchanged, improved, or completely resolved in comparison to the peak frequency after surgery.
Statistical analysis
Quantitative variables are expressed as mean ± standard deviation or median, and qualitative variables are expressed as percentage. Age, gender, defect size, device size, and pulmonary to systemic blood flow ratio (Qp/Qs) were compared across groups using a Wilcoxon rank sum test. Antithrombotic treatment at discharge was compared across groups using a chi square test or Fisher’s exact test. Differences were considered statistically significant if p < 0.05. The data were analyzed using PASW Statistics software version 18 (SPSS Inc, Chicago, IL, USA).
Results
Patient characteristics
A total of 207 patients (84%, mean age 27 ± 20 years, range 4 to 79 years, 61 men) returned the questionnaire and comprised the study population. The response rates of former and latter institutions were 87% and 83%, respectively. Mean follow-up period was 40.6 ± 17.9 months (range 12 to 80 months). The reason for ASD closure was a hemodynamically significant shunt in 206 patients and cerebrovascular accident in one patient. The prevalence of MHA prior to ASD closure was 14%. The prevalence of MHA in patients aged 12–19 years (12%) and ≥20 years (24%) seemed to be higher compared with literature data from the Japanese population after adjustment for gender and age (6% and 9--11%, respectively) (19–21). The transthoracic echocardiography performed within a month after ASD closure showed no significant residual shunt, device thrombosis, or cardiac abnormalities in any patient tested.
Antithrombotic therapy at discharge was aspirin (75–330 mg/day) in 152 patients, aspirin and ticlopidine (100–200 mg/day) in 42 patients, ticlopidine (200 mg/day) in five patients, aspirin and warfarin in four patients, ticlopidine and warfarin in two patients, dipyridamole (150 mg/day) and warfarin in one patient, and clopidogrel (75 mg/day) in one patient. Warfarin was used before the procedure in all seven patients who received warfarin at discharge; six received warfarin for atrial fibrillation and one for history of transient ischemic attack. Antithrombotic therapy added after the procedure was stopped at six months post-ASD closure in all patients. When this study was carried out, superiority of clopidogrel over aspirin was not proved. We therefore prescribed aspirin for many patients. In Japan, clopidogrel is approved only for the prevention of recurrent ischemic heart diseases and ischemic stroke, excluding cardioembolic stroke. Therefore, clopidogrel was seldom used as the first choice for antithrombotic therapy after the surgery.
Characteristics of patients who had no history of migraine headache attacks (MHA), who developed de novo MHA, and who experienced exacerbation, no change or improvement in existing MHA after transcatheter atrial septal defect (ASD) closure.

Data are mean ± standard deviation or %. aIndicates p = 0.03 vs. no MHA group. bIndicates p = 0.05 vs. no migraine. Qp/Qs: pulmonary to systemic blood flow ratio.
Characteristics of de novo MHA patients
Clinical characteristics of each patient who developed de novo migraine headache attacks (MHA) after transcatheter atrial septal defect (ASD) closure.

Duration of follow-up. F: female; M: male; Qp/Qs: pulmonary to systemic blood flow ratio.
In eight de novo MHA patients (35%), MHAs were transient and disappeared within the follow-up period. The time of resolution was 6.0 ± 3.4 months post-surgery. At final follow-up (45 ± 23 months post-surgery, range 12 to 80 months), the remaining 15 patients (65%) continued to suffer migraine. However, most patients reported decreased frequency of MHA with a median of three months (range 0.3 to 12 months). Most de novo MHA patients were treated with nonsteroidal anti-inflammatory drugs or acetaminophen, but two patients (patients 13 and 21) were effectively treated with eletriptan and two patients (patient 1 and 21) were effectively treated with lomerizine, a calcium channel blocker.
Characteristics of patients with history of MHA
Clinical characteristics of each patient who experienced exacerbation of existing migraine headache attacks (MHA) after transcatheter atrial septal defect (ASD) closure.
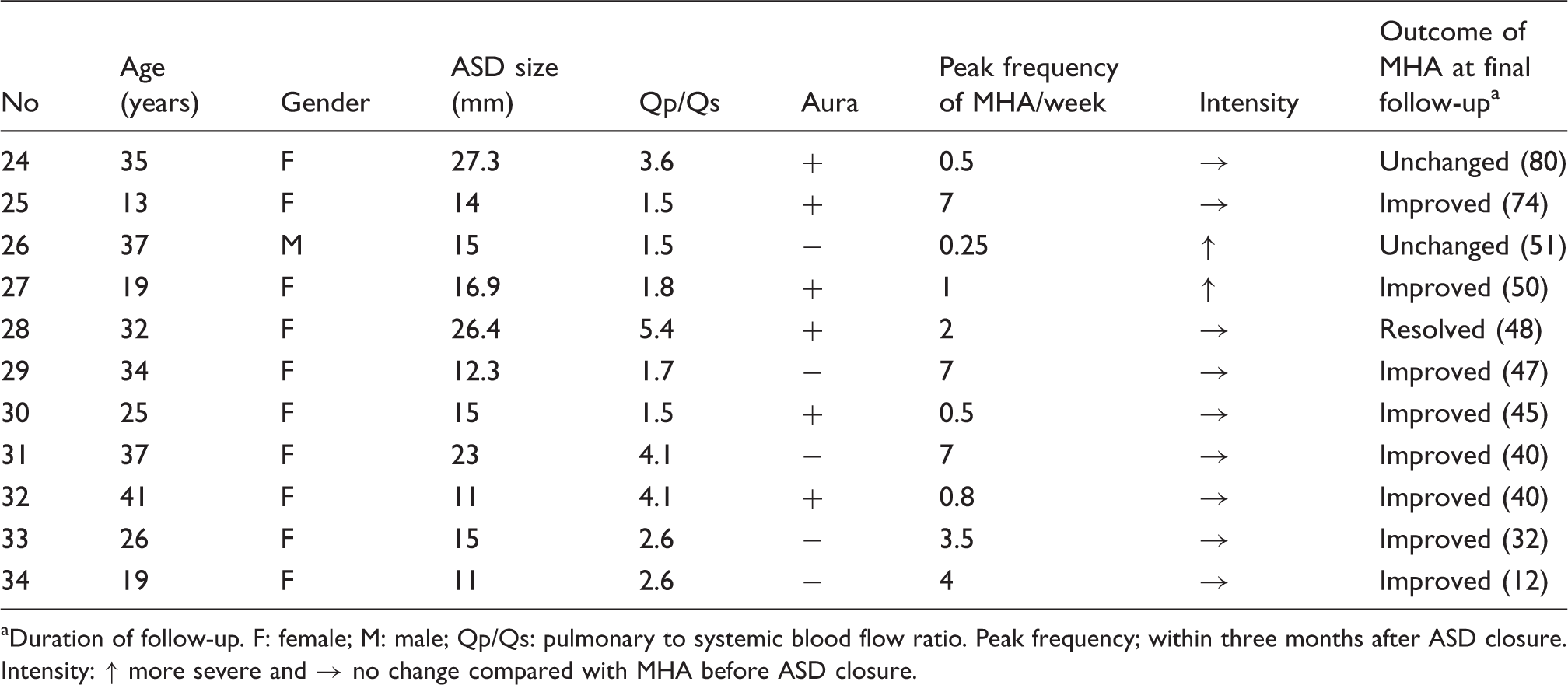
Duration of follow-up. F: female; M: male; Qp/Qs: pulmonary to systemic blood flow ratio. Peak frequency; within three months after ASD closure. Intensity: ↑ more severe and → no change compared with MHA before ASD closure.
Changes in number of patients with migraine after ASD closure (n = 207).

Values are number of patients. ASD: atrial septal defect; MO: migraine without aura; MA: migraine with aura.
Discussion
In this study, we examined MHA in patients who underwent transcatheter closure of ASD. To date, this is the largest cohort in which this relation has been examined. Our results show cases of de novo MHA and exacerbation of existing MHA after ASD closure. In the majority of these patients, MHA were with aura. Interestingly, there were also cases of MHA improvement after ASD closure. Patients with de novo MHA were significantly younger and patients with MHA improvement after the procedure tended to be older than patients with no history of MHA. These results support those of Mortelmans et al., who also reported that the patients with de novo MHA were younger than patients in whom MHA resolved after surgery (2). Together, these results suggest that age may be an important predictive factor of the influence of ASD closure on MHA.
The influence of percutaneous atrial septal defect (ASD) closure on migraine headache attacks (MHA).

n is for de novo MHA and MHA exacerbated combined. bData at two months after the closure. cLonger follow-up of ref. 2. dData at six months after the closure. eExacerbation of MHA was at three months after the closure. Median monthly MHA frequency was reduced after ASD closure (not significantly). A: Amplatzer ASD occluder device; S: Cardioseal/Starflex device; ASA: aspirin; CL: clopidogrel.
Mortelmans et al. found that bigger devices were used in patients with de novo MHA (2); however, we found no differences in device size among groups. Although there are reports suggesting that prognosis for MHA following occlusion of ASD is good (11,22–24), our results showed persistence of de novo MHA for at least six months after ASD closure in the majority of patients, supporting the results of Mortelmans et al. (2) and Rodés-Cabau et al. (12), suggesting that ASD device implantation can act as a permanent trigger for MHA in a high proportion of patients.
The mechanisms of de novo MHA or exacerbation of existing MHA after ASD closure remain unclear. Platelet activation on the surface of the implanted device and subsequent secretion of serotonin by activated platelets have been proposed as one potential mechanism (8–10,28). Our results show that switching medication from aspirin to ticlopidine or clopidogrel rapidly aborted status migrainosus and reduced MHA frequency. In addition, Wilmshurst et al. reported that adding clopidogrel to aspirin in the first month after surgery resulted in fewer MHAs than treatment with aspirin alone (8). These observations can aid our understanding of pathophysiology of MHA. Thienopyridines, including ticlopidine and clopidogrel, specifically inhibit adenosine 5'-diphosphate (ADP)-stimulated platelet function, which is distinct from that of aspirin. Results on a link between MHA and platelet function are conflicting, but there is some evidence that platelets from patients who experience MHA aggregate more readily than normal in response to exposure to several vasoactive amines, including serotonin and ADP (29). Serotonin is involved in the pathogenesis of MHA but its precise role in unclear. Serotonin in venous blood does not normally reach the systemic arterial circulation because it is almost totally destroyed by monoamine oxidase in its first passage through lung tissue. However, serotonin may reach systemic arteries if a large amount is released, if there is a right-to-left shunt that allows the lung tissue to be bypassed, or if platelets are activated on the left atrial disc of a device. Another possibility of mechanisms of MHA is the presence of cerebral embolism due to the formation of thrombus on the surface of the atrial septal device. This seems unlikely in the present study, as no patient developed ischemic stroke after the procedure. Meanwhile, Ferrari et al. reported silent cerebral microemboli in 33 of 35 patients during transcatheter patent foramen ovale and ASD closure (30). Recently, Nozari et al. demonstrated that microemboli triggered cortical spreading depression without causing microinfarction in a mouse model (31). Perhaps implantation of a transcatheter ASD closure device may be a human model showing evidence linking the triggering of MHA with aura to cerebral microembolism. Thienopyridines may reduce microembolization, and it has been shown that combination therapy with clopidogrel and aspirin is more effective than aspirin alone in reducing microembolic signals in patients with symptomatic intracranial and carotid stenoses (32).
A significant release of nickel within the six months after Amplatzer device implantation has been observed (33), and it has been suggested that nickel allergy might be responsible for MHA aggravation after ASD closure (10). However, device endothelialization usually occurs within the first three months after device implantation (34). This might explain why MHA among these patients occurred early after Amplatzer device implantation, and rarely more than three months later. It might also explain why some MHA remitted spontaneously in a few months, although others did not remit during long follow-up.
The implanted device, or deformation or stretching of the atrial septum caused by the device, might induce the release of some vasoactive substances that are associated with MHA, such as calcitonin gene-related peptide (16) or atrial natriuretic peptide (22,24).
This study was retrospective in design, and patients were asked to recall their headaches before and after ASD closure, therefore, results might be influence by a recall bias. To minimize this potentially confounding factor, we regarded the medical records of those days about headache as important. A response bias between the two institutions was considered to be the minimum. The Amplatzer septal occluder device was used in all patients included in the study. Therefore, the results cannot be attributed to the influence of device type. It has been suggested that incidence of de novo MHA might be lower with other atrial septal devices (28), and future study should determine whether MHA after transcatheter ASD closure is influenced by the type of ASD closure device. All patients in this study received antithrombotic therapy after ASD closure, which might influence MHA. If we had used clopidogrel from the beginning, the incidence of de novo migraine might have been reduced. However, the effect of aspirin on MHA seems to be modest and comparable with placebo responder rates (35,36). In addition, antithrombotic therapy was taken only for the six months after therapy, and we observed MHA that persisted for more than 12 months after ASD closure, therefore the occurrence of MHA cannot be attributed solely to antithrombotic therapy. An important limitation of this study was the absence of a placebo group.
Clinical implications
The purpose of our study was to evaluate the effect of atrial septal defect (ASD) closure on migraine headache attacks (MHA). The present study is the largest cohort that provides detailed and systematic descriptions of MHA related to ASD closure.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
