Abstract
Background
Oestrogen receptor 1 (ESR1) is located in region 6q25.1 and encodes a ligand-activated transcription factor composed of several domains important for hormone binding and transcription activation. Progesterone receptor (PGR) is located in 11q22-23 and mediates the role of progesterone interacting with different transcriptional co-regulators. ESR1 and PGR have previously been implicated in migraine susceptibility. Here, we report the results of an association study of these genes in a migraine pedigree from the genetic isolate of Norfolk Island, a population descended from a small number of Isle of Man “Bounty Mutineer” and Tahitian founders.
Methods
A significant number of molecular markers in the ESR1 (143) and PGR (43) genes were evaluated in a sample of 285 related individuals (135 males; 150 females). A pedigree-based analysis in the GenABEL package was used to analyse the results.
Results and conclusions
A total of 10 markers in the ESR1 gene showed association with migraine (p < 0.05) in the Norfolk Island population. No association was detected with PGR. Three haplotypes in ESR1 were found to be associated with migraine (p = 0.004, 0.03, 0.005). Future genetic studies in larger populations and expression analysis are required to clarify the role of ESR1 in migraine susceptibility.
Introduction
Migraine has been defined as a common disabling primary headache disorder by the International Headache Society (IHS) (1). The second edition of the International Classification of Headache Disorders (ICHD-II) drafted by the IHS (1) classifies migraine into two major groups: migraine without aura (MO) and migraine with aura (MA). MO is the most common form, with headache attacks lasting between four and 72 hours. MA sufferers, in addition, often experience aural symptoms preceding the headache phase of the migraine episode (2).
Migraine is more common in those aged between 35 and 45 years although generally attacks start in puberty. Among the most relevant epidemiological observations is the significant sexual dimorphism in the prevalence of migraine in men and women after puberty (three to four times higher in women) (3). European and American studies have shown that 6%–8% of men and 15%–18% of women experience migraine each year. The higher rates in women are thought to be hormonally driven, and numerous epidemiological, clinical and molecular studies support this theory (4–7).
The ICHD-II (1) gives diagnostic criteria for pure menstrual migraine and menstrually related migraine, headaches described as more disabling than those that occur in other stages of the month (8). In addition, changes in migraine frequency occur during pregnancy, lactation, menopause and contraceptive use, supporting a hormonal mechanism (9). Finally, genetic association studies in sex hormone receptor pathways and metabolism have provided some evidence supporting a hormonal role in migraine aetiology.
As part of a systemic review of genetic association studies in hormonal receptors and pathways in migraine, Schurks et al. (4) found that to date eight genes have been investigated, namely ESR1, ESR2, progesterone receptor (PGR), androgen receptor (AR), follicle-stimulating hormone receptor (FSHR), nuclear receptor interacting protein 1 (NRIP1), CYP19A1 and methylenetetrahydrofolate reductase (NAD(P)H) (MTHFR). However, results from these studies are contradictory and need to be replicated in different populations.
The oestrogen receptor 1 (ESR1) gene is located on chromosome 6q25.1 and its protein ER1 (or ERα) (10) mediates the action of 17β-oestradiol or E2, an important endogenous steroid hormone (11). In the human brain the ER1 protein is mainly expressed in frontal and prefrontal cortex, amygdale, mediobasal hypothalamus and locus coeruleus (12). It acts as an activator and regulator of important neurotransmitter systems such as the serotonergic, noradrenergic and gamma aminobutyric acid (GABA)ergic systems. In addition, the PGR gene is located on chromosome 11q22-23 and mediates the role of progesterone interacting with different transcriptional co-regulators. PGR protein is expressed in a variety of human tissues, including the uterus, mammary gland, pancreas, bone, ovary, testes and various regions of the human brain, including serotonin neurons (13).
In the present study we analyse ESR1 and PGR single nucleotide polymorphism (SNP) data from a Genome Wide Association Study (GWAS) to investigate hormone receptor molecular markers with migraine in the genetically isolated population from Norfolk Island, which has previously been shown to have a very high prevalence of migraine (14). The modern Norfolk Island population comes from a settlement of 194 inhabitants who resettled from the Pitcairn Islands in 1856. All of these inhabitants were the descendants of nine “Bounty” mutineers and 12 Tahitian women (15). This population is therefore characterised by founder effect, geographical and cultural isolation, high levels of consanguineous unions during early population expansion, population bottlenecks, admixture and a homogenous environment (16). Combined with recent evidence that shows that the heritability of migraine in the Norfolk Island population is 0.53 (14), these characteristics make Norfolk an excellent model to investigate hormonal-related genes in migraine.
Methods
Population samples
Phenotypic data and biological specimens (venous blood) were obtained from 600 subjects (261 males, 339 females). DNA was isolated using a standard salting-out procedure (17). Phenotypic data were obtained via a medical questionnaire that surveyed migraine family history, symptoms, triggers and medication use. An in-depth interview and comprehensive medical questionnaire were undertaken on all individuals and used to obtain phenotypic data including migraine information regarding family history, symptoms, triggers and medication. Migraine diagnosis was in accordance with ICHD-II guidelines. The inclusion criteria used to select cases were a diagnostic of MA or MO with other non-migraineurs included in the control population. Genealogical data were obtained from multiple sources, including questionnaire, municipal and historical records. Because all the individuals share a common genetic background and under the hypothesis of a common major variant, all individuals diagnosed with subtypes MA or MO were grouped together and phenotyped as being affected with migraine. The study protocol was approved by the Griffith University Human Research Ethics Committee. All subjects provided signed, informed consent prior to participation.
Genotyping
DNA samples from the Norfolk Island population were genotyped according to the manufacturer’s instructions on the Illumina Infinium High-Density (HD) Human610-Quad DNA analysis BeadChip version 1. A total of 620,901 genome-wide markers were genotyped in a sub-sample of 285 related individuals (135 males; 150 females). All samples were genotyped individually but 28 selected samples (5% of cases and controls) were repeated to ensure concordance and accuracy of genotyping. Of these related individuals, 76 are migraine cases (22 males, 54 females). These individuals possessed high inheritance information and were extremely informative for pedigree-based analysis, facilitating cost-effective genotyping. Markers had a median spacing of 2.7 kb (mean = 4.7 kb) throughout the genome. Each Human610-Quad DNA analysis BeadChip employed a four-sample format requiring 200 ng of DNA per sample. Samples were scanned on the Illumina BeadArray 500GX Reader. Raw data were obtained using Illumina BeadScan image data acquisition software (version 2.3.0.13). For the present study, 143 SNPs from the ESR1 gene and 43 SNPs in the PGR gene were selected. These molecular markers are distributed along each gene and in intergenic regions.
Statistical analysis
Data analysis was performed with the GenABEL package (18), a statistical tool implemented as a library in R. R is a free, open-source language and environment for statistical analysis. First, a quality control of the data was performed. GenABEL quality control check the data for SNPs and samples with extremely low call rate (<95%), SNPs with minor allele frequency (MAF) (<0.87%); SNPs out of Hardy Weinberg Equilibrium (HWE), samples with extreme autosomal heterozygosity (first-degree relative (FDR) at FDR 0.01) and those who have genome-wide average identity-by-state (GW IBS) ≥0.95; the software excludes all markers and individuals that fail to pass these tests. The quality control is repeated iteratively until no further errors are found. A polygenic analysis was carried out to account for the pedigree relatedness followed by a family-based association test. The marker-marker (MM) score employed is a score test for association between a trait and genetic polymorphism in samples of related individuals. The p values here presented are unadjusted. To overcome the high penalty incurred by the Bonferroni correction method in pedigree-based populations, the gene-wide significance threshold for ESR1 and PGR genes was estimated empirically using the SimpleM program (19). Based on the average linkage disequilibrium (LD) pattern across both genes, the minimum number of effective tests (Meff) transformed into a gene-wide significance threshold of 1*10−3. Haplotype analysis was carried out in Plink-1.07-dos. Haploview 4.2 was used as a graphical tool to depict results from PLINK.
Results
Genotyping
A total of 186 markers in 285 individuals were analysed using the GenABEL package in the R platform. Among these markers, four were excluded because of a low MAF (<0.086) (rs11967900, rs17081771, rs9397459, rs7739506); one marker was excluded because it was out of HWE (rs9479122); and two markers were excluded because of a low call rate (rs4870053, rs504372). A total of 17 people were eliminated from the analysis because of low call rate (<95%) or high IBS (≥0.95). In total 138 markers and 286 individuals passed all quality control criteria and were included in the analysis.
Top 10 markers analysed in the ESR1 and PGR genes. Because there are some markers located in an intergenic region between the ESR1 and SYNE1 genes, the marker function in this table is given with respect to the position of one gene or another according to the USCS Genome Browser (26).

ESR1: oestrogen receptor gene; SYNE1: spectrin repeat containing, nuclear envelope 1; SNPs: single nucleotide polymorphisms; PGR: progesterone receptor gene; MAF: minor allele frequency.
An association was detected in regions upstream and downstream from the ESR1 gene. Interestingly SNPs rs2813554, located closely to spectrin repeat containing, nuclear envelope 1 (SYNE1) (a neighbour gene), and rs9322361 (p = 0.05) present in an intronic region of SYNE1, also showed marginal association. We also looked at a gender-specific analysis but none of the SNPs included in this study showed a positive gender association with migraine (p > 0.07). This may be because of lower power from the reduced gender-specific number of samples.
Haplotype analysis
MM LD measures D′ and r2 were computed for both the ESR1 and PGR genes. The pairwise D′ results produced by Haploview are presented graphically in Figures 1 and 2. Several regions of high LD were identified within ESR1 (Figure 1). A total of 23 haplotype blocks were identified and a haplotype association analysis was performed. Association was detected in four haplotype blocks in the ESR1 gene. The first block includes nine markers and it spans 25 kb through the 5′ end of the gene. The first SNP in this block, rs10484919, is 2.8 kb upstream from the transcription start site; however, it does not belong to the promoter region (10 kb upstream from the transcription start site). The last marker in block 1 is rs7745737, located at the 5′ end of intron 1. The sixth and seventh block are 2.7 kb and 0.362 kb long, respectively, and are separated by 2.5 kb. The last associated block is number 21 and it spans 24.88 kb through the 3′ end of the gene. This block starts 1774 bp downstream from ESR1 and finishes in the last exon of the SYNE1 gene.
ESR1 structure and associated haplotypes in gene region. Horizontal bold blue lines indicate ESR1 and SYNE1 genes. Vertical bold blue lines indicate the position of the exons in ESR1. Red arrows indicate transcription direction for every of the genes in the graphic. The red box in the 5′ end of ESR1 indicates the position of a promoter associate sequence present in a migraine associated region in a strong LD. The diagram in the lower part is an intermarker linkage disequilibrium plot for the markers with the top fifteen p-values in the polygenic analysis. PGR Intermarker linkage disequilibrium (LD) plot. In the upper part of the graphic the PGR gene structure is shown. Vertical bold blue lines indicate the position of the exons in PGR. The diagram in the lower part is an intermarker linkage disequilibrium plot for the markers with the top fifteen p-values in the polygenic analysis. Areas delimited with a solid black line show haplotypes detected after the analysis performed with Haploview.

Association test for haplotype blocks in ESR1.

ESR1: oestrogen-receptor gene; SNPs: single nucleotide polymorphisms.
D′ values of the ESR1 SNPs in associated haplotype blocks.

ESR1: oestrogen receptor gene; SNPs: single nucleotide polymorphisms.
ESR1 Haplotypes with estimated frequencies.

ESR1: oestrogen receptor gene.
PGR frequency haplotype blocks.
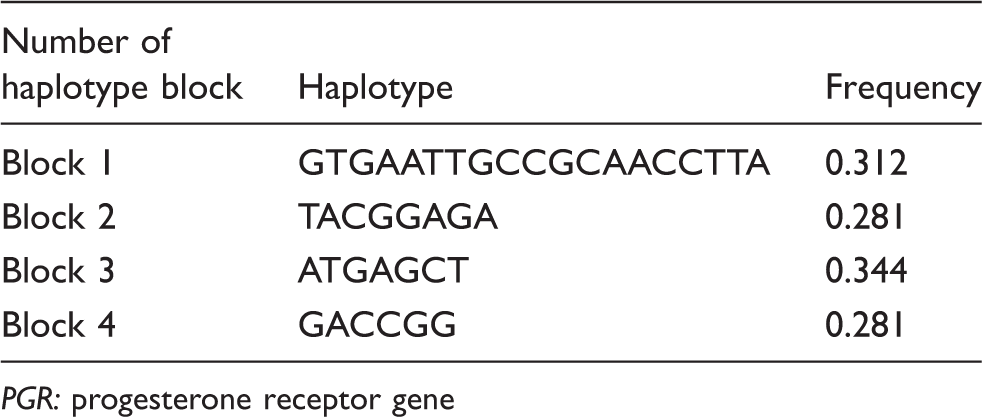
PGR: progesterone receptor gene
Discussion
Previous case-control association studies have demonstrated an association between migraine and the polymorphism G594A in the ESR1 gene (rs2228480) in two different Australian cohorts (p = 0.003 and 8 × 10−6) (20). In contrast, other association studies in Spanish and Finnish populations did not report any association with migraine and rs2228480. However, this polymorphism has been associated with breast cancer (p = 0.05) (16), supporting a role for the gene in diseases with complex hormonal influences on the pathogenic process. In addition, six studies have also reported association results for the polymorphism C325G in the ESR1 gene (rs1801132). Two of these studies suggested an increased risk for migraine under a recessive model which appeared to be strongest among women (p = 0.02 and p = 0.01) (21,22). Joshi et al. (23) also considered the ESR1 Pvu II C > T polymorphism (rs2234693) and detected a significant association of the TT genotype (OR 3.45, confidence limits (CL) = 1.75, 6.81; p = 0.0003) and the T allele (OR 1.729, CL = 1.309, 2.284; p = 0.0001) with migraine. In addition, Colson et al. (20) did not find significant results after analysing this polymorphism (p = 0.31). Finally, SNPs rs6557170, rs2347867, rs6557171, rs4870062 and rs2077647 have been studied on ESR1 without any positive outcome in a Finnish population.
The aim of our study was to investigate ESR1 and PGR genetic variations and migraine in a large Norfolk Island pedigree consisting of 285 related individuals. Our results show strong evidence of ESR1 association with migraine in this population. In this gene, 10 out of 143 SNPs were found to be significantly associated with migraine. Among these markers, seven are located upstream from ESR1 (Table 1), suggesting a regulatory role on the expression of ESR1 transcript. Interestingly, three SNPs (rs2813554, rs2813544 and rs7767143) are downstream, in an intergenic region between ESR1 and SYNE1. SNP rs2228430, previously reported to be associated in an Australian population, showed no association in our study. Other previously associated variants such as rs1801132 and rs2234693 were not included in our analysis.
The haplotype analysis performed in the ESR1 gene supports the use of the Norfolk Island population as a genetically isolated population with a well-conserved homogeneous genetic structure. A previous study reported an increased risk (OR 1.81) in marker rs6557170 in a case-control population from Finland (24). The authors reported this marker to be in a haplotype with an initially significant p value (0.01), but it did not remain significant after taking multiple testing into account. This haplotype includes two markers studied in the present investigation, rs6557170 and rs6557171. Our results show no individual or haplotype association but they demonstrate the presence of several risk haplotypes that should be considered for future studies in terms of SNP selection.
In the haplotype analysis performed in the ESR1 gene considering the 15 most significant associated markers, one block of six SNPs was identified 5.4 kb upstream from the ESR1 transcription start site. This region includes a promoter-associated site with DNAse I-enriched sites. This promoter fragment is 176 bps long, and marker rs9383939 (p = 0.04) is located at position 68 (Figure 1). The presence of a promoter-associated region within the associated haplotype block suggests a possible causal mechanism for the association found in this area upstream from the gene. SNPs mapping to this region have recently been associated with other phenotypes in which oestradiol metabolism is implicated. A study comparing 6q25 breast cancer hits from Asian and European GWAS in the Breast Cancer Consortium (BCAC) found two SNPs, located approximately 170 kb upstream of ESR1, to be associated with breast cancer risk (17). Other SNPs mapping to this region have also been implicated in a GWAS for bone mineral density (25). In addition, a recent paper showed that a number of genes, including ESR1 and C6orf97 (ESR1 neighbour), are co-regulated at this locus although the function of most of these genes is unidentified (26).
A second block, incorporating two markers, is located in intron 2 of the same gene. This implies possible LD of these markers with real causal variants present in splicing sites or coding regions in the gene. The third associated block starts 1174 bp downstream from ESR1; it extends 33.5 kb and finishes with marker rs9322361, located in intron 141 in the SYNE1 gene (see Figure 1). This gene encodes a spectrin repeat-containing protein expressed in skeletal and smooth muscle, and peripheral blood lymphocytes that localises to the nuclear membrane. Mutations in this gene have been associated with autosomal recessive spinocerebellar ataxia 8, also referred to as autosomal recessive cerebellar ataxia type 1 or recessive ataxia of Beauce (27). Alternatively spliced transcript variants encoding different isoforms have been described (28). Interestingly, SYNE1 has been recently reported to be associated with bipolar disorder, recurrent major depression (29) and ovarian cancer (30). Due to the high prevalence of depression in migraine patients reported in the literature (31), common genetic variants in SYNE1 associated with both diseases may suggest common molecular and physiological mechanisms contributing to the comorbidity. Therefore, results presented in this study implicate SYNE1, for the first time, as a new target in migraine. SYNE1 is expressed in different regions in the brain involved with pain processing as well as vascular tissues, possibly affecting vasodilatation or vasoconstriction processes.
The most well-studied polymorphism in the PGR gene is the PROGINS polymorphic 306 bp Alu insertion that occurs within intron 7 (32). Colson et al. showed that individuals who carried the PROGINS insert in an Australian case-control population were 1.8 times more likely to suffer migraine (5). Significant differences in genotypic (p = 0.0001) and allelic frequency (p = 0.0002) were also seen when comparing cases and controls in an Indian population (23).
The present PGR analysis showed no association at the evaluated markers. However, four haplotype blocks were identified showing a very homogenous population and identifying a region with a very low rate of recombination. Overall, these results suggest that PGR is not implicated in the pathogenesis of migraine in the Norfolk Island population.
Conclusions
The results of this study show that there is some evidence of the ESR1/SYNE1 genes’ association with migraine. Association analysis in the ESR1 gene provided strong evidence to support the hormone-mediated pathogenesis model of migraine. However, the presence of a haplotype associated with the disease that includes the SYNE1 gene, in combination with the recent evidence showing association of SYNE1 with other diseases related to migraine such as depression and bipolar disorder, suggest a new target that should be considered in migraine. Future studies are now required in larger populations, potentially including expression analysis to clarify whether the real causal variant contributing to migraine susceptibility is in ESR1/SYNE1 or both.
Clinical implications
Variants in the ESR1/SYNE1 genes may be associated with migraine in the genetically isolated Norfolk Island population. Haplotype analysis in the ESR1 gene in the Norfolk Island population replicates previous associations identified in this gene and demonstrates the presence of several risk haplotypes that should be considered for future studies in terms of SNP selection. Genetic variants in the PGR gene are not associated with migraine in the genetically isolated Norfolk Island population.
Footnotes
Funding
This research was supported by funding from the National Health and Medical Research Council (NHMRC) of Australia. Astrid Rodriguez-Acevedo was supported by a Griffith University International Postgraduate Research Scholarship (GUIPRS). Rod Lea and Miles Benton are supported by Corbett research. Lastly, we extend our appreciation to the people of Norfolk Island who volunteered and participated in this study.
Conflict of interest
None declared.
