Abstract
Background
Migraine is a chronic neurovascular disease characterized by recurrent unilateral headache, which induces incapacity. Despite all the progress that migraine research has provided, the neural mechanisms underlying the onset and maintenance of migraine attacks are poorly understood. Due to the complex characteristics of the disorder, it is difficult to develop a proper animal model that mimics all the clinical manifestations in humans.
Objective
Taking into account the principal characteristics of the disease, the aim of this study is to develop a chronic animal model of migraine in which we can reproduce behavioral and pharmacological phenomena similar to those displayed by migraineurs.
Results
Our animal model displayed behavioral and pharmacological results similar to those experienced by migraineurs. Specifically, there was a decrease in routine physical activity and an increase in resting behavior. Also, the animals exhibited a novel behavior that we called ipsilateral facial grooming behavior provoked by the meningeal nociception. Moreover, one of the drugs used as treatment for migraine reduced the manifestations previously described.
Our results determine that the model mimics many of the clinical features that patients exhibit during migraine attacks. This model can contribute to further understanding of the pathophysiology and the study of novel therapeutic approaches.
Introduction
Migraine is a paroxysmal neurological disorder affecting up to 12% of males and 24% of females, with the highest prevalence occurring between 25 and 55 years of age (1–4). The disease is principally characterized by recurrent attacks of unilateral headache that typically last 4 to 72 hours, associated with other neurological symptoms in addition to pain, such as nausea, photophobia, phonophobia, osmophobia and fatigue as well as numerous disturbances in autonomic, mental, sensory and motor functions (5–8).
The pain produced during the migraine attack is the most disabling of all the symptoms. Among the treatments available for migraine, there is a family of 5HT 1B/1D receptor agonists collectively known as triptans (3,9–13). Other types of drugs can be used, such as nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen and beta-blockers, among others (3,6,7). Even though the treatment based on triptans is a specific pharmacotherapy for migraine, the effectiveness of this treatment is limited, relieving the pain of only 40% of all patients (11–14).
Migraine pain has been proposed to be driven by the activation and sensitization of peripheral trigeminal neurons that innervate the meninges (15). This activation of the peripheral trigeminal neurons is postulated to eventually produce the activation and subsequent sensitization of the central trigeminovascular neurons (15). It has been described that the administration of the triptans must occur at least one hour before the establishment of central sensitization, with the goal of improving triptan efficacy (16).
Despite all the progress that research has accomplished, the neural mechanisms underlying the onset and maintenance of migraine attacks are poorly understood. Due to the complex characteristics of the disorder, it is difficult to develop a proper animal model that mimics all the clinical manifestations in humans.
Inflammatory soup (IS) has been successfully used to study the mechanisms of meningeal nociception. Nowadays, the use of electrophysiology (10), behavioral analysis (18,19) and molecular biology (20) have proven to be useful techniques to study the subsequent changes in the trigeminal sensory processing as a result of the IS infusion. In this respect, different approaches have been used to determine the effects of IS on food intake (21), facial allodynia and free movement (18), facial allodynia and response to commonly used antimigraine drugs (20), cephalic and extracephalic allodynia along with the biochemical mechanisms influencing such behavior (22), and the sex differences in behavior as well as the expression of calcitonin gene-related peptide (CGRP)-related genes in an animal model of chronic migraine (19).
Nevertheless, none of these studies describe if IS infusion, acutely or chronically administered, could generate a spontaneous nociceptive behavior and if these changes could be prevented by the use of antimigraine therapy. Therefore, the aim of this project is to develop an animal model of migraine in which we can study the effect of repeated meningeal nociception on the spontaneous behavior and the possible impact of triptans.
Methods
Ethical concerns
The experiments were conducted in male Wistar rats (250–300 g); the animals were treated and cared for according to the ethical standards prescribed by the Committee for Research of the International Association for the Study of Pain (1983) and with the project commissioning approval of the Instituto Nacional de Psiquiatría Ramón de la Fuente Muñiz.
Since the animals would be in a pain state, the number of rats studied was restricted to the minimum necessary in order to uphold the power of the statistical analysis (n = six per group for effect size = 1.5 SD; α = 0.05; power = 0.80).
Habituation period
All the animals were raised, housed and maintained in a controlled environment with a room temperature of 23 ± 2℃ and humidity of 50 ± 10% in the facilities of the institute. All the rats were kept in transparent acrylic individual cages with light-dark inverted cycles of 12 × 12 hours, and with ad libitum feeding and hydration.
Habituation consisted of a seven-day period before surgery, as follows: each rat was placed daily in the observation cage (transparent glass cage with mirrors that allow the researcher a full view of the animal behavior without any blind spots) for a period of 15 minutes. Subsequently, the animal was manipulated by the researcher and placed again in the observation box for 45 minutes and when this time period lapsed the rat was returned to its individual cage. Manipulation consisted of holding the rat for one minute and touching its head several times.
Surgical procedures
Craniotomy and cannula fixation
After the habituation period, and under general anesthesia (pentobarbital 40 mg/kg), the rat was placed in a stereotactic frame (KOPF instruments, Tujunga, CA, USA), and clorexidine (10% solution) was used as an antiseptic agent before the procedure was commenced. In order to prevent pain as a result of the surgical incision, 1 ml of bupivacaine (5 mg/1 ml) was applied under the skin as a local preventive anesthesia. We performed an incision to expose the skull completely, and after this, using a burr drill a 1 mm in diameter craniotomy was performed in the right frontal bone (+1.5 mm from Bregma and +1.5 mm lateral), taking care not to burn or damage the dura. Sterile petrolatum was placed over the dura to prevent fixation to the calvarium.
Afterward, using stereotactic coordinates we proceeded to place the cannula (18 GA cannula guide and cannula dummy, Plastics One, Roanoke, VA, USA) just above the skull without touching the meningeal tissue, then bone wax was applied surrounding the cannula to prevent leakage of any material into the craniotomy. We applied a glass ionomer to protect and fix the cannula to the skull. Acrylic was used to cover the glass ionomer and to fix the ensemble completely to the skull. All surgery was performed under direct visual control using an operating microscope (WPI, Sarasota, FL, USA).
The cap of the cannula had a tip that goes .5 mm farther than the total length of the cannula, and at the time the cap was placed the skin was sutured with Nylon 4-0. We applied topical neomycin (0.350 g/100 g) to prevent any infection of the surgical area. Breathing and movement were observed until the rat was fully awake and mobile, when it was returned to a clean individual cage. After a period of two days (recovery), the rats were used to proceed with the experiments.
Behavioral recording
Experiments were conducted during a period of between 7:00 and 14:00. Each rat was individually transported in its own box to the observation area, which was in the same room. Light was provided by two 60-W incandescent red bulbs suspended 1 m above the center of the observation area. A video camera was located 60 cm in front of the observation cage and positioned so that the camera could cover all angles of the cage and to obtain a complete image of the free rat behavior.
After preparing the observation area, the rat was placed inside the observation cage for video recording. After a period of 15 minutes, we delivered 2 μl of IS (histamine, serotonin, braykinin 1 mM and prostaglandin E2 (PGE2) 0.1 mM) or saline through the cannula, and recorded for another 45 minutes after the infusion. All the videos were recorded with a high-definition camera and in night vision mode in order to observe the rats in the dark (Handycam Sony, HDR-CX12, Japan). After this manipulation the rat was placed again in its own cage.
The same procedure was repeated for seven days and videotaped observations were analyzed as described in the Behavioral analysis section below. At the end of the behavioral recording the animals were euthanized with an overdose of pentobarbital and then underwent histological examination of the meninges (see below) to observe if there was any damage to the tissue.
Behavioral analysis
Specific behaviors have been described previously as those that should be observed for the analysis of trigeminal nociception (23). These behaviors are:
Exploration behavior: walking, running, climbing, rearing and sniffing. Freezing behavior: immobile posture, with the four paws in contact with the floor and no movement of the vibrissae. Resting behavior: head resting on flexed forepaws with eyes opened or closed. Face-grooming behavior: movement patterns in which paws contact facial areas. Body-grooming behavior: paws, tongue or incisors are brought in contact with a body area other than the face or the forepaws. Additional behavior observations: Due to the fact that it is not possible to predict how the behaviors will be modified, we also took into account possible aberrations or changes in another behavior besides those described above. In order to do this, all the analyzers were instructed to describe if other behavior was initiated by the animals.
The total time spent in each behavior was measured manually with a chronometer in periods of three minutes, and it was further measured in seconds. All the videos were analyzed by the researchers in a blind manner.
Nociceptive behavior analysis
At the end of the behavioral analysis, we analyzed the nociceptive behavior induced by the IS infusion and the effect of the pharmacological therapy.
Groups
All the animals were divided into equally sized groups.
Behavioral groups
Behavioral Control Group (Con-B) (n = six)
The rat was placed in the observation cage for video recording, and after 15 minutes we delivered 2 µl of saline through the cannula and recorded events for another 45 minutes.
Behavioral IS Group (IS-B) (n = six)
Same procedure as control, but we delivered 2 µl of IS (histamine, serotonin, braykinin 1 mM and PGE2 0.1 mM) through the cannula.
Behavioral Sham Group (Shm-B) (n = six)
A sham surgery was performed. The cannula was fixed with a glass ionomer and acrylic. The rat was placed in the observation cage and video-recorded for 15 minutes, then the animal was manipulated (see above) and recorded for another 45 minutes.
Behavioral groups with pharmacological intervention
In separate groups of rats, the IS infusion was preceded by a subcutaneous injection of either zolmitriptan (Zol-B group: 100 µg/kg), acetaminophen (Act-B group: 250 mg/kg) or ketorolac (Ket-B group: 5 mg/kg) (n = six for each group). The time of the injection was one hour prior to the IS infusion for zolmitriptan and ketorolac, and 45 minutes prior to the IS infusion for acetaminophen.
Statistical analysis
To assess potential baseline differences between the groups, a repeated-measures analysis of variance (ANOVA) was performed pre- and post-meningeal infusion. A Tukey test was used for post hoc multiple comparisons between groups, considering p < 0.05 as statistically significant. All the analyses were performed with the Statistical Package for the Social Sciences Software (SPSS) (LEAD technologies v 12.0).
Histological analysis
At the end of the experimentation period, the rats were euthanized with an overdose of pentobarbital and perfused with formol 10%. The meningeal tissue was obtained without damage and submerged in 10% ethanol to dry it. The tissue was then observed under a light microscope to analyze its continuity, in order to determine if the dura was damaged during the surgical procedure.
Results
Behavioral results
Results will be depicted in two different ways:
Mean of behavior during seven days: the mean ± SD of the time that each group displayed a specific behavior during the seven days of experimentation. Recordings were divided into blocks of three minutes, thus the pre-infusion period is shown as a five-block period and the post-infusion period is shown as a 15-block period. Day-by-day analysis: the average ± SD of the time that each group displayed a specific behavior day by day.
During the pre-infusion period there were no significant differences among groups in any of the analyzed behaviors (repeated-measures ANOVA p > 0.05). It is also relevant to mention that the Shm-B group did not show any significant differences as compared to the Con-B group in any of the analyzed behaviors (repeated-measures ANOVA p = 8.75; data not shown).
Resting behavior
Mean of resting behavior during seven days
The mean of resting behavior during the seven days of experimentation was significantly increased (72.23 ± 2.81 seconds) in the IS-B group (91.68 ± 2.50 seconds) during the post-infusion period as compared to the Con-B group (19.44 ± 2.35 seconds) (repeated-measures ANOVA p < 0.05; post noc Tukey p < 0.05) (Figure 1(a)),
The figure depicts the mean of resting, freezing and exploration behavior during the seven days of experimentation, divided into pre-infusion period (15 minutes) and post-infusion period (45 minutes). Each period was analyzed in blocks of three minutes and measured in seconds. As a result of inflammatory soup (IS) infusion (IS-B group), there was a significant increase of resting (a) and freezing behavior (b), and a significant decrease of exploration behavior (c) as compared to Con-B group (*). The administration of zolmitriptan reduces the IS-evoked response (^) without reaching control group values (+).
Day-by-day analysis
After analyzing the behavior day by day in the post-infusion period, it became evident, as shown in Figure 2(a), that the IS-B group (641.81 ± 3.02 seconds) significantly increased (505.67 ± 3.10 seconds) the resting behavior among the seven-day period as compared to the Con-B group (136.13 ± 2.10 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05).
The figure depicts the average in seconds of resting, freezing and exploration behavior analyzed day by day during 45 minutes after meningeal nociception. The inflammatory soup (IS) infusion (IS-B group) produces a significant increase of resting (a) and freezing (b) behavior and a significant reduction of exploration behavior (c) as compared to Con-B group (*). The treatment based on zolmitriptan significantly reduces the behavior evoked by IS (^), but never reaches control group values (+).
Freezing behavior
Mean of freezing behavior during seven days
The mean of freezing behavior during the seven days of experimentation was significantly higher (1.11 ± 0.03 seconds) in the IS-B group (1.24 ± 0.02 seconds) as compared to the Con-B group (0.13 ± 0.01 seconds) on the post-infusion period (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 1(b)).
Day-by-day analysis
After the meningeal infusion, the IS-B group (8.70 ± 0.02 seconds) displayed a significant increase (7.77 ± 0.03 seconds) of the freezing behavior among the seven days as compared to the Con-B group (0.92 ± 0.03 seconds) (repeated-measures ANOVA p < 0.05; post Hoc Tukey p < 0.05) (Figure 2(b)).
Exploration behavior
Mean of exploration behavior during seven days
Concerning the mean of exploration behavior in the post-infusion period, the IS-B group (50.81 ± 2.18 seconds) displayed a significant decrease (72.56 ± 2.79 seconds) as compared to the Con-B group (123.37 ± 2.20 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 1(c)).
Day-by-day analysis
The day-by-day analysis during the post-infusion period, as seen in Figure 2(c), showed a significant decrease (507.96 ± 3.81 seconds) in exploration time in the IS-B group (355.67 ± 3. 21 seconds) as compared to the Con-B group (863.63 ± 3.52 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05).
Facial and body grooming behavior
In both behaviors, there were no significant differences, neither in the mean time of the behavior during the seven days of experimentation among groups (ANOVA p > 0.05; data not shown) nor in the day-by-day analysis (repeated-measures ANOVA p > 0.05; data not shown).
Additional behavior observations: ipsilateral hindpaw facial grooming
Besides the behaviors previously described in the methods, we also found that the animals that received the IS infusion displayed an unusual pattern in facial grooming that we called ipsilateral hindpaw facial grooming.
According to the facial grooming behavior, instead of performing it with the two forepaws at the same time as described by Vos (1994), the rat performed a more intense facial grooming behavior with only one hindpaw and always ipsilateral to the place of the cannula implantation. The time the animal spent performing this behavior was measured and compared among groups.
The IS-B group (74.80 ± 4.50 seconds) showed a significant increase (58.77 ± 5.68 seconds) in time spent performing this behavior as compared to the Con-B group (16.03 ± 2.06 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 3).
The figure shows the mean of time each group performed “hindpaw ipsilateral facial grooming” after meningeal infusion. This novel behavior is characterized by intense facial grooming behavior with only one hindpaw and always ipsilateral to the place of the cannula implantation. As shown in the figure the IS-B group performed this behavior for a longer time period as compared to the Con-B group (*). The treatment based on zolmitriptan and ketorolac reduced this effect significantly (*). Acetaminophen was the least effective of all the tested drugs (*).
Nociceptive behavior analysis
According to the behavioral analysis previously described, we identified three behaviors mainly modified by the IS infusion: freezing, resting and the ipsilateral hindpaw facial grooming. Considering the fact that the ipsilateral hindpaw facial grooming is a novel behavior, we analyzed it separately.
We defined the nociceptive response as the total time spent in resting plus freezing behavior above the control level. The data show the percentage inhibition of the nociceptive response induced by the IS.
Pharmacological intervention results
Zolmitriptan: behavioral results
Resting behavior
Mean of resting behavior during seven days:
When we analyzed the mean of resting behavior during the seven days of experimentation, the Zol-B group (52.50 ± 4.18 seconds) showed a significant reduction (39.18 ± 4.68 seconds) in resting behavior as compared to the IS-B group (91.68 ± 2.50 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) and also a significant increase (33.05 ± 4.33 seconds) as compared to the Con-B group (19.44 ± 2.35 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 1(a)).
Day-by-day analysis:
Upon analyzing the behavior day by day, the Zol-B group (367.51 ± 2.50 seconds) showed a significant reduction (274.29 ± 2.92 seconds) of resting behavior as compared to the IS-B group (641.81 ± 3.02 seconds), but also a significant increase (231.38 ± 2.29 seconds) as compared to the Con-B group (136.13 ± 2.10 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05). The Zol-B group also showed a tendency to diminish resting behavior over the seven days of observation (Figure 2(a)).
Freezing behavior
Mean of freezing behavior during seven days:
The Zol-B group (0.45 ± 0.02 seconds) showed a significant increase (0.32 ± 0.01 seconds) as compared to the Con-B group (0.13 ± 0.01 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05). In addition, the Zol-B group (0.45 ± 0.02 seconds) displayed a significant reduction (0.78 ± 0.02 seconds) as compared to the IS-B group (1.24 ± 0.02 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05), when we analyzed the behavior during the seven days of experimentation (Figure 1(b)).
Day-by-day analysis:
When we performed the day-by-day analysis, the Zol-B group (3.17 ± 0.06 seconds) showed a significantly higher amount of freezing behavior (2.24 ± 0.03 seconds) as compared to the Con-B group (0.92 ± 0.03 seconds) solely during the first three days (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05), and after this period there were no differences between these two groups (repeated-measures ANOVA p > 0.05) (Figure 2(b)).
Exploration behavior
Mean of exploration behavior during seven days:
When we analyzed the behavior during the seven days of experimentation, the Zol-B group (87.14 ± 3.11 seconds) showed a significant increase (36.33 ± 3.10 seconds) in the mean of exploration behavior as compared to the IS-B group (50.81 ± 2.18 seconds) but also a significant reduction (36.23 ± 3.87 seconds) as compared to the Con-B group (123.37 ± 2.20 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 1(c)).
Day-by-day analysis:
Upon analyzing the behavior day by day, the Zol-B group (610.00 ± 4.13 seconds) displayed a significant reduction (253.62 ± 4.53 seconds) of the exploration behavior as compared to the Con-B group (863.63 ± 3.52 seconds), but also a significant increase (254.33 ± 4.91 seconds) as compared to the IS-B group (355.67 ± 3. 21 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 2(c)).
Facial and body grooming behavior
There were no significant differences regarding both behaviors, as compared with the other groups, neither in the mean of seven days of experimentation nor in the day-by-day analysis (data not shown).
Ipsilateral hindpaw facial grooming
The Zol-B group (43.76 ± 3.51 seconds) displayed a significant decrease (31.04 ± 6.57 seconds) in the time period taken for performing this behavior as compared to the IS-B group (74.80 ± 4.50 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05), also the Zol-B group is significantly higher (27.73 ± 3.45 seconds;) than the Con-B group (16.03 ± 2.06 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 3).
Nociceptive behavior
The rats treated with zolmitriptan displayed a significant decrease (62.65%) in the total nociceptive behavior in a significant manner (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 4).
The figure shows the percentage of reduction of the nociceptive behavior (freezing plus resting behavior) after the meningal infusion of every tested drug. Zolmitriptan, ketorolac and acetaminophen diminished the total nociceptive behavior in a significant manner (*).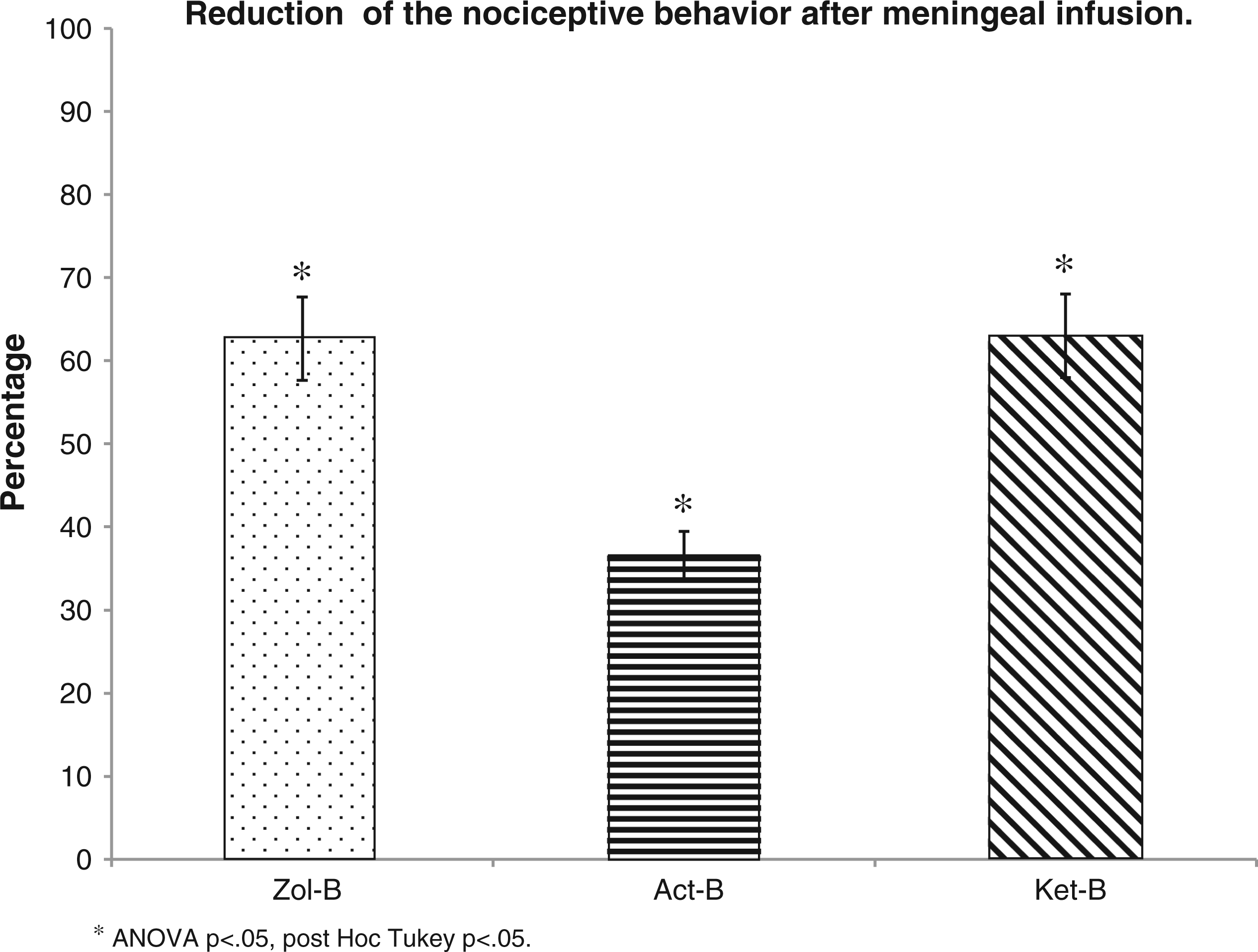
Ketorolac: behavioral results
There were no significant differences when comparing the rats treated with ketorolac in any of the analyzed behaviors (repeated-measures ANOVA p > 0.05; data not shown).
Ipsilateral hindpaw facial grooming
The Ket-B group (50.4 ± 4.38 seconds) showed a significant decrease (24.44 ± 3.57 seconds) in the mean of time as compared to the IS-B group (74.80 ± 4.50 seconds) (repeated-measures ANOVA p > 0.05; post hoc Tukey p < 0.05), and also was significantly higher (34.37 ± 4.54 seconds) than the Con-B group (16.03 ± 2.06 seconds) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 3).
Nociceptive behavior
The rats treated with ketorolac showed a significant reduction of the total nociceptive behavior (63.01%) (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 4).
Acetaminophen: behavioral results
There were no significant differences when analyzing the rats treated with acetaminophen in any of the analyzed behaviors (repeated-measures ANOVA p > 0.05; data not shown).
Ipsilateral hindpaw facial grooming
The Act-B group (71.56 ± 5.48 seconds) did not show any changes as compared to IS-B group (repeated-measures ANOVA p > 0.05) (Figure 3).
Nociceptive behavior
When analyzing the rats treated with acetaminophen, there was a decrease in the total nociceptive behavior (36.53%) as compared to the Con-B group (repeated-measures ANOVA p < 0.05; post hoc Tukey p < 0.05) (Figure 4).
Histological results
Under light microscopy the meningeal tissue showed damage as a result of the surgical procedure in four out of the 58 animals. Those four animals were not considered for further analysis. The rest of the animals did not present any evidence of damage in the meningeal tissue.
Discussion
There are some characteristics that an experimental model should meet in order to be useful as a research tool. First, the model must conform to the standard ethics for animal research. Second, the model must be as similar as possible to the phenomenon of the study. Finally, the model must be reproducible and statistically stable.
The present animal model emulates inclusion of different migraine characteristics with the purpose of having a model that can correlate to human migraine attacks. In order to achieve this, we analyzed the behavior exhibited by the model, and the possible behavioral changes induced by triptans.
The behavioral results showed that the infusion of IS produced a spontaneous increase of nociceptive behavior, such as resting and freezing (behavior that is always elicited by both pain and anxiety), while it decreased the exploration behavior. These observations correlate to the clinical symptoms seen in migraineurs, characterized by a reduction of routine physical activity and lack of movement, because of pain provoked by migraine (5–7,17).
Another pain characteristic during migraine is its unilateral quality (5–7,17). The animals that underwent IS infusion displayed a novel behavior that we called “ipsilateral hindpaw facial grooming.” This spontaneous behavior is characterized by a more intense facial grooming performed with the hindpaw, ipsilateral to the place of cannula implantation and IS infusion. In this context, migraineurs during migraine attacks display a behavior characterized by intense hemifacial touching (7).
Regarding antimigraine pharmacological therapy, triptans are one of the available treatments; nevertheless, patients also commonly use NSAIDs (3). One of the most important facts for the effectiveness of antimigraine therapy is to administer the drug before the establishment of central sensitization (16). Therefore, in order to mimic the timing for drug administration in patients, we treated the animals with zolmitriptan, ketorolac and acetaminophen before the infusion of the IS.
Results from the pharmacological intervention showed that the animals treated with zolmitriptan showed reduced nociceptive behaviors in a significant manner, but the nociceptive behavior is not extinguished completely, as shown in Figure 1 (a–c). After analyzing the model longitudinally, we noticed that the effect of the zolmitriptan gained effectiveness over time, promoting a complete abolishment of the freezing behavior, induced by IS, at day four and thereafter until the end of the experimentation period.
Similarly, ketorolac induced a significant reduction in the nociceptive behavior and ipsilateral hindpaw facial grooming, but never caused them to be discontinued completely. As shown by our results, acetaminophen was the least effective of all the tested drugs when compared to zolmitriptan and ketorolac.
The results acquired by the administration of the drugs are similar to those demonstrated by patients (9,11–13), in which treatment is not completely effective, but helps to diminish pain during migraine attacks, principally by triptans.
This model shows that repeated nociceptive stimulation of the trigeminovascular system modifies the behavior as well as the pharmacological effectiveness, as shown in the rats treated with zolmitriptan, which reduces the freezing behavior completely after day four. In addition, we found a novel behavior, demonstrated in the rats that underwent IS infusion.
The results show that the animal model shares some measurable characteristics that may resemble those observed in humans during migraine. The above reasoning allows us to propose our technique as a new animal model for the study of migraine that could contribute to further understanding of the pathophysiology and study of novel therapeutic approaches for treating migraine.
Clinical implications
A murine model of chronic meningeal nociception that depicts similar manifestations as shown in migraineurs. A rat animal model that can be used to develop new pharmacological approaches for migraine.
Footnotes
Funding
Funding for this study was provided by the Instituto Nacional de Psiquiatría Ramón de la Fuente Muñiz projects: NC093530.0, NC093530.1 and NC113530. ECOS NORD project: M10 S01. CONACYT funding project NC093530 and CONACYT scolarship: 228001/295692. The Institute and CONACYT had no further role in study design, nor in the collection, analysis and interpretation of data or in the writing of the report.
Conflict of interest
None declared.
Acknowledgments
We thank Francisco Merari Aguirre Barrios, Arturo Castillo Tovar and Paul Lamothe Molina for their invaluable support during the course of this project. We also thank the program: Doctorado en Ciencias Biomédicas, Universidad Nacional Autónoma de México.
