Abstract
The study was designed to compare the response to almotriptan in migraine patients who take medication early in the course of the attack with that when medication is taken after pain has become moderate or severe. A randomized, four-arm, multicentre, multinational, double-blind, placebo-controlled trial of almotriptan (12.5 mg) comparing treatment administration when pain intensity was mild and within 1 h of headache onset vs. pain that had become moderate or severe was conducted. Of 491 migraineurs enrolled, 403 were evaluable [intention-to-treat population (ITT)]. Their mean age was 38 years, 84% were female and they had a mean of 3.7 attacks/month. Of these patients, 10% did not take medication according to their randomly allocated basal pain intensity (mild or moderate/severe) and were subsequently reassigned to that group for this analysis —'Act when Mild (AwM)' group. In the almotriptan arms, 53% of mild basal pain and 38% of moderate/severe basal pain patients were pain free at 2 h (P = 0.03; primary end-point). Corresponding proportions in the placebo groups were 25% and 17% (statistically significant vs. respective almotriptan arms). Secondary end-points (ITT) were also significantly in favour of early intervention with almotriptan, both between and across treatment groups, such as sustained pain free: 45.6% vs. 30.5% (P = 0.02). Adverse events were reported in < 5% of treated patients in all groups (NS), with no serious events. Treatment with almotriptan while migraine pain is still mild provides statistically significant and clinically relevant enhancements in efficacy compared with treatment when pain has reached higher severity levels.
Introduction
Migraine is a highly disabling (1), neurological condition (2) that has been estimated to be the most costly neurological disorder in the European Community at more than €27 billion per year (3). The development of the serotonin, 5-HT1B/1D receptor agonists—the triptans (4)—represents a landmark in the field when a specific, effective, acute therapy offered reliable responses with an excellent evidence base (5). An important issue arises in clinical practice as to the optimal timing of the use of these medicines to maximize the potential benefit to patients.
The triptan clinical trial programmes used a substantially uniform approach that typically involved asking patients to wait for the development of moderate or severe pain prior to treating an attack (6). Although this approach was clearly successful (7), the issue of patients who treat attacks when pain is mild now arises. Protocol violators in clinical trials who treated mild pain were noted to have improved pain-free outcomes at 2 h compared with those typically seen when attacks had developed to moderate or severe pain (8, 9), suggesting this may offer an avenue to improve outcomes. This opportunity has been balanced by concerns that patients who treat mild attacks may not be treating migraine, rather tension-type headache, and that the approach itself may promote medication overuse (10).
A further development that drove re-evaluation of the issue of when to treat was an effort to study allodynia in migraineurs during attacks (11). It has been recognized for some time that perhaps two-thirds of patients with migraine have allodynia, pain in response to normally painless cutaneous inputs (12). Open-label studies have suggested that the presence of allodynia predicted a poor outcome to triptan therapy (13). Moreover, triptans are unable to reverse sensitization in experimental studies of trigeminovascular nociception (14). Studies of allodynia suggest that the time to treatment in an attack may be crucial, predicting that patients would have superior outcomes if treatment was initiated within 60 min of onset of the acute attack. We therefore set out to study whether early treatment, within 1 h of onset, of mild pain would produce a superior pain-free response at 2 h to treatment of moderate or severe pain. The work has been reported in preliminary form at the 16th Migraine Trust International Symposium (London, UK 18–20 September 2006 (15)).
Methods
The study was a single-attack randomized, double-blind, placebo-controlled, parallel group, multicentre, multinational clinical trial conducted according to Good Clinical Practice guidelines (16). Forty-one centres in five countries (Belgium, France, Germany, Italy and Portugal) were involved. Ethical approvals were obtained according to appropriate national regulations. The aim was to compare treatment with almotriptan 12.5 mg in terms of pain-free responses at 2 h in patients treating when pain was still mild and within 1 h of onset compared with when pain was moderate or severe.
Design
Patients were invited to participate and, after obtaining written informed consent, were randomized to one of four groups in equal proportions:
Almotriptan 12.5 mg when pain is mild, and within 1 h of onset;
Placebo when pain is mild, and within 1 h of onset;
Almotriptan 12.5 mg when pain is moderate or severe;
Placebo when pain is moderate or severe.
Patients were asked to treat their next eligible attack and return within a week a treatment. We allowed 60 days after entry for the attack to occur. Rescue medication should it be required was one of aspirin, paracetamol or a non-steroidal anti-inflammatory drug. When permitted, preventive medications should have been stable for 1 month prior to entry, and for the duration of participation.
Inclusion criteria
We wished to include male or female patients, aged 18–65 years, in good general health. They should have migraine, with or without aura, by the definition of the second edition of the International Classification of Headache Disorders (17) for at least 1 year. By history, attacks of migraine should have had at least moderate pain intensity within the past year. Patients should have had an average migraine frequency of two to six episodes per month during the last 3 months and the ability to distinguish migraine from interval headaches, as well as being able to provide informed consent. In addition, patients should have had a history of untreated or unsuccessfully treated migraine headache pain of > 4 h duration.
Exclusion criteria
Patients with contraindications to almotriptan according to the summary of product characteristics in Europe or a history of discontinuation of almotriptan were excluded. We excluded patients with chronic daily headache or migraine frequency more than six episodes per month. Onset of migraine after age 50 years, or headache predominantly occurring on waking in the morning also excluded patients. Pregnant or breastfeeding patients were excluded, as were patients who planned to begin non-pharmacological approaches to treatment during the period of the study.
End-points
The primary end-point was a comparison of the proportion of pain-free patients at 2 h after treatment comparing those treated early and at mild pain vs. those treating moderate or severe baseline pain with almotriptan 12.5 mg. There were predefined secondary end-points as described: the proportion of patients pain free at 0.25, 0.5, 1, 1.5 and 24 h postdose; the proportion of patients with pain relief at 0.25, 0.5, 1, 1.5, 2 and 24 h postdose in the moderate/severe baseline pain arms comparing placebo and almotriptan; the proportion of patients who had a sustained pain-free response at 24 h: pain free at 2 h, without return of headache and not using rescue medication in the following 24 h; the proportion of patients taking rescue medication; the proportion of patients with relapse within 24 h, and between 24 h and 48 h postdose; the total attack duration in hours and time lost to the attack in hours; patients were asked to rate treatment satisfaction on a visual analogue scale; migraine-associated symptoms: nausea, vomiting, photophobia, phonophobia and osmophobia at baseline and 2 h after treatment; presence of cutaneous allodynia by questionnaire (18) at baseline, 2 h after treatment and its effect on treatment outcome.
Allodynia questions
The subjects were asked the following questions selected prior to publication of a fuller series (18):
Would you describe your headache as ‘throbbing’?
Was your headache worse if you changed position or moved?
Did you have soreness or achy pain in the shoulder or neck areas?
Did you have head or face pain or scalp tenderness in response to touch or pressure that usually does not cause pain?
Statistical considerations
We estimated that a sample size of 388 evaluable subjects, 97 subjects per treatment/regimen group, was required for an 80% power to detect a 20% difference in the proportion of subjects pain free at 2 h between almotriptan 12.5 mg treated when early/mild and almotriptan 12.5 mg treated when moderate to severe: 60% vs. 40%, respectively. To account for approximately 19% of expected dropouts and non-evaluable subjects after randomization, a total of approximately 480 subjects, 120 subjects per treatment/regimen group, were randomized. Data are presented as proportions of patients in the groups under study. The data were analysed for the intention-to-treat (ITT) population and for the ‘Act when Mild (AwM)’ population. The AwM population consisted of a per protocol analysis such that patients were re-assigned to the group corresponding to pain severity. This analysis was posthoc, since we had not anticipated the substantial number of patients who did not comply with the protocol. The primary end-point was analysed by means of a logistic regression model including centre and treatment as covariates. All tests were two-tailed with the significance level set at α= 0.05. Tolerability and safety data are presented in summary form.
Results
Four hundred and ninety-one patients were screened and all were randomized to one of four groups (Fig. 1). Eighty-seven patients discontinued without treating an attack, with the chief reason being that they did not have an attack to treat within 60 days of randomization. One subject had an adverse event without taking medication, which by current guidelines does not constitute an adverse event for study reporting (16). The distribution of discontinuations was similar in each group (Fig. 1). Four hundred and four patients treated one attack constituting the ITT and safety populations (19). This group was largely White and substantially female, in keeping with triptan studies done in Europe. They had comparable ages and body mass indices (Table 1). For the AwM analysis patients were reassigned to their true headache severity at baseline, consistent with the primary hypothesis that the study sought to test (Fig. 1).
Demographic data for the treated population

Mod/Sev, moderate or severe pain.
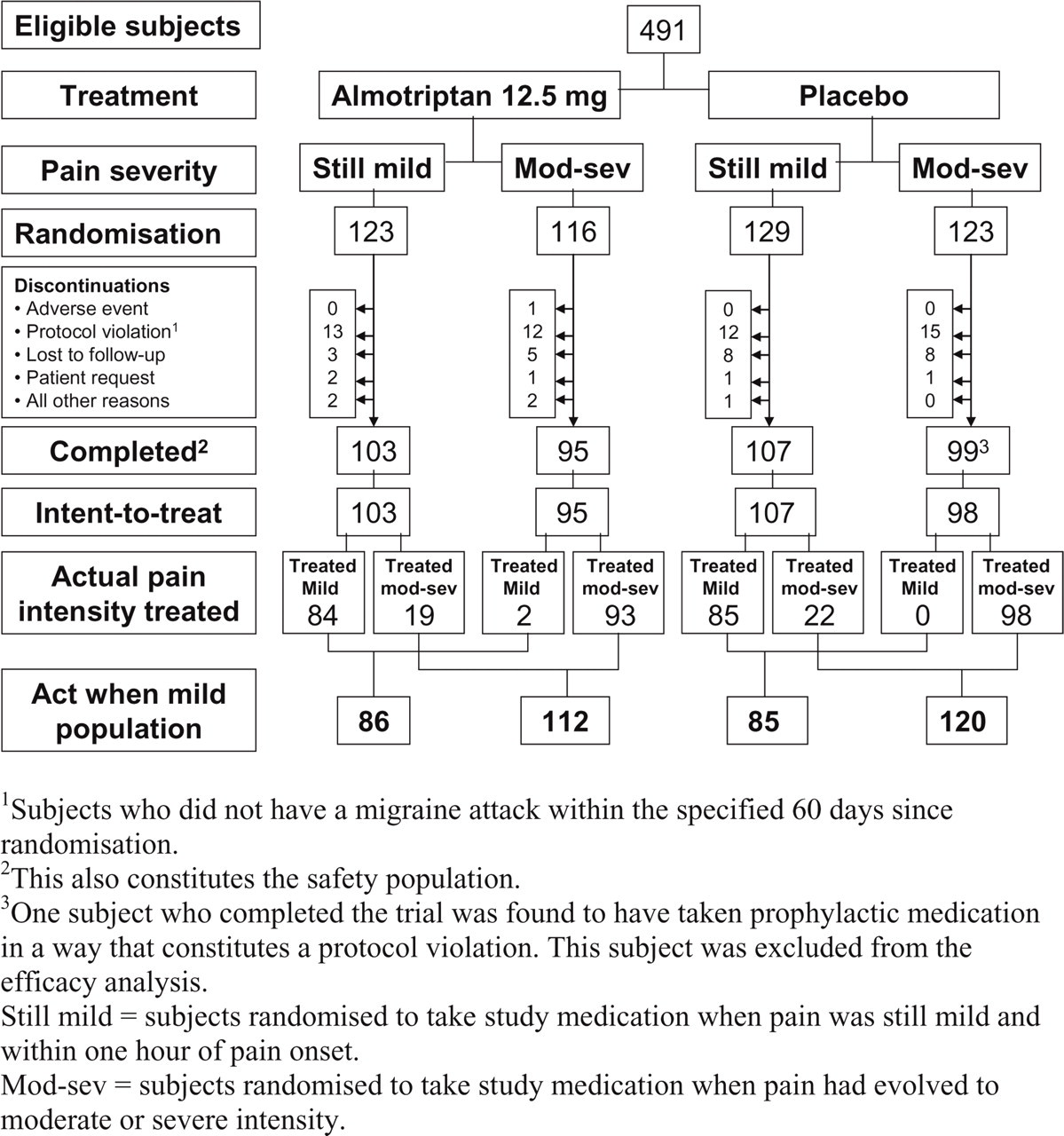
Disposition of patients. 1Subjects who did not have a migraine attack within the specified 60 days since randomization. 2This also constitutes the safety population. 3One subject who completed the trial was found to have taken prophylactic medication in a way that constitutes a protocol violation. This subject was excluded from the efficacy analysis. Still mild = subjects randomized to take study medication when pain was still mild and within 1 h of pain onset. Mod-sev = subjects randomized to take study medication when pain had evolved to moderate or severe intensity.
Primary end-point
In the almotriptan 12.5 mg mild group, a total of 50/103 (49%) subjects reported that they were free from pain at 2 h following drug administration. Slightly fewer subjects were pain free at 2 h in the almotriptan 12.5 mg moderate to severe group [38/95 (40%)]. In the placebo mild group, 27/107 (25%) subjects were pain free and 15/98 (15%) subjects in the placebo moderate to severe group. With respect to the primary treatment comparison for the ITT population, the almotriptan 12.5 mg mild group was not statistically significantly different from almotriptan 12.5 mg moderate to severe group (P = 0.2154). The odds ratio (OR) was 1.44 [95% confidence interval (CI) 0.81, 2.56]. For both the almotriptan 12.5 mg mild and moderate/severe groups there were significant differences from the respective placebo groups. The OR for pain free at 2 h for the mild group comparing almotriptan 12.5 mg and placebo was 2.93 (95% CI 1.62, 5.31, P = 0.0004), and for the comparison in the moderate/severe groups 3.71 (95% CI 1.85, 7.45, P = 0.0002) in favour of almotriptan 12.5 mg.
AwM group
Results were significantly impacted by the number of subjects in the ITT population who did not treat themselves according to the intensity of pain they were randomly assigned to treat. According to the review of data before the blind was opened (based on clinical report form information), 38 subjects assigned to treat a mild pain had missed the correct timing and treated a moderate or severe pain, whereas two subjects assigned to treat a moderate or severe pain treated a mild pain. Reassigning patients to the AwM groups in the almotriptan 12.5 mg treated mild group, 46/86 (54%) were pain free by 2 h, whereas 42/112 (37.5%) in the almotriptan 12.5 mg moderate/severe group were pain free at 2 h (P = 0.02; OR 0.54; 0.29, 1.01; Fig. 2). In relation to the duration of migraine attack, when measuring from the time of dosing, the median time to the end of the attack was significantly shorter when almotriptan 12.5 mg was taken early and when pain was still mild (1.5 h) compared with when taken after the pain had become moderate–severe (2.1 h; P < 0.004).
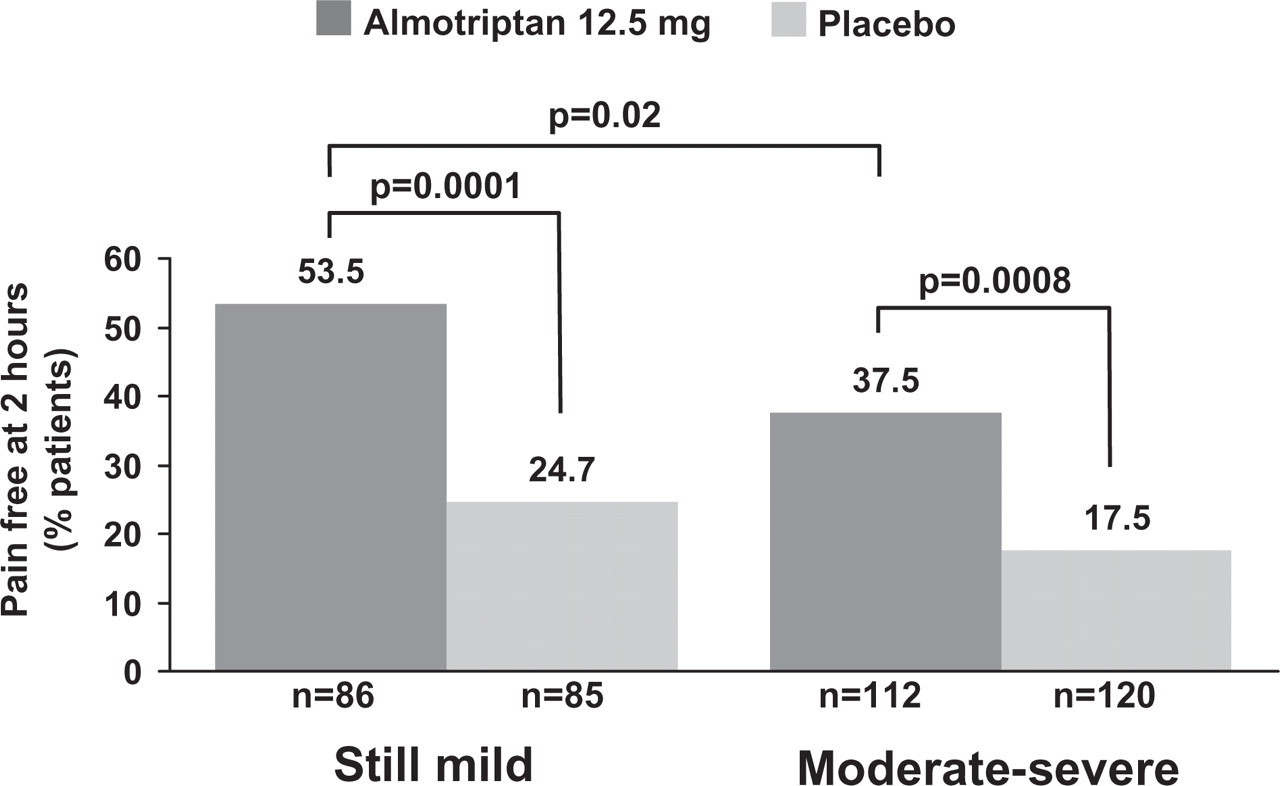
Pain-free data at 2 h in the Act when Mild (AwM) analysis demonstrating a significant benefit for treatment with almotriptan 12.5 mg when taken early, within 1 h, and when pain is still mild compared with when pain is moderate or severe.
Secondary end-points
Sustained pain free
A total of 104/403 (25.8%) subjects were free from pain for 2–24 h without the use of rescue medication: almotriptan 12.5 mg mild 47/103 (46%); almotriptan 12.5 mg moderate to severe 29/95 (30%); placebo mild 17/107 (16%); and placebo moderate to severe 11/98 (11%; Fig. 2). Patients in the almotriptan 12.5 mg mild group had sustained ‘pain free’ rates higher than the almotriptan 12.5 mg moderate to severe group (P = 0.024), with an OR of 1.99 (95% CI 1.10, 3.63) in favour of the mild group. In patients in the moderate to severe group, almotriptan 12.5 mg was better than placebo at maintaining a pain-free state, with an OR of 3.4 (95% CI 1.59, 7.47; P = 0.0018) in favour of almotriptan treatment. Similarly for the patients with mild pain, the almotriptan 12.5 mg group did better than the placebo group at maintaining a pain-free state, with an OR of 4.8 (95% CI 2.45, 9.22; P < 0.0001) in favour of almotriptan. There was no statistically significant difference between the placebo mild and placebo moderate to severe groups (P = 0.38).
Duration of migraine attack
The median duration of the migraine attack from onset to resolution of pain was significantly shorter in patients who took almotriptan 12.5 mg early and when pain was still mild (2.0 h) than in those who took almotriptan 12.5 mg after pain had become moderate–severe (5.0 h; P = 0.0005). This includes the difference in time to intake of medication between each pain severity group. Measuring from the time of dosing, the median time to the end of the migraine attack was shorter when almotriptan 12.5 mg was taken early and when pain was still mild (1.6 h) compared with when taken after the pain had become moderate–severe (1.9 h; NS). Expressing the duration of the migraine attack in terms of a Kaplan–Meier plot confirms the overall lower burden of pain in patients who took almotriptan early and when pain was still mild compared with those who took almotriptan after pain had become moderate–severe (Fig. 3).
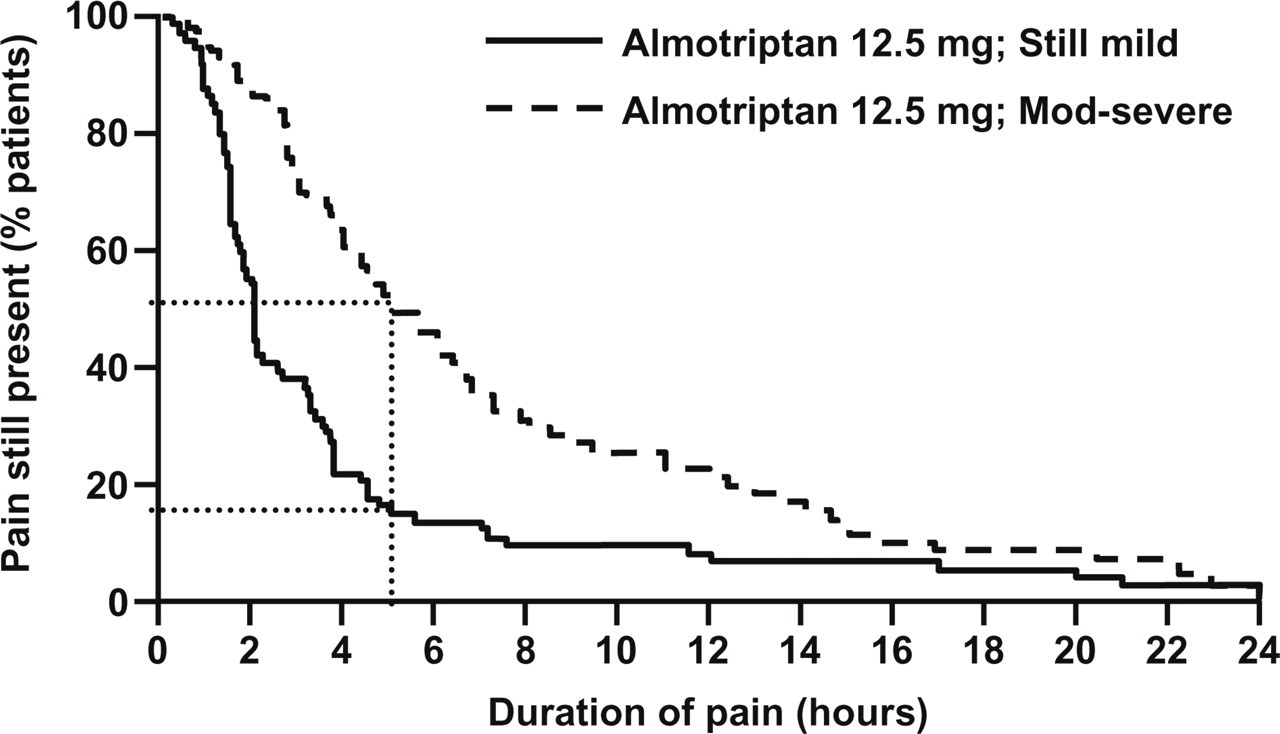
Kaplan–Meier plot of duration of migraine attack pain in patients treating with almotriptan 12.5 mg early (within 1 h of onset) while pain is still mild vs. when pain is moderate or severe. The data are based on the Act when Mild (AwM) Group (see Methods) and the difference between the curves illustrates the benefit of the AwM strategy.
Time lost in daily activities
The shorter duration of migraine attacks when almotriptan 12.5 mg was taken early and when pain was still mild compared with when taken after pain had become moderate/severe impacted positively on patients' ability to resume daily activities at 48 h after dosing. Although there was no difference between the placebo groups in median time lost (2 h), in the almotriptan 12.5 mg treatment groups the median time lost was significantly lower in the early/still mild group than in the moderate–severe baseline pain group (0 vs. 2 h, respectively; P = 0.0015).
Headache recurrence
Reflecting the sustained pain-free findings, significantly fewer patients had recurrence within 24 h of dosing following early treatment with almotriptan 12.5 mg and when pain was still mild (3/50, 6.0%) compared with those treated with almotriptan 12.5 mg when pain had become moderate–severe (9/38, 24%; P = 0.0124). In contrast, there was no significant difference in the proportion of patients with recurrence at 24 h in the two placebo groups (10/27, 37% and 4/15, 27%; P = NS). Moreover, the difference in relapse rates between almotriptan 12.5 mg and placebo in the early/still mild groups was significant (6% vs. 37%; P = 0.0006). There were no significant differences in relapse rates at 24–48 h postdose across the treatment arms.
Use of rescue medication
When comparing treatments, the number of subjects taking rescue medication in the almotriptan 12.5 g mild group was no different from those in the almotriptan 12.5 mg moderate to severe group (P = 0.1921). When comparing the almotriptan 12.5 mg moderate–severe group with the placebo moderate–severe group, there were more subjects in the placebo group who took rescue medication (P < 0.0001, OR 4.14, 95% CI 2.25, 7.60). With respect to the mild groups, there were more subjects who took rescue medication in the placebo mild group compared with the almotriptan 12.5 mg mild group (P < 0.0001, OR 0.28, 95% CI 0.16, 0.51). The difference between the two placebo groups was not significant (P = 0.056).
Allodynia
Of the patients who treated with almotriptan 12.5 mg when pain was mild (n = 86), 53% were pain free at 2 h and 92% of these were allodynic at treatment (answered ‘yes’ to at least one question). In comparison with those who treated with placebo when pain was mild (n = 85), 26% were pain free and 91% were allodynic at treatment. For those who treated with almotriptan 12.5 mg when pain was moderate or severe (n = 112), 38% were pain free at 2 h and 96% had been allodynic at treatment. Again, for comparison of patients treating moderate or severe pain with placebo, 18% were pain free at 2 h and 98% had been allodynic at treatment. Table 2 sets these data out in more detail. Allodynia did not predict a poor outcome, rather baseline headache severity seemed Key predictor.
Migraine-associated symptoms and signs of allodynia at baseline and 2 h postdose

BL, baseline; 2 h, 2 h postdose; TG, therapeutic gain (active response minus placebo response).
Safety and tolerability—adverse events
During the treatment phase, five of 103 (4.9%) subjects had a total of eight adverse events (AEs) in the almotriptan 12.5 mg mild group; four of 95 (4%) subjects had four AEs in the almotriptan 12.5 mg moderate to severe group; five of 107 (4.7%) subjects had nine AEs in the placebo mild group; and four of 99 (4%) subjects had five AEs in the placebo moderate to severe group. All AEs during the study were of mild to moderate severity. All AEs had resolved by the end of the study.
Discussion
This study has demonstrated using a rigorous double-blind placebo-controlled design that almotriptan is effective in the acute treatment of migraine when the attack is in its early stages and pain is mild, and when the attack is more developed and the pain is moderate or severe. Moreover, if it is accepted that the AwM group is the best vehicle to question that issue, the study has also demonstrated that early treatment when pain is mild has benefit over the following 24 h with reduced time with symptoms. Interestingly, the presence or absence of allodynia did not predict the response to treatment when compared with baseline severity. Furthermore, there is no AE penalty, so that for many patients it seems logical to advise them to proceed to treatment as soon as they are sure they have a migraine.
The issue of how best to use triptans, serotonin 5-HT1B/1D receptor agonists, is not trivial given the high prevalence of migraine (20). Triptan studies were initially done using a rather uniform clinical trial design, where patients were asked to wait and treat attacks when the pain was at moderate or severe intensity (6). One important reason for this was to try to avoid the treatment of tension-type headache, although this design itself would not necessarily avoid that complication given the complexity of migraine. It had been the standard of care in the ergotamine era to treat attacks as early as possible (21). This was based largely on the dual issues that it was slowly absorbed and poor absorption itself was a feature of acute migraine. Three areas of consideration suggest that triptans may be better used earlier in migraine. First, clinical experience tells us that treating milder attacks seems to be efficacious. Many clinicians have advocated this over time in order to try to turn off the brain mechanisms involved in the attack as soon as possible. Second, retrospective analyses of protocol violators from placebo controlled-trials, patients who treated when their attack pain was mild as opposed to moderate or severe, reported higher pain-free rates (8, 9). Third, as interest has grown in allodynia (11, 22), a clinical symptom well recognized for many years (12), open-label studies have suggested that the presence of allodynia takes time to develop, and that the presence of allodynia predicts a poor outcome from triptans (13). Thus, two hypotheses have developed: first, that treatment of migraine earlier in the acute attack course when pain is mild would be more effective than treating it when pain is moderate or severe, and second, that the development of allodynia predicts a poor outcome from acute migraine treatment.
Several studies (Table 3) have been conducted with randomized, double-blind, placebo-controlled designs. A number of open-label or unblinded studies have been reported and suggest that earlier/mild stage treatment is useful, including those done with almotriptan (23). Klapper and colleagues (24) tried to address the issue of treating the attack when pain is mild using zolmitriptan 2.5 mg comparing it with placebo in patients who would typically have a window of mild headache. Zolmitriptan was more effective than placebo at 2 h. It is interesting in the context of our study that some patients still waited for the attack pain to become moderate or severe. A further study with zolmitriptan 2.5 mg has reported results from two attacks pooled, again asking patients to treat early. A similar superiority for zolmitriptan was demonstrated (25). Remarkably, the therapeutic gains, difference between the active response rate and the placebo response rate, were almost identical in these studies, 25% and 24.4%, and were very similar to what has been reported for the more traditionally designed studies, 21.1% (5). A further study with a crossover design, where only the first dose was reported, showed superiority for frovatriptan 2.5 mg on the 2-h pain-free end-point when compared with placebo in the treatment of migraine when the pain is still mild (26). However, the therapeutic gain was rather modest at 8%. A study by Brandes and colleagues (27) attempted to address the intertwined issues of severity of pain and timing of treatment. Using eletriptan 40 mg they demonstrated superiority for the triptan in the 2-h pain-free outcome. Patients were encouraged to treat when pain was mild, but it was not mandated, so some treated when it was moderate or severe. The response rate for treating mild pain was higher than for moderate or severe, while the time to treatment, within 1 h or after 1 h, did not alter the outcome. Finally, Cady and colleagues (28) have reported two large placebo-controlled trials treating mild pain in migraine within 1 h, again demonstrating comparable therapeutic gains of 26–28%. Our new data support and extend the position, using almotriptan 12.5 mg, that treatment of the attack when pain is mild is more helpful than when it is moderate or severe. However, it is one thing to terminate the attack in a timely fashion—does treatment of the attack when pain is mild secure a more extended benefit?
Controlled trials in acute migraine of the treatment of mild pain or of ‘early’ treatment reporting 2-h pain-free rates∗

All trials listed are double-blind and placebo-controlled.
TG, therapeutic gain = active response minus placebo.
Sustained pain free is an excellent metric of outcome for the attack, combining an initial pain-free response at 2 h with no recurrence of pain and no need for rescue medication within the following 24 h. In the eletriptan study cited above (27), sustained pain-free responses appeared superior, although the study was not apparently powered to show a difference. Our new data explore this issue, demonstrating that almotriptan when used to treat migraine while pain is still mild results in a significantly higher sustained pain-free rate at 24 h than is the case if it is used when pain is moderate or severe. This finding is important, because if treatment of mild pain only resulted in higher 2-h pain-free outcomes without sustained effect, it might produce many more attacks where a second dose was needed, and thus raise the possibility of developing medication overuse problems. The data suggest this should not occur.
We have explicitly studied allodynia using part of a questionnaire developed to explore the issue (18), which had not been fully published when this study commenced. It has been suggested by an open label study that the presence of allodynia predicts a poor outcome from triptan treatment (13). A recently published study using cutaneous sensitivity as a surrogate marker of allodynia has suggested that pain-free rates for rizatriptan 10 mg were not altered by its presence (29). The new study explores this question and again we find no link. It remains clear that very early treatment, at 20 min or less, is unexplored and will be somewhat difficult in clinical practice to test.
Some limitations of this study and of the issue of the treatment of migraine while headache is mild need to be considered. The fact that so many patients chose to ignore the dosing instructions and wait until pain was moderate or severe represents an issue in clinical practice—many patients feel they must save their triptan for the worst attacks—and a trial design issue. We looked at the issue of non-compliance prior to the blind being broken and resolved to explore this. If the hypothesis that treating when pain is moderate or severe will reduce response rates is correct, then it is in principle a problem to ignore the issue in the analysis. We felt it correct to report the ITT population and very clearly label the AwM group so that we could understand the implications. Were the attacks that patients chose to delay treating different? We shall not know. Certainly, there was no country bias or other demographic issue that could be detected. A further issue is whether a study should be conducted that randomizes to both early and late treatment of both mild and moderate/severe pain. This is probably logistically impossible and complicated by the pressure to keep placebo groups to a reasonable minimum.
