Abstract
The extent and nature of triptan use for headache relief has been evaluated in a large epidemiological survey in the French general population. Over 25 000 individuals were screened for headache and for triptan use. Of this sample, 290 triptan users were identified from whom extensive data on headache characteristics and healthcare resource consumption were obtained. The use of triptans is relatively infrequent, 0.2% in the general population, with only 7.5% of migraine sufferers using these drugs. The majority of triptan users were female (80%) and presented headache characteristics typical of migraine (80%). The remaining 20% of subjects were thus using triptans for headache types in which the utility of these drugs has not been demonstrated. Among migraineurs, triptan consumers reported more frequent and severe headaches than non-consumers, and reported a higher incidence of nausea and vomiting. The principal determinant of triptan prescription was consultation with a general practitioner (GP), which may itself have been triggered by the severity of the headaches. GPs, rather than specialists, are the primary prescribers of triptans in France.
Introduction
During the 1980s, a new class of drugs for the acute treatment of migraine attacks was developed, the triptans (1). The first of these was sumatriptan, followed more recently by zolmitriptan, naratriptan, rizatriptan, eletriptan and almotriptan. These agents are selective 5-HT1B/1D serotonin receptor agonists, and were developed in order to address the underlying biological mechanism of the migraine attack (2). The pathophysiology of migraine is believed to involve local vasodilatation of the cranial vessels irrigating the dura mater and antidromic activation of nociceptive sensory afferents running in the trigeminal nerve. Activation of these neurones leads to local release of neuropeptides such as substance P, calcitonin gene-related peptide and neurokinin A, which cause neurogenic inflammation and further vasodilatation. Activation of serotonin receptors in these cranial vessels leads to vasoconstriction, whilst in the trigeminal nerve, excitability is attenuated, thus reducing neuropeptide release (3). This theoretical schema has been confirmed in an extensive series of clinical trials with sumatriptan, which has demonstrated powerful efficacy in the acute relief of migraine attacks (4). The triptans are thus a major advance in the rational treatment of migraine and are considered to be the most efficacious treatment of choice for severe migraine.
Over the last 12 years, several triptans have been made commercially available. These differ principally in their pharmacokinetic properties or in their potency. However, from a clinical point of view, only minor differences in efficacy have been demonstrated between the different drugs (5, 6). In France, triptans were introduced later than elsewhere, and their cost reimbursed (65% of cost by national health service and a variable amount by private insurance) only since June 1998. At the time of the study, three triptans were available for the treatment of migraine: sumatriptan (Imigrane®, subcutaneous form, oral form, nasal spray), zolmitriptan (Zomig®, oral form) and naratriptan (Naramig®, oral form).
It is timely to evaluate with hindsight what place these much heralded new drugs occupy in the everyday management of migraine, and what impact they have had. Information on this can be obtained from patient registry surveys, from studies of central prescription registers in those countries where this information is available, or from general population surveys. In reality, there is very little such information on triptan use accessible in the public domain. A prescription register survey in Denmark (7) revealed that nearly 1% of Danes had recourse to sumatriptan over a 1-year period, compared with an overall prevalence of migraine in Denmark of 13% (8). An important finding of this study was that a minority of sumatriptan users (5%) were taking this medication on a daily basis, accounting for 38% of all sumatriptan use. To the best of our knowledge, no general population data have been reported.
We have recently performed a survey in France to study the epidemiology and clinical presentation of migraine in the general population (GRIM2000) (9). The prevalence of migraine [International Headache Society (IHS) categories 1.1 and 1.2] was 7.9% and the prevalence of migrainous disorder (IHS category 1.7) was 9.1%. In an ancillary population, we have now extended this survey to users of triptans, in order to evaluate the extent and appropriateness of triptan use in the general population. The results of this analysis are presented here.
Methods
Screening of the general population
Data were generated from a nationwide survey of headache sufferers in France conducted by ISL (Institut de Sondage Lavialle, Issy-les-Moulineaux, France), an international polling institute. All data were collected between October and December 1999. The use of Computer-Assisted Personal Interview technology ensured optimal data collection from the subjects interviewed.
During a first screening phase, a national representative sample of the general population was screened in order to identify headache sufferers and triptan users. A sample of 10 575 individuals from the general population aged ≥ 15 years was taken from a panel of 21 000 individuals using a random sampling method (FLASH sample). The structure of the sample was controlled using a stratified method based on national population data from the 1996 INSEE (Institut National des Statistiques et des Etudes Economiques) report to ensure representativity with regards to sex (two categories), age (five categories), socio-professional status (seven categories), regional distribution (nine regions), and size of the community (five categories). Screening questions were asked during a face-to-face interview conducted by trained interviewers as part of an omnibus survey that asked specific health-related questions within a much wider multisubject questionnaire. Overall, the demographic characteristics of the subjects screened were representative of national population data (9). In the initial screening sample, subjects were selected for the data collection phase if they responded positively to the question ‘Have you ever suffered from headaches?’. However, since the proportion of triptan users in this sample was very low (84 out of 1486 interviewed headache sufferers), a separate second panel of 15 000 individuals (ISOSCOPE sample) was screened by a postal omnibus survey which included the question ‘Have you taken one of the following drugs: Imigrane (as a tablet, nasal spray or injection), Naramig or Zomig?’. This second sample identified 347 subjects reporting triptan use.
Data collection
In the second data collection phase, face-to-face interviews were carried out with individuals identified as headache sufferers in the screening phase (1486 from the FLASH sample and 347 from the ISOSCOPE sample). These interviews were conducted by trained interviewers in the subject's home by appointment. Information was collected on the symptoms and clinical description of the headaches, on quality of life using the QVM disease-specific measure (10), on disability using the MIDAS scale (11), and on healthcare consumption. During the interview, subjects were asked if they had been prescribed a triptan for their headaches. Background demographic data were also obtained.
Data analysis
The clinical data generated were used to classify the headaches as migraine using a specific algorithm based on the diagnostic guidelines established by the IHS (12). This algorithm has been described in detail elsewhere (9). Briefly, the subjects’ replies to certain questions about the nature of their headaches allowed the four IHS criteria for the diagnosis of migraine (duration of untreated headaches of 4–72 h; presence of at least two of unilaterality, pulsatility, incapacitating intensity and aggravation with effort; associated with either nausea and/or vomiting or photophobia and phonophobia; more than four such headaches experienced) to be assessed. Subjects reporting a headache frequency of on average one every 24 h were classified as chronic daily headache. For subjects to be classified as strict migraine (IHS category 1.1 and 1.2), they had to fulfil all four IHS criteria. If only three conditions were fulfilled, a classification of migrainous disorder was made, in accordance with IHS category 1.7. Subjects fulfilling less than three criteria were classified as ‘other episodic headache’. For certain analyses, the strict migraine and migrainous disorder groups were combined into a total migraine group. This technique of retrospectively assigning diagnosis has previously been validated against expert diagnosis established by neurologists (13). The clinical features of the diagnostic groups so established are described in detail elsewhere (9, 14).
Subjects were questioned about the length of time they had suffered from headaches, and about the frequency of these. Data were collected about the duration, frequency and severity of the headaches by presenting the subjects with a multiple-choice list. The incidence of nausea and vomiting was also determined. A composite measure of headache severity (MIGSEV) (15) was used. This is a three-category scale obtained from three-point measures of the pain intensity, tolerability, nausea and discomfort of the last headache attack. On the basis of this score, subjects were rated as mild (score < 2), moderate (score 2–6) or severe (score > 6).
The degree of disability attributable to the headaches was also evaluated using the MIDAS questionnaire (11, 16). This contains seven questions. The first five (M1–M5) assess the number of days in which professional, domestic, social and recreational activities have been diminished by headaches. From these items a global score is calculated, which can then be categorized into four grades of increasing severity. The sixth question (M6) measures the number of days over the previous 3 months that the subjects have suffered a headache, and the seventh question (M7) the average severity of these headaches rated on a scale of 1–10.
Part of the interview was devoted to healthcare resource utilization, and subjects were asked specifically whether they had taken any of the five triptan preparations available in France, identified by their trade names. Those replying in the affirmative to both the questions ‘have you been prescribed a triptan for your headache?’ and ‘have you taken a triptan for your headache?’ were defined as triptan users. Subjects were questioned about the frequency and nature of physician consultations. For the healthcare consumption data, where a subject could not provide specific information, it was assumed that no resources had been used. Data were also collected on handicap and quality of life, but these will not be described.
Statistical analysis
All data were controlled, validated and analysed centrally. As a preliminary step, the principal demographic and nosological characteristics of the FLASH and ISOSCOPE populations were compared to ensure that the two populations were comparable using a test for homogeneity (χ2 test). Although the principal data are presented separately for the FLASH and ISOSCOPE populations, intergroup comparisons between the triptan consumers and non-consumers were performed using the pooled data from the two populations in order to optimize statistical power. The χ2 test was used except where otherwise stated. Missing data corresponded to < 10% of the sample size, with the exception of the healthcare resource consumption data, for which missing values were assimilated to ‘no consumption’. For the other items, missing responses were assumed to be distributed identically to declared responses. A probability level of P < 0.01 was considered statistically significant.
Results
Identification of study population
During the screening phase of the survey, 10 585 subjects in the FLASH sample were interviewed with the omnibus questionnaire. Of these, around one-third (n = 3087) reported that they suffered from headache and were retained for the second data collection stage. Exploitable data were successfully collected from 1486 individuals. Triptan use was reported by 84 subjects. These were entered into the ‘Triptan consumer’ population. The remaining 1402 subjects constituted the ‘Triptan non-consumer’ population. Regular use of headache relief medication, principally analgesics and non-steroidal anti-inflammatory drugs (NSAIDs), was high (67% of subjects) in the FLASH sample (15).
In the ISOSCOPE sample, 15 000 subjects were initially interviewed with the omnibus questionnaire. Of these, 347 (0.2%) reported that they used triptans, of whom 235 were successfully interviewed and yielded exploitable data. All 235 subjects reported suffering from headaches and triptan use was confirmed by 206 subjects. These were also entered into the ‘Triptan consumer’ population. This population therefore contained a total of 290 subjects. The 19 remaining subjects in whom triptan use was not confirmed in the follow-up interview were discarded. The flow of subjects through the selection process is illustrated in the Venn diagram in Fig. 1.

Venn diagram illustrating patient numbers at the different steps of the population survey.
Comparison of triptan consumers in the FLASH and ISOSCOPE populations
Given that the ‘triptan consumer’ population were derived from two different source populations (FLASH and ISOSCOPE), it was important to verify that these two subject populations were comparable. This was indeed the case for age, gender, migraine diagnosis, duration of headache pathology and MIDAS severity score (Table 1). We also evaluated the representativity of the subjects who provided headache data with respect to the target population that was originally screened in terms of age, gender, occupation and geographical region. In the FLASH sample, the participants were indeed representative of all parameters with the exception of gender, where women were slightly over-represented (9). In the ISOSCOPE sample, the 235 participants were comparable to the target population of 347 for all parameters (no significant difference at P < 0.05; χ2 test). However, the 112 non-participants did differ somewhat from both the target population and the participants, since males and the older age groups (>50 years) were over-represented among the non-participants.
Comparison of triptan consumers from the FLASH and ISOSCOPE sample populations

∗Student's t-test. MIDAS, Migraine Disability Assessment.
Diagnostic features of triptan consumers
The subjects were classified into four diagnostic categories using the diagnostic algorithm based on the IHS criteria (Table 2). In the FLASH sample, the large majority (75.0%) of triptan users were in the strict migraine and migrainous disorder categories (corresponding to IHS categories 1.1, 1.2 and 1.7). The 63 migraineurs using triptans represent 7.5% of the total migraineur population. Subjects with chronic daily headache were also over-represented in the triptan consumers. On the other hand, subjects with other forms of episodic headache were rarely triptan consumers (< 5%), even though these individuals represent around one-third of all headache sufferers. This distribution of diagnostic categories was replicated in the larger ISOSCOPE sample (Table 2). When pooled triptan consumers were compared with non-consumers, there was a significant difference on distribution of diagnosis between the two populations (P < 0.01; Wilcoxon test).
Distribution of triptan consumers and non-consumers according to headache diagnosis
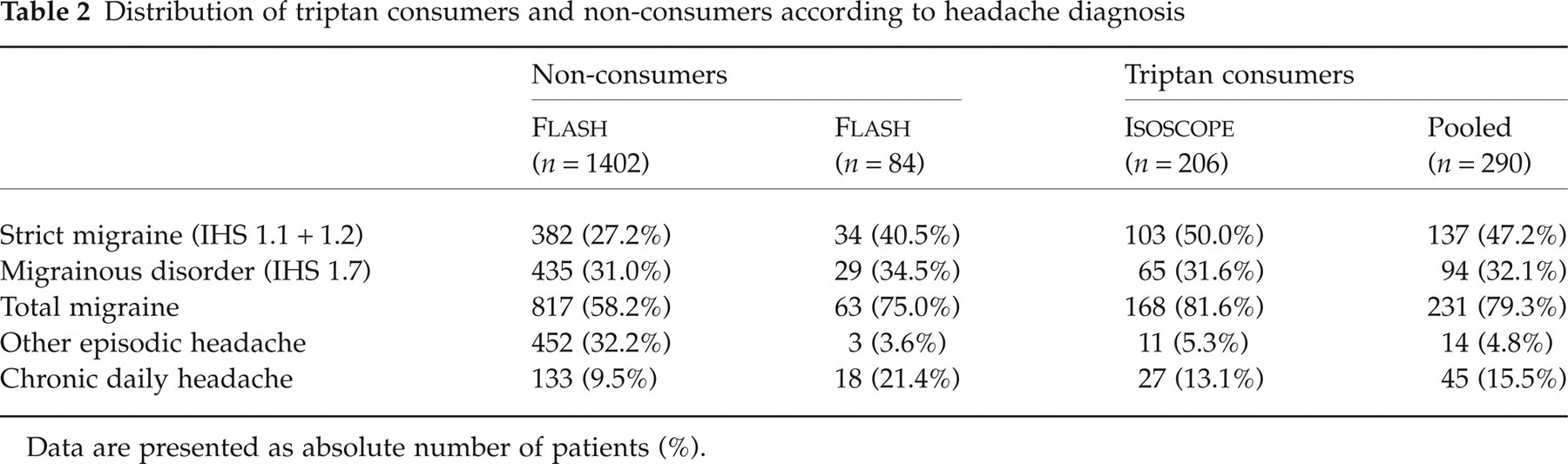
Data are presented as absolute number of patients (%).
Demographic features of triptan consumers
Since age and gender had been shown to be comparable in the FLASH and ISOSCOPE samples, the two samples were pooled for this analysis. Triptan consumption was somewhat more frequent in females than in males, with 232 females (18.1% of the female population) and 58 males (14.1% of the male population) reporting triptan use. Eighty percent of triptan users were thus female. This gender difference was not significantly different (χ2 test) from that seen in the triptan non-consumers (74.9% female). When the comparison was restricted to the migraineur population only (IHS categories 1.1, 1.2 and 1.7 combined), a similar gender distribution was seen (22.7% consumers among females, 19.7% consumers among males). The mean age of the triptan consumers was 38.5 (SD 11.0) years and that of the triptan non-consumers 39.6 (SD 16.6) years. The two groups are not significantly different with respect to age distribution (Mann–Whitney U-test). Again, essentially similar data were obtained when non-migraineurs were excluded from the analysis [mean age of 37.7 (SD 10.3) years and 38.1 (SD 13.5) years for consumers and non-consumers, respectively].
Headache features of triptan consumers
The headache features of migraineurs (IHS categories 1.1, 1.2 and 1.7 combined) consuming triptans were compared with those of migraineurs who were non-consumers. In general, all the subjects interviewed had suffered from headaches for many years. There was no significant difference in disease duration between the triptan consumers and non-consumers. The mean duration of illness was 18.9 (SD 12.3) years in the former and 18.2 (SD 13.7) years in the latter. Most subjects in all groups experienced headaches at least monthly, if not more often (Fig. 2). There was no significant difference in the distribution of frequency of headache attacks between triptan consumers and non-consumers. Nausea associated with headaches was encountered more frequently in triptan consumers from both the FLASH and ISOSCOPE populations than in the non-consumers from the FLASH sample (Fig. 2). Vomiting was reported in only three subjects (all triptan non-consumers). The distribution of nausea and vomiting was significantly different in the triptan consumers compared with non-consumers (P < 0.001).
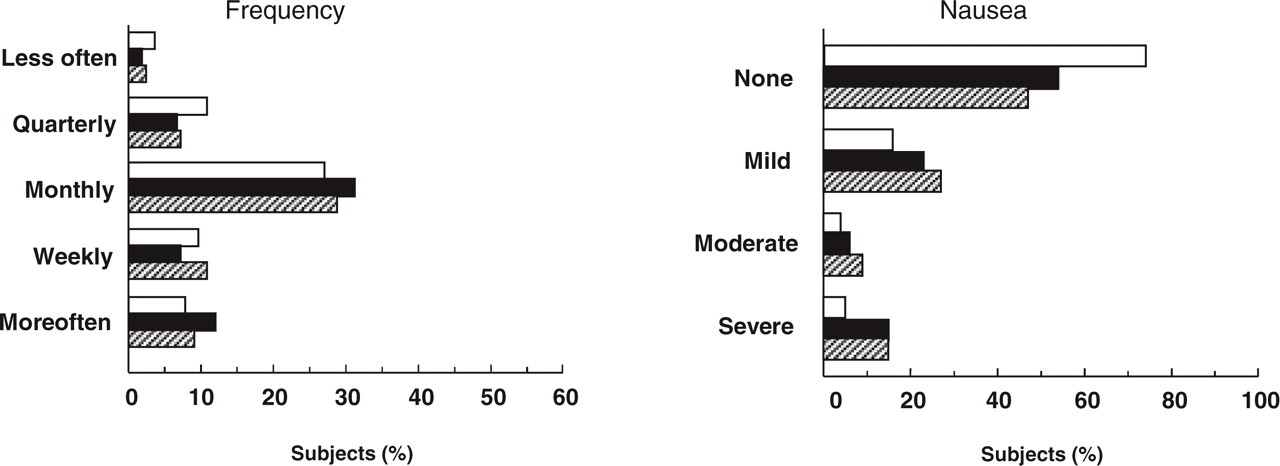
Clinical characteristics of headaches in migraineurs. Left: frequency of attacks. Right: prevalence of associated nausea. Values are given as percentage of subjects in each category. □, Triptan non-consumers from the FLASH sample (n = 817); ▪, triptan consumers from the FLASH sample (n = 63); ▨, triptan consumers from the ISOSCOPE sample (n = 168). There were no more than five missing values in any group.
Data on pain intensity, tolerability, nausea and handicap of the last headache were used to obtain the consolidated MIGSEV score for headache severity. The distribution of MIGSEV scores was unequally distributed between triptan consumers and non-consumers (P < 0.001; χ2 test), with the former group being more highly represented in the moderate and severe MIGSEV categories (Fig. 3). Similarly, the distribution of MIDAS grades was significantly different, with triptan consumers being over-represented in higher MIDAS grades (Fig. 3).
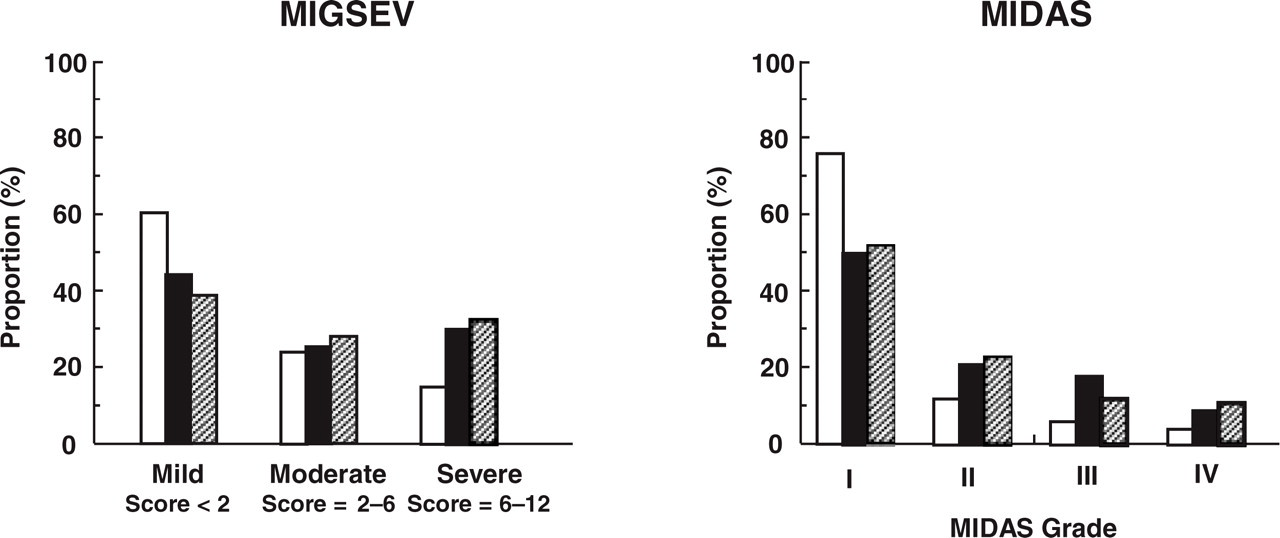
Distribution of MIGSEV category and MIDAS grade in migraineurs according to triptan consumption. Left: MIGSEV category. Right: MIDAS score. Values are given as percentage of subjects in the sample in each MIGSEV category. □, Triptan non-consumers from the FLASH sample (n = 817); ▪, triptan consumers from the FLASH sample (n = 63); ▨, triptan consumers from the ISOSCOPE sample (n = 168). There were seven missing values for MIGSEV categories (all in the non-consumer group) and 62, eight and nine missing values for MIDAS grade in the triptan non-consumers from the FLASH sample, triptan consumers from the FLASH sample and triptan consumers from the ISOSCOPE sample, respectively.
Physician visits by triptan consumers
When the extent of medical intervention was compared between triptan consumers and non-consumers, important differences between the two groups emerged. Triptan consumers were nearly four times as likely to have consulted their general practitioner (GP) about their headaches than non-consumers (Table 3). However, the frequency of GP visits was similar in triptan consumers and non-consumers (median = 2 visits in the previous 6 months). The proportion of subjects having consulted a neurologist or other specialist was also significantly higher in the triptan consumers. It should be noted that only a minority (less than one-third) of specialist consultations involved a neurologist. Those subjects using triptans who specified the source of the prescription (>80.0%) all reported that this medication had been prescribed by the GP. Medical consultation patterns were essentially identical in the FLASH and ISOSCOPE samples.
Comparison of headache-related physician visits between triptan non-consumers and consumers

Values are given as absolute number of patients (%) who reported seeing a physician at least once in the previous 6 months.
∗These data are calculated only for those subjects who consulted a general practitioner at least once.
Discussion
This population-based survey has evaluated the use of triptans in headache sufferers in France. Two different representative samples of the general population were studied, the FLASH sample of 10 585 subjects screened by self-report of headache, and the ISOSCOPE sample of 15 000 subjects, initially screened by self-report of triptan use. These two samples were comparable in terms of age and gender distribution, and treated identically in terms of the face-to-face interview and data analysis. Although the two samples were superficially comparable, they were analysed separately in order not to introduce bias due to differences in the sample selection. This enabled characteristics of triptan consumers identified in the smaller comparative FLASH sample to be replicated in the larger ISOSCOPE sample.
From a methodological point of view, the use of a general population survey has a number of advantages and disadvantages compared with patient registry and population register studies. The interrogation of a representative sample of the general population limits the risk of selection bias potentially encountered in patient registry studies. The use of face-to-face interviews enables information on medication use to be correlated with diagnosis and clinical features of disease. This information is often lacking in prescription register studies. On the other hand, data collection by patient self-report rather than from medical records may compromise the accuracy of the medical information obtained. Moreover, the one-off nature of the study provides cross-sectional data, but no longitudinal or historical information is available. An additional potential source of error arises from any selection bias due to the incomplete participation rate. Participation in this study was 48% in the FLASH sample and 68% on the ISOSCOPE sample. These participation rates are in themselves unsurprising for this kind of survey in general population studies. However, the representativity of the participants with respect to the total target population was acceptable in both the FLASH (9) and the ISOCOPE samples.
The overall prevalence of triptan use in the general population obtained from the ISOSCOPE sample was around two per thousand. This figure is quite low compared with data from population surveys or prescription registry studies in other countries (e.g. 7.8 per thousand in the Danish study by Gaist (7)). This difference cannot be explained by a lower prevalence of migraine in France, since the figure of 7.9% obtained for IHS migraine categories 1.1 and 1.2 in the GRIM2000 study (9) was similar to that obtained in other countries (18). Moreover, only 7.5% of migraineurs reported using triptans for acute relief of headache attacks. This figure is very similar to the proportion of triptan users amongst migraineurs (8%) reported in another recent survey of migraine in France (19). A drawback of the present study compared with prescription register studies is that triptan use is identified by subject self-report. The extent of use may thus be underestimated due to subjects’ failure to recognize the medication that they use as a triptan. However, this is unlikely to triple the prevalence of triptan use, and the conclusion that triptan use in France is comparatively low appears robust. It is possible that the more recent introduction of these drugs to the French market compared with other countries may contribute to this difference, since only a little over a year separated the reimbursement of triptans in France and the start of this survey. This explanation is supported by another survey (FRAMIG study) showing that triptan use in France rose from 3% in 1998 to 8% in 2000 (19, 20).
Concerning diagnosis, 80% of triptan users in the combined FLASH and ISOSCOPE samples were identified as suffering from migraine (IHS categories 1.1, 1.2 or 1.7), 15% from chronic daily headache and 5% from other forms of episodic headache. The prevalence and clinical features of migraine and of chronic daily headache in the diagnostic groups identified by our algorithm, described extensively elsewhere (9, 14), are consistent with those described elsewhere in the literature. Currently, triptans have demonstrated their efficacy only in migraine headaches, and their use in chronic daily headache or non-migraine episodic headaches may not be justified. Indeed, two randomized clinical trials have shown sumatriptan to be inefficacious in episodic tension-type headaches, at least in individuals without co-morbid migraine (21, 22). Another form of potential triptan misuse identified elsewhere (e.g. (7)) is the frequency (daily) of triptan consumption. There is no evidence that triptans are of use in the prophylaxis of migraine, and indeed excessive use may favour headache development. Headache medication misuse has been incriminated as a possible cause of transformation of classical migraine into chronic daily headache (23), and recent data from France have demonstrated that chronicity and treatment resistance of headaches are frequent in subjects over-using triptans (14). In the present study, 45 (25%) of 178 patients with chronic daily headache were taking triptans. However, potential causal relationships between triptan misuse and development of chronic daily headache cannot be addressed in the present study due to the lack of longitudinal data.
When the populations of migraineurs using triptans and those that do not are compared, no striking differences in demographic features are seen. The principal factor that seems to condition triptan use is medical consultation. Since these drugs are available only on prescription, seeing a doctor is a prerequisite to using triptans. In the FLASH sample, only 23% of migraineurs reported having consulted their GP at least once in the previous 6 months. However, for those that did consult, around half were prescribed a triptan. Triptan prescription was made by GPs, rather than by neurologists or other specialists. The vast majority of subjects are thus treating their headaches, if at all, with over-the-counter medications such as NSAIDs and mild analgesics, which may be less efficacious than the triptans. The triptan users tended to have more frequent and more severe headaches than non-users, with a higher prevalence of nausea and vomiting. These factors may have incited subjects to consult their doctors about their headaches.
Although there is a higher proportion of MIDAS grades III and IV (i.e. more severe migraine) in triptan users than in non-users, subjects in all MIDAS grades used triptans. A recent study in the USA has demonstrated that triptans are most appropriate for the treatment of severe (MIDAS grades III and IV) migraine headaches (24), and current American treatment guidelines recommend a stratified approach to migraine relief based on MIDAS grades (25). These advocate the use of triptans as first-line treatment for severe migraine, and of NSAIDs or mild analgesics for milder (MIDAS grades I or II) migraines. In France, previous guidelines, now under re-evaluation, recommended a step-by-step treatment strategy, whereby all patients were treated initially with NSAIDs or mild analgesics, and non-responders then prescribed triptans. The data from our study suggest that severe migraineurs in France are probably under-prescribed triptans, whilst subjects with mild migraine are over-prescribed these drugs. Moreover, given the overall low level of GP consultation for headaches, a stratified treatment approach may be more appropriate than a serial one. Future treatment guidelines in France should take these considerations into account.
In conclusion, triptan use by migraineurs is relatively low in France. This can be attributed to limited physician consultation. Once entered in the healthcare system, prescription of triptans to these patients is frequent. The majority of prescription was to migraine sufferers, although 20% went to individuals whose headache forms have not been shown to benefit from treatment with triptans.
