Abstract
Objectives
The aim of this study was to evaluate the angiotensin II receptor antagonist candesartan as prophylactic medication in patients with episodic cluster headache.
Methods
This study comprised a prospective, placebo-controlled, double-blind, parallel-designed trial performed in seven centres in Scandinavia. Forty (40) patients with episodic cluster headache (ICHD-2) were recruited and randomised over a five-year period to placebo or 16 mg candesartan in the first week, and placebo or 32 mg candesartan in the second and third week.
Results
The number of cluster headache attacks (primary efficacy variable) during the three-week treatment period was reduced from 14.3 ± 9.2 attacks in week 1 to 5.6 ± 7.0 attacks in week 3 (−61%) in the candesartan group and from 16.8 ± 14.1 attacks in week 1 to 10.5 ± 11.3 attacks in week 3 (−38%) in the placebo group. The difference between the candesartan and placebo group was not significant with the pre-planned non-parametric ranking test, but a post-hoc exact Poisson test, which takes into account the temporal properties of the data, revealed a significant result (p < 0.0001).
Conclusions
This was a negative trial. Post-hoc statistics suitable to describe the temporal changes in cluster headache indicate that conduction of future larger studies may be justified.
Introduction
Prophylactic treatment studies on episodic cluster headache using ICHD criteria.

Difference from baseline. bAll included patients had major protocol violations. cCessation of attacks in 15% in active group and 14% in placebo group. d‘Subjective substantial improvement.’ eThe difference from run-in was 55% in active group and −5% in placebo group. fReduction in headache severity rating of 30% in active group and 27% in placebo group from run-in to treatment week. E: episodic cluster headache (CH); C: chronic CH; ICHD: International Classification of Headache Disorders.
Candesartan cilexetil (Atacand™), an angiotensin II receptor blocker, has been shown to prevent migraine attacks with few side effects (8). The present trial is the first study with the aim to evaluate candesartan as a prophylactic treatment in patients with episodic CH (ECH).
Methods
This was a multi-national, randomised, placebo-controlled, parallel study conducted at seven different centres, in Norway (n = five), Denmark (n = one) and Sweden (n = one) from 2004 to 2010. A flowchart for the study is provided in Figure 1. The trial was performed according to the guidelines for controlled trials of drugs in cluster headache (9) with one exception. In order to decrease the risk of spontaneous remissions before starting on medication, there was no medication-free baseline period. This was a recommendation from a previous large study on ECH (10).
Study flowchart.
Recruitment was performed at outpatient clinics and the diagnosis was verified by a neurologist. Inclusion criteria were ages 18–75 years, and a diagnosis of ECH was made according to the International Classification of Headache Disorders, second edition (ICHD-2) (1). In addition all patients should have had a period of at least one month with ECH previously, and no one should have had attacks for more than three weeks in the present cluster period before inclusion. Patients should also have been able to differentiate between CH attacks and other types of headache. Exclusion criteria were pregnancy, nursing or inability to use contraceptives in women; decreased hepatic or renal function; systolic blood pressure below 110 mmHg; hypersensitivity to active substance; previous history of angioneurotic oedema; use of daily cluster prophylactics in the four weeks prior to inclusion for lithium and two weeks for other medications; psychiatric illness preventing full participation in this study; heart condition; use of antihypertensives; use of specific attack treatment other than sumatriptan injections or oxygen; chronic CH; abuse of alcohol or narcotics; medication-overuse headache; and use of antipsychotic or antidepressive medication in the four weeks prior to inclusion.
Eligible patients underwent physical and neurological examinations. Medical history and demographics were obtained. Patients were instructed to keep headache diaries recording headache duration and severity, level of disability, presence of one or more autonomic symptoms (conjunctival injection, lacrimation, ptosis, miosis, eyelid oedema, nasal congestion, rhinorrhoea, forehead sweating), acute medication use and adverse events (free text).
After randomisation (computer-generated randomisation scheme), patients started immediately on either placebo (one tablet/day week 1 and two tablets/day weeks 2 and 3) or candesartan cilexetil 16 mg (one tablet/day) week 1 and 32 mg (two tablets/day) weeks 2 and 3. One week after randomisation all patients were contacted by phone, by the study nurse, to record side effects (checklist and open-ended questioning). After the medication period the participants continued to keep the diary for one week, using only acute medication. Active and placebo tablets, provided for the study by AstraZeneca, had the same size, weight, taste and appearance to ensure blinding. Patients were allowed to use sumatriptan injections or oxygen inhalation for acute attacks during the study. Randomisation and blinding were performed in accordance with the Consolidated Standards of Reporting Trials (CONSORT) statement (11).
Approvals
The study was approved by the regional ethics committees and National Medicines Agencies in all three countries. Written informed consent was obtained from all patients (EudraCT nr: 2004-002737-39, Clinical Trial Gov nr: NCT00184587).
Statistical analysis and efficacy variables
Considering an overall difference between the two treatment groups of 0.5 standard deviation (SD) of the mean of the primary efficacy variable to be a meaningful effect, and assuming a 15%–20% dropout rate, it was calculated that 64 patients would be needed to reach a power of 80% with a two-sided α of 0.05. Descriptive statistics are for continuous variables given as mean ± SD with minimum and maximum, or medians and quartiles, depending on whether symmetric distribution was present or not.
The primary efficacy variable was the number of attacks per week. Secondary efficacy variables included (full list at clinicaltrials.gov) days and hours with CH, duration of attacks, number of treatments with sumatriptan or oxygen, level of disability (0 = no disability, 1 = mild, 2 = moderate, 3 = severe, and 4 = unbearable), headache severity index (product of level of disability and duration of attacks), patient satisfaction with the treatment (scale from 1 to 10 with 1 = very poor effect and 10 = very good effect), analgesics use, autonomic symptoms, and responder rate. A responder was defined as a patient with a 50% or more reduction in attack frequency in week 3 compared to week 1. Treatment effect was evaluated using week 1 as “pseudo-baseline” (active group on candesartan 16 mg × 1) comparing it with week 3 (active group on candesartan 32 mg × 1). According to the protocol, differences in the primary efficacy variable were planned to be evaluated with non-parametric tests. However, when looking at the data before unblinding, it was evident that one should take into account the temporal properties in the attack pattern. The pre-planned statistical tests would not be sufficient (12), and due to the nature of ECH, spontaneous regression occurs and these patients do not continue to contribute data, making it more difficult to evaluate the real treatment effect. Hence, we decided to include post-hoc analyses with the exact Mantel-Haenzel test for common Poisson relative risk, and a time-to-event model. The Mantel-Haenzel is an exact test of the ratio of the rates (attack frequency over time) for the two treatment groups (13) stratified by centre, resulting in a more suitable description of clinical efficacy given the temporal properties of ECH, as stated above. The time-to-event model tested the probability for further attacks from the time until the patient ceased reporting headache episodes during the 28-day observation period, with randomisation group and centre as explanatory factors. A two-sided p < 0.05 was considered significant. Summary statistics and graphs were produced with SPSS 17.01 (SPSS Inc, IL, USA) and R 2.13.2 (Hornik (2011) ISBN 3-900051-08-9). Statistical estimates were obtained from SPSS 17.1 and SAS 9.2 (SAS Inst., Cary, NC, USA).
Results
Recruitment had to be stopped after five years, at which time point 40 patients had been randomised. Thirty-two of them returned fully evaluable reports (candesartan = 19; placebo = 13). The distribution (19 versus 13) is precisely within one SD and is an example that exact balance is not easy to achieve in multicentre studies of small volumes.
Baseline data.
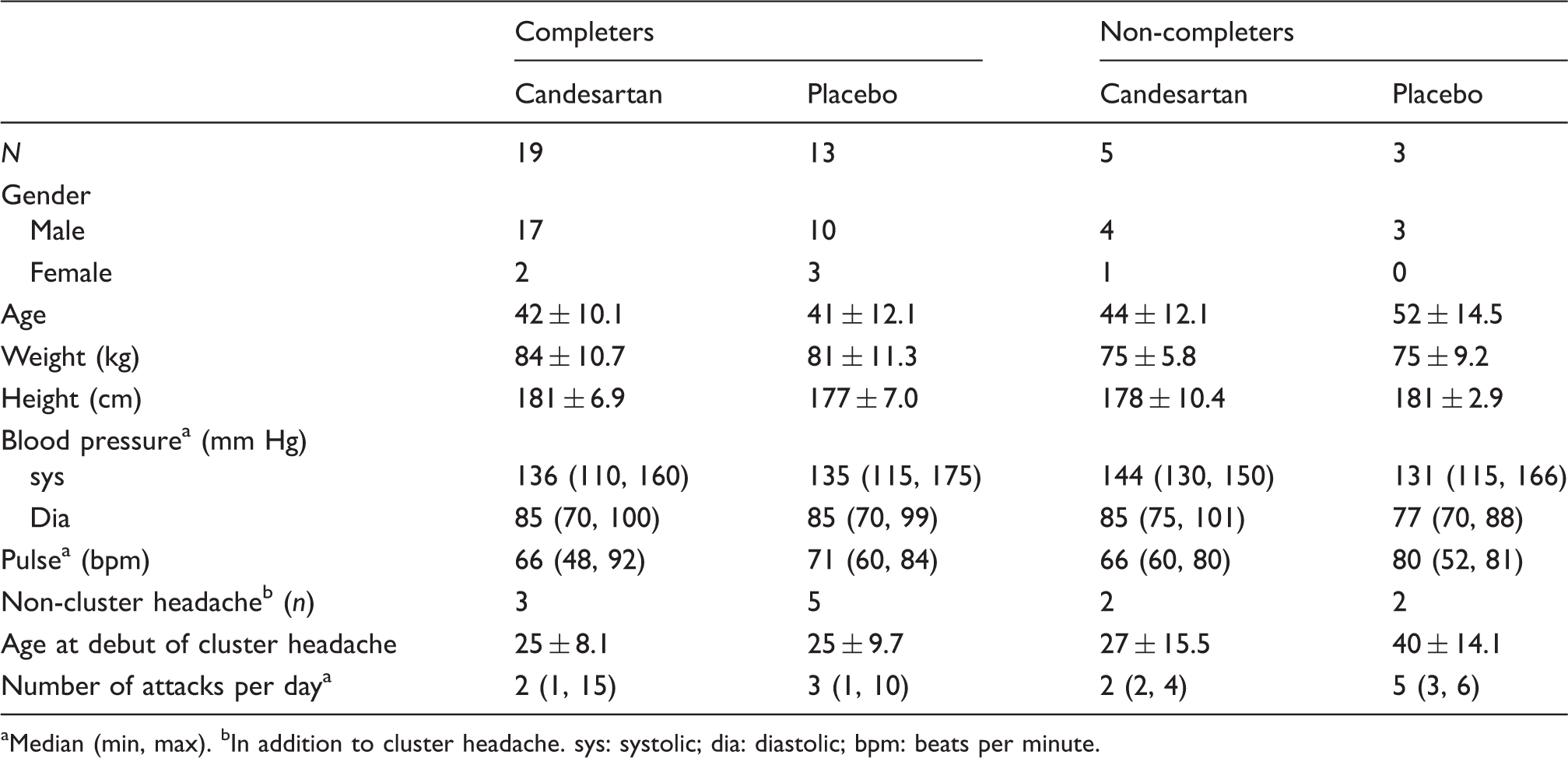
Median (min, max). bIn addition to cluster headache. sys: systolic; dia: diastolic; bpm: beats per minute.
Primary efficacy variable
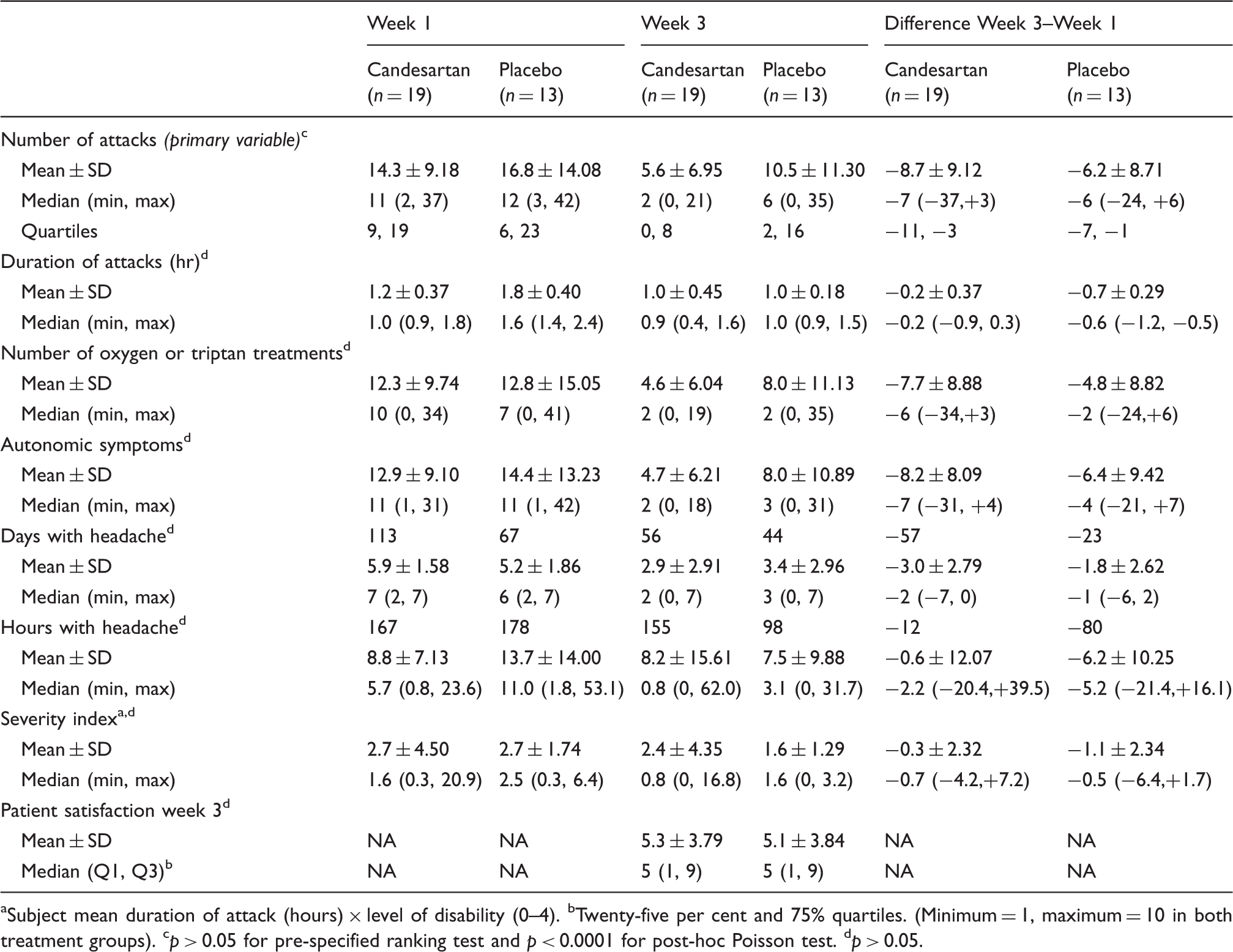
Summary statistics (based on group sizes) on number of attacks for week 1 and 3 are provided in Table 3. The difference in attack numbers between week 1 and 3, when tested with a non-parametric test (Wilcoxon rank-sum test), was not statistically significant (p = 0.38). However, while all patients reported daily attacks during the first study week, only a subsample experienced attacks during week 3. The number of patients still reporting attacks (Figure 2) and the number of attacks reported (Figure 3) are shown to decline over time in both groups, but more so in the candesartan group (attack frequency reduction for candesartan 61% and placebo 38%). To take these properties of the data fully into account, the pre-specified statistical analysis on number of attacks was supplemented with an exact Mantel-Haenzel test of common Poisson relative risk (RR) over the whole treatment period (21 days). With this analysis the difference between the groups was highly significant (RR = 1.3; 95% confidence interval (CI) = 1.17–1.48; p < 0.0001).
Number of patients experiencing cluster headache each day in the 21-day treatment period (candesartan 16 mg week 1, 32 mg weeks 2 and 3). Mean number of cluster attacks per day (candesartan 16 mg week 1, 32 mg weeks 2 and 3). Efficacy measures in study weeks 1 and 3. Subject mean duration of attack (hours) × level of disability (0–4). bTwenty-five per cent and 75% quartiles. (Minimum = 1, maximum = 10 in both treatment groups). cp > 0.05 for pre-specified ranking test and p < 0.0001 for post-hoc Poisson test. dp > 0.05.

Secondary efficacy variables
Data on secondary efficacy variables are provided in Table 3. Some of the variables described in the protocol were difficult to estimate without extensive assumptions outside the protocol. None of these were significant with the protocol-described statistical methods. Only the variables that could be assessed with a higher degree of certainty are provided in the paper. The rest of the data on outcomes (full list at clinicaltrials.gov) can be provided in full with information on limitations from the main author upon request. Numerically there is a larger reduction in the number of headache hours in the placebo group in week 3 compared to week 1 (not significant). The mean number of headache hours per patient is much lower for active substance (8.8 hours) than for placebo (13.7 hours). The percentage of patients with a 50% or more reduction in attack frequency in week 3 compared to week 1 was 63% for candesartan and 46% for placebo (p = 0.61, X2 test).
The number of patients continuing to report attacks declined in both groups from the second treatment week, but at a higher rate in the candesartan group (Figure 2). At the end of the treatment period, nine of the 19 patients (47%) in the candesartan group and nine of the 13 patients (69%) in the placebo group were still reporting headache attacks (p = 0.29; 95% CI = 0.07–2.16, Fisher’s exact test). A Cox proportional hazards regression (HR) analysis was fitted to estimate the risk of experiencing cluster headache attacks over time, using candesartan or placebo. A non-significant difference (HR = 0.48; 95% CI = 0.20–1.18; p = 0.10), favouring candesartan, was observed for the entire 28-day study period (Figure 4).
Days to freedom from headache.
Number (n) of patients with adverse reactions. a

A total of 14 patients reported adverse events (nine of 19 in the active group and five of 13 in the control group). Odds ratio (OR) 1.42; 95% confidence interval (CI) = 0.28–7.80, p = 0.73.
Discussion
In this first prospective, placebo-controlled, double-blind study on angiotensin receptor blockade in patients with CH, candesartan failed to reach statistical significance in the pre-specified statistical test, but was associated with a significant reduction in cluster attacks over the course of the study (p < 0.0001) and with a trend for shorter cluster period duration, when applying post-hoc statistical methods more appropriate for the studied variables.
Designing and performing studies on ECH is difficult (Table 1) because of both the rarity of the condition and the varying clinical expression among individuals within a cluster bout. However, it is nevertheless important to conduct studies on this condition as it is one of the worst pain conditions known, and with a significant impact on patients’ life (7).
The prophylactic treatment options available today are few and with limited efficacy, and only small studies (Table 1) are available with the exception of one relatively large study with a negative result. Most previous studies have reported effect as difference from baseline, defined as the period before start of the study drug, whereas we used the first week of drug treatment (half dose) as baseline. Since active treatment was given also during the baseline period, our calculations are more conservative than calculations using a placebo baseline. This has to be taken into account when conclusions on the trial are drawn. Figure 3 indicates that there is indeed a marked decline in attacks already in week 1 in the active group, not seen in the placebo group, and it may explain why the reduction in headache hours from weeks 1 to 3 was larger in the placebo group than in the candesartan group. The pre-planned statistics (week 1 vs. week 3) were therefore less suitable, and the post-hoc statistical method (13), which takes into account the development in attack frequency during the whole period from day 1, gives a better picture of the difference.
Lack of power may be another reason why the pre-specified analyses failed to show an impact of the active treatment. The study was calculated for 54 patients completing the trial period. However, despite a five-year recruitment period, we were able to include only 40 patients, and only 32 of them completed the study. Additional loss in power came from study group size imbalance, as 19 were randomised to candesartan and only 13 to placebo. Hence, the major limitations of the study were the lack of power and that the pre-planned statistical test was not optimal. Strengths of the study were the strict criteria for participation, inclusion of only ECH patients, and that it followed the guidelines for controlled trials of drugs in cluster headache (9). In addition it was a randomised, placebo-controlled, multicentre trial performed in three different countries.
The pathophysiological mechanism that could explain an effect of angiotensin II receptor blockers in CH is unknown. Several studies suggest that the posterior hypothalamus may play a part as a generator or modulator in trigeminal autonomic cephalalgias (14). The suprachiasmatic nuclei (SCN) of the hypothalamus are the main centres for the generation and regulation of circadian rhythms in mammals, and these are also thought to be important in the striking circadian pattern of CH attacks. SCN neurons synthesise a number of different neuropeptides, including angiotensin II (15). Circadian rhythms are generated in individual SCN neurons via transcription of so-called clock genes. In a peripheral tissue model, angiotensin II type 1 (AT1) receptor activation by angiotensin II increases expression of the clock gene Per2, suggesting that this neuropeptide can regulate the molecular clock in the SCN as well (16). Theoretically, the angiotensin II receptor blocker candesartan may therefore exert an effect on these areas and mechanisms thought to be important in the pathophysiology of CH.
Based on the experience we have gained in this trial, we have some comments on how future CH studies may be planned and analysed. An essential aspect of ECH is its episodic (periodic) nature. A time-to-remission design offers several advantages and is probably the most efficient trial model for this condition. This study provides such an analysis. The major drawback is that a simple time to disappearance of attacks does not account for impact on frequency or severity and it is less useful in chronic CH, for which disappearance of attacks may not be a realistic goal. Another important aspect of CH is that the attacks occur over a period of time; i.e. as counts over a unit, which is relevant for both ECH and chronic CH. This count will frequently be zero or missing, making use of classical non-parametric tests sub-optimal. Cases with missing data will be lost, giving those who fail to provide data with a high impact, and a high number of zero values results in many ties and loss of the non-parametric characteristics. To evaluate the attack rate in smaller CH trials, Poisson-based methods, which we used in the post-hoc analysis, are probably preferable.
In a previous study (10) the use of run-in periods was advised against in CH studies because the short duration of the periods may not allow for sufficiently long treatment periods. Use of a baseline period with active medication, as we used in this study, is clearly not advisable since medication may have an effect from early in the treatment period. One should therefore use a design not requiring a baseline period at all.
Recruiting enough ECH patients and difficulty with including them immediately after the start of a cluster period poses a challenge for the researcher as reflected in the small clinical trials available. We recommend that an international research consortium on CH be established so that future trials can be expected to have sufficient power and be executed during a shorter period.
Conclusion
This was a negative study, but lack of power and statistics less suitable to find true differences with temporal changes made it impossible to make decisive conclusions on the efficacy of candesartan in ECH prophylaxis. Post-hoc statistics suitable to describe the temporal changes in CH did, however, indicate a possible therapeutic effect, so future studies on candesartan, perhaps also including patients with chronic CH, may be justified.
Clinical implications
Candesartan was not significantly different from placebo in episodic cluster headache. Post-hoc statistics indicating a possible therapeutic effect and lack of study power may justify larger future studies.
Footnotes
Acknowledgement
The authors would like to acknowledge the contribution of study nurse, Gøril Gravdahl, for her work during this trial.
Funding
An unrestricted grant and study drugs were provided by AstraZeneca. The grant was provided to the Norwegian National Headache Centre and not to a single researcher. From the Norwegian National Headache Centre, each participating centre received 2000 NOK (275€) per patient included to cover expenses.
Conflict of interest
Sven-Olof Jansson is an employee of AstraZeneca. The other authors have nothing to declare.
