Abstract
Background
In a previous study we demonstrated that high-frequency oscillations (HFOs) elicited by median nerve stimulation are significantly correlated to clinical fluctuations of migraine. We aimed at verifying whether clinical fluctuations and HFO changes are correlated to N20 somatosensory evoked potential (SEP) recovery cycle, which is likely to reflect the functional refractoriness of primary somatosensory cortex neurons.
Methods
We analysed both HFOs and N20 SEP recovery cycle to paired stimulation in 21 migraine patients and 18 healthy volunteers.
Results
Shortened recovery cycle correlated with low-amplitude HFOs as well as with clinical worsening. By contrast, prolonged recovery cycle correlated with enhanced HFOs, as well as with spontaneous clinical improvement.
Conclusions
In our migraine patients the strict relationship between presynaptic HFO amplitude and N20 recovery function suggests that changes of both parameters might be caused by modifications of the thalamo-cortical drive. Our findings suggest that the thalamo-cortical drive during interictal stages could fluctuate from abnormally high to abnormally low levels, depending on mechanisms which reduce cortical excitability in spontaneously improving patients, and increase cortical excitability in spontaneously worsening ones.
Keywords
Introduction
Brain cortex in migraine patients is hyperresponsive between attacks to external stimulation. For instance, migraine patients show high susceptibility to light stimuli and acoustic noise (1,2). From a neurophysiological point of view, a possible correlate of these clinical findings is the habituation deficit, which represents a typical hallmark of migraine (3). During interictal stages, the cortical response evoked by repeated sensory stimuli does not change, whereas repeated stimulations elicit progressively smaller responses in the normal brain. In principle, two main mechanisms could account for the lack of habituation in migraine patients. Cortical sensory neurons may be hyperexcitable because of a lack of inhibitory inputs from γ-aminobutyric acid-releasing (GABAergic) cortical interneurons. Alternatively, the hyperexcitability of neurons in primary sensory cortices can be caused by their reduced refractoriness, in turn due to a reduction of the physiological thalamo-cortical drive. Earlier studies on habituation in migraine patients, based on the evaluation of cortical response to visual stimulation, seem to lend substance to the latter hypothesis. For instance, when the response to repeated blocks of stimuli is analysed, evoked potentials are characterised by the low amplitude of the first block of averagings (4). This finding, which tends to exclude that cortical sensory neurons are merely disinhibited, has been further confirmed by Bohotin et al. (5), who evaluated the amplitude of cortical visual-evoked response (VERs) after repetitive transcranial magnetic stimulation (rTMS) in migraineurs. After excitatory (10 Hz) rTMS, habituation became similar to that observed in healthy subjects, thus suggesting that habituation does not occur in migraine because the amplitude of the scalp response does not reach the threshold where habituation usually starts. Although the abovementioned findings might also be caused by paradoxical effects of rTMS in migraine patients (6), these data seem to confirm that cortical sensory neurons in migraineurs are hyperexcitable because of a deficitary thalamo-cortical drive, instead of reduced intracortical inhibition. According to this interpretation, which recalls the earlier “ceiling theory” (7), Coppola et al. (8) proposed using the term “hyperresponsivity” instead of hyperexcitability to better define interictal cortical changes in migraineurs.
The hypothesis that the excitability of primary somatosensory cortex neurons is increased in migraine because of a reduced thalamo-cortical drive has found possible further support from the analysis of 600 Hz high-frequency oscillations (HFOs) after upper limb electrical stimulation (9–11). Several studies have demonstrated that scalp-recorded HFOs are formed by two subsequent bursts, the former probably generated in thalamo-cortical radiations, and the latter generated postsynaptically by cortical neurons able to fire repetitively (12,13). It is still debated whether the activity of the postsynaptic burst is generated by excitatory glutamatergic neurones or by inhibitory GABAergic interneurones (14,15); by contrast, it is quite generally accepted that the activity of the earlier or “presynaptic” burst is linked to the excitatory thalamo-cortical drive, since it is significantly enhanced by single-dose administration of rivastigmine, an inhibitor of central acetylcholinesterase (14). Until now two different studies reported that the presynaptic thalamo-cortical burst is abnormally reduced during interictal periods in migraineurs (16,17), and, in one of them (17), also the postsynaptic burst was reduced. These results obviously suggest that the reduction of thalamo-cortical drive plays a major role in determining cortical excitability changes in migraine patients.
Cortical excitability in migraineurs has been also evaluated by the analysis of somatosensory evoked potential (SEP) recovery cycle (18,19). When SEPs are obtained by two paired electrical shocks at different interstimulus intervals (ISIs), the SEP amplitude evoked by the later stimulus is smaller than the one recorded from a single stimulus. The longer the ISI, the higher the amplitude of the SEPs recorded after the second stimulus, until full amplitude recovery is observed (20,21). Although the precise mechanisms by which the refractoriness of cortical neurons reflected by recovery cycle can vary among subjects are not yet completely understood, shortened cortical SEP recovery cycle is generally considered as a marker of cortical hyperexcitability. For instance, shortened recovery cycle has been previously demonstrated in cortical myoclonus and subacute spongiform panencephalitis (22,23). Valeriani et al. (18) found in a population of 15 children suffering from migraine without aura that the recovery of the N20 SEP amplitude took a shorter time in migraineurs than in healthy subjects, thus suggesting that the excitability of the SI area is increased in migraine.
In a previous study (24) we found that HFO reduction was correlated to spontaneous clinical worsening, whereas patients in spontaneous clinical improvement showed enhanced HFOs. To verify whether interictal HFO changes were caused by modifications of cortical excitability, in turn related to spontaneous clinical fluctuations of the disease, we evaluated not only HFO area under the curve (AUC), but also the recovery cycle of the N20 primary response to median nerve stimulation after paired stimulation at three different ISIs (5, 10 and 20 ms).
Methods
Subjects
Patients were recruited among the members of the staff of the Hospital “A. Gemelli”, Rome (Italy) and of the Scientific Institute “E. Medea”, Udine (Italy). Only patients who fulfilled diagnostic criteria for migraine with aura (MA) (International Classification of Headache Disorders, II (ICHD-II), Headache Classification Committee of the International Headache Society, 2004: code 1.2) and migraine without aura (MO) (ICHD-II: code 1.1) were included in the study. We therefore recorded 21 migraine patients, 17 of whom belonged to a group of patients previously examined (24): five MA (mean age: 34.8 years; five women) and 16 MO (mean age: 30.7 years; 12 women and four men). None of them were ever administered prophylactic treatments for migraine in their life. No patients were put on any prophylactic drug during the present study. They were compared with 18 healthy volunteers of comparable age (mean age 31.6 years, range 21–51) and gender distribution (12 women and six men) without personal or family history of migraine and devoid of any detectable medical condition. In all 21 migraine patients, SEP recording was performed at least three days before or after their last or their next migraine attack, on the basis of migraine diaries regularly checked by two authors (DR and SZ). All participants gave informed consent in accordance with the Declaration of Helsinki and the study was approved by the local ethics committee.
Clinical data.
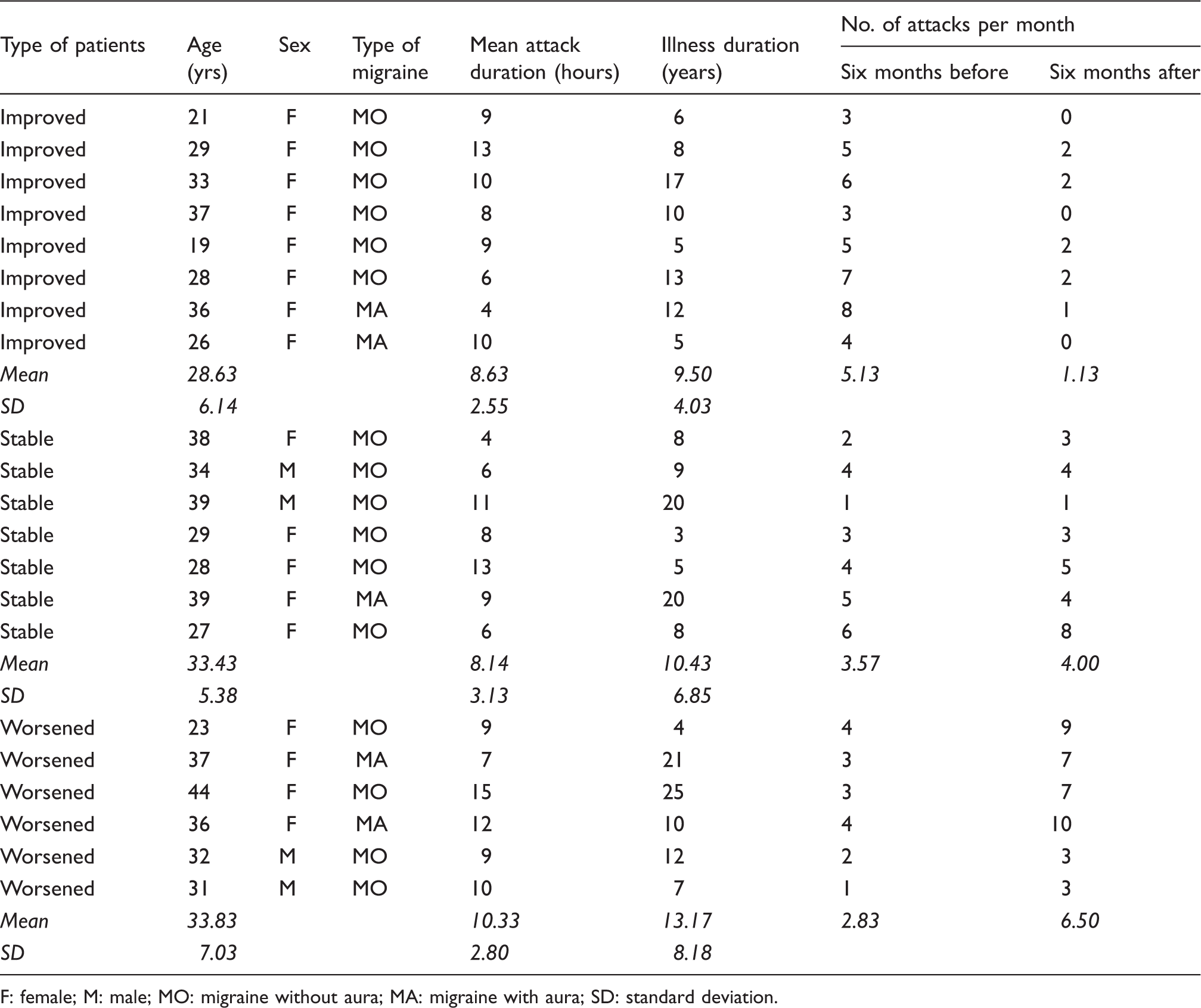
F: female; M: male; MO: migraine without aura; MA: migraine with aura; SD: standard deviation.
SEP recording
SEP recording was performed by using a commercially available five-channel Medelec™ Synergy apparatus (Viasys Health Care). SEPs were elicited on electrical stimulation of the right median nerve on the wrist by using a constant current square wave pulse (0.2 ms width, cathode proximal) and a stimulus intensity set just above the motor threshold. Active electrodes were placed over the contralateral parietal area (P3) referenced to F3; on the sixth cervical spinous process (Cv6) referenced to an electrode located immediately above the thyroid cartilage (Anterior Cervical, AC); and in the supraclavicular fossa (Erb’s point) referenced to Fz. Subjects were asked to lie with eyes closed on a couch in a warm and darkened room. Since high-frequency SEPs are largely influenced by drowsiness or sleep (15), they were asked to signal the occurrence of drowsiness, in order to temporarily stop the examination. However, this manoeuvre was never necessary. The analysis time was 50 ms, with a sampling rate of 20,000 Hz. The amplifier bandpass was 3–3000 Hz (12 dB roll off). An automatic artefact-rejection system excluded from the average all runs containing transients exceeding ±65 µV at any recording channel. In order to ensure baseline stabilisation, SEPs were filtered off-line by means of a digital filter with a bandpass of 10–3000 Hz. Each subject underwent two successive protocols, designed as follows: (a) four consecutive blocks of 500 sweeps were collected at a repetition rate of 5 Hz and then averaged. To analyse HFOs, each P3-to-F3 trace was digitally filtered with a bandpass of 400 to 800 Hz, by means of automated signal analysis software (Autosignal, version 1.6). (b) For investigating the SEP recovery cycle, we used a paired stimulation technique, similar to that of previous studies (18). SEP recording to a single stimulus was taken as control. Moreover, three further recordings were performed by using two paired stimuli at different ISIs (5, 10 and 20 ms). Each recording was obtained by the average of 1000 sweeps.
Data analysis
Data were loaded by technicians on a storage device as anonymous ASCII files and then analysed on separate computers. Data analysis was performed in a blinded manner by two of the authors of the study (DR and SZ); results were then compared. Data obtained by SEPs recorded from Erb’s point and cervical electrodes are not considered in the present study, since they are out of its main aim.
The amplitude of the primary N20 response on the P3-to-F3 traces was measured from the baseline. For evaluating HFO AUC, we used the same method previously described in a former paper (24). The beginning of the burst was detected approximately in correspondence with the N20 onset on the corresponding LF-SEP trace, measuring from the first wavelet having a 50% larger amplitude than the electrical noise to the burst ending at the last wavelet having a 50% larger amplitude than the electrical noise. The burst recorded at the P3-to-F3 array, consisting in a train of about five to eight wavelets, was further divided into two subsequent segments, respectively ending and beginning in correspondence with the N20 peak. The N20 peak latency has already been utilised as a cut-off point to measure subsequent components within the cortical burst (25). By considering that both bursts had an approximate duration of 5 ms, we calculated the electrical noise in a 5 ms interval (from 35 to 40 ms) where the signal was presumably not generated by neural activity. Since the early burst is thought to be generated in terminals of thalamo-cortical radiation fibres, whereas the late burst is probably generated by postsynaptic contributions from intracortical neurons (15,26,27), these bursts will be termed “presynaptic” and “postsynaptic” respectively throughout the rest of the paper. Burst amplitudes were measured as the area under the rectified waveform. AUC values were then corrected by subtracting the AUC of the electrical noise calculated within the 35–40 ms interval from presynaptic as well as postsynaptic burst areas.
In the double-stimulus traces, the responses following the second stimulation were obtained by subtracting the control SEP waveforms from the waveforms following each double stimulus. SEP amplitudes in the double stimulus conditions were normalised as the percentage of the corresponding SEP amplitude obtained after a single stimulus, which was assumed as 100%. Results of paired stimulation at each ISI were thus expressed by an index, according to the following formula: (N20 amplitude after paired stimulation/N20 amplitude to single stimuli) × 100.
MO versus MA
Clinical and neurophysiological data: MO patients versus MA patients.

HFO: high frequency oscillations; ISI: interstimulus interval; SD: standard deviation.
Statistical analysis
Comparison between migraineurs and control subjects
We first compared SEP data (N20 amplitude, AUC of both presynaptic and postsynaptic HFOs, recovery indexes at each ISI separately) between healthy volunteers and the whole population of migraine patients. Comparisons were performed by means of independent two-tailed t tests.
Correlation between recovery cycle and HFOs’ AUC
To verify whether the behaviour of N20 recovery and HFOs’ AUC were significantly related to each other, we performed correlation tests by means of a linear regression model between presynaptic and postsynaptic HFOs’ AUC and recovery indexes at each ISI. Comparisons were performed separately in healthy subjects and in migraine patients.
Clinical-neurophysiological correlations
To perform correlation tests between clinical fluctuations and neurophysiological data, clinical changes were expressed as the percentage changes of the number of migraine attacks, according to the following formula: 100–(number of attacks six months after the SEP recording/number of migraine attacks six months before the SEP recording) × 100. Clinical scoring was correlated to: 1) both presynaptic and postsynaptic burst AUC, separately; 2) recovery cycle (at each ISI separately). When significance level was reached, further comparisons were performed by means of one-way analyses of variance (ANOVAs). To perform ANOVAs between neurophysiological parameters and clinical scoring, patients were divided into three groups: 1) improving patients: the number of migraine attacks after the test was at least 50% smaller than before; 2) worsening patients: the number of migraine attacks increased by at least 50% after the test; 3) stable patients: no variations (frequency of migraine attacks ranging from less than 50% decrease and less than 50% increase). Such a classification was performed in analogy with therapeutic trials, in which response to drugs is commonly defined as at least a 50% reduction in average monthly migraine frequency (28). One-way ANOVA ((healthy, stable, improved, worsening) condition × burst AUC) was then performed. Post-hoc analysis was performed by means of a t test for independent samples with Bonferroni’s correction for multiple comparisons (p value set at 0.008).
Results
Comparison between migraineurs and control subjects
Statistical analysis failed to find significant differences in neurophysiological parameters between the whole population of migraine patients and healthy controls (N20 amplitude, migraineurs 1.18 ± 0.57, controls 1.02 ± 0.58, p = 0.40; presynaptic HFOs, migraineurs 0.23 ± 0.17, controls 0.2 ± 0.11, p = 0.48; postsynaptic HFOs, migraineurs 0.18 ± 0.18, controls 0.16 ± 0.12, p = 0.68; ISI 5, migraineurs 3.66 ± 15.54, controls 20.32 ± 18.52, p = 0.06; ISI 10, migraineurs 38.7 ± 24.6, controls 57.45 ± 34.9, p = 0.06; ISI 20, migraineurs 71.7 ± 57, controls 71.9 ± 17.4, p = 0.99).
Correlation between recovery cycle and HFOs’ AUC
In healthy subjects, correlation tests (by means of a linear regression model) failed to find significant correlation between AUC of the pre- and postsynaptic bursts and recovery indexes at 5, 10 and 20 ms ISIs. In particular, no significant correlation was found between presynaptic burst AUC and recovery index at 20 ms ISI (F = 0.03, r2 = 0.002, p = 0.86), and between postsynaptic burst AUC and recovery index at 20 ms ISI (F = 2.38, r2 = 0.13, p = 0.14). By contrast, in migraine patients we found significant correlation between presynaptic burst AUC and recovery index at 20 ms ISI (F = 9.778, r2 = 0.34, p = 0.005), and between postsynaptic burst AUC and recovery index at 20 ms ISI (F = 5.544, r2 = 0.226, p = 0.029).
No correlations were found between HFO AUC and recovery indexes at 5 and 10 ms ISIs in migraine patients. Figure 1 shows the correlation between HFO AUC and recovery index at 20 ms ISI in healthy subjects as well as in migraine patients. Due to the presence of an outlier, correlation tests were repeated after removal of this subject, with no substantial changes in terms of significance (presynaptic burst AUC and recovery index at 20 ms ISI: F = 20.68, r2 = 0.5347, p = 0.0002), and between postsynaptic burst AUC and recovery index at 20 ms ISI: F = 9.699, r2 = 0.3502, p = 0.006).
Correlation test (by means of a linear regression model) between HFO AUC and recovery index (N20 amplitude after paired stimulation at 20 ms ISI/N20 amplitude to single stimuli) × 100). The figure clearly shows the significant correlation between HFO AUC and recovery index in migraine patients (presynaptic HFOs: r2 = 0.34, p = 0.005. Postsynaptic HFOs: r2 = 0.226, p = 0.029).
Clinical-neurophysiological correlations
Correlation test (by means of a linear regression model) failed to find significant correlation between clinical fluctuations and recovery indexes at 5 (F = 0.60, r2 = 0.031, p = 0.44) and 10 ms ISI (F = 0.497, r2 = 0.02, p = 0.49). Conversely, we found significant correlation between clinical trend of migraine frequency and pre-synaptic burst AUC (F = 18.15, r2 = 0.488, p = 0.0004), between clinical trend of migraine frequency and post-synaptic burst AUC (F = 8.43, r2 = 0.307, p = 0.009), and between clinical trend of migraine frequency and recovery indexes at 20 ms ISI (F = 7.309, r2 = 0.278, p = 0.0014). Figure 2 clearly shows the correlation between frequency decline of migraine attacks and decrease of recovery index, as well as between frequency decline of migraine attacks and increase of HFO AUC.
Correlation test (by means of a linear regression model) between clinical trend of migraine frequency and neurophysiological data. On the left, the figure clearly shows the significant correlation (r2 = 0.278, p = 0.0014) between frequency decline of migraine attacks and decrease of recovery index at 20 ms ISI. In the middle, the figure shows the significant correlation (r2 = 0.488, p = 0.0004) between frequency decline of migraine attacks and increase of presynaptic HFO AUC. On the right, the figure shows the significant correlation (r2 = 0.307, p = 0.009) between frequency decline of migraine attacks and increase of postsynaptic HFO AUC.
We further analysed HFO AUC and recovery index at 20 ms ISI by comparing healthy subjects and patients. They were divided into three subgroups: eight patients showed a reduction of 50% or more of the number of migraine attacks during the six months following the test (average number of attacks before the test: 5.13/month; average number of attacks after the test: 1.13/month), and were thus classified as “improving”. Seven patients did not show changes in migraine attack frequency greater than 50% (average number of attacks before the test: 3.57/month; average number of attacks after the test: 4.0/month), and were therefore classified as “stable”. Lastly, six patients showed a 50% or higher increase of the number of migraine attacks during the six months following the test (average number of attacks before the test: 2.83/month; average number of attacks after the test: 6.5/month), and thus were classified as “worsening”. See Table 1 for analytical data.
As far as presynaptic burst AUC is concerned, ANOVA showed significant difference between the four groups (F3,35 = 8.23, p = 0.0003). Post-hoc analysis by means of independent samples t tests with Bonferroni’s correction for multiple comparisons (p value set at 0.008) showed that presynaptic burst AUC differed between control subjects and improving patients (p = 0.001; burst AUC: 0.20 ± 0.11 μV × ms in healthy controls, 0.38 ± 0.11 μV × ms in improving patients), and between improving and worsening patients (p = 0.0003; burst AUC: 0.1 ± 0.09 μV × ms in worsening patients, 0.38 ± 0.11 μV × ms in improving patients).
As far as postsynaptic burst AUC is concerned, ANOVA showed significant difference between the four groups (F3,35 = 4.34, p = 0.01). Post-hoc analysis by means of independent samples t tests with Bonferroni’s correction for multiple comparisons (p value set at 0.008) did not show any significant difference between groups. However, a strong trend to statistical difference was noticed between control subjects and improving patients (p = 0.03; burst AUC: 0.16 ± 0.12 μV × ms in healthy controls, 0.31 ± 0.22 μV × ms in improving patients), and between improving and worsening patients (p = 0.02; burst AUC: 0.06 ± 0.04 μV × ms in worsening patients, 0.31 ± 0.22 μV × ms in improving patients). Figure 3 shows the AUC of presynaptic as well as postsynaptic bursts in healthy subjects and migraine patients.
Comparison between migraine patients and control subjects. Histograms show the mean AUC of both pre- and postsynaptic HFOs in controls and migraine patients. Vertical bars represent standard deviations. Remarkably, improved patients show clearly enhanced HFOs, whereas HFO AUC is reduced in worsening ones. Post-hoc analysis showed a significant difference between the AUC of the presynaptic burst in healthy subjects and improving patients (p = 0.03) and in improving and worsening patients (p = 0.02; asterisks).
As far as recovery index at 20 ms ISI is concerned, ANOVA showed significant difference between the four groups (F3,35 = 4.71, p = 0.007). Post-hoc analysis by means of independent samples t tests with Bonferroni’s correction for multiple comparisons (p value set at 0.008) showed that recovery index differed between control subjects and improving patients (p = 0.001; recovery index: 71.93 ± 17.4 in healthy controls, 39.5 ± 26.28 in improving patients). No other post-hoc analyses reached significance level. Figure 4 shows the recovery indexes at different ISIs in healthy subjects and migraine patients.
Comparison between migraine patients and control subjects. Histograms show the recovery index (N20 amplitude after paired stimulation/N20 amplitude to single stimuli) × 100) at 5, 10 and 20 ms ISIs in controls and migraine patients. Vertical bars represent standard deviations. Remarkably, recovery index at 20 ms ISI is clearly reduced in improved patients whereas it is enhanced in worsening ones. Post-hoc analysis showed a significant difference between the recovery indexes in healthy subjects and improving patients (p = 0.001; asterisk).
Neurophysiological data in patients and healthy subjects.
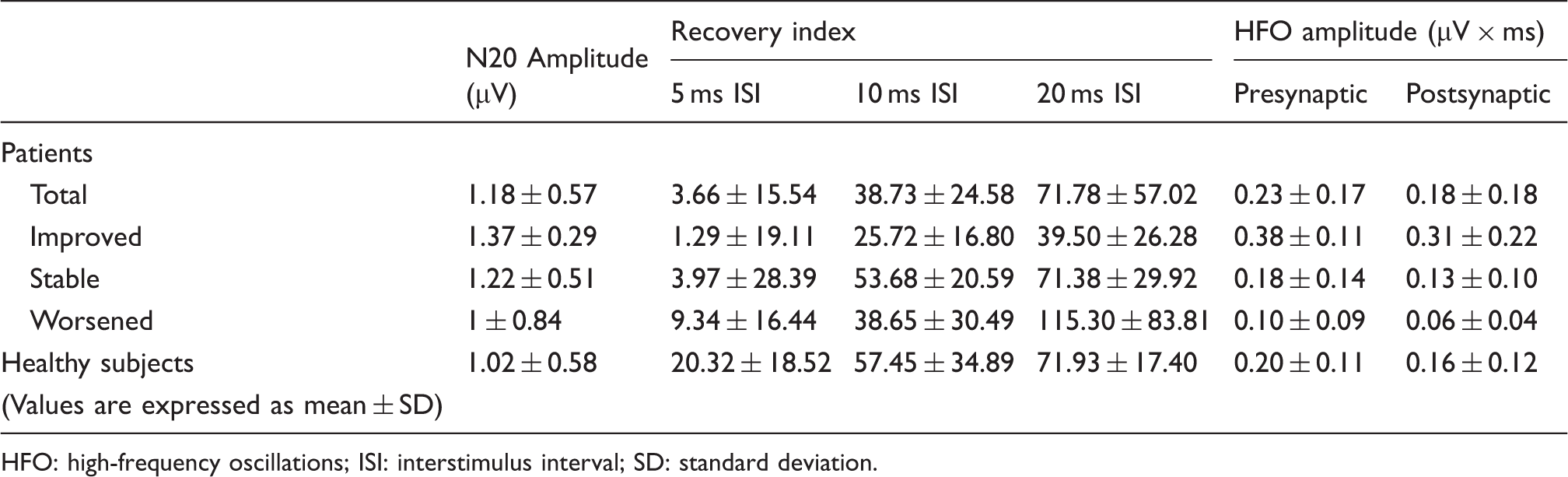
HFO: high-frequency oscillations; ISI: interstimulus interval; SD: standard deviation.
For illustrative purposes, grand averages of SEP traces in worsening and improving patients are shown in Figures 5 and 6. Before grand averaging, all traces were carried out coincident at the peak latency of the N20 potential. We performed this normalisation of latencies to avoid artificial ‘smoothing’ due to different latencies of the responses between the subjects. Figure 5 show grand-average traces obtained from total, improving and worsening patients after narrow bandpass filtering of 2000 stimuli, in order to selectively evaluate HFOs. Figure 6 compares traces obtained from single stimulation to that obtained after 20 ms ISI paired stimulation, in order to evaluate the strongly different recovery index between worsening and improving patients.
Grand average of SEPs obtained from 18 healthy volunteers (top), from all 21 migraine patients (middle), and from eight patients classified as spontaneously improving and from six patients classified as worsening (bottom). P3-to-F3 recordings, wide-bandpass standard SEPs showing the N20 response on the top, narrow-bandpass (400–800 Hz) recordings showing both pre- and postsynaptic HFOs on the bottom. HFOs are clearly enhanced in improving patients, whereas they are reduced in patients showing a worsening evolution of the migraine. No significant changes are observed between the whole population of patients and healthy subjects. Grand average of SEPs obtained from 18 healthy volunteers (top), from all 21 migraine patients (middle), and from eight patients classified as spontaneously improving and from six patients classified as worsening (bottom). P3-to-F3 recordings. SEPs were recorded after a single stimulus (baseline, red traces), and after two paired stimuli at 20 ms ISI. Black traces (paired stimulation) have been obtained by subtracting baseline recording from paired stimuli traces. To permit a direct comparison between N20 amplitudes, N20 peak latencies were carried out concurrently. Notice that N20 after paired stimulation is still suppressed in improving patients, whereas it is fully recovered in worsening ones. No significant changes are observed between the whole population of patients and healthy subjects.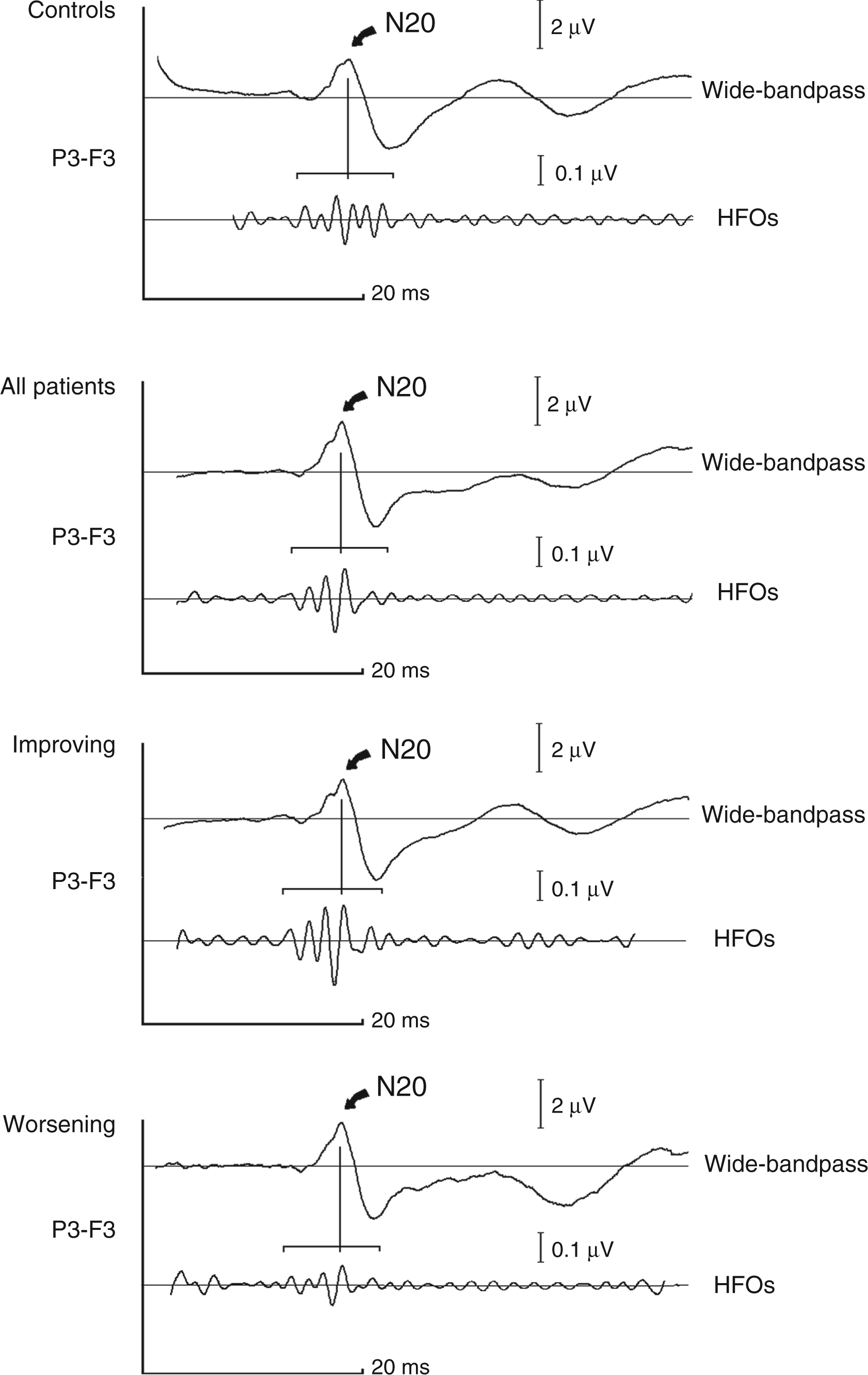

Discussion
The most relevant findings of our study can be summarised as follows: 1) in healthy subjects, the AUC of both presynaptic and postsynaptic HFOs does not show a significant correlation with the recovery cycle of the N20 primary response. By contrast, in migraine patients, regardless of their clinical condition, the AUC of both presynaptic and postsynaptic HFOs shows a significant correlation with the N20 recovery functions; 2) both HFO AUC and recovery cycle show significant correlation with clinical fluctuations occurring in the six months preceding and following the test; 3) both HFO AUC and recovery cycle do not show any significant difference between healthy subjects and migraine patients as a whole.
Correlation between HFOs and recovery cycle
Recovery cycle is commonly considered as a marker of cortical neurons refractoriness. In principle, refractoriness should reflect the neuronal excitability; for instance, shortened cortical SEP recovery cycle has been previously demonstrated in diseases which are characterised by central nervous system hyperexcitability, such as cortical myoclonus or subacute spongiform panencephalitis (22,23), or by disinhibition of the somatosensory system, such as dystonia (29). However, the precise mechanisms by which target neurons are less or more excitable during paired-pulse protocols are not yet completely understood. In theory, two main mechanisms could explain why cortical neurons can modify their refractoriness. First, hypoactivity of GABAergic intracortical circuitries can cause a disinhibition of neurons in primary sensory cortices; second, a reduction of thalamo-cortical drive can cause a reduced refractoriness of the same cells. As a matter of fact, paired-pulse protocols are commonly used to evaluate glutamate release probability in thalamo-cortical neurons, although in controlled conditions such as in vitro studies (30,31). Recent studies (32,33) demonstrated that recovery functions are independent of the number of synapses interposed between stimulus and the generator of the examined SEP component, since the N20 primary response needs a longer time for recovery than the subsequent P30 component. A number of factors, among which extracellular sodium concentrations, multiple generators for the same wave, interactions between different evoked responses, or jitter effects, have been claimed to explain this finding. However, these data reinforce the view that a complex interplay between inhibitory and excitatory inputs modulates the refractoriness of primary sensory neurons.
Looking at our present data, the recovery cycle showed a clear-cut correlation with HFO AUC in migraine patients, whereas the same finding was not noticed in healthy subjects. This finding is clearly explained by the low dispersion of the recovery index in healthy subjects, which tends to minimize any significant correlation with HFO AUC. By contrast, migraine patients can show extremely shortened and extremely prolonged recovery cycles (Figure 1).
Taking into account that presynaptic HFOs are probably generated by glutamatergic thalamo-cortical neurons since they are significantly enhanced by single-dose administration of central acetylcholinesterase inhibitors (14), the strong correlation between shortened recovery cycles, expressed by larger recovery index, and reduced presynaptic HFOs (or, alternatively, between prolonged recovery cycles and enhanced presynaptic HFOs) might suggest that, differently from healthy subjects, modifications of the glutamatergic thalamo-cortical drive play a major role in determining cortical neuron refractoriness in our migraine patients.
It is important to highlight that “prolonged” recovery cycle is parallel to a “smaller” recovery index, meaning “enhanced refractoriness”; and a “shortened” recovery cycle is parallel to a “larger” recovery index, indicating “reduced refractoriness”.
Abnormalities of the recovery cycle have been demonstrated in the whole central nervous system, including also the spinal cord (18). As clearly stated above, abnormal shortening of the recovery cycle can be in theory due either to insufficient inhibitory mechanisms, or to reduced excitatory drive to target cells. These two mechanisms are not mutually exclusive; as a matter of fact, it is possible that insufficient inhibitory drive could account for abnormal excitability in other parts of the nervous system. On the other hand, in our migraine patients reduced thalamic HFOs correlated with shortened N20 recovery cycle, expressed by larger recovery indexes, while increased thalamic HFOs correlated with prolonged N20 recovery cycle, expressed by smaller recovery indexes. Taking into account that presynaptic HFOs clearly reflect thalamo-cortical drive, it is conceivable that a reduction of this drive plays a major role in determining the shortening of the N20 recovery cycle and vice versa.
As far as postsynaptic HFOs are concerned, in this study its behaviour parallels that of presynaptic HFOs. Intuitively, assuming that presynaptic and postsynaptic burst are consecutively generated in the same neural pathway, the notion that presynaptic HFO reduction can entail a decrease of postsynaptic HFOs while presynaptic HFO increase could be paralleled by an analogous increase of postsynaptic ones is not surprising. However, in our previous study (24) and in the study by Coppola et al. (16), significant changes of presynaptic HFOs were not paralleled by analogous changes of postsynaptic ones. This finding, however, could be explained by technical reasons. Postsynaptic HFO AUC is physiologically lower than that of presynaptic ones. This low amplitude obviously entails a worse signal-to-noise ratio, so that, in relatively small samples, slight changes of postsynaptic HFO AUC can be underestimated.
As far as the generators of postsynaptic HFOs are concerned, our present data cannot confirm the hypothesis of Hashimoto et al. (15) on their origin from cortical GABAergic interneurons, since recovery cycle does not directly evaluate cortical GABAergic circuitries. Our present data, however, warrant some general considerations. In the present study, we postulate that the reduced glutamatergic release from thalamo-cortical neurons entails a reduction of the refractoriness of target cells, whereas increased glutamatergic release from thalamo-cortical neurons increases the refractoriness of the same target neurons. Looking at the strong correlation between HFO amplitudes and N20 recovery cycle in migraine patients, this finding reinforces the view that the net result of the HFO activity is represented by cortical inhibition. A consistent bulk of data suggests that HFOs reflect the activity of a system by which arousal-related structures modulate the somatosensory input processing (34,35). Taking into account the role played by lateral inhibition in sharpening the cortical response to sensory stimulation (36), it is conceivable that, as a whole, the HFO system is devoted to enhance the efficacy of the somatosensory input processing.
Clinical-neurophysiological correlations
In the present study, spontaneously improving patients show prolonged recovery cycle, expressed by smaller recovery indexes, whereas worsening patients show abnormally shortened recovery cycle, expressed by larger recovery indexes. Our present data therefore suggest that primary somatosensory cortex neurons which receive glutamatergic input from thalamo-cortical cells are hyperexcitable in spontaneously worsening patients and hypoexcitable in spontaneously improving ones. Some earlier data in the literature, however, make this finding not entirely new. Migraine is now conceptually considered as a chronic disorder with episodic attacks. Patients often move from a stage in which they have low-frequency episodic headaches to a high-frequency stage and eventually to chronic migraine, but spontaneous remissions are not uncommon (37). However, the reasons why the attack frequency can spontaneously change are far from being completely understood. Although the mechanisms underlying the transformation of migraine from episodic to chronic are obviously different from those causing temporary worsening, data recently obtained in chronic migraineurs might be helpful in understanding our present findings. Aurora et al. (38), using transcranial magnetic stimulation, demonstrated that magnetic suppression of perceptual accuracy, an index of intracortical inhibition mechanisms, was significantly diminished in 25 patients with chronic migraine compared with patients with episodic migraine and control subjects. Positron-emission tomography (PET) scan studies performed in some of their chronic migraine patients suggested that cortical excitability is raised in these patients. Aurora et al. (38) thus concluded that high cortical excitability may cause an abnormal susceptibility to migraine triggers in chronic migraineurs, thus explaining the high frequency of migraine attacks. Therefore, it is possible that increased cortical excitability can play a significant role in migraine worsening. The net inhibitory effect of HFO activity on the cortex is reduced in worsening patients, but forcedly enhanced in spontaneously improving ones. As a matter of fact, a similar increase of HFOs’ amplitude has been noticed in patients with remitted idiopathic generalised epilepsy (39).
Brainstem dysfunction
Looking at our present data, changes of the thalamo-cortical drive seem to play a pivotal role in determining presynaptic HFO reduction and shortened recovery cycle, expressed by larger recovery index, in worsening migraine patients, as well as presynaptic HFO increase and prolonged recovery cycle, expressed by smaller recovery index, in improving ones. Interestingly, Torres-Escalante et al. (40) recently demonstrated that presynaptic inhibition of glutamatergic thalamo-cortical neurons is regulated by a complex interaction between serotonergic and GABAergic receptors. The activation of serotonergic receptors, in fact, inhibits glutamate release; however, when the effect of GABAB receptor activation is maximal, the influence of serotonin changes to the opposite direction, increasing glutamate release through activation of 5-HT2 receptors. A considerable bulk of data (8,41–43) suggests that serotonergic disposition reaches its minima during preictal stages and increases during the attacks. It is thus conceivable that in our worsening patients serotonergic disposition tends to progressively decrease. This progressively lower serotonergic disposition could determine reduced thalamo-cortical activity, which in turn decreases both the refractory period of neurons in the somatosensory cortex and the amplitude of presynaptic HFOs.
HFOs and recovery cycle in migraine patients as a whole
In the present study, neither N20 recovery cycle nor the AUC of both presynaptic and postsynaptic HFOs show significant differences between healthy subjects and the whole population of migraine patients. Thus far, two earlier studies reported HFO reduction in migraineurs compared with normal subjects (16,17), while more recently Lai et al. (44) reported that presynaptic HFOs are enhanced. Regarding SEP recovery cycle, earlier studies have been performed in children and agree in concluding that recovery cycle is abnormally shortened in children with migraine. Several reasons could account for these discrepancies with our own data. First of all, small differences in recording technique or the use of small samples can lead to an underestimation of slight intergroup differences. However, in our opinion, the most reasonable reason for these discrepancies is related to a theoretical problem. Our findings strongly suggest that fluctuations in serotonergic disposition are the main cause of the neurophysiological changes we observed in migraine patients. Although it seems firmly established that serotonergic disposition is decreased in preictal stages and is normalised during attacks, little is known about its behaviour during spontaneous, progressive worsening or improvement of the disease. On the basis of our present data, we postulate that serotonergic disposition is progressively reduced during spontaneous worsening, and progressively increased during spontaneous improving stages of the disease. Although simplistic and somewhat arbitrary, the clinical subdivision in worsening and improving patients takes into account this possibility. As a matter of fact, in two previous HFO studies, patients were recruited from specialised headache clinics. Since patients with long-term spontaneous improvement are sometimes lost in regular clinical follow-up in headache clinics, it is possible that most of them in these two studies were in a worsening stage of the disease. As far as studies using SEP recovery cycle are concerned, it is also possible that this discrepancy was due to differences between the physiopathology of migraine between children and adult patients; this possibility, however, should be verified in further studies.
Conclusions and limitations of the study
The main limitation of this study is represented by the small subject number after categorisation by clinical fluctuation. Moreover, as stated above, such a categorisation is probably simplistic and somewhat arbitrary. On the other hand, our data confirm that reduced preactivation level of sensory cortices due to inefficient thalamo-cortical drive, rather than a mere change in cortical excitability, plays a major role in the pathophysiology of migraine (8). Moreover, in this study we demonstrated that spontaneous clinical fluctuations of migraine can be assessed by means of simple neurophysiological techniques. For instance, both N20 recovery functions (19) and habituation (45) became normal after effective treatment in migraine patients. In this view, neurophysiological follow-up of migraine patients could be a useful tool to objectively assess the evolution of the disease and to decide the need for prophylactic treatment.
Clinical implications
Our data confirm that a reduced preactivation level of sensory cortices due to inefficient thalamo-cortical drive plays a major role in the pathophysiology of migraine. Spontaneous clinical fluctuations of migraine can be assessed by means of simple neurophysiological techniques. Neurophysiological follow-up of migraine patients could be a useful tool to objectively assess the evolution of the disease and to decide the need for prophylactic treatment.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
