Abstract
Background: In previous studies we found that high-frequency somatosensory oscillations (HFOs) reflecting thalamo-cortical activation were decreased in migraineurs between attacks and that high-frequency repetitive transcranial magnetic stimulation (rTMS) was able to normalize the habituation deficit of visual evoked potentials (VEPs). Here we study the effects of activating (10 Hz) or inhibiting (1 Hz) rTMS on conventional low-frequency (LF) and high-frequency somatosensory evoked potentials (SSEPs).
Subjects and methods: rTMS was applied on the motor cortex of 13 healthy volunteers (HVs) and 13 migraine without aura (MO) patients. We measured N20-P25 LF-SSEP amplitude and habituation, and maximal peak-to-peak amplitude of early and late HFOs before and after rTMS.
Results: In HVs, 1 Hz rTMS significantly reduced the amplitude of the first LF-SSEP block and its habituation. In MO patients, 10 Hz rTMS increased the amplitude of the first block and induced habituation. Ten Hz rTMS produced an increase of late HFO in both groups, but more interestingly, in MO patients also significantly increased the early HFOs, which are reduced at baseline compared to those of HVs.
Conclusions: These data confirm for SSEP that excitatory rTMS can normalize habituation in migraine patients and show that this is accompanied by early an HFO increase, which is thought to reflect thalamo-cortical activity. Taken together with similar effects we observed for VEPs, this finding supports the hypothesis that dysfunctioning thalamo-cortical loops may be responsible for the interictal habituation deficit in migraine.
Keywords
Introduction
Migraine patients are characterized interictally by a habituation deficit of evoked cortical potentials during continuous stimulus repetition (1,2). The exact neurobiological mechanisms underlying this abnormality remain speculative. Two main hypotheses are under debate. Some studies suggest that increased cortical excitability (3), possibly due to deficient intracortical inhibition (4,5), might be the culprit. There is, on the other hand, converging evidence from evoked potential studies that reduced pre-activation level of sensory cortices could be responsible for the habituation deficit (2). The pre-activation excitability level of sensory cortices is set by thalamo-cortical loops that are under aminergic, in particular serotonergic and noradrenergic, control from the brain stem (6,7).
One method to explore thalamo-cortical afferents and cortical activation at the same time is to record high-frequency oscillations (HFOs) embedded in the common somatosensory evoked potentials (SSEPs). Two bursts of HFOs have been identified in bipolar scalp recordings in humans: an early component thought to be generated by pre-synaptic thalamo-cortical afferents, and a late component reflecting post-synaptic inhibitory fast-spiking neurons in parietal area 3b (8–14). Low amplitude of early HFOs of SSEPs was found between attacks in two studies (15,16), supporting the hypothesis that the habituation deficit in migraineurs is due to a reduced pre-activation level of sensory cortices. In one study, SSEP HFOs were increased in migraine patients (17).
Repetitive transcranial magnetic stimulation (rTMS) allows the transient activation of the underlying cortex with high-frequency (HF) stimulation (≥5 Hz) or to reduce its excitability at low-frequency (LF) stimulation (≤1 Hz). We have shown that 10 Hz rTMS over the occipital cortex is able to normalize the interictally reduced habituation of visual evoked potentials (VEPs) in migraineurs (18,19). The effect of rTMS on SSEPs and HFOs has not been studied yet. Such studies might shed light on the neuronal mechanisms of the habituation deficit found in migraine. While the primary visual areas can be localized by the production of magnetophosphenes, magnetic stimulation of the somatosensory cortex does not induce paresthesias and is more difficult to stimulate selectively. We chose therefore to target the adjacent motor cortex, where TMS produces a motor response and reciprocal interactions with the somatosensory cortex are well documented.
We decided to explore simultaneously the effect of rTMS on amplitude and habituation of conventional wide-band SSEPs, and on SSEP HFOs. We hypothesized that 10 Hz rTMS would revert deficient SSEP habituation in migraineurs between attacks by increasing HFOs and, therefore, thalamo-cortical activity.
Material and methods
Subjects
Thirteen migraine without aura (MO) patients were recorded (International Classification of Headache Diseases [ICHD]-II code 1.1; mean age: 35.6 ± 11.7; 8 women and 5 men). They were compared to 13 healthy volunteers (HVs) of comparable age and gender distribution (mean age: 33.8 ± 7.4; 9 women and 4 men) without personal or familial history of migraine and devoid of any detectable medical condition. Subjects taking any medication on a daily basis except for the contraceptive pill and subjects having a personal or a family history of epilepsy were excluded. To avoid sex-hormone influences on cortical excitability (20), we studied women during the follicular phase (days 8 to 10 from the first day of menses).
We recruited patients who had between one and four attacks per month (mean: 2.6 ± 0.9) and included their data only if they had an interval of at least three days between the recording and their last or their next (verified by a telephone call) migraine attack.
All of the participants were naive for TMS and neurophysiological investigations, and signed informed consent in accordance with the Declaration of Helsinki. The study was approved by the Ethics Committee of the Faculty of Medicine, University of Liège.
Recordings of somatosensory evoked potentials
SSEPs were elicited after electrical stimulation of the right median nerve at the wrist using a constant-current square wave pulse (0.1 ms width, cathode proximal) and a stimulus intensity set at two times the motor threshold. For each recording session, two series of 500 sweeps, before and after rTMS, were collected at a stimulation rate of 4.4 Hz and then averaged. The active electrodes were placed over the contralateral parietal area (C3’, 2 cm posterior to C3 in the international 10-20 system) and on the fifth cervical spinous process (Cv5), both referenced to frontal central zero (Fz); the ground electrode was on the right arm. The evoked potential signals were amplified with CED™ 1902 pre-amplifiers and recorded with a CED™ 1401 device (Cambridge Electronic Design Ltd., Cambridge, UK). Subjects were seated in a comfortable chair in an illuminated room with eyes open and asked to fix their attention on the wrist movement. Fifty milliseconds of the post-stimulus period were sampled at 5000 Hz to collect a wide range of frequencies. All recordings were averaged offline using the Signal™ software package version 3.10 (Cambridge Electronic Design Ltd).
Transcranial magnetic stimulation
We used the Magstim™ Rapid magnetic stimulator (Magstim Co. Ltd., Whitland, Dyfed, UK), connected to a double 7.0 cm figure-of-eight-shaped coil, with a maximal stimulator output of 1.2 Tesla. The coil was positioned over the left motor area. We first identified the motor threshold (MT), using single TMS pulses and the same procedure as previously described (21). For rTMS we used two different stimulation frequencies: 1 Hz (LF-rTMS) without interruption for 15 minutes and 10 Hz (HF-rTMS) partitioned in 18 trains of five seconds with inter-train intervals of 10 seconds. For each frequency, 900 pulses were thus delivered. When LF-rTMS was performed, stimulus intensity was set to 120% of the MT, whereas 90% of the MT was used with HF-rTMS.
Data analysis
SSEPs were recorded before and immediately after 1 or 10 Hz rTMS. The two rTMS sessions were performed in random order at ≥1 week intervals by the same investigators (G.C. and V.DP.). All recordings were numbered anonymously and analyzed blindly offline (see below) by one investigator (G.C.).
Broadband somatosensory evoked potentials
For broadband unfiltered SSEPs (0–2500 Hz) 500 artefact-free evoked responses recorded in each subject were averaged (“grand average”). The various SEP components (N13, N20, P25 and N33) were identified according to their respective latencies. We measured peak-to-peak amplitudes of the cervical N13 component (recorded under the active Cv5 electrode), and the cortical N20-P25 and P25-N33 components (recorded under the active C3′ scalp electrode).
Thereafter, in order to analyze habituation, the first 200 evoked responses were partitioned in two sequential blocks of 100 responses, since a clear-cut lack of habituation from the second block of stimulation onward was observed previously in migraineurs (22,23). Thereafter, each block was averaged offline (“block averages”) and analyzed for N20-P25 amplitudes. Habituation was defined as the percentage change in N20-P25 amplitude between the two blocks.
High-frequency oscillations
According to the method described elsewhere (16), digital zero-phase shift band-pass filtering between 450 and 750 Hz (Barlett-Hanning window, 51 filters’ coefficients) was applied offline in order to extract the HFOs embedded in the N20 SSEP component. In the majority of recorded traces, we were able to identify two separate bursts of HFOs: the first or early burst occurred in the latency interval of the ascending slope of the conventional N20 component and the second or late burst occurred in the time interval of the descending slope of N20, sometimes extending into the ascending slope of the N33 peak. In general the frequency of oscillations was higher in the first than in the second HFO burst, and in between the early and late bursts there was a clear frequency and amplitude decrease, which allowed the separation of the two bursts. In recordings in which a clear distinction between the two components was not possible, we considered as early burst those HFOs occurring before the N20 peak and as late burst those after the N20 peak.
After elimination of the stimulus artefact, we measured the latency of the negative oscillatory maximum and the maximum peak-to-peak amplitude. All measurements were performed separately on the two HFO bursts.
Statistical analysis
We used the Statistical Package for the Social Sciences (SPSS) (SPSS Inc., Chicago, IL, USA) for Windows, version 15.0 for all analyses. A one-way analysis of variance (ANOVA) test was used to compare the variables at baseline. Paired student's t-tests were used to compare the variables before and after rTMS at 1 or 10 Hz, for each component and for each group. Moreover, we performed a multivariate analysis of variance taking as a within-subject factor “block” (for wide-band SSEP) or HFOs (early or late) and as between-subject factors “group” (HVs, MO patients), “time” (before, after rTMS), and “session” (1 Hz, 10 Hz), in order to verify the interaction between them. p < 0.05 was considered significant.
Results
All SSEP components, both for broadband recordings and HFO, were clearly identified in all subjects. The mean TMS motor threshold to activate the first dorsal interosseous muscle at rest was not significantly different between subject groups: 56.5 ± 9.4% for MO patients, and 56.7 ± 7.9% for HVs.
Broadband somatosensory evoked potentials
Grand average SSEP recordings after electrical median nerve stimulation had similar latencies and amplitudes in both groups [for each measure F(1,24), p > 0.05]. No significant changes in SSEP latency or amplitude were detected after rTMS.
By contrast, repetitive ANOVA for the amplitude of SSEP block averages disclosed a main effect for factors group [F(1,98) = 23.85, p < 0.001], block [F(1,98) = 4.51, p = 0.036], and a significant interaction of group by block [F(1,98) = 8.97, p = 0.003], block by group and time [F(1,98) = 5.87, p = 0.017] and block by session and time [F(1,98) = 16.62, p < 0.001].
Before rTMS, SSEP N20-P25 amplitude was lower in the second block than in the first block in HVs, which reflects habituation (−14.2% before 1 Hz, −6.1% before 10 Hz rTMS), while there was no habituation in MO patients (+3.0% before 1 Hz, +12.2% before 10 Hz rTMS). This difference in habituation profile at baseline between HVs and MO patients was significant in both recording sessions [F(1,24) = 8.98, p = 0.006 before 1 Hz and F(1,24) = 12.26, p = 0.002 before 10 Hz rTMS] (Figure 1).
Amplitude of the broadband SSEP N20-P25 component (mean ± SEM) in two successive blocks of 100 averaged responses before and after 1 Hz or 10 Hz repetitive transcranial magnetic stimulation (rTMS). HV: healthy volunteers (N = 13); MO: migraine without aura patients (N = 13); SSEP: somatosensory evoked potential; SEM: standard error of the mean.
rTMS profoundly modified both SSEP amplitude and habituation. LF-rTMS reduced N20-P25 amplitude in block 1 (t = 2.131, p = 0.05) and induced a habituation deficit in HVs (+4.7% t = 4.057, p = 0.002). It had no significant effect on habituation in MO patients (+6.6%, t = 0.544, p = 0.596). On the contrary, after 10 Hz rTMS in migraineurs block 1 amplitude was significantly increased (t = 3.619, p = 0.003) and the baseline deficient habituation was replaced by normal habituation (−10.0%, t = 2.357, p = 0.036). In HVs there was no significant effect of 10 Hz rTMS, either on block 1 amplitude (t = −0.36, p = 0.725) or on habituation (−1.2%, t = 0.922, p = 0.375) (Figure 1).
High-frequency oscillation
Latency of the negative oscillatory maximum for both early and late HFO bursts was similar between the subject groups.
Repetitive ANOVA for early burst of HFO disclosed a main effect for factors group [F(1,48) = 9.63, p = 0.003] and a significant interaction of group by session and time [F(1,48) = 3.50, p = 0.03], but not of group by time [F(1,48) = 3.02, p = 0.08], or time by session [F(1,48) = 0.350, p = 0.552].
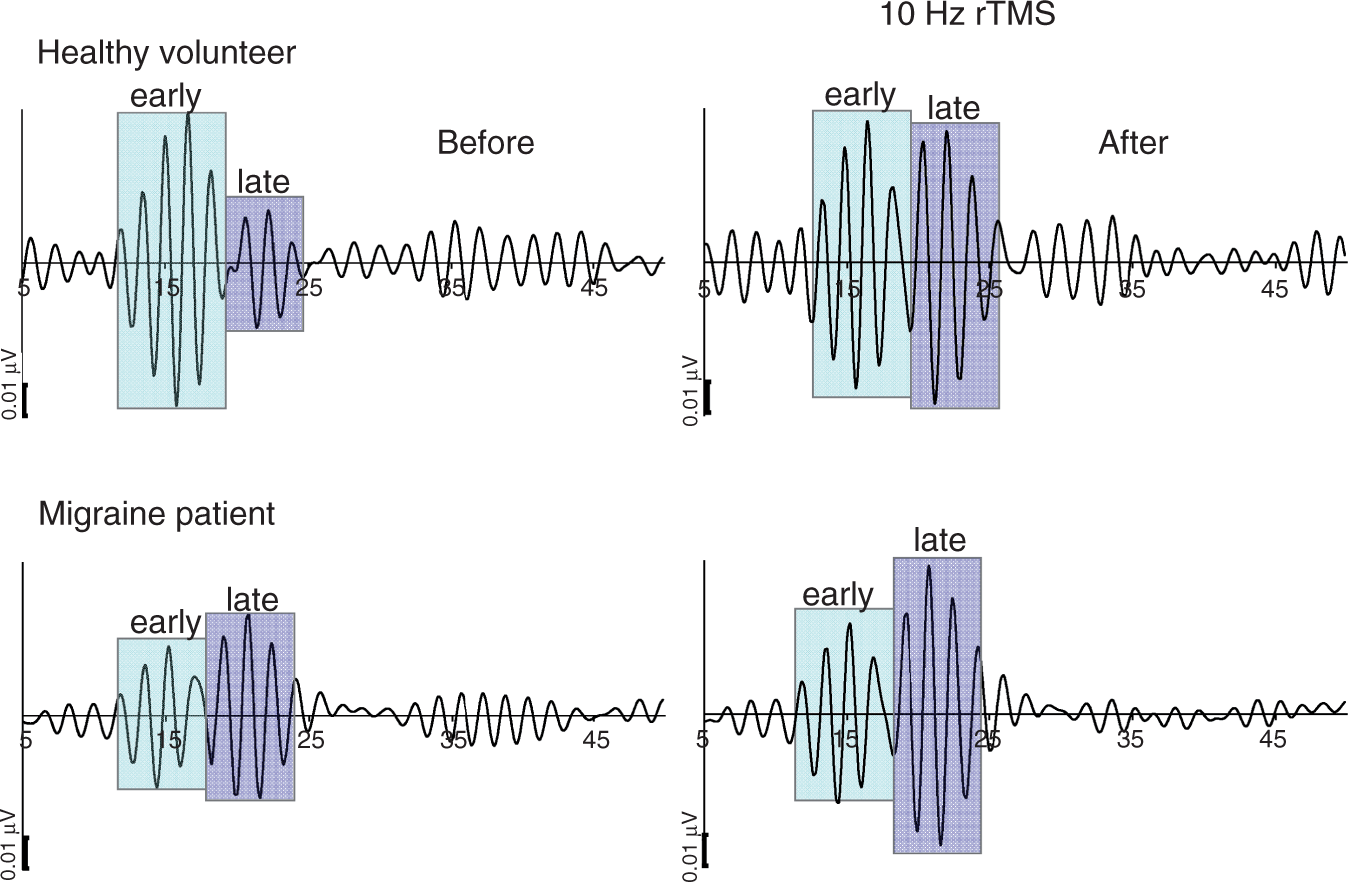
Post-hoc analysis revealed that, before rTMS, maximum peak-to-peak amplitude of the early burst of HFO was significantly smaller in MO patients than in HVs [F(1,24) = 5.276, p = 0.031 before the 1 Hz session and F(1,24) = 6.606, p = 0.017 before the 10 Hz session]. By contrast, there was no significant difference between HVs and MO patients in amplitude of the late HFO burst (Figures 2,3 and 4).
Illustrative traces of SSEP HFOs in a healthy volunteer and a migraine without aura patient before and after 1 Hz rTMS. Note the reduced amplitude of the early but not of the late HFO burst in the migraine patient, and the absence of an rTMS effect. SSEP: somatosensory evoked potential; HFOs: high-frequency oscillations; rTMS: repetitive transcranial magnetic stimulation. Illustrative traces of SSEP HFOs in a healthy volunteer and a migraine without aura patient before and after 10 Hz rTMS. Note the rTMS-induced increase of the early HFO burst in the migraine patient and the increase of the late HFO burst in both subjects. SSEP: somatosensory evoked potential; HFOs: high-frequency oscillations; rTMS: repetitive transcranial magnetic stimulation. Changes in amplitude (mean ± SEM) of early and late HFO bursts in healthy volunteers (HV) and migraine without aura (MO) patients before and after 1 Hz or 10 Hz rTMS of the motor cortex. SEM: standard error of the mean; HFO: high-frequency oscillation; rTMS: repetitive transcranial magnetic stimulation.

Synoptic table of electrophysiological measures (µV: mean ± SD) of SSEP HFO.

p values are given for comparison of HFO amplitudes between migraine without aura (MO) patients and healthy volunteers (HVs) at baseline [a], and between pre- and post- 1 Hz or 10 Hz rTMS [b]. HFO: high-frequency oscillation; rTMS: repetitive transcranial magnetic stimulation.
HF-rTMS, however, modified HFO. It significantly increased the pre-rTMS reduced amplitude maximum of early HFO in MO patients (t = 2.332, p = 0.036), but had no effect on early HFO in HVs (t = 1.358, p = 0.199). In both subject groups, 10 Hz rTMS also enhanced late HFO (HVs t = 2.680, p = 0.020; MO patients t = 2.688, p = 0.019) (Figures 3 and 4).
Discussion
The main findings of our study can be summarized as follows: (1) rTMS of the motor cortex modifies amplitude and habituation of conventional broadband SSEPs in an opposite way depending on its frequency and on the basal cortical activation level that differs between healthy subjects and migraineurs; (2) excitatory, that is, HF-rTMS, increases the early burst of SSEP HFOs in migraine patients and the late HFO burst in both groups.
Our rTMS protocol clearly influences the motor cortex in addition to the adjacent somatosensory areas, contrary to other protocols where stimulation intensities below the motor threshold are used to reduce motor cortex activation (24). It is well known that the motor cortex has a strong modulatory effect on somatosensory responses. Enomoto et al. showed that 1 Hz rTMS of the motor cortex at 110% of the active MT reduced N20-P25 amplitude in broadband SSEP, without affecting latency (25). These effects on SSEPs disappear when LF-rTMS is applied at stimulation intensities below the MT (26,27). In accordance with Enomoto et al., we find in healthy subjects that slow suprathreshold rTMS of the motor cortex significantly reduces N20-P25 SSEP amplitude in the first block of averagings. Ragert et al. reported that 5 Hz rTMS, supposed to activate the underlying motor cortex, had no effect on the unconditioned N20-P25 SSEP amplitude, although recovery of the N20 amplitude after paired stimulations of the median nerve was faster (28). Concordantly, we show that HF-rTMS fails to induce changes in amplitude of broadband SSEP compared to baseline in healthy subjects. This might be due to the fact that in healthy subjects the activation level of the somatosensory cortex is maximal and therefore cannot be increased.
In migraine patients, we confirm the interictal lack of habituation of the N20-P25 SSEP component (22,23). LF-rTMS does not change the already-low first block amplitude nor the deficient habituation, while 10 Hz rTMS increases first block amplitude and normalizes habituation. We have reported similar results with rTMS over the visual cortex. Depressing visual cortex excitability with 1 Hz rTMS decreased first block amplitude of VEPs and habituation in HVs, but did not affect VEPs in migraine patients (18,19). Conversely, enhancing visual cortex excitability with 10 Hz rTMS increased first block amplitude and restored normal VEP habituation in migraineurs, leaving VEPs unchanged in healthy subjects (18,19). Taken together, these studies support the concept that the effects of rTMS depend on the background activation level of the underlying cortex, the so-called “context dependency” (29). Following this concept, LF-rTMS that reduces the level of neuronal discharges can produce effects in systems with a high level of basal activity, as in visual or somatosensory cortices of healthy subjects. If, however, the basal condition is characterized by low levels of spontaneous discharges, as underlined by the lower first SSEP amplitude block in migraine patients between attacks, LF-rTMS has little effect, while HF-rTMS produces maximal activation. For the sake of completeness, it must be mentioned that a paradoxical effect of LF-rTMS on intracortical facilitatory circuits, that is, activation rather inhibition, was previously described in the motor cortex of migraine with aura patients (30). As previously reported by others (30–33), we found similar MTs between migraineurs and healthy controls. SSEP N20-P25 amplitude, however, was smaller in migraineurs than in controls, which is yet another argument in favor of interictal cortical hypoexcitability in migraine.
In the second part of our study, we confirm previous results showing that thalamo-cortical activity, as indexed by the early HFO burst in SSEPs, is significantly reduced interictally in migraineurs compared to HVs (15,16). The reason that Lai et al. (2011) found opposite results may be related to patient selection and to the fact that subjects were recorded with eyes closed, which suppresses early HFO (17). Our study shows, in addition, that LF-rTMS has no significant effect on HFO, either in HVs or in migraineurs, but that HF-rTMS increases late HFO in both groups, and early HFO only in migraine patients. Previous reports on rTMS effects on HFO in healthy subjects are partly discordant, probably because of several technical discrepancies. After LF-rTMS, late HFOs were reduced in one study (24) but increased in another (34). Early HFO amplitude was enhanced (24,34) or decreased in a study by Murakami et al. (2008) using continuous theta burst rTMS, which inhibits the underlying cortex (35). These discrepancies are likely due to methodological differences in stimulation site (S1 instead of M1), threshold (80% instead of 120% MT) or protocol (classic rTMS vs. theta burst). The fact that we found an amplitude decrease of wide-band SSEPs after 1 Hz rTMS but no effect on HFO supports the assumption that the generators for the two activities differ (36,37).
Early HFOs in healthy subjects were found increased after activating rTMS in studies by others (24,38). This contrasts with our data and may be related to the differences in protocol and/or intensity of TMS which can be associated with differential influences on motor and somatosensory cortex. Contrary to HVs, however, migraineurs had a significant increase in amplitude of their low-baseline early HFOs after 10 Hz rTMS in our study. We suggest that the difference between the two groups is due to a different thalamo-cortical activation at baseline allowing for an increase after 10 Hz rTMS in migraineurs, where it is reduced, but not in healthy subjects, where it is normal, confirming that the effects of rTMS depend on the actual level of activity of the cortex and its connected areas (29).
That TMS modifies a neuronal activity (early HFO) that is generated in the thalamus is not surprising. Modifications of cortical excitability induced by TMS are able to induce plastic changes in remote regions (39). It has been shown, for example, that stimulation of the forepaw region in the M1 cortex of mammals, prior to electrical stimulation to the forepaw, suppresses neuronal responses to touch by more than 40% in the ventroposterolateral nucleus of the thalamus (40) and by approximately 10% in brain-stem nuclei (41,42). In humans, rTMS over the motor cortex changed neurotransmitter release in subcortical structures (43), and the ventrolateral thalamic nucleus was found to facilitate TMS-induced EEG oscillations in the motor cortex through cortico-subcortico-cortical feedback loops (44).
The cellular and molecular mechanisms underlying the rTMS-induced changes are not completely understood. Low- or high-frequency stimulations, respectively, decrease or increase cortical excitability for some time in a way reminiscent of long-term depression (LTD) and long-term potentiation (LTP) (45,46). According to the Hebbian model of synaptic plasticity, rate-dependent repetitive TMS can modify plasticity at excitatory glutamatergic synapses (39). Hence, LTD induced by slow rTMS arises from decreased effectiveness of weakly activated glutamatergic synapses, rather than by increased effectiveness of GABAergic inhibition (47,48). LTP induced by rapid rTMS is due to the synchronous up-regulation of weakly and strongly activated excitatory synapses (39). Translated to our results, this would suggest that LF-rTMS decreased broadband SSEP amplitude by inducing LTD in excitatory intracortical glutamatergic synapses, and left late HFOs unmodified because they reflect activity in GABAergic inhibitory interneurons (10,49). Although LF-rTMS had no effect on the otherwise normal late HFOs, we cannot exclude that weakly activated glutamatergic synapses could down-regulate not only excitatory but also inhibitory mechanisms. This may be reflected in HVs in a decreased early excitation (cortical preactivation), manifesting as a reduced first amplitude block on the one hand, and in a late reduced lateral inhibition, leading to deficient habituation over subsequent blocks of averagings on the other hand (50). By contrast, HF-rTMS, by boosting glutamatergic transmission, increases broadband SSEP and late HFO amplitudes, the latter because activation of GABAergic interneurons arises from lateral glutamatergic afferents. In concordance with this concept, Restuccia et al. (2003) found an increase in amplitude of the late HFO component (9) after administration of the central acetylcholinesterase inhibitor rivastigmine, an indirect way to modulate glutamatergic transmission (51). Moreover, in migraine HF-rTMS may exert an excitatory action on GABAergic cortical cells as an indirect consequence of the increased early presynaptic HFO drive. This in turn may enhance late lateral cortical inhibition, which may contribute to the reducing response in later blocks of SSEP averagings (50).
Some authors have proposed that the deficient habituation in migraineurs is due to a basal cortical hyperexcitability (4,5) and/or insufficient homeostatic plasticity (52). The concept of cortical homeostatic metaplasticity postulates that in an activated cortex an excitatory intervention like HF-rTMS or anodal trancranial direct current stimulation triggers a homeostatic mechanism by which the cortex tends to maintain its excitability level within a physiological range (53,54). The abovementioned intracortical facilitatory circuits, which are paradoxically facilitated in the motor cortex of migraine with aura patients (30), depend on glutamatergic transmission amenable to homeostatic plasticity. Consequently, HF-rTMS could paradoxically reduce the supposedly higher level of cortical excitation in migraine by inducing compensatory homeostatic inhibitory mechanisms (52). If this were the case, one would expect that both N20-P25 SSEP amplitudes and HFOs would be increased at baseline, reflecting genuine cortical hyperexcitability and that HF-rTMS would reduce this increased excitation. The fact that in our patients amplitudes, either of broadband SSEPs or early HFO, were significantly reduced at baseline compared to HVs (see Figures 1 and 2) and increased, rather than decreased, after HF-rTMS does not favor this hypothesis. Moreover, one cannot exclude the possibility that thalamic structures may be down-regulated because of persistent cortical hyperexcitability as a secondary compensatory mechanism, explaining low first block SSEP amplitudes and reduced HFO. In this case, the thalamic down-regulation would be expected to be enhanced by further cortical activation with HF-rTMS and mitigated by cortical inhibition with LF-rTMS. Our data, however, do not favor this hypothesis, as HF-rTMS increased late HFO in both subject groups and early HFO in patients, and LF-rTMS produced no detectable change in either group.
To conclude, we found that thalamo-cortical drive can be increased in migraineurs by 10 Hz rTMS-induced activation of the motor (and sensory) cortex, because it is low at baseline. At the same time 10 Hz rTMS normalizes the deficient habituation of SSEPs. This may not be possible in healthy subjects, in whom thalamo-cortical activity and cortical pre-activation is maximal before the rTMS.
Taken together with similar effects we observed for VEPs, these findings support the hypothesis that reduced cortical pre-activation due to insufficient thalamo-cortical drive could be responsible for the abnormal information processing (i.e. habituation deficit) found in migraine patients between attacks. We cannot definitively exclude that cortical homeostatic metaplasticity and/or thalamic down-regulation secondary to cortical hyperexcitability may explain part of these findings, although our data do not support such mechanisms.
Footnotes
Funding
This study was supported by the research convention 3.4.650.09 from the National Fund for Scientific Research–Belgium to J.S. and by research grants from the Faculty of Medicine–University of Liège.
