Abstract
Introduction
Aura occurs in 20–30% of patients with migraine. Some descriptions of aura go far beyond the most frequent visual and sensory symptoms, suggesting the involvement of different cortical areas. The aim of this prospective study was to evaluate the frequency and types of disorders of higher cortical functions (HCF) that occur during visual and/or sensory aura.
Methods
We interviewed 60 patients with visual and/or sensory aura about HCF disorders of praxia, gnosia, memory, and speech, during aura. Patients were divided into two groups, with and without HCF disorders, and were compared in terms of demographic data and aura characteristics.
Results
From all 60 patients, 65% reported at least one HCF disorder during aura. The patients with HCF disorders had longer-lasting auras (28.51 ± 16.39 vs. 19.76 ± 11.23, p = 0.016). The most common HCF disorders were motor dysphasia (82.05%) and dysnomia (30.74%). Motor dysphasia was more often reported by patients with visual as well as sensory aura (p = 0.002). The number of HCF disorders correlated with the aura duration (p = 0.003).
Conclusion
According to our results, HCF disorders during aura occur more often than previously thought. The aura duration has some influence on the HCF disorders.
Introduction
Migraine is a primary headache with an estimated annual prevalence of 11% of the adult population (1). Aura occurs in 20–30% of migraine patients, usually preceding the headache, sometimes occurring during or after the headache, or even without a headache. According to the diagnostic criteria of the International Classification of Headache Disorders, migraine aura develops gradually over five to 20 minutes and lasts up to 60 minutes. Typical symptoms of aura are homonymous visual symptoms with positive (shimmering lines or points in the visual field) or negative phenomena (loss of vision); less frequent are one-sided sensory symptoms with positive (tingling, pricking) or negative phenomena (numbness), or dysphasia. Patients with motor aura are separated into a special group because of a proven genetic basis (2).
The exact mechanism of aura is still not completely understood (3). Cortical spreading depression (CSD) is demonstrated during aura (4). CSD originates in the occipital region and propagates proximally (5–8). The involvement of other cortical areas beyond the occipital region could be assumed because of the existence of sensory and motor clinical features during aura in some patients (9). The anatomical counterparts of higher cortical functions (HCF) are polymodal associative areas that integrate sensory information of different modalities (10). The exact localization of lesions for the majority of HCF disorders has been known since Brodmann (11).
In clinical practice, some patients report speech and memory difficulties during aura (12–14). To the best of our knowledge, there has been only one study exploring HCF disorders during migraine (9). HCF disorders were recorded more frequently in migraine with aura than in migraine without aura (9).
The aim of this study was to evaluate the frequency and types of HCF disorders that occur during visual and/or sensory aura.
Methods
Study questionnaire.

Question to differentiate dysphasia from dysarthria.
Of those 89 patients, 60 agreed to participate in this study and completed the questionnaire in the presence of a doctor (I.P.). The questionnaire had 17 questions regarding color naming, object and face recognition, memory, speech, calculation, spatial orientation, and praxia, as well as the occurrence of hallucinations during the aura. The patients were divided into two main groups. The patients who reported at least one HCF disorder symptom were labeled as Group I; patients with no HCF disorder symptoms comprised Group II. These two groups were compared in terms of demographic data (age, gender, and education level) and aura features (age at migraine onset, frequency expressed in number of auras per year, and aura duration expressed in minutes).
For further analysis, Group I was divided into two subgroups: Subgroup Ia included the patients with visual aura only, and Subgroup Ib included the patients with visual as well as sensory symptoms during aura. There were two (3.3%) patients with sensory aura only; they were added to Subgroup Ib for statistical analysis. The differences in the occurrence and type of HCF disorder were analyzed by comparing these two subgroups.
The HCF disorders were classified, according to their presumed cortical localization, as disorders of the occipital lobe (color-naming disorder, prosopagnosia, and visual dysgnosia, corresponding to questions one through three from the study questionnaire), temporal lobe (retrograde and anterograde amnesia, déjà vu phenomenon and auditory agnosia, corresponding to questions four through seven), fronto-temporo-parietal lobe (motor and nominal dysphasia, corresponding to questions 10 and 11), temporo-parieto-occipital associative region (dyslexia, dysgraphia, and dyscalculia, corresponding to questions 12–14), and parietal lobe (astereodysgnosia, manual dyspraxia, and spatial disorientation, corresponding to questions 15–17). The two subgroups were compared according to the involvement of the cerebral lobe.
The data are presented as arithmetic mean ± SD or as percentages. Independent samples t test, chi squared test, Fisher’s exact test, and the Mann-Whitney U test were used to compare the data between the groups. Spearman’s test was used to assess correlation. The significance level for the analysis was set beforehand at 5% (p < 0.05).
Results
The study included 44 (73.3%) females and 16 (26.7%) males, aged 39.0 ± 13.1 (range 21–70) years, who experienced migraine with aura. Twenty-six (43.3%) patients had visual aura only; 34 (56.7%) patients had visual and sensory aura. Disorders of the tested HCF were reported by 39 (65.13%) patients, classified as Group I. The 21 patients who did not experience HCF disturbance during aura were classified as Group II.
Comparison of demographic data and aura features between Group I and Group II.

The majority of patients reported one, two, or three HCF disorders, as is shown in Figure 1. The most common HCF disorder was motor dysphasia, reported by 32 patients (82.05%), followed by dysnomia, reported by 12 patients (30.77%), and retrograde amnesia, reported by nine patients (23.08%), as is shown in Figure 2.
Distribution of patients by number of HCF disorders. Distribution of patients by HCF disorder type.
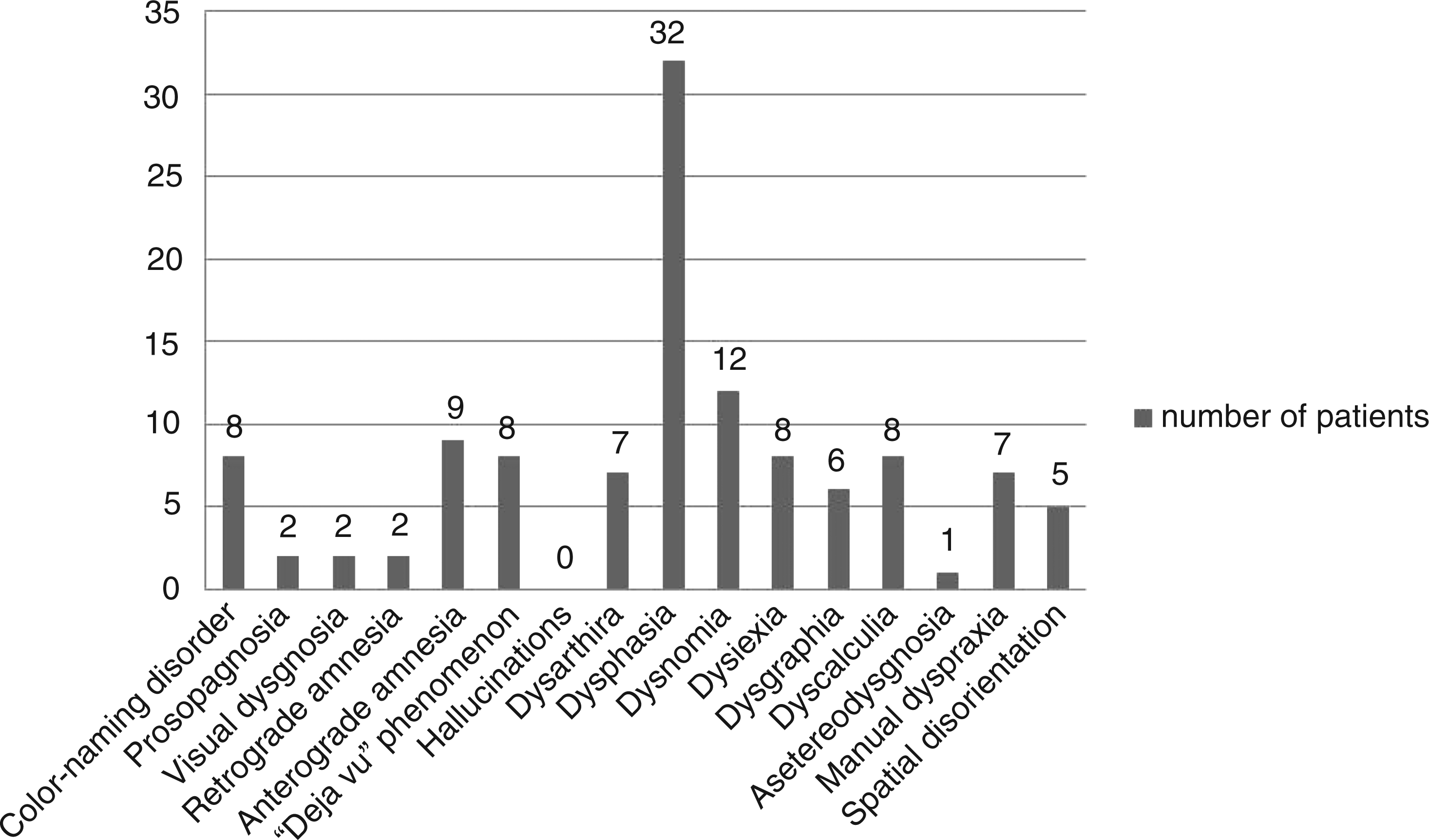
Comparison of the occurrence of HCF disorders between Subgroup Ia and Subgroup Ib.

HCF: higher cortical function.
Comparison of the occurrence of HCF disorders between Subgroup Ia and Subgroup Ib according to presumed localization in cerebral lobes.
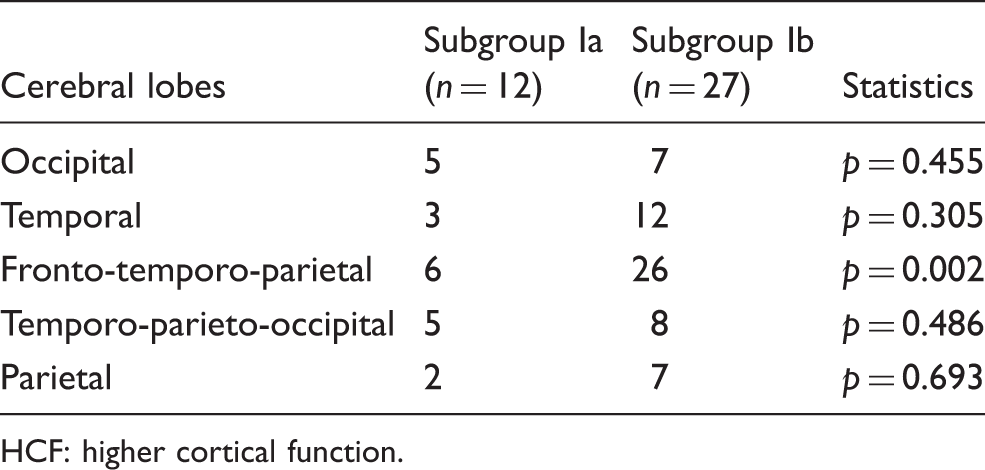
HCF: higher cortical function.
There was a positive correlation between aura duration and the number of HCF disorders (ρ = 0.467, p = 0.003).
Discussion
This study analyzed the presence of HCF disorders during migraine aura. At least one HCF disorder was reported by 65% of the patients. This is not the first study investigating HCF disorders in migraine patients. Olfactory and/or gustatory hallucinations and distortions of body image were reported by 15% of the patients with migraine in a study by Morrison (15). Beyond the visual and sensory symptoms, phenomena reported during migraine include misperceptions, impaired gnosis, praxis, and memory; these were more frequently reported by patients with aura than without it in a study by Vincent and Hadjikhani (9). Acute (16,17) and recurrent (18) pain were shown to have a significant impact on cognitive test performance, particularly on selective attention and executive functions. We decided to study HCF disorders during migraine aura and before the painful stage of migraine in order to exclude the potential influence of pain on HCF.
The patients without HCF disturbances did not differ from the patients with HCF disorders in terms of gender, age, education level, age at migraine onset, or aura frequency. It is likely that other factors are relevant to the occurrence of HCF in patients with migraine with aura. Aura lasted nearly 10 minutes longer in the patients with HCF disorders and positively correlated with the number of HCF disorders. Dysphasia was also more common in patients with longer aura duration.
Neuroimaging studies support the hypothesis that CSD is the initial pathophysiological event of migraine, which originates in the occipital lobe (5). The most common manifestation of migraine with aura is visual phenomena, reported by 58 of our 60 patients. Besides the simple positive or negative phenomena, 20% of the studied patients reported more complex visual disorders. Visual agnosia is a disorder of recognizing objects, faces (prosopagnosia), and linking objects with their functions, with preserved acuity and visual field width; this HCF is localized in the frontal occipital lobe (19,20). Color-naming disorder during aura was the most common visual dysfunction reported in our patients. The decrease of color intensity was frequently reported during epileptic seizures (21) and was also identified by migraine patients with aura (9). The small number of patients with prosopagnosia can be explained by the fact that this function is localized bilaterally (19).
Motor dysphasia and dysnomia were the most common HCF disorders, reported by half of our patients. Dysphasic aura is characterized by an aphasia that can progress later in dysarthria (22,23). The study questionnaire enabled the distinction between aphasic and dysarthric disorder. Dysarthria was noted by 12% of our patients. We did not examine the timing and sequence of aura events, so it is possible that dysarthria progressed from dysphasia. The aphasic phenomena were more frequently reported by patients with complex visual and sensory aura. The existence of complex aura could indicate the spreading of CSD from the cortical occipital to parietal lobe regions, and then further rostrally to the centers for speech. We classified HCF disorders according to their presumed lobar localization with the hypothesis that a wave of depolarization propagates through contiguous regions of the cortex. It was expected that patients with complex aura had more frequent disorders of the temporal and parietal lobes than patients with visual aura only. However, these differences were not found, and the only HCF disorder more frequently reported by patients with complex aura was dysphasia. We can only speculate that propagation of CSD could involve associative pathways or that CSD could start simultaneously in more than one cortical region. Magnetic resonance imaging (MRI) tractography results suggest that many complex functions rely on the activity of distributed networks rather than on single cortical areas (13).
Nearly a third of our patients had memory disorders during aura, such as difficulties in remembering the events during aura or more frequently in recalling past events. Déjà vu is a widespread, fascinating, and mysterious human experience, reported in patients with temporal lobe epilepsy, but also in healthy subjects (24). Déjà vu was reported by 13% of our patients, mostly by patients with complex aura. Morphological studies in healthy subjects revealed decreased gray matter volume in cortical mesiotemporal regions, thus demonstrating the neurological basis of this phenomenon (25). Hallucinations were not reported by our patients.
The main limitations of our study are that the data were collected between attacks and that the HCF disorders were determined by patients' reports. This is the only investigation of HCF disorders occurring during aura. As the majority of our patients had an average migraine disorder duration of 20 years with a monthly appearance of aura, we could suppose that their descriptions are relevant. It is possible that the longer aura duration in some patients allows them to notice more HCF disorders, and it is possible that these disorders are even more frequent than we estimated.
The results obtained by our study strongly suggest that HCF disorders represent an important part of migraine aura. The frequent occurrence of these disorders during aura adds more questions to the puzzle of the pathophysiology of migraine. The transient neuropsychological disturbances were more frequent than had been considered, and these data are important for the patient as well as for the physician who diagnoses migraine. Neuropsychological testing during aura could give more accurate data in this field, as could aura descriptions correlated with functional neuroimaging performed during aura.
Clinical implications
Disorders of higher cortical functions during aura occur more often than it was previously assumed. The duration of aura has some influence on the disorders of higher cortical functions.
Footnotes
Funding
This work was supported by a grant from the Ministry of Science and Technology of the Republic of Serbia (grant no. 175022).
Conflict of interest
None declared.
