Abstract
Background:
Migraine has been considered a vascular risk factor especially in young women. Factors predisposing to endothelial damage in migraine are still being debated. The insufficiency of circulating endothelial precursor circulating cells (EPCs) suggested a link between migraine and cardiovascular risk. This research aimed to study a subtype of EPCs, those expressing e-selectin, to assess endothelial activation and, therefore, endothelial dysfunction in migraine.
Methods:
Consecutive headache patients (n = 99) and 35 adjusted controls were recruited. Total EPCs, defined as CD34+/KDR+ cells, and EPC colony-forming units (CFUs) were assayed. We identified as “early” EPCs those CD62E– EPCs, and “late” EPCs, CD62E+, a surrogate marker for endothelial damage. Plasmatic calcitonin-gene related protein (CGRP) and vascular-endothelial growth factor (VEGF) were analyzed.
Results:
We did not find differences in the total number of CFUs among clinical groups. Means of total CD34+/KDR+ and “early” EPCs were not significant among clinical groups. Nevertheless, the mean of “late” EPCs was lower (log10-transformed mean = 1.715; SD = 0.393) in the control group than in the migraine patients (log10-transformed mean = 2.167; SD = 0.685), even after adjustment by VEGF plasma level and other confounding factors. Linear regression analyses disclosed significant predictors for “late” EPCs for controls vs migraine (β = 0.452 SE ± 0.13; p = 0.001). We did not observe differences between migraine with or without aura.
Conclusion:
We observed higher number of activated EPCs in migraine patients than in controls. CD62E+ EPCs might be considered a marker for vascular damage in migraine patients.
Introduction
Migraine carries an increased risk for vascular disorders, comprising stroke, myocardial infarction, angina and death due to ischemic cardiovascular disease. The increased vascular risk assigned to migraine has been considered slight in population- and hospital-based studies. The relative risk (RR) of ischemic stroke in migraine patients is double (RR 2.1; 95% confidence interval (CI) 1.9–2.5) for migraine in general, and higher for migraine with aura (MA) (RR 2.9; 95% CI 1.9–4.4) than for migraine without aura (MO) (RR 1.6; 95% CI 1.0–2.4). This risk is even more consistent for young females (less than 45 years; RR 2.7, 95% CI 2.2–3.5) (1). In spite of this body of evidence, the absolute risk of cerebrovascular events among women remains low (3.8/100,000 women/year) (2). Although the intrinsic mechanism has not yet been fully elucidated, changes in vascular reactivity (3), endothelial disturbance (4,5), and even changes in platelet function have been addressed as potential pathways for vascular damage in migraine patients. In addition, several vascular disorders increase the risk for suffering from migraine, mainly MA, as occurs in cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) (6), an autosomal vascular disorder caused by mutations in the NOTCH3 gene, or in hereditary endotheliopathy with retinopathy, neuropathy and strokes (HERNS) syndrome (7). In these conditions a primary vascular disorder triggers migraine. Migraine prevalence is increased among patients having Raynaud phenomenon (8).
Lee et al. (9) reported a decreased number and function of a subclass of hematopoietic circulating cells preprogrammed to renew the endothelium, named endothelial precursor circulating cells (EPCs), first described in 1997 (10), in migraine patients, particularly those with MA. The number of these cells in peripheral blood is very low and the cell phenotype defined by cell-surface antigens has not been uniformly used. These angiogenic cells express three surface markers (CD133, CD34, and the vascular-endothelial growth factor (VEGF) receptor-2) that define the early functional angioblast. Later in the systemic circulation, EPCs gradually lose their progenitor properties and start to express endothelial markers such as VE-cadherin, E-selectin, endothelial nitric oxide synthase and von Willebrand factor (vW) (11). Lee et al. used the number of CFUs as the biomarker for their study. EPCs forming colonies express acLDL/ulex-lectin in addition to CD34/KDR/CD31 antigens. The number and function of EPCs have also been used as a surrogate for cardiovascular risk in several vascular disorders (12), but also in inflammatory disorders like systemic lupus erythematosus (SLE) (13) and rheumatoid arthritis (RA) (14). Endothelial dysfunction is an early marker of vascular damage preceding clinically overt cardiovascular disease (15) and strongly predicts coronary disease (16); nevertheless, this correlation is still under debate for migraine.
Here we analyzed the number of subsets of EPCs and their capacity to make colony-forming units (CFUs) in migraine patients and controls in order to compare the level of endothelial activation. We hypothesized that vascular damage must be greater in those more disabled migraine patients.
Methods
This research has been approved by the Ethics Committee for Clinical Investigation of our hospital. Patients and controls were recruited in an outpatient clinic specially dedicated to more disabled migraine patients. Patients having more than 15 days/month of headache, being at least 8 of migraine type, for at least the last three months, were assigned to the “chronic migraine” (CM) group, and those who had fewer than 15 episodes/month were assigned to the “episodic migraine” (EM) group, according to the International Headache Society ad hoc Committee criteria (17) and the new criteria for chronic migraine (18). Most controls were recruited from patients’ friends.
Inclusion criteria for the migraine group were: 1) age older than 18 and younger than 65; 2) they must fulfill MA or MO criteria according to the IHS-2004 classification, and 2006-criteria for chronic migraine; and 3) they must give and sign their informed consent. Exclusion criteria for this group included: 1) any other active inflammatory process at the moment of inclusion; 2) any other unstable cardiovascular disorder active at the moment of inclusion; 3) previous diagnosis of cancer or treatment with antimitogen agents; and 4) pregnancy in the last year.
For controls, we instructed patients to select these from among their friends, with the same age and sex, and living in the same area, and with no headache complaints. After this, all candidates for controls were interviewed by one of us (AO). Exclusion criteria were the same as for the migraine group.
Period of recruitment
This study began at the end of 2009. From November 2009 to March 2010, we set up the conditions and procedures in order to optimize the required techniques. A total of 20 volunteers were recruited from hospital staff members for this purpose, but they were not included. Recruitment of cases and controls began in March 2010 and finished in February 2012.
After informed consent, a total of 24 ml of blood with heparin was drawn. Given the frequent number of any headache type, those subjects with CM were drawn irrespective of the moment of the last migraine bout. However, those patients with EM were instructed to be drawn at least three days after the last migraine crisis. For EPC culture and CFU counting, we followed the manufacturer’s instructions (StemCell Technologies catalogue #2871, manual version 3.0.0, available at www.stemcell.com). Mononuclear cells were obtained by light density separation using Ficoll-Paque TM PLUS (GE Healthcare Bio-Science AB, Uppsala, Sweden). Samples were centrifuged at 300× g for 25 min. The mononuclear layer was collected into a new tube and washed twice in phosphate-buffered saline (PBS) + 2% fetal bovine serum (FBS). The pellet was re-suspended in 3 ml of CFU-Hill liquid medium (StemCell Technologies Inc. Vancouver, Canada) and nucleated cells were counted using Trypan Blue at a 1:2 dilution. A total of 5 × 10E6 cells were plated per well containing 2 ml of CFU-Hill Liquid Medium in fibronectin-coated plates, and incubated for two days at 37°C, 5% CO2 and 95% humidity. Non-adherent cells were collected and transferred into a polypropylene tube. Nucleated cells were then counted using Trypan Blue at 1:2 dilution. A total of 1 × 10E6 cells were plated per well into a 24-well fibronectin-coated plate, adding fresh media to a final volume of 1 ml/well. Samples were incubated again for three days at 37°C, 5% CO2 and 95% humidity. CFUs were then counted. CFUs were defined as a central core of round cells radiating elongated spindle-like cells at the periphery. For flow-cytometry (FC), we followed the same protocol as for cell cultures to obtain mononuclear cells from peripheral blood. Mononuclear cells were then counted up to 1 × 10E7 per 100 µl of PBS. After having added 1 µL of each of the fluorochrome-dyed antibodies (CD62E-fluorescein isothiocyanate (FITC) (Immunostep, Salamanca, Spain); CD34-PE and KDR-APC (Miltenyi Biotech, Auburn, California, USA)) for each 1 × 10E6, samples were incubated in the dark at room temperature. After incubation, cells were washed by adding 5 ml of PBS + 2% FBS to each tube. The pellet was resuspended in PBS and filtered using a 5 µm-cup filter to avoid aggregated cells. The two cell populations under study were then identified as “early” EPCs (CD34+/KDR+/CD62E−), and “late” EPCs (CD34+/KDR+/CD62E+) according to Hristov and Weber (11) and Yin et al., (19) since the proportion of CD133+/KDR+ or CD34+/KDR+ represent early, bone marrow-derived EPCs.
Commercially available enzyme-linked immunosorbent assay (ELISA) methods were used to determine serum levels of CGRPα (Peninsula Laboratories LLC, Bachem, San Carlos, CA, USA) and VEGF-165 (Life Technologies, Invitrogen, Carlsbad, CA, USA). The mean minimal detectable level was determined by adding two standard deviations (SD) to the mean optical density value of 20 zero standard replicates and calculating the corresponding concentrations. Detectable values for VEGF ranged from 0 pg/ml to 1500 pg/ml; and for CGRP, from 0 to 10 ng/ml.
Statistical analysis
All computations were performed with the SPSS (15.0, SPSS Inc., Chicago, IL, USA) package. Continuous variables were expressed as mean ± SD. Categorical variables were analyzed using the Chi (2) method (α = 0.05). Non-normally distributed variables were log10-transformed for further comparisons. We used the Student’s t test for comparing continuous variables, and the Pearson method for bivariate correlations (continuous variables). Post hoc corrections were made using the Bonferroni method. Linear regression models were constructed to determine which terms uniquely predicted variance in the final regression model to support interaction at p < 0.10. Reproducibility of EPCs counting was evaluated in a subset of 20 subjects using the intra-class coefficient method (ICC) for individual measures, available in SPSS 15.0.
Power calculations
For comparison of means, the expected power was 86%, using a sample of 30 controls and 30 patients and variances observed here for late EPCs.
Results
Clinical characteristics regarding the chronicity of the migraine and control groups. IHS criteria were used to define “chronic migraine”.

IHS: International Headache Society; df: degree of freedom; MA: migraine with aura; MIDAS: migraine disability assessment; OH: alcohol; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood, pressure; DM: diabetes mellitus; N/A: not appropriate; ns: not significant.
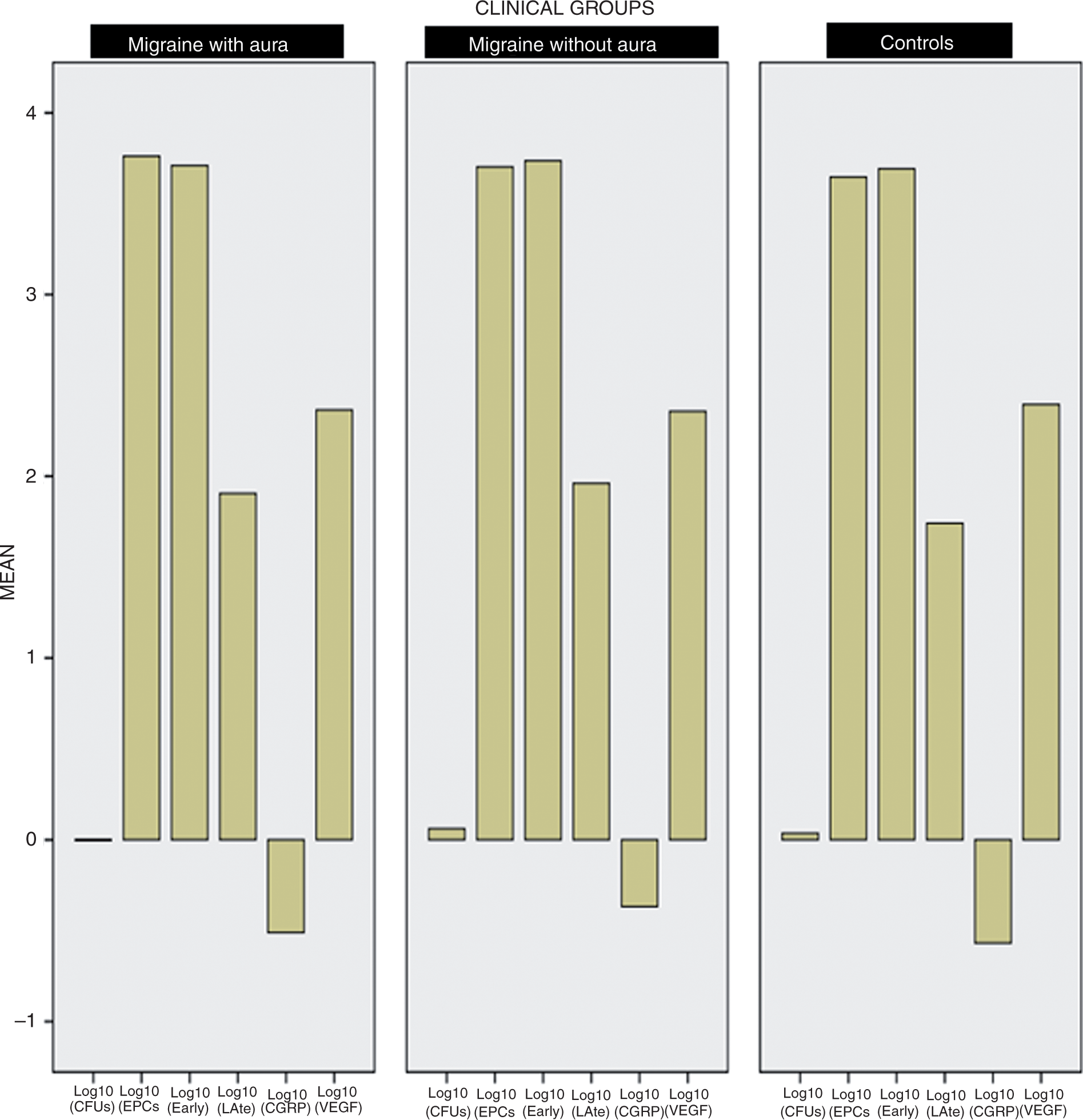
At the beginning of the study, we analyzed intraclass variability to test the reproducibility of the EPC-counting method and CFU-culture and -counting methods. There were no differences in the number of EPCs or counts of CFUs measured in 20 repeated samples drawn two weeks after (EPCs, ICC = 0.985 (95% CI = 0.968–0.993); CFUs, ICC = 0.714 (95% CI = 0.408–0.876)). There were no differences in the number of CFUs among clinical groups (Figure 1) and the total number of EPCs (Figure 2). Nevertheless, we observed a significantly lower number of CD34+/KDR+/CD62E+ EPCs (late EPCs) (p = 0.001, Figure 2) in controls (log10-transformed mean = 1.715; SD = 0.393) than in migraine patients (log10-transformed mean = 2.167; SD = 0.685). The number of CFUs did not correlate well with subtypes of EPCs. Neither the number of EPCs nor CFUs correlated with age, smoking, plasmatic lipids (except high-density lipoprotein (HDL), which correlated with early EPCs; see Table 2), systolic or diastolic pressure. Body mass index (BMI) inversely correlated with total EPCs (CD34+/KDR+; r = −0.264; p = 0.018) and with early EPCs (CD62E−; r = −0.254; p = 0.023). Plasmatic VEGF inversely correlated with EPCs (r = −0.232; p = 0.022), mainly with early EPCs (r = −0.249; p = 0.018). There were no differences in the plasma VEGF or CGRP among clinical groups (Figure 3). CGRP did not correlate either with the severity of migraine, measured with the migraine disability assessment (MIDAS), or the type of migraine (MA, MO; not shown). Linear regression analyses disclosed significant predictors for late EPCs for controls vs migraine groups (EM + CM; β = 0.452 SE ± 0.13; p = 0.001), between the CM and control group (β = 0.430 SE ± 0.14; p = 0.004; corrected p = 0.013), and between the EM and control group (β = 0.476 SE ± 0.15; p = 0.002; corrected p = 0.006). After adjustment for VEGF, results remained significant (p = 0.014 for controls vs migraine (CM + EM), for controls vs CM (p = 0.011; corrected p = 0.033), and for controls vs EM (p = 0.009; corrected p = 0.028). Further adjustment for BMI disclosed significant predictors for late EPCs for controls vs migraine (EM + CM; β = 0.444 SE ± 0.16, p = 0.010), and for controls vs EM (β = 0.510 SE ± 0.17, p = 0.006). Uncorrected results were still significant for controls vs CM (β = 0.369 SE ± 0.8, p = 0.047; corrected p = 0.141). There were no differences between migraine subgroups (for all analyses, p > 0.05).
These point plots represent the distribution of the number of colony-forming units (CFUs) regarding clinical groups. There were no significant differences among groups. These box-plots represent median, 25, 75, 95 percentiles, and outliers (circles) corresponding to the distribution of the log10-transformed number of EPCs (total, CD4+/KDR+), early EPCs (plus CD62E–) and late EPCs (plus CD62E+). There were no significant differences among groups with respect to the total count of EPCs or early EPCs. Nevertheless, we observed a significantly (p = 0.001) lower count of late EPCs in the control group. Identical values were observed regarding the type of migraine (MA or MO) with respect to the control group (not shown). These three panels represent the log-transformed values of total CFUs, total EPCs, early-EPCs, late-EPCs, and plasmatic CGRP and VEGF in the clinical groups regarding their type of migraine (with or without aura) and controls. None of the small differences observed were significant. Bivariate correlations using the Pearson method with those variables implicated in vascular risk. Only VEGF and HDL-cholesterol showed some correlation with EPCs. VEGF: vascular-endothelial growth factor; HDL: high-density lipoprotein; EPCs: endothelial precursor circulating cells; CFUs: colony-forming units; CGRP: calcitonin-gene related protein; BMI: body mass index; MIDAS: migraine disability assessment.


Discussion
The main finding of our study was that CFUs and the total number of EPCs were no different in the three main clinical groups (CM, EM, and controls). Interestingly, the number of those EPCs showing CD62E positivity, which identifies more mature EPCs, was significantly lower in controls than in any type of migraine subset (CM, EM, MA or MO). Our sample was composed of subjects with a narrow age range, having a low number of cardiovascular risk factors (20/134 smokers; six having a BMI > 32; two with diabetes mellitus (DM), four with hypertension, 20 dyslipemics). We did not find any correlation of EPCs – any type – with MIDAS, and, as observed by Lee et al., (9) there was no correlation of CFUs and headache scores. Nevertheless, there are striking differences between Lee et al. and the present report. Subjects under study were older (mean difference >13 years for all equivalent clinic groups), the number of subjects with EPC culture was very low (six in each group), and the number of cardiovascular risk factors was higher in Lee et al. with respect to the present report. All of these circumstances may affect the number of EPC CFUs. As the number of migraine bouts was high, even for EM, we could not avoid the influence of migraine crises in our study for all migraine subjects. After a vascular injury, there is evidence that EPCs transitorily mobilize, increasing the circulating pool of EPCs from bone marrow that will form CFUs (CD133+), and that this ability depends on the presence of chronic vascular pathology (20). Therefore, it is recommended that EPC levels must be carefully interpreted in the combined setting of acute vascular ischemia and chronic vascular injury. Then there are several factors influencing the number of EPC CFUs that are most likely acting on the Lee et al. sample rather than the present study.
We selectively included a CM group with the intention of enhancing possible differences in total EPCs and CFUs linked to the severity of migraine, but it failed in this purpose. We had hypothesized that more disabled patients might have more endothelial damage. As our results suggest, there were no differences between chronic and EM patients, as they were grouped according to clinical criteria. Although “chronic” and “episodic” migraine are conventional definitions, biological consequences, at least regarding endothelial dysfunction, are quite similar. However, clinical status, as a predictor for the ratio of CD34+/KDR+/CD62E+, correlated better with CM than with EM.
In the definition of EPCs as used here, the phenotype CD34+ (hematopoietic stem)/KDR+ (endothelial stem) identified a subtype of EPCs that are not mobilized from the bone marrow, but are mostly generated from circulating multipotent CD34+ stem cells at sites of vascular injuries (21); therefore, the number of circulating CD34+/KDR+ cells may serve as a marker for vascular injury. On the other hand, CD62E marker is present in endothelial microparticles after endothelial activation (22). This marker identified more mature EPCs that could be associated with potential endothelial damage. As expected, we failed to correlate any EPC subtype with the total number of CFUs because the EPC definition we used here did not include earlier EPC forms that are responsible for their capacity to generate CFUs, identified by CD133, a marker that is lost in the early process of cell maturation (11). After adjusting for other vascular risk factors, only the number of CD62E+ EPCs remained significantly lower in the control group, suggesting that migraine (irrespective of the subtype) is itself a vascular risk factor. This finding suggests that CM patients have a higher endothelial turnover than controls and a persistent state of endothelial activation.
Genetic studies of endothelial function in migraine have yielded overall negative results. In young women, there was no relation between EDN and EDNRB genes mediating the association of migraine and stroke (23), even when those genes disclosed some association in white, but not black, women. Other genetic studies failed to demonstrate any association of migraine with the endothelial nitric oxide synthase gene (NOS3) (24,25). Therefore, the genetic explanation for impaired NO-dependent vasodilation in migraine patients (26) remains unresolved. Nevertheless, in premenopausal women with migraine, vW factor activity, along with other inflammation and coagulation markers, was increased, particularly in MA women, which is evidence for endothelial activation and therefore, endothelial dysfunction (27). E-selectin microparticles were found to increase cardiovascular events in patients with stroke (28). The overexpression of vW factor and e-selectin microparticles is considered as a biomarker of endothelial dysfunction, a phenotypic shift to procoagulant and proadhesive endothelial cell state (29). Taking these data into account, the present study reinforces the hypothesis of endothelial involvement in the pathogenesis of migraine.
Our study has some strengths. First, the sample was composed of adjusted clinical groups for vascular confounders; all subjects were personally interviewed by two experts in headache. Second, the person who performed EPC determination and CFU counting was blinded to clinical status. This study also has some limitations. The sample size is relatively small, especially the control group. We included an EM group having a high frequency of migraine bouts per month; therefore differences in variables comparing this group with the CM group were not as sharp as might be desired.
In conclusion, the total number of EPCs did not differ (CD34+/KDR+) between migraine and control groups, but the ratio of activated EPCs (CD34+/KDR+/CD62E+) was significantly lower in controls than in migraine groups. There were no differences in this parameter among subtypes of migraine (chronic/episodic; MA/MO). We propose here that CD62E could be a marker for vascular damage in migraine patients.
Clinical implications
In young women, migraine has been considered a vascular risk factor acting through endothelial damage. We found a significant increase in circulating CD62E+ endothelial precursor cells (EPCs), both in episodic and chronic migraine. Our results suggest that CD62E might be a biomarker of endothelial activation in migraine, but it did not distinguished episodic from chronic migraine.
Footnotes
Funding
This work has been funded by ISCIII-FISS PI08/0387, PI11/1232, and IFIMAV number ‘CI 12/03’.
Conflict of interest
None declared.
