Abstract
Background: Endothelial dysfunction could be involved in the pathophysiology of migraine. The results obtained from a few studies on endothelial dysfunction in migraine are controversial. We investigated brachial flow-mediated dilatation (FMD), which reflects systemic endothelial dysfunction, in migraine patients without comorbidities. By employing strict inclusion criteria we avoided the possible changes to FMD from confounding factors.
Methods. Forty migraine patients without comorbidities (20 with and 20 without aura) and 20 healthy subjects were included. FMD of brachial arteries and carotid intima-media thickness were measured by using standard procedures.
Results. We did not find any difference in FMD between migraine patients and healthy subjects (p = .96). Also, no differences were found among healthy subjects, migraine patients with aura and without aura (p = .99).
Conclusion. Our study showed that systemic endothelial function is not impaired in migraine patients without comorbidities, neither in those with or without aura. Considering these findings, the investigation of cerebral endothelial function would be useful in a further investigation of the role of endothelial (dys)function in migraine pathophysiology.
Introduction
Migraine has long been defined as a neurovascular disorder without long-term consequences to the brain or possibly other organs. However, in recent years several studies have associated migraine with cerebrovascular disorders (1–5). The increased prevalence of white matter lesions on MRI and several cases of ischemic strokes during and between migraine attacks indicated an association between migraine and ischemic strokes (6–12). Therefore, migraine has come to be a known “risk factor” for brain ischemia. Furthermore, epidemiological studies suggest that migraine is associated not only with disorders of the cerebral but also coronary, retinal, dermal and peripheral vasculature (13). Some studies have also shown a procoagulatory and proinflammatory state in migraine patients (13). All of these findings suggest a role of endothelial dysfunction in migraine. It is speculated that endothelial dysfunction could lead to migraine or vice versa, migraine could lead to endothelial dysfunction (14–20). Also, it is proposed that migraine may be a local manifestation of systemic vascular abnormality (21,22).
Studies on endothelial function in migraine patients are rare, and they have shown conflicting results. For these purposes flow-mediated dilatation (FMD), a well-known functional method that reflects the endothelial function of systemic arteries through relative change of the brachial artery diameter after ischemia, was used (23,24). Some authors have described decreased FMD in migraine patients (14,21,22,25); others have not found any difference in systemic endothelial function compared to the healthy subjects (26). But none of them employed strict inclusion criteria such as carotid intima-media thickness (IMT) to avoid the possible changes to FMD from confounding factors. Thus, the existence of endothelial dysfunction in migraine patients has yet to be shown.
The aim of our study was to assess systemic endothelial function with FMD in migraine patients without comorbidities during an interictal period by employing strict inclusion criteria and comparing it with that of healthy subjects.
Methods
Biochemical and morphological parameters of healthy subjects, MwA and MwoA
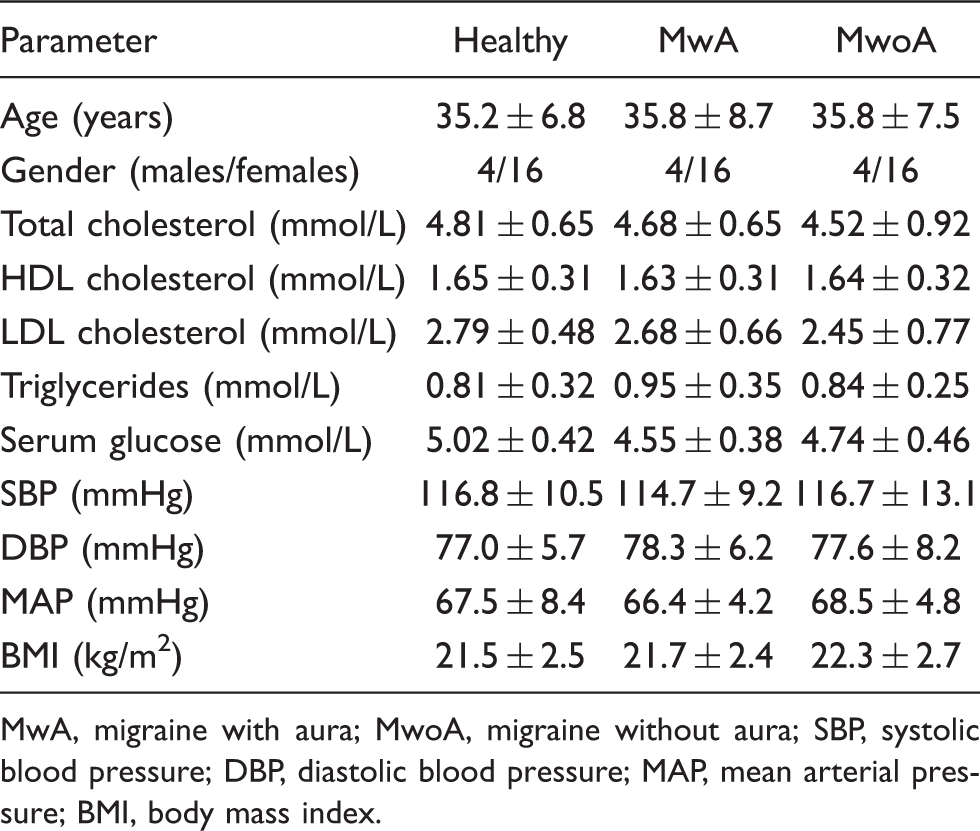
MwA, migraine with aura; MwoA, migraine without aura; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; BMI, body mass index.
For all subjects, the major exclusion criteria were body mass index (BMI)<18 or ≥25 kg/m2, history of cardiovascular disease, arterial hypertension (systolic blood pressure >140 mmHg or diastolic blood pressure >90 mmHg), diabetes, hypercholesterolemia (total cholesterol >5.5 mmol/L), pregnancy or lactation and regular use of vasoactive drugs (except triptans or other kinds of transient vasoactive antimigraine drugs). Based on medical history, physical and neurological examination and routine laboratory tests, all subjects were in good health.
The study was approved by the National Medical Ethics Committee of the Republic of Slovenia (and all subjects gave written informed consent before being included).
Color-coded duplex sonography of the carotid and vertebral arteries was performed in all patients. IMT was measured according to the Mannheim Intima-Media Thickness Consensus (28) on both sides 2 cm below the bifurcation on the far wall of the common carotid artery. The distance between the characteristic echoes from the lumen-intima and media-adventitia interfaces was measured. Our final IMT value was based on the mean value of three maximal IMT measurements. Subjects with plaques (focal structures that encroach into the arterial lumen of at least 0.5 mm or 50% of the surrounding IMT value or demonstrate a thickness >1.5 mm) were excluded from the study. Based on this criterion, only one person was excluded.
The study was performed in a quiet room under constant conditions between 7.30 and 10.30
In the next step systolic blood pressure (SBP), diastolic blood pressure (DBP) and mean arterial blood pressure (MAP) were measured continuously using noninvasive plethysmography (Colin 7000, Komaki-City, Japan).
The following variables were statistically analyzed by the statistic software SPSS 15.0: FMD, EMAD, ADC and IMT. Before using one-way analysis of variance (ANOVA) test a clearly normal distribution of FMD, EMAD, ADC and IMT was found in all three groups. One-way ANOVA with Bonferoni correction was used to compare the relative responses of FMD, EMAD, ADC and IMT among the groups of healthy subjects, and those who had diagnoses of either migraine with aura (MwA) or migraine without aura (MwoA). An independent t-test was used to compare FMD, EMAD, ADC and IMT between the group of healthy subjects and migraine patients and between MwA and MwoA. We also analyzed the association between FMD and IMT in healthy subjects and all migraine patients.
Results
The median monthly headache attack frequency in migraine patients over the previous year was 2.0 ± 1.1 (MwA 1.7 ± 1.0, MwoA 2.3 ± 1.1). The mean duration of the migraine attacks was 10.9 ± 7.6 years (MwA 11.5 ± 8.1 years, MwoA 10.3 ± 7.3 years).
All subjects had normal values of systolic blood pressure, diastolic blood pressure, mean arterial pressure, total cholesterol, HDL, LDL, triglycerides, glucose and body mass index (Table 1). Furthermore, we did not find any difference in the basal EMAD in the right brachial artery between healthy subjects, migraine patients with MwA and MwoA (Table 2).
We did not find any statistically significant difference in values of IMT between the group of healthy subjects and the group of migraine patients (p = .90; Table 2). In the next step of analysis, one-way ANOVA with Bonferoni correction did not show significant any difference in carotid IMT between the group of healthy subjects, MwA and MwoA (p = .76; Figure 1). Also we did not find any difference in IMT between MwA and MwoA (p = .43; Table 2). In that way we convincingly excluded the possibility of early atherosclerotic process in all groups of tested subjects.
Comparison of IMT in healthy subjects, migraine patients with aura (MwA) and without aura (MwoA) (p = 0.76). EMAD (basal, peak), ADC, FMD and IMT in all migraine patients, MwA and MwoA and healthy subjects MwA = migraine with aura, MwoA = migraine without aura, EMAD = end-diastolic mean arterial diameter, ADC = absolute diameter change, FMD = flow mediated vasodilatation, IMT = intima media thickness.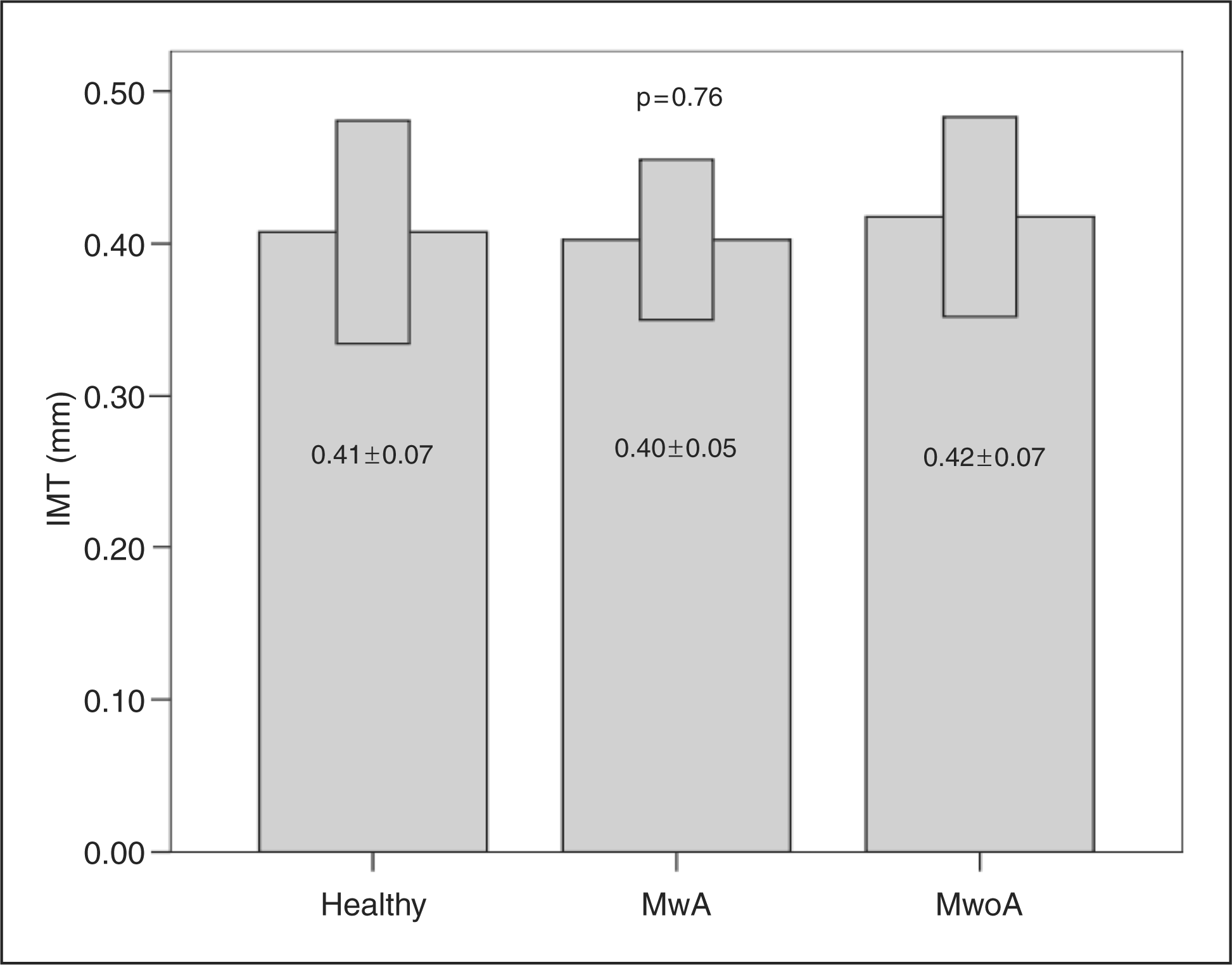

There were no differences in post-hyperemic EMAD between healthy subjects, migraine patients with MwA and MwoA (Table 2). In addition, we did not find any difference in ADC between healthy subjects and all migraine patients (p = .97; Table 2), the group of healthy subjects, migraine patients with MwA and MwoA (p = .98; Table 2) and the group of migraine patients with MwA and MwoA (p = .87; Table 2).
The most important finding was that we did not find any difference in FMD between healthy subjects and migraine patients (p = .96; Table 2). One-way ANOVA with Bonferoni correction did not show any difference between the group of healthy subjects, migraine patients with MwA and MwoA (p = .99; Figure 2). In addition, we did not find any difference between the group of migraine patients with MwA and MwoA (p = .99; Table 2).
Comparison of FMD in healthy subjects, migraine patients with aura (MwA) and without aura (MwoA) (p = 0.99).
In the last step, analysis of correlations did not show any significant association between IMT of the common carotid artery and FMD in the group of healthy subjects (p = .145; r2 = 0.114; Figure 3), and in all migraine patients (p = .148; r2 = 0.054; Figure 4).
The linear regression analysis of FMD to IMT in healthy subjects (p = 0.145; r2 = 0.114). The linear regression analysis of FMD to IMT in all migraine patients (p = 0.148; r2 = 0.054).

Discussion
The main finding of our study was the absence of any difference in FMD between migraine patients without comorbidities and healthy subjects, or between migraine patients without comorbidities with and without aura.
In comparison to the other studies that evaluated systemic endothelial function in migraine patients, our study strictly excluded all confounding factors. To the best of our knowledge, previous studies did not measure and compare carotid IMT in migraine patients and healthy subjects. It is well known that carotid IMT excludes the possibility of early atherosclerotic process and associated endothelial dysfunction (30–32). In our study we did not obtain any difference in carotid IMT between healthy subjects and migraine patients without comorbidities. As no differences in FMD were noted between healthy subjects and migraine patients without comorbidities during the interictal period, evidently those migraine patients had intact systemic endothelial function.
Our findings of non-altered FMD in migraine patients without comorbidities are in agreement with the results of Silva et al., who also did not find altered FMD during the interictal period in migraine patients (26). They concluded that migraine might not be related to endothelial dysfunction. On the contrary, de Hoon and Vanmolkot showed altered reactivity of the systemic vasculature (14,25). Yetkin et al. also proposed that migraine might be a local manifestation of systemic vascular abnormality (21,22). Association of migraine with ischemic heart disease in both women and men could confirm these speculations, but Schurks et al. made a systematic review and meta-analysis of current evidence and they did not find an association between any migraine type and myocardial infarction (3–5,33). They found only the known association between ischemic stroke and MwA. It is known from previous studies that stroke can occur during and between migraine attacks (1,2). Some studies reported that young women and patients with MwA are more prone to this kind of stroke and that the association is independent of other cardiovascular risk factors (34–37). Nevertheless, the risk of stroke in patients with migraine is not restricted to women. A large-scale epidemiologic study in the United States, the National Health and Nutrition Evaluation Survey (NHANES), reported the risk of stroke to be doubled in patients with migraine, both male and female (38). But there is probably an underestimation of the real extent of brain injury in migraine patients in the general population.
On the basis of our own and previous findings, we can say that there is no strong evidence for impaired FMD in migraine patients. FMD, procoagulatory and proinflammatory states as markers of endothelial activation could reflect endothelial dysfunction (13). However, we did not measure circulation markers of endothelial dysfunction, as we believe that FMD is a more direct reflection of endothelial dysfunction.
On the other hand, we cannot exclude isolated cerebral endothelial dysfunction. Recently, Pretnar-Oblak et al. showed that cerebral and systemic endothelial function may not be closely associated (39). Similarly, Kruit et al. reported an increased prevalence of posterior circulation hyperintensive, ischemic-like lesions in migraine patients (40). They also reported that patients with MwA had a higher prevalence of these lesions compared with MwoA and controls. Again, the prevalence of cardiovascular risk factors was not higher in migraine patients with posterior circulation infarct-like lesions compared with those without lesions. This may be due to low cerebral flow, below an ischemic threshold, during and after migraine attacks (41–45). Subclinical cerebellar dysfunction has been also reported in migraine patients (46,47). A significantly higher prevalence of brain stem hyperintensive lesion was also described in migraine patients compared to controls (48). The precise etiology of these lesions in migraine, and the reasons why migraine patients seem to be more susceptible to these kind of vasculo-ischemic brain changes, remain unknown. There are some studies that compared cerebrovascular reactivity (CVR) to carbon dioxide (CO2) levels in migraine patients (49–55). Hyper-reactivity, hyporeactivity and no difference in reactivity were observed. However, there are few studies that have specifically compared CVR to CO2 between both cerebral circulations. Silvestrini et al. have shown that migraine patients with MwA had significantly lower vascular reactivity in the basilar artery (56). These findings suggest that endothelial dysfunction may be primarily located in the arteries of the cerebral circulation, particularly in the territory of the posterior cerebral circulation.
Taking these and our results together, we can conclude that systemic endothelial function is not altered during the interictal period in migraine patients without comorbidities, with and without aura, compared to healthy subjects. In light of these findings, however, the investigation of cerebral endothelial function in migraine patients would be beneficial.
Footnotes
Acknowledgments
The author expresses gratitude to Valentin Beznik and Marjeta Švigelj for technical assistance, and special thanks to all study volunteers.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflicting interests
The author declares no conflict of interest.
