Abstract
Background
Change in headache diagnoses over time within the same individual is not well studied in the adult population. In this study, we prospectively examined the individual variation of migraine and tension-type headache (TTH) diagnoses over time.
Methods
As part of the epidemiological Deutsche Migräne und Kopfschmerzgesellschaft (DMKG) headache study, 1312 participants were personally interviewed and 1122 responded to a second mailed questionnaire 2.2 years later. Headaches were assigned to migraine or TTH at two different points in time using the International Headache Classification, ICHD-II. We used broad (definite and probable subtypes) and strict (only definite type) definitions of migraine and TTH.
Results
Using the broad definition increased the reproducibility of migraine diagnosis from 48.0% to 62.0% and of TTH from 59.0% to 65.0%. A constant TTH diagnosis was related to a higher social status (OR 2.81; 95% CI 1.43–5.53) a higher level of education (OR 1.96; 95% CI 1.00–3.85) and physical inactivity (OR 2.28; 95% CI 1.16–4.49). A constant diagnosis of definite migraine was associated with severe headache (OR 2.64; 95% CI 0.97–7.21) and frequent use of headache medication (OR 4.73; 95% CI 0.95–23.60). The result that coexisting TTH decreased the likelihood of a constant migraine (OR 0.29; 95% CI 0.10–0.85) is assumed to indicate response variability.
Conclusions
In epidemiological studies, definite and probable subtypes should be included in the diagnosis to increase the diagnostic accuracy.
Introduction
Change in headache diagnoses over time in the same individual is a known phenomenon (1,2), but has been rarely studied in the adult population. In general, a headache diagnosis is based on assigning headache characteristics and accompanying features to diagnostic criteria. The latter, as presented in classification systems, describes a specific cluster of features which are characteristic for a specific headache type, such as migraine or tension-type headache (TTH). The only accepted headache classification system worldwide was first introduced in 1988 by the International Headache Society (IHS), and an updated edition was published in 2004 (ICHD-II).
In epidemiological studies, headache features are reported by the respondent in interviews or questionnaires and subsequently assigned to the respective headache category, if the criteria are fulfilled. Variability in the diagnostic classification of headaches, reported by the same individual at two or more different points in time, may occur for two major reasons. First, a decrease or a progression of headache characteristics may cause a ‘true’ evolution from one headache category to another (1). Age, female gender, low education and coexisting further headaches have been reported to be associated with a worse prognosis of migraine and TTH (1,3), meaning the evolution from a less to a more disabling headache subtype. Second, individual response variability over time may cause inconsistent diagnostic classifications (4). The heterogeneous character of migraine, which simply means that migraine features may vary in severity between and within individuals (4–7), and an overlap as well as the possible coexistence of migraine and TTH characteristics, have been suggested to increase response variability (4).
The purpose of this study was to examine change in migraine and TTH classifications over 2.2 years of follow-up within the Dortmunder Health Study (DHS) using the ICHD-II. Sociodemographic factors and specific headache characteristics were analysed to determine if variability in diagnostic assignment was related to response variability or to a natural evolution of the initial headache symptoms.
Methods
Data collection procedure
The DHS is part of the Deutsche Migräne und Kopfschmerzgesellschaft (DMKG) headache study. The detailed methodology of this study has been previously described (8–10). In short, between 2003 and 2004 an age- and gender-stratified random sample, restricted to the age groups 25–75 years, was drawn from the population register of the city of Dortmund, a town with more than 500,000 inhabitants in western Germany. Persons who did not participate in the personal interview plus examination in the study centre were asked to fill in a mailed questionnaire. The overall response rate was 66.9%, yielding 2291 participants (1312 interview and 979 questionnaire participants). As the questionnaire did not allow headache classification according to the IHS criteria, only interview participants (n = 1312) were included in this analysis. Participants of the interview did not differ from questionnaire participants in terms of age and gender (52.1 vs 53.0 years), but 12-month headache reports were significantly different (52.8% vs 68.8%, p < 0.001) (10).
In 2006, a follow-up of the DHS was conducted via mailed questionnaire on average 2.2 years after the baseline assessment. This questionnaire included a headache question module that was identical to the headache questions of the baseline interview. The follow-up response among those initially interviewed was 85.5% (n = 1122). Participants of the follow-up assessment were significantly older than non-participants (52.9 vs 47.5 years, p < 0.001), and the prevalence of current headaches at baseline was lower in the former group (12-month baseline prevalence, 51.2% vs 62.6%, p = 0.003) (10).
Headache assessment and diagnosis
Based on the ICHD-II criteria of the IHS (11,12), a headache question module was developed that allowed retrospective assessment of headaches. This module was used in the baseline (interview) and follow-up (questionnaire) assessment. It included 20 items on headache characteristics necessary to classify the common primary headache types, migraine and TTH. In the first question, participants were asked if they have experienced headaches during the 12 months prior to the assessment. Those with headaches were asked to report the features of their headaches in subsequent questions.
Based on the answers, the reported symptoms were assigned to migraine, TTH or unclassified headaches using the ICHD-II. If ICHD-II criteria 1.1 and 1.2 (migraine without and with aura) were fulfilled, headaches were assigned to definite migraine (definition 1). A probable migraine was present if the headaches met all but one criteria of definite migraine (ICHD-II criteria 1.6). Headaches were assigned to definite TTH (definition 1), if ICHD-II criteria 2.1 to 2.3 (infrequent episodic, frequent episodic and chronic TTH) and an assignment to probable TTH if all but one criteria of definite TTH (ICHD-II criteria 2.4) were met. As probable migraine and probable TTH criteria are not mutually exclusive, a clear assignment to either probable migraine or probable TTH is not possible (8). As a consequence, participants fulfilling criteria of probable migraine and of probable TTH, were assigned to the category of ‘probable migraine plus probable TTH’, which we also referred to as mixed probable subtypes.
Headaches that were not assigned to any of these categories were referred to as unclassified headaches. If no headaches were reported during the 12 months prior to the interview, participants were categorised as headache-free.
We used two definitions of migraine and TTH. The broad definition included definite and probable subtypes of migraine or TTH, respectively. The strict definition included participants that were assigned to the definite subtype only.
Assessment of covariates
Information on sociodemographic data included age, gender, immigrant background and partnership status. Immigrant background was determined by assessing own and parental place of birth and nationality. Partnership status included the two categories ‘living in partnership’ and ‘not living in partnership’.
Social status was quantified using the social class index by Winkler (Winkler index) (13). The index score was calculated from education (school and vocational), occupation and income and ranged from 3 to 21. A score from 3 to 8 indicates a lower social status, whereas a middle social status is scored from 9 to 14. A score of more than 14 corresponds to a higher social status. Given the few participants in the lower category of social status, we dichotomised social status by merging the low and middle categories.
Education was assessed by asking the participants’ highest level of school education. An education of more than 10 years was defined if participants finished at least secondary school. Otherwise, less than 10 years of education was assigned.
A more detailed description on the assessment of lifestyle factors can been found in Winter et al. (14). In brief, smoking status encompassed the three categories never, ex or current smoker. We compared current smokers and non-smokers (including never and ex-smokers). The body mass index (BMI) was calculated by weight and height. BMI was dichotomised into the categories of ≥ 30 kg/m2 and <30 kg/m2. Physical activity was assessed by grouping the participants’ weekly levels of activity into four categories (never, less than 1 hour, 1–2 hours, more than 2 hours per week) In this study, active participants were those who reported regular activity of more than 2 hours per week. Alcohol consumption was assessed by the participants’ drinking behaviour of the last work day and of the last weekend, and included the amount of wine and beer in litres and spirits in number of glasses. Alcohol consumption was then calculated in grams per day. A daily alcohol consumption of more than 20 g per day in women and more than 30 g per day in men was considered as a risky level (14).
Headache characteristics included headache frequency, headache severity and the frequency of used headache medication. In migraine, further characteristics were the presence of aura symptoms, of coexisting TTH and of a familial history of migraine. In TTH the presence of migraine symptoms was considered as a further headache characteristic. A frequent headache was defined if the diagnosed headache occurred on at least 3 days per month. Frequent use of headache medication was defined as taking medication on at least 3 days per month.
Data analysis and statistical methods
Headaches were categorised at baseline and at follow-up. If a headache was assigned to the same category at both time points, a constant diagnosis was assumed; otherwise a change in headache diagnosis was accepted. We calculated the proportion of constant migraine and TTH diagnoses for the broad and the strict definitions. The chance corrected agreement between the baseline and follow-up diagnoses was calculated using Cohen’s kappa statistic.
We investigated the relationship between a constant headache diagnosis of definite migraine (ICHD-II code 1.1 and 1.2) or definite TTH (ICHD-II code 2.1-2.3) and sociodemographic, lifestyle as well as headache characterising variables, which were selected on the basis of results reported by other studies (3).
Differences in categorical variables were compared using the chi-square test or Fisher’s exact test (if cell number was five or less). Continuous variables were described with means and standard deviations, and compared using the Student’s t test. To evaluate factors assumed to be associated with a constant diagnosis of definite migraine/definite TTH, we performed multivariable logistic regression models. All data analyses were carried out using SAS statistical software, version 9.2.
Results
Baseline characteristics of the study population.
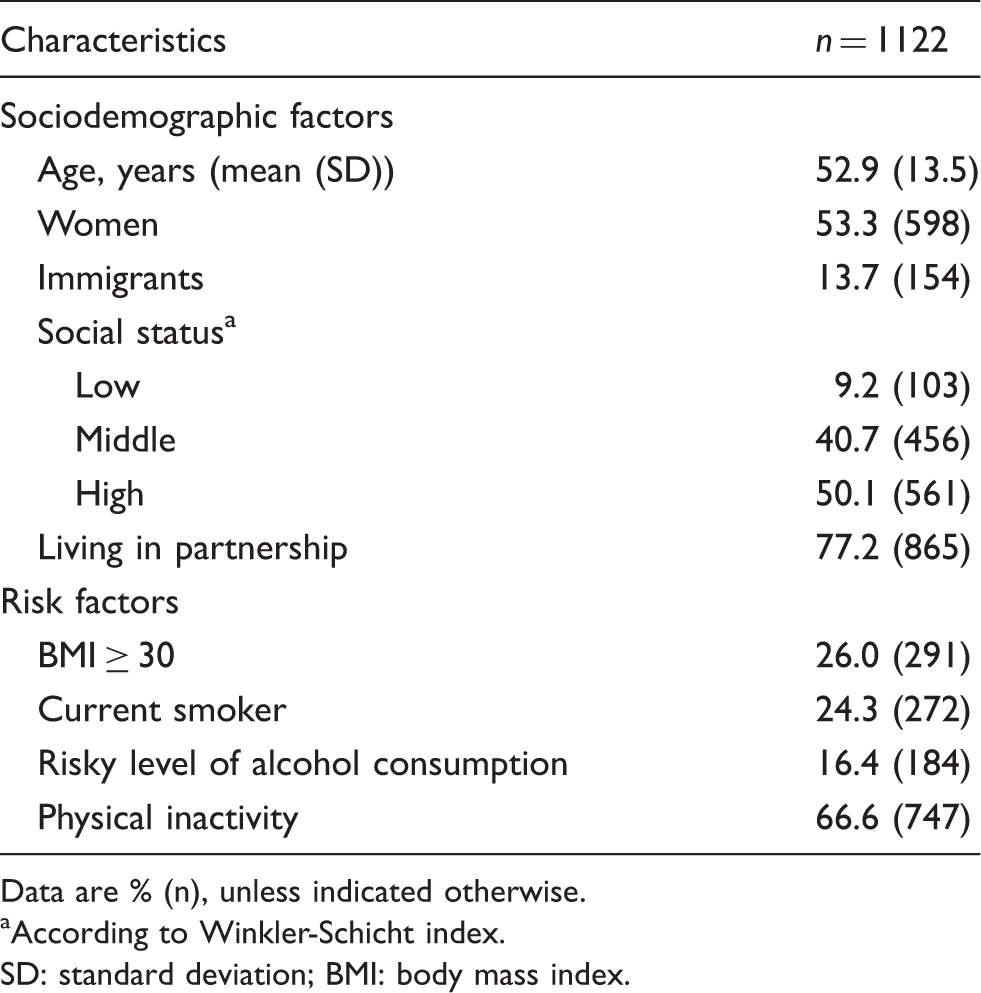
Data are % (n), unless indicated otherwise.
According to Winkler-Schicht index.
SD: standard deviation; BMI: body mass index.
Of the 1122 participants with baseline and follow-up assessment, 51.2% (n = 573) reported headaches during the year prior to the baseline assessment. Of those with headaches, 68.8% (n = 394) reported headache characteristics that fulfilled the criteria of either definite TTH (35.3%; n = 202), probable TTH (21.3%; n = 122), or probable TTH plus probable migraine (mixed probable TTH, 12.2%; n = 70). Characteristics that met criteria of migraine (broad definition) were reported by 37.4% (n = 214) of participants, including 16.6% (n = 95) participants assigned to definite migraine, 8.6% (n = 49) to probable migraine and 12.2% (n = 70) to mixed probable migraine.
Number of participants assigned to tension-type headache and its subcategories at baseline and follow-up.

κ-coefficient is shown for the broad and strict definition of tension-type headache.
Headaches were classified according to the 2nd edition of the IHS criteria (ICHD-II).
TTH: definite tension-type headache; PROTTH: probable tension-type headache ; Mixed PTTH: ‘probable migraine plus probable tension-type headache’.
Number of participants assigned to migraine and its subcategories at baseline and follow-up.

κ-coefficient is shown for the broad and strict definition of migraine.
Headaches were classified according to the 2nd edition of the IHS criteria (ICHD-2).
MIG: migraine; PROMIG: probable migraine; Mixed PMIG: ‘probable migraine plus probable tension-type headache’.
Chance corrected agreement between baseline and follow-up diagnoses
We calculated the kappa-coefficient of the broad and strict definition of migraine and TTH using Cohen’s kappa statistic. Out of the group of ‘no or other headaches than TTH’ at baseline (n = 728), 78.6% (n = 572) of participants were reassigned to that group, whereas 21.4% (n = 156) were assigned to TTH. In TTH, the kappa-coefficient of the broad definition was κ = 0.44 (Table 2a). Focussing on the strict definition slightly increased the kappa-coefficient to κ = 0.50.
Out of the 908 participants without migraine at baseline, 91.3% (n = 829) were reassigned to that group and 8.7% (n = 79) were assigned to migraine at follow-up, yielding a kappa-coefficient of κ = 0.54, indicating moderate agreement. The strict definition resulted in a decreased agreement of κ = 0.47 (Table 2b).
Determinants of a constant diagnosis
To investigate potential determinants of a constant diagnosis of definite migraine, we compared the baseline characteristics of participants with a constant versus a changed headache diagnosis. Participants with a constant diagnosis tended to be younger (43.6 vs 47.8 years, p = 0.068), were more likely to have severe headaches (78.3% vs 53.2%, p < 0.010) and to more frequently use headache medication (30.4% vs 12.2%, p = 0.030). Across the 95 participants assigned to definite migraine, based on symptoms of their most severe headache, 67 (70.5%) additionally fulfilled three criteria of TTH. We called this group ‘participants with coexisting TTH symptoms’. We found that coexisting TTH symptoms were inversely associated with a constant migraine diagnosis at follow-up (56.5% vs 83.7%, p = 0.004).
Odds ratios for potential determinants of a constant migraine diagnosis (strict definition).
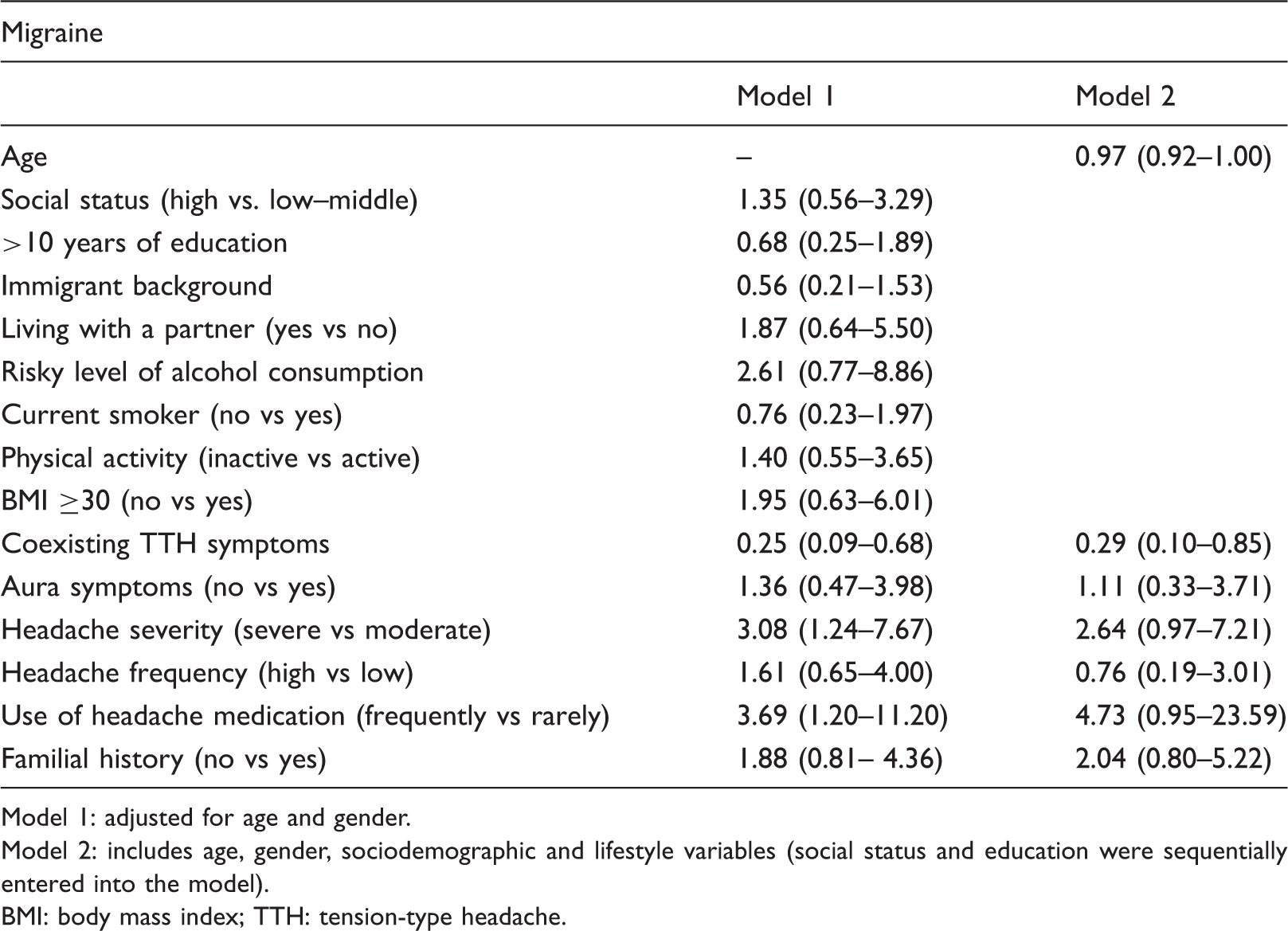
Model 1: adjusted for age and gender.
Model 2: includes age, gender, sociodemographic and lifestyle variables (social status and education were sequentially entered into the model).
BMI: body mass index; TTH: tension-type headache.
No association was found for sociodemographic or lifestyle variables (data not shown), but BMI tended to increase the likelihood of a constant migraine (OR 3.40; 95% CI 0.97–12.14; p = 0.056).
Odds ratios for potential determinants of a constant tension-type headache diagnosis (strict definition).

Model 1: adjusted for age and gender.
Model 2: includes age, gender, sociodemographic and lifestyle variables (social status and education were sequentially entered to the model).
BMI: body mass index; TTH: tension-type headache.
Reproducibility of IHS criteria for migraine and TTH presented as amount (number) of participants fulfilling a criterion at baseline and at follow-up.

TTH: tension-type headache.
In migraine, ICHD-II criterion B (headache attacks lasting 4–72 hours) was the least reproducible criterion (63.2%), whereas criterion A (at least five attacks in life) was the most reproducible (93.7%).
Discussion
In this population-based study, we examined the diagnostic agreement of migraine and TTH in the same individual assessed at two points in time, 2 years apart, using two different definitions. The broader definition of migraine increased the reproducibility of an initial migraine diagnosis, because half of participants with inconstant definite migraine drifted into the category of mixed or pure probable migraine. Thus, our results support the introduction of probable migraine into the ICHD-II, to account for some degree of heterogeneity in migraine (7). A higher reproducibility between the broad and strict definitions was also observed in TTH, where again half of inconstant definite TTH drifted to mixed or pure TTH. However, it should be noted that the kappa-coefficient indicated a better agreement for the strict definition of TTH after accounting for chance.
Our results agree with previous studies, reporting variability in headache diagnosis over time (1,5). In a study by Nachit-Ouinekh et al., the stability of headache symptoms was investigated in 1733 participants over a 1-year period (5). The authors used two different migraine definitions; the first included definite, and the second definite plus probable migraine. Using the first definition, 62.2% of participants were reassigned to migraine (IHS code 1.1 or 1.2), whereas the second definition resulted in 82.0% of reassignment. As in our study, the most irreproducible criterion was ‘duration of headaches lasting 4–72 hours’ (5), supporting the suggestion by Rasmussen et al. to reduce the lower limit to 2 hours to improve specificity and sensitivity of this feature (5,15).
Coexisting TTH in migraine has been reported to increase response variability, because patients cannot clearly tell symptoms apart (4,5,16,17). Features of the most recent headache are also more likely to be remembered, which has been described as ‘recency effect’ by Lipton et al. (4). As we were able to show that migraine diagnoses were less constant if TTH coexisted, we assume that in our study coexisting TTH decreased the diagnostic accuracy of migraine due to response variance. However, this does not necessarily mean that the two different headache types switch within one patient. Alternatively, a probable subtype of migraine or TTH is a milder biological form of the other headache type. As, in our data, the likelihood that a probable TTH turned out to be another or no headache was higher than that a probable migraine did so (55% vs 43%), it is more likely that a probable TTH is a milder form of migraine than vice versa. Thus, it might be justified in a concurrent diagnostic situation between probable TTH and probable migraine to define probable migraine as the leading diagnosis. On the other hand, it was more likely that a mixed probable TTH (meaning probable TTH plus probable migraine) turned out to be TTH, than that a mixed probable migraine turned out to be migraine. However, 73% of participants with this mixed form at baseline did not meet full migraine criteria because of headaches that lasted 30 minutes to 4 hours. If criteria were reduced from 4 to 2 hours as suggested by Rasmussen (see above) (15), these participants were assigned to migraine. Furthermore, the uncertainty in the differentiation between probable migraine and probable TTH also supports the idea of some shared biological mechanisms in both headache types, which might explain symptom overlaps.
We found that a constant diagnosis of definite migraine was associated with severe (compared to moderate) headache. Severe attacks are more likely to be remembered and some mild migraine attacks are sometimes misinterpreted as TTH (4,17). Thus, a severe migraine can either be better distinguished from a coexisting TTH or it decreases the variability of symptom reporting due to better remembering symptoms. In this case, a decrease in response variability would explain the association between severe migraine and a constant diagnosis. But it is also possible that severe migraine predicts a worse prognosis simply because of a higher severity level of migraine.
We observed that BMI tended to increase the likelihood of constant migraine. In a study by Winter and colleagues (18) an association between specific migraine features and BMI was reported in women. To control for BMI as a potential confounder, we adjusted headache characteristics for BMI which did not change the estimates, indicating that the reported effects of headache characteristics were independently associated with constant migraine.
A constant diagnosis of TTH was related to a higher level of education, a higher social status and physical inactivity. In contrast to our result, Lyngberg et al. reported that a poor outcome of TTH was associated with marital status but not with education or physical activity (3). However, some published studies reported an association between high (19) or low (20) educational level and prevalent TTH. Our results indirectly support the assumption that TTH prevalence is higher in higher educated or socioeconomically higher stated individuals. In the here reported situation, the higher prevalence of TTH would be explained through a longer disease duration in socioeconomically higher stated groups, probably because of a more constant exposure to trigger factors.
Our study has strengths and limitations. Among the limitations is the fact that headache diagnoses were not made by a headache specialist. This may result in diagnostic inaccuracy. However, data on headache diagnoses in epidemiological studies are generally obtained by using interviews and questionnaires. Another limitation is that the follow-up assessment was based on self-completion questionnaires, whereas baseline assessment was done by face to face interviews. Although the number and wording of questions in the headache module was identical in both assessments, it is possible that the precision of headache type classification (e.g. TTH or migraine) might be lower using questionnaires. This would result in higher response variance due to the different assessment techniques used at baseline and at follow-up. Furthermore, follow-up response was lower in participants with headaches at baseline. This may result in an overestimation of constant diagnoses, because it is possible that these non-responders no longer had headaches at follow-up.
Among the strengths of this study is the high response rate of 85.0%. Furthermore, this is a population-based sample that also included participants with light to moderate headaches and thus greater symptom variability can be assumed.
In summary, we observed that definite migraine accompanied by coexisting TTH, and less severe forms of definite migraine are prone to response variability and subsequent misclassification of headache types, supporting the suggestion to exclude TTH from the population at risk of migraine and migraine from the population at risk of TTH in population-based incidence studies (10). We believe that the inclusion of probable subtypes into the definition of migraine and TTH increases the diagnostic accuracy in the setting of epidemiological studies.
Footnotes
Moderate migraine and coexisting TTH decrease the diagnostic accuracy of migraine and need special attention.
Non-fulfilment of complete migraine criteria is mainly caused by attacks lasting less than 4 hours.
In epidemiological settings, probable subtypes should be included in the definitions of migraine and TTH to increase diagnostic accuracy.
Funding
The Dortmund Health Study was supported by the German Migraine and Headache Society (DMKG) and by unrestricted grants of equal share from Almirall, Astra Zeneca, Berlin Chemie, Boehringer, Boots Health Care, Glaxo-Smith-Kline, Janssen Cilag, McNeil Pharma, MSD Sharp & Dohme and Pfizer to the University of Muenster.
Conflict of interest
L. Khil has nothing to disclose. Dr Straube has received honoraries by Allergan, Berlin Chemie, Desitin, Boehringer Ingelheim, MSD, Pfizer, and grants from the German Science foundation, German Minister of Research and Education and the Kröner-Fresenius foundation. Dr Evers has received unrestricted grants and honoraria in the last 2 years by Addex, Allergan, Berlin Chemie, Boehringer, Colucid, GSK, Ipsen, Merz, MSD, Pfizer, Eisai, UCB. Dr Berger has received the following research support: for the conduction of DMKG Headache Study unrestricted grants of equal share from the German Migraine and Headache Society and a consortium formed by Almirall, Astra-Zeneca, Berlin-Chemie, Boehringer Ingelheim Pharma, Boots Healthcare, GlaxoSmithKline, Janssen Cilag, McNeil Pharmaceuticals, MSD Sharp & Dohme, Pfizer to the University of Muenster; for the ‘Course of Restless Legs Syndrome Study’ unrestricted grants from the German Restless Legs Society and a consortium formed by Boehringer Ingelheim Pharma, Mundipharma Research, Neurobiotec, UCB (Schwarz Pharma) and Roche Pharma to the University of Muenster; from the German Minister of Research and Education for several research projects within the German Competence Nets Stroke, Diabetes, Multiple Sclerosis and an ongoing cohort study on depression and subclinical arteriosclerosis.
