Abstract
Background
Medication overuse headache (MOH) is a disabling health problem. Convincing evidence attributes a pathophysiologic role to central sensitization. By recording somatosensory evoked potentials (SSEPs) in patients with MOH, we observed increased sensitization and deficient habituation to repetitive sensory stimuli consistent with drug overuse. The renin–angiotensin system in the brain seems to play a relevant role in neural plasticity and dependence behavior. We therefore sought differences in SSEP sensitization and habituation in patients with MOH who underwent angiotensin converting enzyme (ACE) I/D polymorphism analysis.
Methods
We recorded median-nerve SSEPs (two blocks of 100 sweeps) in 43 patients with MOH. We measured N20–P25 amplitudes, and assessed sensitization using the first block amplitudes, and habituation using amplitude changes between the two sequential blocks. According to their genotype, subjects were divided into three groups: “D/D”, “D/I” and “I/I” carriers.
Results
The habituation slope of the two SSEP block amplitudes was significantly increased in the D/D subgroup (n = 16) with respect to that of the I/I subgroup (n = 6), with the D/I subgroup (n = 21) falling in between. In D/D carriers, the habituation slope correlated positively with the duration of the overuse headache, and the first SSEP block amplitudes, a measure of sensitization, increased in strict relationship with the type of overused medication in the MOH patients overall and in the D/D subgroup; this was not so in the D/I and I/I subgroups.
Conclusion
In patients with MOH, the homozygote D/D ACE polymorphism influences habituation and sensitization to repeated sensory stimuli in strict relationship with medication overuse. We suggest that angiotensin peptides influence neuronal mechanisms of plasticity by interacting with central monoaminergic synaptic transmission.
Keywords
Introduction
The 2nd Edition of the International Classification of Headache Disorders (ICHD-II), published in 2004, recognized medication overuse headache (MOH) as a secondary form of headache usually developing in episodic migraine patients after prolonged (over 3 months) symptomatic medication overuse. Although the original criteria for MOH have already been revised (1), the classification of MOH still remains a debated topic (2). Because one subgroup of MOH patients is characterized by a tendency to take acute medications frequently and compulsively, MOH was thought to belong to the spectrum of addictive behaviors (3,4). As a matter of fact, MOH may be complicated by the presence of other substance abuses, including alcohol or cigarettes, which, among others, were recognized as risk factors for relapse into overuse within 1 year after medication withdrawal (5). How medication overuse leads to chronic headache is still unknown; however, possible culprits for pain chronification include defective central pain control systems (6) and nociceptive system sensitization (7).
Evidence has been recently found in favor of central sensitization in MOH by recording cortical somatosensory evoked potentials (SSEPs). Whereas in episodic migraine patients an initial decreased response amplitude and a subsequent lack of habituation is detectable during the pain-free period (8), in MOH patients persistent sensitization of the somatosensory cortex was found as reflected by an initial increased response amplitude with a further increase during stimuli repetitions. This phenomenon was dependent on the drug of overuse, as it is maximal in patients overusing non-steroidal anti-inflammatory drugs (NSAIDs) and almost non-existent or masked in those who overuse only triptans (9).
Habituation and sensitization are two opposing processes that compete to determine the final behavioral outcome after a stimulus sequence (10). These two phenomena are considered most useful for studying the biological substrates of behavior as well as the mechanisms of learning and neuronal plasticity (11).
The renin–angiotensin system (RAS) plays an important role in cardiovascular and fluid homeostasis through the highly active octapeptide angiotensin II, which is synthesized from the renin-derived decapeptide angiotensin I (ANG I) by the cleaving activity of the angiotensin converting enzyme (ACE). Excessive upregulation of the RAS is capable of promoting inflammation and endothelial dysfunction, and the latter effect has been claimed to explain the association of migraine with several cardiovascular disorders, including ischemic stroke, ischemic heart disease, and vasospastic disorders (12). In addition to the peripheral RAS, a complete RAS was described in the brain (13), where several studies have indicated that it plays a relevant role in neural plasticity and dependence behavior. Research suggests that ANG II has a number of actions in the brain (14). It works as a neurotransmitter and interacts with catecholamines, serotonin, and other peptides, and its receptors are located in widely distributed areas, such as the locus coeruleus, and dopaminergic regions (15), which are all known to be involved in migraine pathophysiology (16) and in its chronification (7). In migraine patients, during the interictal phase, angiotensin brain activity seems to be enhanced (17). Furthermore, ACE inhibitors (18,19) and ANG II receptor blockers (20) exert a prophylactic effect on migraine.
Angiotensin converting enzyme activity has a strong genetic determination (21). Carriers of the D/I and I/I polymorphisms of the ACE I/D gene polymorphism express reduced and intermediate angiotensinergic activity, respectively, whereas homozygotes for the “D” allele (D/D) are characterized by higher enzyme activity, with higher angiotensin activation. The incidence of the homozygote D/D polymorphism of the ACE gene was found to be higher in episodic migraine with (22) and without aura (23) than in controls.
The ACE D/D genotype seems to be an influencing factor in migraine attack frequency (24) and also in substance abuse behavior (25,26).
The aim of our study was to verify whether ACE polymorphisms could influence neural plasticity (habituation/sensitization) and the clinical features of MOH. Therefore, in the present study we measured SSEP amplitudes to low numbers of stimuli, as a marker of sensitization, and SSEP habituation over two sequential blocks during uninterrupted peripheral stimulation in a well-characterized group of MOH patients who underwent ACE I/D polymorphism analysis. We reasoned that the homozygote ACE D/D polymorphism would have a role in modifying some of the pathophysiologic features of MOH, acting on both the biological and behavioral nature of the disease by favoring somatosensory response sensitization and lack of habituation in strict relationship with the type of over-consumed drug and the duration of overuse headache.
Methods
Subjects
Forty-three consecutive patients diagnosed, during their first visit to our headache clinic, as affected by MOH, according to the revised ICHD-II criteria (1), were considered eligible for this study. The study was approved by the local ethics committee, and the patients were asked to give their informed consent to undergo study procedures. During the first visit stable blood pressure was measured. All MOH patients underwent the SSEP recordings in a pain-free state. Because MOH patients tend to take acute medications compulsively and frequently during the day, it was impossible to prevent them from taking medication on the day of recordings. However, we managed to perform the recordings at least 3 hours after the last medication intake.
The physicians, the molecular biologists, and the neurophysiologists were blinded to the ACE gene determination, the electrophysiology, and the clinical history, respectively.
Neurophysiologic measurements
The SSEPs were recorded according to methods described elsewhere (9). Briefly, we elicited SSEPs by electrical stimulation of the right median nerve at the wrist using constant current square wave pulses (0.1 ms width, cathode proximal) and a stimulus intensity set at two times the motor threshold. The active electrode was placed over the contralateral parietal area (C3′) referenced to ipsilateral ear lobe; the ground electrode was on the right arm. We collected 500 evoked responses for each subject at a stimulation rate of 4.4 Hz and digitally filtered the signal off-line between 0 Hz and 450 Hz, to identify the various SSEP components (N20, P25, and N33) according to their respective latencies. We measured the peak-to-peak amplitudes of the cortical N20–P25 component with a grand average. Thereafter, the early 200 evoked responses were partitioned into two sequential blocks of 100 responses. Each block was averaged (“block averages”) and analyzed for N20–P25 amplitudes. Habituation was expressed as the change in the N20–P25 amplitude in block 2 compared to block 1 and the slope of the linear regression line over the two blocks.
Genetic analysis
Patients underwent a 3 ml peripheral blood pick-up to perform the genomic DNA extraction from leukocytes. Genetic analysis was conducted with a standard method, as described in a previous paper (27), to detect the presence of the 287-base pair (bp) insertion (I)/deletion (D) polymorphism of intron 16 (rs4646994) of the ACE gene – located on chromosome 17 (17q23.3). According to their genotype, subjects were divided into three groups: “D/D” (homozygous for the D allele), “D/I” (heterozygous), and “I/I” (homozygous for the I allele) (22–25).
Statistical analysis
We used the Statistical Package for the Social Sciences (SPSS) for Windows, version 19.0 for all analyses. Neurophysiologic independent variables (SSEP N20, P25, and N33 latencies, N20–P25 slope of the linear regression line over the two blocks) and the clinical data (disease duration, days with headache, number of tablets taken per month, duration of overuse headache, systolic and diastolic blood pressure) were compared, adopting the analysis of variance (ANOVA) test taking “genotype” as dependent factor. As N20–P25 grand-average amplitude is not distributed normally, it was compared between subgroups adopting a non-parametric Kruskal-Wallis test. Pearson’s correlation coefficient was calculated to test correlations between SSEP amplitudes or habituation and clinical data (disease duration, days with headache, number of tablets taken per month, duration of overuse headache, type of overused medication, systolic and diastolic blood pressure).
Results
Clinical and demographic data for MOH patients carrying D/D, D/I and I/I.
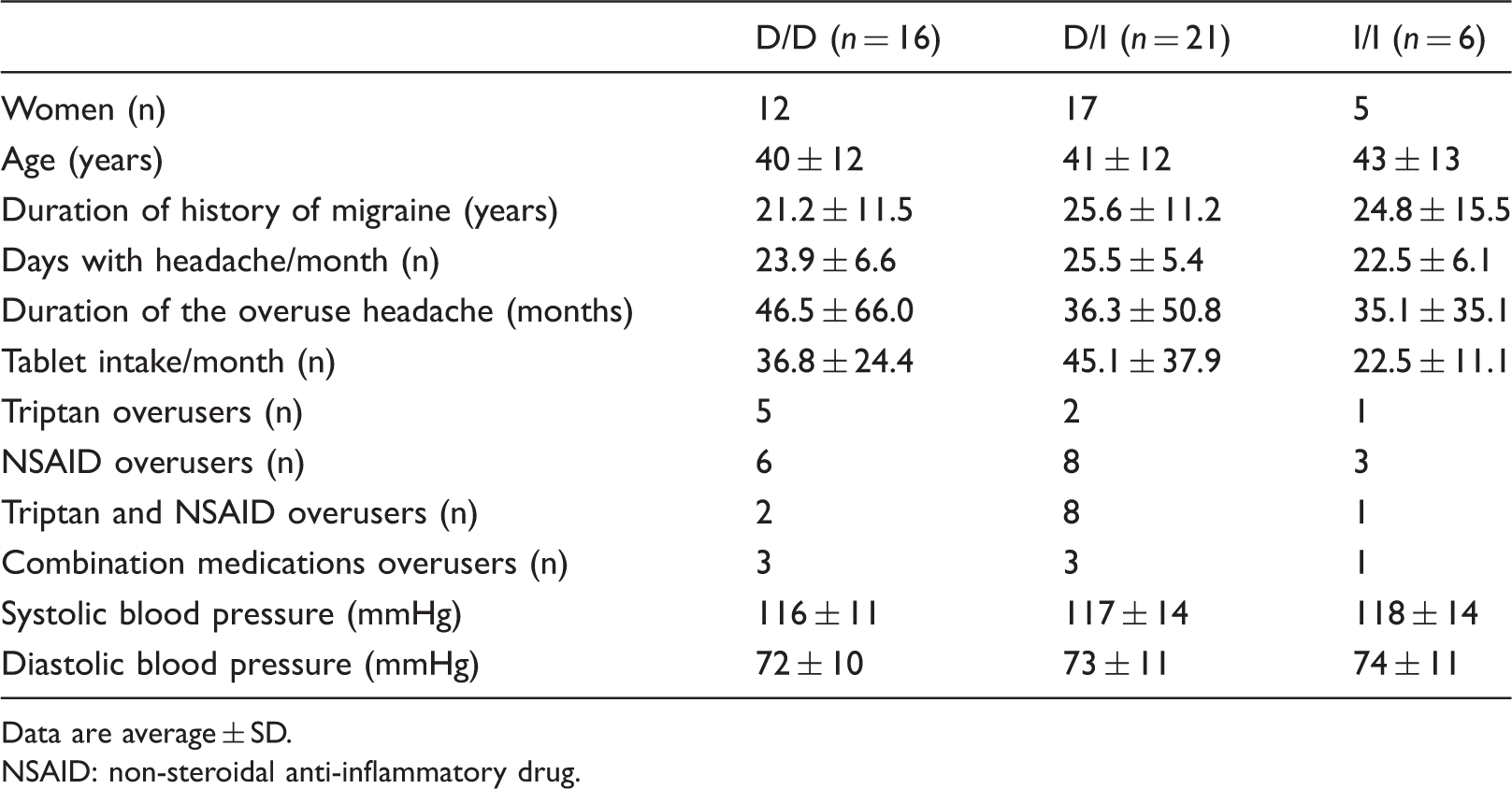
Data are average ± SD.
NSAID: non-steroidal anti-inflammatory drug.
When considering individual MOH patient subgroups, the SSEP N20, P25, and N33 latencies and the N20–P25 amplitudes were not different between D/D, D/I, and I/I (for each measure F(2,40), p > 0.05). ANOVA testing of SSEP block amplitude averages disclosed a significant interaction of polymorphism by block (F(2,40) = 3.233, p = 0.05), but not a main effect of block (F(2,40) = 0.005, p = 0.946) and polymorphism (F(2,40) = 1.210, p = 0.309) alone. Post hoc analysis showed that the slope of the two blocks of SSEP amplitudes was highest in the D/D subgroup (+0.45 ± 0.60), and smallest in the I/I subgroup (–0.49 ± 0.79, p = 0.02 vs. D/D), with the D/I subgroup falling in between (−0.004 ± 0.85, p = 0.07 vs. D/D) (Figure 1). Mean values of both systolic and diastolic blood pressures were not different between subgroups (F(2,40), p > 0.05) (Table 1).
Somatosensory evoked potential (SSEP) amplitude change over two blocks of averages in each angiotensin converting enzyme (ACE) polymorphism subgroup (D/D, D/I, and I/I). Somatosensory evoked potentials data of MOH patients carrying D/D, D/I, and I/I. ANOVA vs I/I, p < 0.05. Data are average ± SD. MOH: medication overuse headache.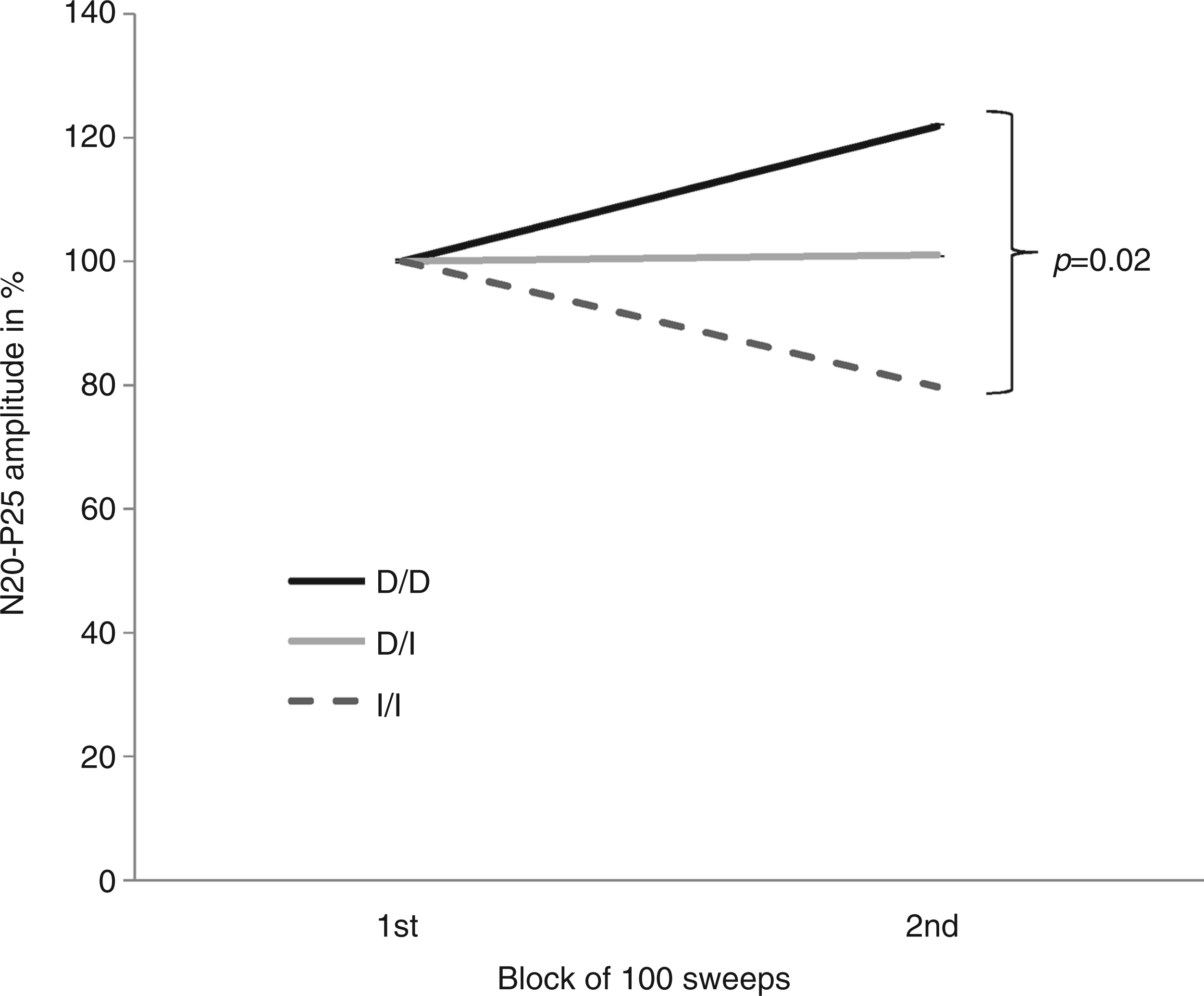

Pearson’s test disclosed a positive correlation between the SSEP amplitude slope and the duration of the overuse period in the whole MOH group (r = 0.315, p = 0.040). Focusing the analysis on the genetic subgroups, this positive relationship was highly significant in the D/D carriers (r = 0.751, p = 0.001), but not in the D/I (r = 0.00, p = 1.00) and I/I carriers (r = 0.674, p = 0.212) (Figure 2).
In the D/D medication overuse headache (MOH) subgroup the N20–P25 amplitude slope correlated positively with duration of the overuse headache (months). This relationship was absent in both the D/I and I/I subgroups.
No differences were found, in terms of the type of drugs overused, between subgroups (Pearson χ2 = 4,132; Montecarlo Exact Test p = 0.25). However, the SSEP amplitude of the first block increased in strict relationship with the type of overused medication in the MOH patients overall (r = 0.338, p = 0.02) and in the D/D (r = 0.534, p = 0.03) subgroup. In fact, the first amplitude block was lowest in the triptan overusers and progressively increased in NSAID, and combined triptan and NSAID overusers and reached the highest values in those overusing combination medications (Figure 3). The latter relationship was absent in both the D/I (r = 0.230, p = 0.304) and I/I (r = 0.256, p = 0.678) subgroups. Pearson’s test disclosed no significant correlation between clinical characteristics and both systolic and diastolic values in MOH patient subgroups.
In the D/D medication overuse headache (MOH) subgroup the N20–P25 first amplitude block increased in strict relationship with the type of overused medication. This relationship was absent in both the D/I and I/I subgroups.
Discussion
Our findings are consistent with the hypothesis that the angiotensinergic system influences neurophysiologic patterns in MOH patients in strict relationship with the type of over-consumed drug and duration of overuse headache. We observed that D/D homozygote carriers differ from D/I and I/I carriers in terms of response to repeated stimuli and the effects of the type of drug overused. In fact, although MOH patients overall did not habituate, as we were unable to find a significant repetition (block) effect on performing ANOVA, D/D carriers presented the highest grand-averaged amplitudes and the most severe deficit in habituation. This abnormal neurophysiologic pattern gradually disappeared in the D/I and in I/I carriers where the cortical response habituated normally.
It is well known that genetics strongly influences ACE activity (21). A 287-bp I/D polymorphism in intron 16 of the ACE gene was found to be associated with cellular and plasma ANG II levels (28), and several reports suggest that D/D genotype carriers have higher ACE activity with respect to I/I and D/I carriers (24,29,30). Although ACE polymorphisms account for only half of all of the variance in serum ACE levels, a significant relationship has been described between the polymorphism and the serum ACE level (29,30). Higher angiotensinergic activity was found to be associated with an increased risk of various diseases, such as diabetic nephropathy in type I diabetic patients (31), renal insufficiency (32), bipolar disorder (33), and schizophrenia (34). A complete angiotensinergic system seems to exist in the brain (13) and angiotensin, particularly its octapeptide ANG II, seems to have several functions in the central nervous system (15). There is an interaction between ANG II and catecholamines in the brain. Centrally injected renin increases dopamine and decreases norepinephrine turnover in the paraventricular and hypothalamic nuclei (35), as well as in the locus coeruleus (36), nucleus tractus solitarius, nucleus commissuralis and dorsal motor nucleus of the vagus (37). The latter effects are specifically mediated by ANG II and closely associated with blood pressure control (15). Angiotensin II affects 5-HT synthesis in the hypothalamus, brain stem and pineal gland of animals (38,39), but it does so dose-dependently as low ANG II concentrations decreased and high concentrations increased the rate-limiting enzyme of serotonin synthesis, tryptophan hydroxylase (38). These experimental observations are of particular interest because the strongest lack of habituation was seen in MOH D/D carriers. It is indeed well known that the so-called state-setting, chemically addressed connections originating in the brain stem and using, among others, dopamine, noradrenaline and serotonin as neurotransmitters, are crucial for the modulation of cortical excitability (40) including sensitization and habituation processes (41). The effects of these aminergic connections on the habituation/sensitization processes in animals vary with the concentration and duration of neurotransmitter exposure (11,42). In particular, persistently low 5-HT concentrations cause persistent response facilitation, which is associated with decreased synaptic effectiveness (42). Furthermore, the serotonergic, dopaminergic, and noradrenergic systems interact, contributing to behavioral sensitization (43). Brain circuits involved in addictive behavior include the ventral and dorsal striatum, amygdala, and orbitofrontal cortex and are heavily modulated by dopaminergic projections from the ventral tegmental area of the midbrain, serotonergic projections from the median and dorsal raphe nuclei, and noradrenergic projections from the locus coeruleus (7,44). We therefore suggest that high ACE activity, which characterizes the D/D subgroup of patients, may contribute to induce SSEP lack of habituation, on the one hand, by an ANG II-related increase of dopamine and decrease of norepinephrine turnover (35). On the other hand, steadily high ANG II concentrations, by increasing the efficiency of the rate-limiting enzyme of serotonin synthesis, may chronically reduce serotonin disposition (38). The final consequence of these neurotransmitter interactions on the functional properties of the cerebral cortex is a persistently heightened response to repeated stimuli. This view finds support from experimental evidence that ANG II can interfere with long-term learning mechanisms (45–47).
Moreover, it emerges from animal models that ANG II is involved in long-lasting neural changes subtending behavioral sensitization to substances (amphetamine) (26). These neural plastic modifications seem to be proportional to the degree of angiotensinergic stimulation (47). In agreement with these experimental data, here we have observed that ACE polymorphisms influence overuse behavior, as, in patients carrying the D/D genotype, the more prolonged the duration of medication overuse, the greater the deficit in habituation. This probably means that carrying the D/D genotype, which we have observed to be an independent contributing factor in inducing lack of SSEP habituation, predisposes to a more pronounced deficit of habituation that may in turn also be promoted or reinforced by the duration of medication overuse and then of the chronic headache.
The other distinctive finding in our study is that the interaction between ACE polymorphisms and drug overuse influences not only habituation, but also sensitization mechanisms. We have previously observed that sensitization, measured by the amplitude of the first SSEP block, was lower in MOH patients overusing triptans and progressively increased in those overusing NSAIDs or both anti-migraine medications combined (9). Here we have confirmed our previous findings adding that sensory sensitization is further increased in patients overusing combination medications containing psychoactive compounds (such as caffeine), which are intrinsically associated with higher behavioral sensitization (48). Moreover, we have observed that the relation between the type of drug and sensitization is influenced by ACE polymorphisms, as, among the overall MOH patients, those carrying the D/D genotype express the highest sensitization, reinforcing the concept that, in MOH, sensory sensitization parallels behavioral sensitization. As research suggests that ANG II modulates dopamine turnover in the nigrostriatal and mesolimbic reward dopaminergic system (44,49), we suppose that higher ACE activity influences the way in which drugs induce sensorial sensitization, chiefly through a dopamine-mediated mechanism.
In common with other chemical addictions, our results support the vision of MOH as a bio-behavioral disorder in which chronic head pain results from a combination of behavioral (drug overuse) and biological (central nervous system dysfunction) factors (50). Among the biological factors, inheritance seems to play a relevant role as it can predispose to specific clinical manifestations and progression of the MOH (51–54). The findings of the present study, furthermore, suggest that, in MOH, genetics can also influence the way the brain responds plastically to chronic head pain and excessive drug consumption.
Finally, certain limitations of the present study should be acknowledged. First, the number of patients is too small to generalize our results, although our cohort was sufficient to disclose strong statistical significance. Another shortcoming of our study is the absence of a control and/or of an episodic migraine group, even though the major objective of our study was not to explore whether MOH and episodic migraine patients were associated with genetic variants in the ACE polymorphism distribution with respect to healthy subjects.
In conclusion, ACE polymorphisms influence cortical plastic changes to repeated sensory stimuli in MOH. Excessive angiotensinergic activity, associated with the D/D polymorphism, increases sensitization as a result of drug overuse and determines the strongest lack of habituation, which is proportional to the duration of medication overuse. The different electrophysiologic patterns characterizing different ACE polymorphisms suggest that angiotensin peptides influence neuronal mechanisms of plasticity by interacting with monoaminergic synaptic transmission (15,55). To conclude, in view of the potential role for ACE inhibitors (14) and ANG II receptor blockers (26) as prophylactic therapy to decrease medication overuse and craving, controlled studies of these classes of agents in MOH patient population may be promising.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
No conflicts of interest have been declared.
