Abstract
Background:
Migraine patients report psychosocial stress to be among the major triggers for their migraine attacks. The mechanisms underlying the onset of migraine attacks are not yet fully understood. Neuroimaging studies have shown changes in the cortical excitability of migraine patients. Here, we investigated cortical activation related to processing of emotional stimuli in individuals with migraine.
Method:
Twenty-four participants suffering from migraine attacks and 25 healthy volunteers had to passively observe pictures of emotional facial expressions (angry, happy and neutral). Electro-cortical activity was continuously recorded by means of an electroencephalogram (EEG), and ratings of valence (unpleasant vs. pleasant) and arousal (calm vs. exciting) were collected.
Results:
The migraine and control group did not differ in their ratings of valence and arousal of the visual stimuli. However, participants with migraine, in contrast to healthy controls, showed larger N170 amplitudes toward angry facial expressions compared to neutral ones.
Discussion:
Individuals with migraine may have an altered cortical activity linked to the processing of emotional information. Thus, these individuals may process high arousing and threatening events preferentially, and this facilitated processing may be related to their already high cortical excitability.
Introduction
Migraine is a disabling headache disorder affecting 12% of the Western population (1). Emotional stress is considered one of the potential triggers for migraine attacks (2). Here we investigated whether persons with migraine show altered central nervous processing of emotional stimuli.
Electrophysiological and imaging studies support the idea of disturbed cerebral processing of sensory stimuli in migraine patients, probably resulting from an enhanced cortical excitability (3,4). Supportively, electroencephalogram (EEG) studies provide evidence for increased amplitudes and reduced habituation of the electro-cortical signals (5). Such increased amplitudes may result from the malfunctioning of cortical information-processing due to a metabolic disequilibrium (for a review, see (6)). Similarly to chronic pain patients (7), migraine patients showed a reduced ability to shift their attention away from a painful stimulus (8,9) as indicated by larger N2 (related to frontal inhibition or conflict monitoring mechanisms (10,11)); and P300 (related to detection of rare and task-relevant stimuli (12)); amplitudes in response to painful stimuli. Chronic pain (7) and migraine (13) patients have an altered function of the anterior cingulate cortex (ACC) in common. Notably, the ACC mediates affective responses to noxious stimuli (7,14). Thus, because of the importance of emotional stimuli and the overlap of neuronal networks involved in emotion, pain and migraine, we hypothesize that persons suffering from migraine would show alterations in their processing of emotional stimuli.
The human face and especially threatening expressions have been suggested as powerful stimuli able to activate the fear system (15). Facial stimuli elicit a negative peak, known as N170, which reflects the early visual processing of structural features of faces prior to face recognition (16). Several studies could not detect a modulation of the N170 amplitude by emotional facial expressions (16,17). However, Wieser and colleagues (18) could show larger N170 amplitudes for angry compared to happy and neutral facial expressions but only in participants who were waiting to give a public speech afterwards (i.e. a high anxiety group), and not in control participants. Hence, the threatening stimuli engaged more attention especially for people with enhanced social anxiety. Processing of emotional stimuli is also associated with an early posterior negativity (EPN) whose amplitude is enhanced by emotional pictures compared to neutral pictures (19,20). EPN reveals an early “tagging” mechanism of emotionally significant stimuli that occurs independently from stimulus novelty and also while participants hold unrelated task goals in mind (20). Accordingly, threatening relative to friendly and neutral expressions elicit a more pronounced EPN and an augmented late positive potential (LPP) (17). The LPP is a component of the evoked related potentials (ERPs) considered an index for enhanced processing of salient stimuli, especially aversive ones (17,19).
The present study was designed to investigate whether persons with migraine display an altered central nervous processing of emotional stimuli. To this end, we presented emotional facial expressions (21) and recorded the electro-cortical signal by means of the EEG in participants with migraine and healthy controls. We expected higher sensibility to angry facial expressions in participants with migraine compared to controls.
Subjects and methods
Participants
The study was approved by the ethics committee of the German Society of Psychology (Deutsche Gesellschaft für Psychologie; DGPs). Twenty-nine female participants responded to advertisements in local media describing that we were searching for female participants with migraine. The Migraine Disability Assessment (MIDAS) questionnaire (22) was used to certify if participants suffer from migraine attacks according to the International Headache Society (IHS). Exclusion criteria were chronic diseases or disabilities (e.g. other headache disorders, asthma, diabetes, rheumatoid arthritis); severe neurological disorders; mental retardation; acute depressive episode; or psychotropic medication. Three subjects were excluded because they did not meet the IHS criteria. Then, healthy control participants matched for age and level of education to the migraine participants but without history of migraine were recruited. All participants were right-handed.
Summary data of the migraine group.

MIDAS: Migraine Disability Assessment.
Before the experimental task, participants completed the German version of the trait part of the State-Trait Anxiety Inventory (STAI) (23) and the Beck-Depression Inventory (BDI) (24). The two groups did not differ in trait anxiety (migraine: M = 42.92, SD = 6.44; control: M = 42.52, SD = 6.99; F(1,48) = 0.04, p = 0.837), but participants with migraine reported a slightly higher depression scores than the healthy controls (migraine: M = 10.67, SD = 5.04; control: M = 7.52, SD = 6.93; F(1,48) = 3.28, p = 0.076).
Procedure
Upon arrival at the laboratory, participants read and signed the informed consent. Afterwards participants were seated in a comfortable chair in an electrically shielded room, and electrodes were attached. Participants were told to keep the pictures in their visual focus and to move as little as possible.
Stimulus presentation, EEG recording and behavioral responses were controlled by personal computers located in an adjacent room. Presentation software (Presentation 14.9; Neurobehavioral Systems, Albany, CA, USA) was used to control stimulus delivery and synchronization with the EEG recording. Brain Vision Recording software (Version 1.20) was used for recording the EEG signal. The experimental session consisted of a passive-viewing task during which facial stimuli (21) with angry, happy or neutral expressions were presented (28 times for each category) on a computer screen. Three additional pictures of facial expressions served as example trials. Stimuli lasted 1000 ms and were presented in a pseudo-random order with the restriction that no more than two stimuli with the same emotional expression were presented subsequently. The inter-trial interval (ITI) varied between 1750 and 2250 ms (mean duration was 2000 ms (17,18)).
After the passive-viewing task, all pictures were presented again, and participants rated their valence and arousal on scales ranging from one to nine. One meant “very unpleasant” for the valence and “calm” for the arousal, while nine meant “very pleasant” and “exciting,” respectively.
Apparatus and data analysis
The EEG was recorded with Ag/AgCl active electrodes from 28 sites according to the international 10–20 system (Fp1, Fp2, F7, F3, Fz, F4, F8, FC5, FC1, FC2, FC6, T7, C3, Cz, C4, T8, CP5, CP1, CP2, CP6, P7, P3, Pz, P4, P8, O1, Oz and O2) using a Brain-Amp-MR amplifier (Brain Products GmbH, Germany). The reference and the ground electrodes were placed on the left mastoid (A1) and on the right mastoid (A2), respectively. Four electrodes were placed below and above the right eye and the right and left outer canthi of the eyes for recording vertical and horizontal eye movements (EOGs). The impedance for all electrodes was kept below 10 kΩ. The electro-cortical signal was continuously recorded with a sampling rate of 1000 Hz and notch filtered (50 Hz).
Offline analysis was performed with the computer software Brain Vision Analyzer Version 2.0 (Brain Products GmbH). Data were off-line mathematically re-referenced to an average reference (17,18,25–27). The EEG was bandpass filtered (0.1–35 Hz) and independent component analysis (ICA) corrected for horizontal and vertical ocular artifacts. Trials with more than ± 50 µV amplitude were excluded from further analysis. Altogether, 17% of the trials in the migraine group and 22% of the trials in the control group had to be excluded (p = 0.315). Ten additional participants were excluded from further analyses because of their high range of rejection (more than 50% of the trials).
Epochs were registered 500 ms before to 1000 ms after stimulus onset and baseline-corrected (100 ms before stimulus onset). Each experimental condition (angry, happy and neutral) was then averaged for each participant. We analyzed: the N170 defined as maximal negative peak 140 ms to 190 ms after stimulus onset at the parietal sites (P7, P8); the early posterior negativity (EPN) defined as mean activity over the time interval between 240 ms and 280 ms after stimulus onset at the parietal sites (P7, P8) and the occipital sites (O1, O2); and the late positive potential (LPP) defined as mean activity over a time window from 400 ms to 600 ms after stimulus onset over the midline electrode sites (Fz, Cz, Pz).
Separate analyses of variance (ANOVAs) were calculated for subjective ratings and for ERP components including group (migraine, control) as between-subjects factor and emotion (angry, happy, neutral) as within-subjects factor. For the N170 and the LPP components, the ANOVA contained the additional within-subjects factor site (N170: P7, P8; LPP: Fz, Cz, Pz). For the EPN component, the ANOVA contained the additional within-subjects factors hemisphere (left, right) and site (parietal, occipital). If necessary, Greenhouse-Geisser correction of degree of freedom (GG-ε) was applied for violation of sphericity. The significance level was set at 0.05 (two-tailed) for all analyses. The effect size for all analyses is reported as partial
Results
Valence and arousal ratings
Mean scores and SDs of the valence and arousal ratings for emotional facial expressions separated for the migraine and control group.

Valence scale ranged from one (very unpleasant), through five (neutral) up to nine (very pleasant). Arousal scale ranged from one (calm) up to nine (exciting).
EEG measures
N170
As index for encoding of structural facial features, the analysis of the N170 returned a significant main effect for emotion (F(2,74) = 5.71, GG-ε = 0.864, p = 0.007,
Waveforms of the N170 to emotional facial stimuli. The graph depicts the waveforms of the N170 at the parietal electrodes (P7 and P8) during the viewing of angry (black line), happy (gray line) and neutral (dotted line) facial expressions in the control (on the left) and migraine (on the right) group. Surrounded peaks indicate the N170. In the migraine group, the N170 amplitude was significantly increased toward angry facial expressions compared to neutral ones. No distinct modulation of the N170 amplitude by emotional facial expressions was found in the control group. Mean amplitudes and SDs of N170, early posterior negativity (EPN) and late positive potential (LPP) in response to emotional facial expressions separated for migraine and control group. EPN amplitude are reported for the two groups separately only for descriptive reasons (in fact, the interaction Emotion × Group did not reach the significant level).
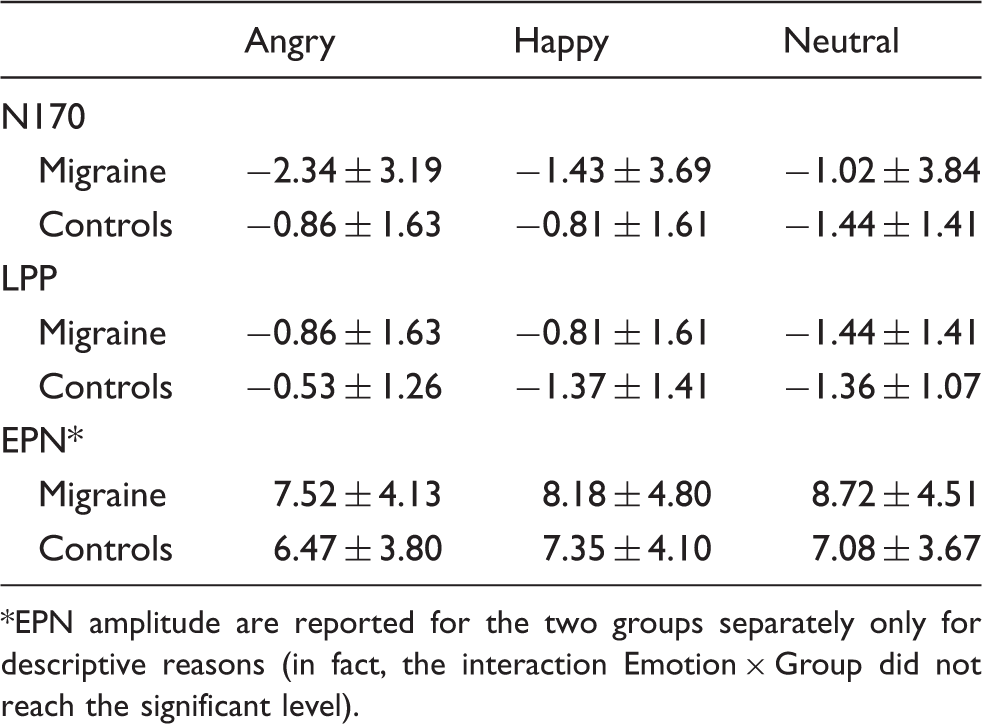
EPN
As index of automatic selective attention toward biologically salient stimuli, the analysis for the EPN revealed significant main effects of emotion (F(2,74) = 4.39, p = 0.016,
LPP
As an index of motivated attention, LPP analysis revealed significant main effects of emotion (F(2,74) = 7.03, p = 0.002,
Waveforms of the late positive potential (LPP) to emotional facial stimuli. The graph depicts the waveforms of the LPP during the viewing of angry (black line), happy (gray line) and neutral (dotted line) facial expressions in the control (on the right) and migraine (on the left) group. The LPP was defined as mean amplitude over a time window from 400 ms until 600 ms after stimulus onset. In both groups, the LPP amplitude was larger toward negative and toward positive pictures compared to neutral pictures (bottom panels).
Discussion
To our knowledge, this is the first study investigating the processing of emotional facial stimuli in people suffering from migraine. We found that these individuals are characterized by an altered electro-cortical activity linked to the processing of emotional facial expressions compared to healthy individuals. The N170 amplitude was modulated by the emotional content of the facial expressions in the migraine group but not in the control group. Specifically, migraine participants exhibited enhanced N170 amplitudes triggered by angry faces, suggesting thus that they process angry facial expressions preferentially and more intensively than healthy individuals. These results fit well with previous findings (18,27). Recently, we were able to demonstrate that the N170 in response to facial emotional expressions is modulated by the level of social anxiety (18). Namely, participants with higher levels of social anxiety showed enhanced early processing of threatening facial expressions. Based on these findings, we assumed that the enhanced N170 amplitude in response to angry faces observed in individuals with migraine indicates an enhanced early processing of threatening social stimuli. In support of this, Kolassa and Miltner (27) found similar preferential elaboration of phobic-related stimuli in phobic participants. Given that faces are important stimuli in social interactions and that emotional facial expressions may be detected pre-attentively (28), it is then tempting to suppose that people with migraine are particularly sensible to threatening social stimuli similar to other individuals with emotional disorders (18,27). Furthermore, threat may also be related to a higher stress response (29), and individuals with migraine often report stress as a trigger for a migraine attack (2). Therefore, the facilitated processing of angry facial expressions in the migraine group may suggest a higher sensibility of these participants for stressful events, which is also supported by their ratings of arousal (Table 2). Arousal indicates an activation of the motivational defensive system (30), and it is therefore conceivable that individuals with generally higher cortical excitability (3) also present greater and facilitated processing of high arousing facial expressions (31).
Our healthy controls did not show differences in N170 amplitudes related to emotional facial stimuli, which is in line with most previous studies (17,32). According to these studies, the N170 encodes the structural representation of a facial stimulus without encoding its affective content (16). Contrasting with our results in healthy controls, however, other studies (25,33) found a modulation of the N170 by emotional stimuli, in particular enhanced N170 amplitudes triggered by angry facial expressions. The discrepancy between the present and these findings may depend on the task. In fact, participants in our study had to passively look at emotional facial stimuli, whereas in the other studies participants had to actively focus toward or away from these stimuli (32) or to perform a behavioral response (33). To account for these discrepancies, Vuilleumier and Pourtois (34) suggested that mechanisms associated with the modulation of the N170 amplitude may partially overlap but still independently operate from affective aspects of the processing of facial expressions.
The LPP has been found to be modulated by motivated attention, in particular its amplitude seems to be enhanced in response to high arousing stimuli (19,35,36). Confirming these previous findings, the present study found larger LPPs toward high arousing facial expressions (i.e. angry and happy) in comparison to neutral expressions in both groups, and very likely this reflects motivated attention attracted by salient stimuli. In addition, we found tentative evidence for enhanced LPP in response to angry facial expressions in our control group, confirming Schupp and colleagues (17). These greater positive LPP amplitudes have been interpreted as results of a facilitated processing of threatening facial expressions linked to greater activation of the defensive motivational system (26). In contrast, the migraine group showed enhanced LPP amplitude toward happy facial expressions only. Considering that friendly facial expressions are more arousing than neutral ones, it may be possible that the enhanced LPP to the friendly stimuli is due to an arousal effect more than to a valence effect. In fact, LPP amplitude in response to the happy facial expressions was very similar to the LPP amplitude in response to the angry facial expressions (which just failed to reach the significance level). Interestingly, De Tommaso et al. (36) reported enhanced LPP amplitude toward both negative and positive emotional pictures in the migraine group as well as in the control group. However, the migraine group showed reduced LPP amplitudes toward the picture stimuli compared to the control group. The authors interpreted this reduced LPP amplitude as “distracting” effect from pain. In other words, the migraine patients may have allocated their attention toward the painful stimulation, which was delivered during picture viewing. In the same vein, Kenntner-Mabiala and colleagues (37,38) found that painful stimuli and high arousing picture stimuli compete for attentional resources. Presumably, the distraction induced by painful stimuli in migraine patients (36) is linked to their sensitization toward high arousing events. Moreover, stress is one of the most reported triggers of migraine attacks (2), high arousing emotional events are linked with higher levels of stress (29), and individuals with migraine showed a neural hyperexcitability (3,4), which impacts cognitive functions (31). Therefore, it is tempting to speculate that the processing of highly arousing stimuli in participants with migraine may be linked to the mildly altered electro-cortical excitability of participants in our study.
Regarding the EPN, we observed no specific emotion modulation in the migraine group, meaning that all participants showed more negative EPN amplitudes toward high arousing stimuli (i.e. angry and happy expressions) than toward low arousing stimuli (i.e. neutral expressions). These results are in line with previous findings (19) and indicate that this ERP component is not specifically compromised in people who suffer from migraine. Considering that the EPN reflects a fast and automatic processing of emotional stimuli and is mediated by the amygdala (17,19), we concluded that participants in the migraine group may not show any different modulation of this electro-cortical potential because the amygdala may not play a particular role in this neurological disorder.
Despite the different emotion-related cortical activity, participants in the migraine and in the control group did not differ in their explicit ratings of the emotional stimuli. Thus, angry facial expressions were rated as negative and as high arousing compared to neutral facial expressions, while happy facial expressions were rated as more positive (Table 2). This lack of a group difference may be linked to the fact that our migraine group did not consist of severely affected migraine patients but instead of individuals who suffer from relatively few migraine attacks. Possibly the relatively low frequency of migraine attacks in our sample may not have really compromised their social and/or emotional life. It would be interesting to investigate the processing of emotional stimuli in a group of patients with high-frequency migraine attacks in order to verify whether these patients might also have altered self-reports. This would shed light on the question whether the altered electro-cortical signal is related to an altered behavioral response.
To summarize, our study revealed in individuals with migraine altered mechanisms in the processing of emotional facial stimuli. Possibly, because of their high arousal and salience, angry facial expressions are quickly and strongly processed by the migraine group, while happy facial expressions may be highly salient or particularly high arousing for individuals with migraine and therefore catch their attention with preference. Therefore, high arousing stimuli may be processed preferentially in individuals with a high cortical excitability.
Footnotes
Funding
This work was supported by the
Acknowledgements
We thank A. Baur und V. Schneider for their fundamental help in the recruitment of the participants and in the collection of the data. MA, AKP, PW and AM designed the experiment. MA analyzed the data. MA, PW, CS, PP and AM contributed to data interpretation and wrote the manuscript.
Conflict of interest statement
None declared.
