Abstract
Objective: To summarize and evaluate proton (1H) and phosphorus (31P) magnetic resonance spectroscopy (MRS) findings in migraine.
Methods: A thorough review of 1H and/or 31P-MRS studies in any form of migraine published up to September 2011.
Results: Some findings were consistent in all studies, such as a lack of ictal/interictal brain pH change and a disturbed energy metabolism, the latter of which is reflected in a drop in phosphocreatine content, both in the resting brain and in muscle following exercise. In a recent interictal study ATP was found to be significantly decreased in the occipital lobe of migraine with aura patients, reinforcing the concept of a mitochondrial component to the migraine threshold, at least in a subgroup of patients. In several studies a correlation between the extent of the energy disturbance and the clinical phenotype severity was apparent. Less consistent but still congruent with a disturbed energy metabolism is an observed lactate increase in the occipital cortex of several migraine subtypes (MwA, migraine with prolonged aura). No increases in brain glutamate levels were found.
Conclusion: The combined abnormalities found in MRS studies imply a mitochondrial component in migraine neurobiology. This could be due to a primary mitochondrial dysfunction or be secondary to, for example, alterations in brain excitability. The extent of variation in the data can be attributed to both the variable clinical inclusion criteria used and the variation in applied methodology. Therefore it is necessary to continue to optimize MRS methodology to gain further insights, especially concerning lactate and glutamate.
Keywords
Introduction
Migraine is a complex neurovascular disorder, involving complex genetics, multiple neurotransmitter systems, and multiple cortical and subcortical regions of the brain (1). Experimental migraine models have been useful but incomplete in reflecting the full scope of the human condition (2). At present, no integrative model exists to explain this multifaceted syndrome. Over the past 25 years, several magnetic resonance spectroscopy (MRS) studies have been performed in migraine patients. Whereas in the first 10 years these studies comprised mostly phosphorus MRS (31P-MRS), a majority of proton MRS (1H-MRS) studies have been performed since 2003. Although a large number of data have been generated, the contribution to our understanding of migraine pathophysiology by these heterogeneous studies is not straightforward. Of these 31 MRS studies, two handled multinuclear MRS (that is, both 1H-MRS and 31P-MRS) and another three used visual stimulation to excite the brain. Thirty studies covered one or more brain areas, predominantly the occipital cortex, although measurements were also performed in other cortical regions and non-cortical regions, such as the basal ganglia. Only nine studies covered 31P-MRS of muscle tissue. Most studies emphasized on migraine with aura (MwA) and its rare subtype of hemiplegic migraine. To a lesser extent migraine without aura (MwoA) was covered, especially in combination with 1H-MRS. The majority of studies were obtained interictally. 31P-MRS results were mostly depicted as molar concentrations (absolute quantification), whereas 1H-MRS studies used almost exclusively ratios (relative quantification) to represent the results.
A review of MRS data in migraine has not been performed since 2006 (3). We therefore embarked on an updated review of both 31P-MRS and 1H-MRS data. Besides discussing the objective MRS results, a critical review was made of patient and control inclusion criteria (migraine subtypes, regions of interest, age, gender, attack frequency, prophylaxis, and ictal versus interictal) and methodological aspects (field strength, coil, MR parameters, quantification method, and assumptions concerning adenosine triphosphate or ATP) and their respective potential shortcomings. Eligibility criteria for papers to be included in this review were original articles in English on either 31P-MRS and/or 1H-MRS in muscle and/or brain of migraine patients, published before or during September 2011 and available through MEDLINE using the search terms ‘migraine’ and ‘magnetic resonance spectroscopy’. This search yielded 76 papers, of which 30 original articles fulfilled eligibility criteria and were included. Two papers were excluded for language raison (one paper in Polish and one in Japanese), two more papers were excluded owing to co-morbidities (lupus, during transient global amnesia). Among the 76 papers were six review papers on MRS in migraine, and one additional MRS study in a rat model of migraine. A broader MEDLINE search with ‘migraine’ and ‘spectroscopy’ did not yield additional papers, but using the search terms ‘MRS’ and ‘migraine’ one additional eligible paper was identified, bringing the total of original articles to 31. Citation lists of all original articles and review papers were searched for additional references, but none were found. A Web of Science search using search terms ‘migraine’ and ‘magnetic resonance spectroscopy’ did not yield additional original articles.
Results: 31P-MRS findings
31P-MRS is a non-invasive tool to analyse energy metabolism of brain, muscle and other tissues (4). Under normal aerobic circumstances, ATP is produced through glycolysis and oxidative phosphorylation. ATP can also be generated through transfer of Pi from PCr to ADP via the creatine kinase reaction. Anaerobically, the brain switches to the significantly less efficient anaerobic glycolysis to produce ATP. The concentrations of ATP ([ATP]), PCr ([PCr]) and Pi ([Pi]), as well as phospholipids can be derived directly from the 31P spectrum (Figure 1A). Additional parameters can be calculated, such as the PCr/Pi ratio (which reflects the energy status of the cell), the phosphorylation potential defined as ATP/(ADP × Pi), the ADP concentration ([ADP]) and the percentage of v/vmax (the ratio of the actual velocity of oxidative metabolism to the maximum oxidative capacity). The concentration of magnesium ([Mg2+]), which is an important cofactor in the creatine kinase reaction, and the intracellular pH (pHi), can be calculated from chemical shifts in the spectra. Table 1 gives a detailed overview of 31P-MRS studies in the brain of migraine patients.
Brain 31P (A) and 1H -spectrum (B). Several 1H-metabolites are difficult to quantify, e.g. Glu + Gln (difficult to differentiate, small peaks, close to NAA), GABA (very small peak right of NAA), and Lac (low concentration, resonates at same frequency as lipids such as subcutaneous fat). From the 31P-spectrum, pHi can be calculated from the frequency difference between Pi and PCr, and [Mg2+] can be calculated from the frequency difference between PCr and β-ATP. Some metabolites show several resonance peaks in the spectrum originating from different proton or phosphorus nuclei, such as tCr (tCr1 and tCr2) and ATP (α-ATP, β-ATP and γ-ATP). PME: phosphomonoesters, PDE: phosphodiesters, ATP: adenosine triphosphate, PCr: phosphocreatine, Pi: inorganic phosphate, NAA: N-acetylaspartate, tCr: total creatine, Cho: choline, mIns: myo-inositol, Lac: lactate, Glu: glutamate, Gln: glutamine, GABA: y-aminobutyric acid. Overview of brain 31P-MRS studies in migraine patients: migraine types, brain regions and results. MwoA: migraine without aura, MwA: migraine with aura, MwpA: migraine with prolonged aura, MS, migrainous stroke, BM: basilar-type migraine, FHM: familial hemiplegic migraine, other: MS and MwpA, PCr: phosphocreatine, Pi: inorganic phosphate, ATP: adenosine triphosphate, ADP: adenosine diphosphate, PP: phosphorylation potential, v/vmax: rate of ATP synthesis, TP: total phosphorus signal, PDE: phosphodiesters, Mg2+: magnesium, ↑: increased, ↓: decreased. p < 0.05. This study was performed both ictally and interictally. Only pH was measured in this study. 4/12 patients suffered from MwoA as well. MwA patients included typical aura and BM.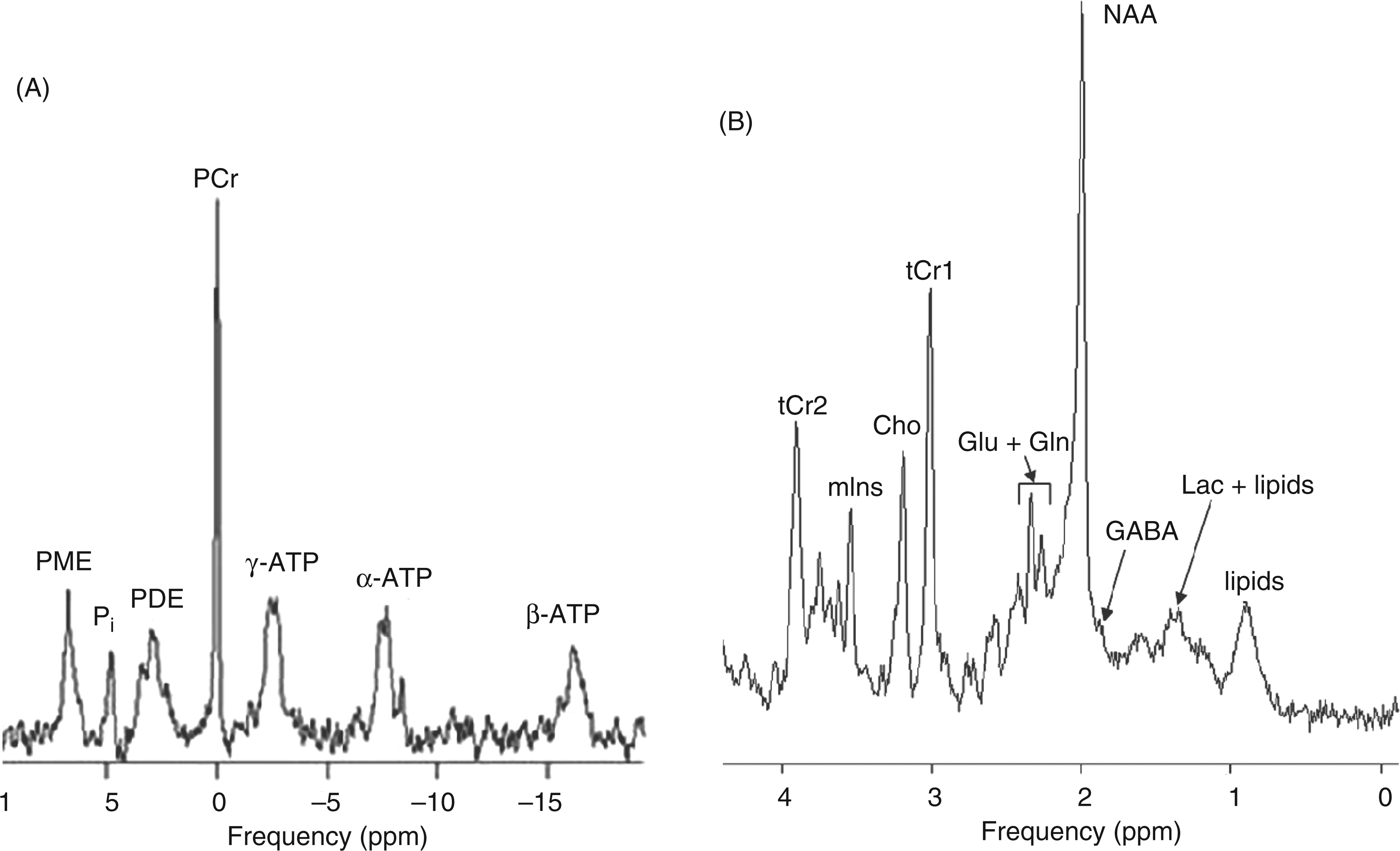

Brain
Ictal changes
Only the group of Welch et al. measured brain energy phosphate metabolism and pHi during migraine attacks in both MwoA and MwA patients (5,6). They observed a significant reduction of the PCr/Pi ratio in both anterior and posterior brain areas of MwA (but not MwoA) patients, indicating a low availability of free cellular energy, but there was no alteration in brain pHi during attacks (as well as interictally). In addition, low magnesium levels were observed during migraine attacks without changes in pHi (7). These early 31P-MRS studies in migraine failed to support the prevailing concept at that time that migraine pain was caused by brain ischemic acidosis, but rather suggested a disordered energy metabolism during a migraine attack.
Interictal changes
Most 31P-MRS studies were performed interictally, but the duration of the headache-free interval is rarely specified. Interictal phosphorus brain abnormalities in occipital lobes were common to different subtypes of migraine, such as MwoA (5,6,8–12), MwA (5,6,9–11,13,14), familial hemiplegic migraine (FHM) (11,15), basilar-type migraine (10,14), migraine with prolonged aura (9,10,16,17) as well as in migrainous stroke (9,10,16,18). A few studies report similar results in other brain regions, including the basal ganglia (19) and frontotemporal (11,20) and parietal brain areas (20).
First of all, a significant decrease in [PCr] (8,9,12–15,17) and an increase in [Pi] (13–15,17) resulting in a reduced PCr/Pi ratio (5,16,17,19,20) were the most generalized observations, regardless of the brain region. One study, Boska et al., reported only a depletion of PCr in anterior brain regions of MwA and the occipital cortex of FHM patients (11). The calculated value of the phosphorylation potential was significantly decreased in migraine patients, as reported in a number of studies (8,9,12–15). The brain [ATP] was always assumed constant and equal to normal controls, being 3 mM or was not mentioned at all, except for Reyngoudt et al., who quantified [ATP] in the occipital lobe of MwoA patients and found a significant decrease compared with controls, at least in a subgroup of patients (12). A significantly increased [ADP], indicating that brain tissue is working at a higher metabolic rate and hence has a lower energy reserve, was also frequently observed (8,9,13–15,18). The percentage of v/vmax was found to be significantly increased in migraine patients (8,9,14,15).
pHi was measured in all abovementioned studies and no significant pHi changes were observed except in three studies (13,14,18). The pHi changes found in these studies should be regarded carefully, however, because of the presence of large standard deviations (13), borderline significance (14) or migrainous stroke (18). The same remark holds true for Mg2+, an essential cofactor for mitochondrial membrane stability and in oxidative phosphorylation, which was found significantly decreased in a minority of studies (7,10,11,14).
The combined abovementioned abnormalities suggest a mitochondrial component in the neurobiology of migraine, at least in a subgroup of patients. Indeed, a similar cerebral 31P-MRS pattern has been observed in some mitochondrial cytopathies (21,22). However, a consistent pattern of mitochondrial abnormalities (e.g. from muscle biopsies or mitochondrial DNA analysis) in migraine has not emerged, and these steady state measurements reflect the balance of ATP demand and mitochondrial function/ATP production (4). In any case, the data suggest that the brain of migraine patients is compromised in its ability to deal with metabolic stress.
It seems that the cerebral 31P-MRS findings correlate with the severity of the clinical phenotype in several studies (10,12,16,19). Reyngoudt et al. observed a reduction in [ATP] in patients with the highest attack frequency and suggested that the existence of one or more subgroup(s) may explain apparent contradictions between previous studies as most were performed on small and/or heterogeneous patient groups (see below) (12).
Muscle
Several 31P-MRS studies also reported a defective energy metabolism in skeletal muscle tissue (always in the gastrocnemius muscle except for two studies that did not specify which calf muscle), including MwoA, MwA, migraine with prolonged aura, FHM and migrainous stroke patients (8,13–18,23,24). The measurement paradigm generally consisted of a rest period, followed by an exercise period, and again followed by a rest period. The (isokinetic) exercise involved pressing a pedal (plantar flexion) connected with a pneumatic ergometer and the rest–exercise–rest cycle was repeated several times. Most 31P-MRS muscle studies showed no significant differences in phosphorus metabolites between patients and controls at rest (8,13–15,17,18). However, following exercise, patients’ muscles had a lower PCr content and a higher Pi content than controls, showing a slower recovery of both PCr and Pi, which is reflected in an elevated recovery time constant. According to Uncini et al., the degree of muscle oxidative metabolism defect in FHM patients seemed to be correlated with the clinical phenotype (15). PCr and Pi concentrations at rest and during work and recovery were calculated assuming an intramuscular ATP concentration of 8 mM (25). These 31P-MRS data suggest a multisystem oxidative metabolism defect, including both muscle and brain tissue.
Results: 1H-MRS findings
Overview of brain 1H-MRS studies in migraine patients: migraine types, brain regions and results.

MwoA: migraine without aura, MwA: migraine with aura, MwpA: migraine with prolonged aura, MS, migrainous stroke, FHM(2): familial hemiplegic migraine (type 2), SHM: sporadic hemiplegic migraine, CM: chronic migraine, other: MS and MwpA, MwAplus: MwA with symptoms of paraesthesia/paresis/dysphasia, Lac: lactate, NAA: N-acetylaspartate, mIns: myo-inositol, Cho: choline, tCr: total creatine, Glu: glutamate, ↑: increased, ↓: decreased.
p < 0.05.
Visual stimulation was performed by projection of a blue/yellow flickering checkerboard (8 Hz).
Visual stimulation was performed by projection of a flashing red light (14 Hz).
This patient suffered actually from migrainous headache with a series of long-lasting visual aura.
Visual stimulation was performed by projection of a black/white flickering checkerboard (8 Hz).
Lac
An early 1H-MRS study showed elevated interictal levels of cerebral Lac in the occipital cortex of a heterogeneous group of migraine patients (three MwA patients, one basilar-type migraine patient, one migrainous stroke patient and one migraine with prolonged aura/migrainous stroke patient) (27). A similar observation was made in the cerebellum of four patients with FHM2, a rare subtype of MwA (23). In a functional 1H-MRS study, visual stimulation resulted in a Lac increase in the occipital visual cortex of MwA patients with both visual symptoms and paraesthesia, paresis and/or dysphasia, but not in MwA patients with only visual symptoms, in which Lac was already higher than normal in the resting state (28). The authors of the abovementioned studies suggested that the accumulation of Lac was a marker for a disturbance in oxidative glycolysis, related to energy metabolism impairment, which is typical for mitochondrial diseases (29). Recent studies, however, performed by Reyngoudt et al. at higher field strength (3 T) in MwoA patients did not demonstrate significant increases in Lac, both at rest (30) and following visual stimulation (31). It was postulated that the lack of stimulation-induced Lac increase in MwA, as reported by Sandor et al. (28), and also in patients with mitochondriopathies might be partially attributed to a saturation of Lac transporter systems (32). In the subgroup of MwA, with high resting-state Lac levels, it was hypothesized that the aura was limited to the visual cortex, whereas in the subgroup of migraine patients with both visual symptoms and paraesthesia, paresis and/or dysphasia, this was not the case. Experiments demonstrated that decreased pHi, which would accompany increased extracellular Lac, is an inhibiting factor for CSD (cortical spreading depression), which is believed to be at the basis of the aura symptoms (33). However, a decrease of pHi has never been observed, and an accurate detection, let alone accurate quantification of Lac, is cumbersome and will be discussed below.
NAA
A functional 1H-MRS study reported significantly reduced NAA levels in the occipital cortex of MwA patients but not in MwoA patients compared with controls, before and following visual stimulation (34). The authors hypothesized the presence of less efficient mitochondrial functioning in MwA patients compared with MwoA patients and controls (34), and that the reversible loss of NAA during visual stimulation is due to a redistribution of NAA from the intra to the extracellular space, which would result in a variation in chemical environment that may alter its magnetic resonance visibility (35). The hypothesis of a mitochondrial deficiency was also put forward by a study in which decreased NAA was found in the (left) thalamus of MwoA patients (36). Finally, a reduced NAA concentration was also found at rest in the cerebellum of FHM1 patients (37), in the left temporoparietal cortex (site of migrainous stroke) of a sporadic hemiplegic migraine (SHM) patient (38) and in the occipital cortex of another SHM patient (39), which has been rather attributed to neuronal impairment or loss. The latter is obvious in migrainous stroke but may also fit with the frequently observed cerebellar atrophy in FHM (40).
Glu/Gln
Glu levels were found to be markedly reduced in the cerebellum of FHM1 patients in a 1H-MRS study performed at 1.5 T (37). However, methodological aspects, including a potential spectral overlap of Gln and GABA resonances at 1.5 T make it difficult to assess these changes. According to Dichgans et al., the reduction of Glu may in part reflect neuronal impairment, as was already indicated by a concomitant NAA reduction (37). Another possibility to explain the decreased Glu levels is an impaired glutamatergic neurotransmission. The latter hypothesis, however, is in contrast with a prediction of enhanced glutamatergic neurotransmission in FHM1 patients, based on a gain of function of pre-synaptic Ca2+ channels by CACNA1A mutations (41). The concomitant decrease in Glu and NAA (described above) are therefore likely to be attributed to atrophy.
Recently susceptibility loci for migraine, both MwA and MwoA, have been described in genome-wide association studies, and some refer to Glu homeostasis (42–44). Although no significant metabolite differences were observed in a study using two-dimensional 1H-MRS, linear discriminant analysis revealed a separation between migraine patients and controls based on Gln and N-acetylaspartylglutamate in the anterior cingulate cortex (ACC) and insula, suggesting glutamatergic abnormalities in the brain of migraine patients compared with controls (45). At present, however, there is no direct proof of altered brain glutamate levels in MwA and MwoA.
mIns
Limited data are reported concerning mIns. A decreased mIns content was found in the left temporoparietal cortex of one SHM patient during prolonged hemiparetic migraine aura (38). Another study in FHM1 patients showed an increased interictal cerebellar mIns concentration (37). As mIns is considered an in vivo glial marker, an increase in this metabolite would signify a regional glial cell proliferation (46), which could be in line with an autopsy report that gave evidence of a proliferation of astrocytes and gliosis in the cerebellum of a CACNA1A mutation carrier (47).
Cho
A single study reports a decreased Cho level in the cerebellum of MwA patients, suggesting a variation in membrane turnover and/or composition (48).
Methods
Demographic and patient information of MRS studies in migraine.


MwoA: migraine without aura, MwA: migraine with aura, MwpA: migraine with prolonged aura, MS, migrainous stroke, BM: basilar-type migraine, FHM(2): familial hemiplegic migraine (type 2), SHM: sporadic hemiplegic migraine, CM: chronic migraine, MwAplus: MwA with symptoms of paraesthesia/paresis/dysphasia, other: MS and MwpA, n: number of volunteers, F/M: female/male ratio, Pφ: prophylaxis, Y: yes: N: no, ‘-‘: information is lacking, n/a: not applicable.
Heterogeneous data: both MwA and MwoA attacks.
12 participants for brain, 15 participants for muscle.
Methodological parameters of MRS studies in migraine.
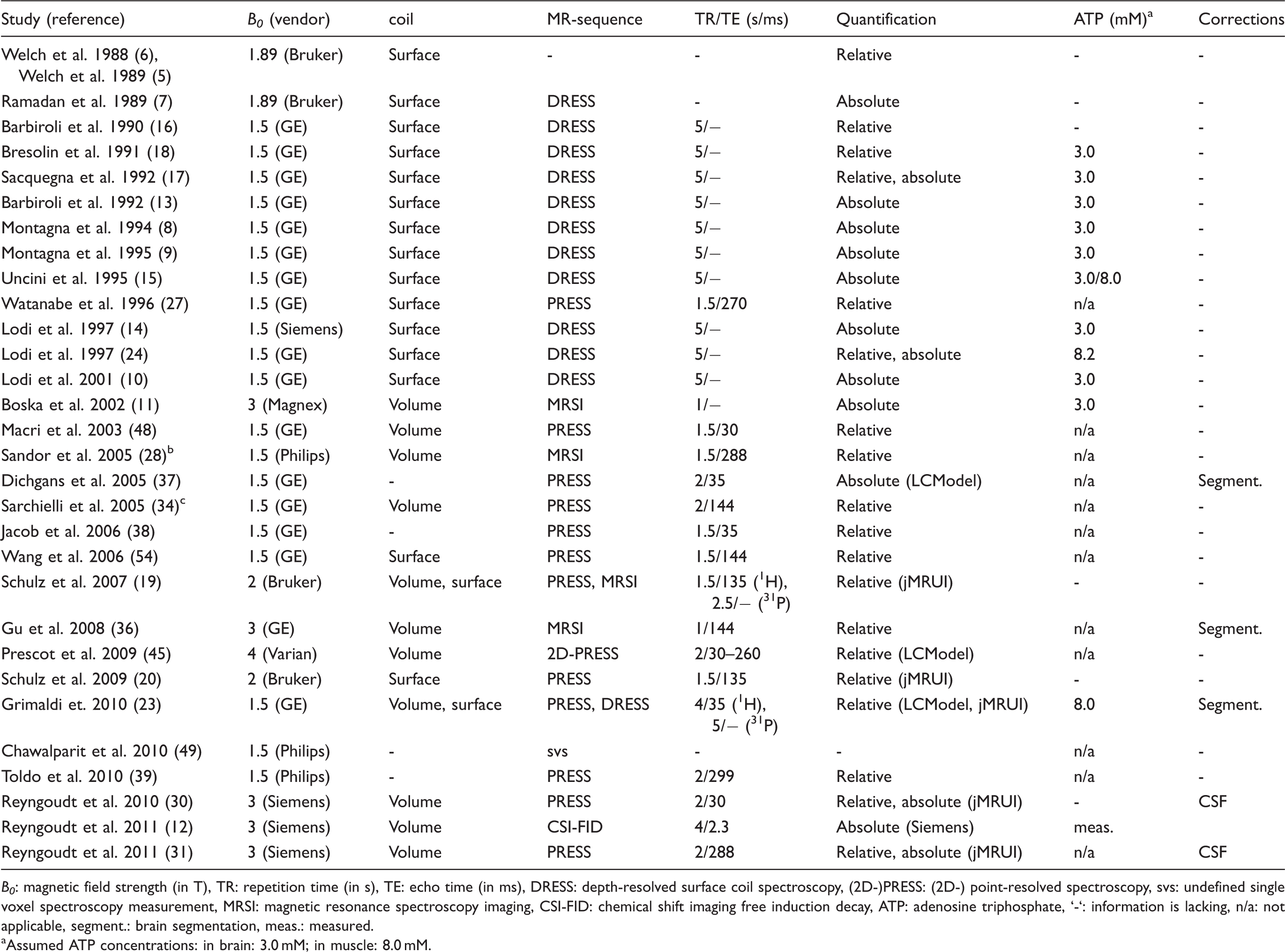
B0: magnetic field strength (in T), TR: repetition time (in s), TE: echo time (in ms), DRESS: depth-resolved surface coil spectroscopy, (2D-)PRESS: (2D-) point-resolved spectroscopy, svs: undefined single voxel spectroscopy measurement, MRSI: magnetic resonance spectroscopy imaging, CSI-FID: chemical shift imaging free induction decay, ATP: adenosine triphosphate, ‘-‘: information is lacking, n/a: not applicable, segment.: brain segmentation, meas.: measured.
Assumed ATP concentrations: in brain: 3.0 mM; in muscle: 8.0 mM.
Regions of interest
Occipital
In most studies the occipital cortex was chosen as the region of interest (5–11,13–18,23,27,28,30,31,34,37,39,49). According to several studies this was due to the fact that aura, with most often visual symptoms, could be attributed to this area in MwA patients (13,27,37,49). However, aura symptoms are experienced by 20–30% of migraine patients. Consequently, other reasons for performing measurements in the occipital lobes are more of a practical nature, including its ideal location for high signal-to-noise ratio when using surface coils, as stated in several early MRS studies (5–7), and the fact that it is easily stimulated (28,31,34).
Frontal regions, anterior cingulate cortex, insula, cerebellum, hypothalami, thalami, and so on
Frontal regions were investigated in some earlier studies based on the hypothesis that CSD progressed anteriorly (5–7). Frontal regions, including the primary motor cortex, were also examined in a few more recent studies, including a study covering FHM and migrainous stroke (11,20). Moreover, in the study of Prescot et al. (45) the ACC and the insula were under investigation because both brain areas are implicated in the perception of pain (50,51). MRS was also performed in the cerebellum of MwA (48) and FHM patients (37). The cerebellum was investigated in FHM patients because a substantial proportion develop cerebellar degeneration (37). Subclinical cerebellar abnormalities have been described and are more pronounced in MwA than MwoA (52), but the cerebellar volume in MwA is unaltered (53). The thalamus and hypothalamus, subcortical structures involved in the pathophysiology of migraine attacks, were investigated in MwoA patients (1,36) and (probable) chronic migraine (54), respectively. Several other studies focused on larger brain regions, both grey and white matter, for different reasons (see Table 1 and 2 for details), including the hemisphere on the side of the headache (5–7), the site of MRI abnormalities (38), or to look at white matter specifically (19).
Muscle
31P-MRS of muscle was always conducted in the right calf muscles (almost exclusively in gastrocnemius muscle except for two studies in which the muscle type was not specified) (8,13–18,23, 24). The reasons for this are practical: surface coils can easily be positioned around the calf, the gastrocnemius is a large enough muscle to perform localized spectroscopy and calf muscles can easily be fatigued by using an ergometer.
Patients and controls
Age and gender
All but five studies (9,18,19,37,39) reported the age ranges of both migraine patients and controls. Overall, most participants were between 20 and 45 years of age, but there are limited data on children and adolescents (14,17,39,49).
Migraine patients and controls were matched for both gender and age in only four studies (10,14,23,31,45), just for age in approximately 50% of the studies (8,11,12,17,19,27,28,30,34,48,54) and just for gender in one study (36).
Migraine subtypes and attack frequency
Patient groups did not always exist of a homogeneous migraine subtype, making interpretations of metabolic alterations not always straightforward. Montagna et al. performed 31P-MRS in a large cohort of migraine patients, including MwoA patients, MwA patients and a group of 'complicated' migraine patients, the latter comprising both migrainous stroke and migraine with prolonged aura (9). In Lodi et al., 31P-MRS was performed in a migraine group consisting of MwoA, MwA, basilar-type migraine, migraine with prolonged aura and migrainous stroke and in a group of cluster headache patients (10). Neither study, Montagna et al. (9) or Lodi et al. (10), mentioned migraine attack frequencies. Watanabe et al. reported elevated lactate (Lac) levels in the occipital cortex of five (two with MwA, one with basilar-type migraine, one with migrainous stroke and one with migraine with prolonged aura/migrainous stroke) out of six investigated migraine patients (27). The migraine patient group in the study of Sandor et al. was also rather heterogeneous, including patients suffering from MwA, migraine with prolonged aura and MwAplus, the latter consisting of patients who had visual aura associated with at least one of the following: paraesthesia, dysphasia or paresis (some of these should perhaps have been labelled hemiplegic migraine) (28). Prescot et al. mention patients who suffered from acute episodic migraine without verifying whether it concerned MwoA or MwA (45). In several studies the criteria for 'migraine attack frequency' were not very stringent (13,19,20,34,37) or were lacking altogether (5–7,9–11,15,24,48,49).
Several studies, though, did cover homogeneous patient groups who experienced a well-defined number of attacks, as was the case for MwoA (8,12,13,30,31,34), MwA (19,34) and FHM1 (37).
Ictal versus interictal
In all but a few studies migraine patients were scanned interictally, mostly for obvious practical reasons but also to explore the interictal character of the disorder rather than the attack itself. Attack-free intervals ranged from 48 hours (12,27,30,31) or a few days (11,28,34,49) to a week (5–7,10,14) or more than a week (17,19,20,23,39). In all other studies no information concerning the attack-free period is specified (8,9,13,15,16,18,24,36,37,45,48,54).
The first 31P-MRS studies in migraine, however, were studies in which both ictal and interictal measurements were performed, but in two different sets of migraine patients (5–7). In these aforementioned studies no patient underwent both an ictal and an interictal measurement or had an aura at the time of the study. These ictal spectra were measured between 3 and 48 hours following the headache onset (5–7). Finally, Jacob et al. obtained an ictal 1H-MR spectrum during a long-lasting migraine attack of a SHM patient (38).
Prophylaxis: yes or no?
Most studies described the lack of use of prophylactic medication in a certain (in most cases not defined) time period before and during the course of the MRS experiments (8,9,11–14,16,18,24,30,31,34,36,39,49). In quite a few other studies, however, no information about the medication history of migraine patients is given (15,17,19,20,27,28,37,48,54). Furthermore, in the earliest MRS studies (5–7) and in Prescot et al. (45), a small minority of patients studied ictally were taking medication for migraine prophylaxis.
MRS methodology
Methodological differences may also explain the discrepancies found in MRS studies in migraine. Methodological aspects include the choice of magnetic field strength, coil, localization technique, repetition time, echo time, flip angle, (pre)processing steps, signal estimation, and quantification procedure. These aspects are summarized in Table 4.
Magnetic field strength and coils
Only a minority of the studies was conducted at higher field strengths (≥3 T) (11,12,30,31,37,45), providing greater spectral resolution, which allows more accurate metabolite quantification and determination of spectral frequencies and is also the basis of pH and [Mg2+] calculations. A potential spectral overlap of Gln and GABA resonances was mentioned in a 1H-MRS study performed at 1.5 T by Dichgans et al. (37). The variability in the MRS data reflects a combination of true variation between study participants and experimental variation, which mainly arises from the signal-to-noise ratio of the acquired spectra. It is possible that the signal-to-noise ratio, in particular in 31P-MRS with the inherent lower sensitivity of the 31P nucleus, did not allow detection of significant differences between controls and some patient subgroups. This also holds true for 1H-MRS. The detection of more subtle metabolic changes, such as a rise in Lac in healthy volunteers (28,34), could be achieved with high-field MR systems and/or the use of surface coils.
Indeed, the detection of Lac has long been a burning issue in MRS. It should be emphasized that Lac is a low-concentration metabolite in healthy brain tissue (on the order of 0.2–1 mM). And although early functional 1H-MRS studies, performed at lower field strength (1.5 T), found Lac increases of 60–150% (55–57), Mangia et al. (58–60) reported in several advanced 1H-MRS studies, performed at 7 T, an increase in [Lac] of 0.1–0.2 mM following visual stimulation and corresponding to an increase of only 20%. A Lac increase of up to 0.2 mM is unlikely to reflect a switch to anaerobic glycolysis, according to Mangia et al. (59,60). Considering these results and the fact there is very little information about absolute Lac values in the migrainous brain, except the 3 T functional 1H-MRS study of Reyngoudt et al. in which Lac concentrations of 0.5–0.6 mM were found in controls and MwoA (31), one should be very careful concerning Lac quantification and the interpretation of the results.
31P-MRS studies were generally performed using surface coils because of a high achievable signal-to-noise ratio (5–10,13–20,23,24). Volume coils were used in most 1H-MRS studies (19,23,28,30,31,34,36,45,48) but in only two studies performing 31P-MRS (11,12). In a few studies information concerning coil specification was lacking (37–39,49).
MRS measurement parameters
Table 4 describes which MRS localization technique was used in each study. The majority of 31P-MRS studies made use of the depth-resolved surface-coil spectroscopy (8–10,13–18,23,24) whereas 1H-MRS was mostly performed using point-resolved spectroscopy (19,20,27,28,30,31,34,36–39,54). The advantages of single voxel applications are a homogeneous magnetic field across the volume, good spectral resolution and the measurement of a well-defined volume, although the latter can simultaneously be seen as a main drawback because other important areas may be missed. This can be solved by multivoxel applications, although this increases measurement times significantly. Moreover, magnetic field inhomogeneities across an entire object, inherent to multivoxel measurements, make quantitative spectroscopy challenging.
The choice of specific MRS measurement parameters, such as echo time and repetition time, may also have a substantial effect on the acquired spectra. Spectra acquired at short echo time often reveal a complex spectral pattern and severe overlap of broad resonances from macromolecules and lipids, further complicating the accurate assessment of changes in metabolites. Moreover, long echo times are often preferred for the detection of low-concentration resonances such as Lac (19,20,27,28,31,34,36,39,45).
Signal processing
Signal processing procedures differed extensively between studies over the years. The Welch group initially used basic triangulation to determine signal areas of 31P-peaks (5–7), whereas the Bologna group quantified phosphorus data using a user-interactive curve fitting program (8–10,13–18,24). Later 31P-MRS studies used scanner software (11,19,20,34,45), LCModel (23,37,45) or the AMARES (30,48) and/or QUEST (31) algorithm (jMRUI). Information regarding signal estimation was not reported in the remaining studies (27,28,38,39,49,54).
Quantification
Relative quantification was performed by calculating the ratios between peaks and was conducted in approximately half of the studies (Table 4). In 31P-MRS studies this involved in most cases the phosphocreatine (PCr)/inorganic phosphate (Pi) ratio, whereas in 1H-MRS studies metabolites were generally normalized to tCr, which is assumed to be relatively constant. However, changes in the tCr content have been observed in several conditions and one should be cautious when using tCr as an internal reference (see review in (46)).
In the remaining studies attempts were made to perform ‘absolute' quantification. In almost all 31P-MRS studies in migraine (8–10,13–15,17,18) metabolites were assessed by assuming a constant cytosolic ATP concentration of 3 mM (61), because no absolute data on ATP concentration in human brain of migraine patients exist. In Reyngoudt et al. (12), however, [ATP] was quantified, based on the fact that β-ATP is proportional to the total cellular ATP content (62,63). Similarly, muscle ATP concent was assumed to be a constant 8 mM (15,23). Dichgans et al. (37) and the more recent studies of Reyngoudt et al. (12,30,31) are the only 1H-MRS studies reporting 'absolute' metabolite values, and also included the incorporation of some additional correction factors (see Table 4).
Functional paradigm
Three different functional 1H-MRS studies used three different functional paradigms, as can be seen in Table 4 (28,31,34,64). Stimulation duration varied from around 13–15 min (28,31) to 25 min (34). In comparison, the studies of Mangia et al. mentioned earlier (58–60), performing functional 1H-MRS at 7 T using a radial red/black checkerboard (8 Hz), observed a significant increase in Lac and Glu of approximately 20% and 3%, respectively, following a stimulation of only 2 min. These Lac increases were reduced over time, implying an adaptation of Lac changes following visual stimulation.
Conclusions
Clearly, MRS studies in migraine have yielded disparate information on brain metabolism. The data regarding 31P-MRS in muscle are far scarcer, yet the results seem to be relatively reproducible. The variation found throughout these studies can be attributed to a number of factors, including heterogeneous migraine populations, migraine severity (attack frequency), medication history, time of measurement (ictal versus interictal), regions of interest, magnetic field strength, signal processing and/or quantification strategies. Sometimes important study details (such as attack frequency, age, gender and quantification steps) were not recorded and therefore it is not possible to determine the extent to which they may have affected the results.
Synthesis of major interictal brain MRS findings in migraine.

MwoA: migraine without aura, MwA: migraine with aura, S/FHM: sporadic and familial hemiplegic migraine, ATP: adenosine triphosphate, PCr: phosphocreatine, PP: phosphorylation potential, Lac: lactate.
We would like to stress that the depletion of brain high-energy phosphates reflects an imbalance between ATP production and ATP use in migraine patients, but we do not know whether this is due to a primary mitochondrial dysfunction or secondary to alterations in brain excitability. The 31P-MRS muscle studies support the former hypothesis. It is interesting to speculate that abnormalities are more apparent in the brain because it is under constant activation. Only when duly exercised, in fact, were muscle energetics similarly abnormal in migraine patients. In general, migraine can be considered a 'threshold disorder' or a 'biobehavioural disorder' (70), implying that an intrinsic metabolic defect may render the brain more susceptible to various factors triggering an attack. Triggering factors would act by increasing the metabolic demand or hampering ATP production, and when a certain threshold is reached in an organ that is rather near its maximum capabilities, such as the brain, they would then induce a metabolic crisis responsible for the headache attack.
Potential future research may comprise further robust, quantitative and multinuclear brain MRS studies in migraine, including patients with a single phenotype who experience a well-defined number of attacks and are not using prophylaxis, compared with a gender- and age-matched control group. Another possible next step could be the absolute quantification of muscle phosphorus metabolites, especially ATP, in migraine patients, because [ATP] was assumed to be constant (8 mM) in the muscle of migraine patients and controls in previous 31P-MRS studies. Finally, 13carbon-MRS could provide further insight into possible Lac and Glu dynamics in migraine pathophysiology.
Footnotes
Funding
This research is funded by the Special Research Fund PhD (grant no. B/07768/02).
Acknowledgements
The authors wish to thank Leslie Vlerick and Marjan Acou for critically reviewing the manuscript.
