Abstract
Background: Evidence for optimal medication-overuse headache treatment is lacking. Some experts suggest reduced symptomatic medication with prophylactics from the start of withdrawal, while others suggest a two-month drug-free period with multidisciplinary education.
Aim: To examine the acceptability, feasibility and outcome of these two regimes in a non-randomised open-label study.
Methods: Patients able to undergo outpatient detoxification, with medication-overuse headache that had previously been unsuccessfully treated by specialists and without significant co-morbidities were treated with (A) individual withdrawal with restricted symptomatic medication and prophylactics from Day 1 or (B) a two-month drug-free period and multidisciplinary education in groups. All patients received close one-year follow-up.
Results: Eighty-six of 98 patients completed follow-up. Both treatments proved highly effective—80.0% of Group A and 85.4% of Group B were cured of medication-overuse headache. Headache-frequency reduction was 40.2% in Group A and 38.4% in group B. In 48.9% of group A and 48.8% of group B, headache-frequency reduction was >50%. Programme B required fewer resources from the staff and only 61.9% of these patients needed prophylactics after two months compared with 84.8% in programme A.
Conclusions: Both structured detoxification programmes proved highly effective with one-year close follow-up in previously treatment-resistant patients with medication-overuse headache. We suggest multidisciplinary education for patients in groups and delaying initiation of prophylactics until after the detoxification.
Keywords
Introduction
While medication-overuse headache (MOH) is well recognised, it remains uncertain how best to treat it. Only a few studies have compared different MOH treatments, and agreement on how to treat these severely affected patients is lacking. While it is agreed that highly motivated patients who do not suffer from significant co-morbidities and who do not overuse strong opioids, tranquilizers and/or barbiturates should be offered outpatient detoxification (1), recommendations for outpatient management differ considerably between headache centres and no internationally accepted guidelines exists. At the Danish Headache Centre (DHC), we have used abrupt discontinuation of the overused medication followed by a two-month drug-free period (2) where the patient is allowed to take only specified sedatives and no analgesics. We subsequently allow restricted intake of symptomatic medication—up to nine days/month for ergotamine, triptans, opioids and/or combination analgesics and up to 14 days/month for simple analgesics—and use prophylactic treatment as needed (3). Others suggest a short withdrawal phase where a reduced amount of analgesics is allowed and prophylactic treatment is initiated from the start (4,5). Clinical trials comparing a short withdrawal phase with a long drug-free period have, to our knowledge, not been conducted.
It has previously been argued that prophylactic medications have no or little effect in patients with MOH (6). One study demonstrated that patients who had not experienced any effect of prophylactic medications during the medication overuse, did experience an effect of prophylactics after a two-month drug-free period (7). A few recent studies show a modest effect of prophylactic medications in MOH patients (8–12) but these studies are hampered by a small number of patients, a very short follow-up period, or do not sufficiently distinguish between chronic migraine and MOH.
We hypothesized that a two-month drug-free period would educate the patients by showing that headaches can be endured without analgesics and that close follow-up by a multidisciplinary team would result in fewer drop-outs and relapses and less consultations with the physicians. Further, we hypothesized that initiating prophylactic therapy from the beginning of withdrawal would reduce headache frequency in the first two months but would imply a risk of overmedicating patients who do not need prophylactics after detoxification.
We aimed to examine the acceptability, feasibility and outcome of two different outpatient detoxification programmes for patients with MOH previously unsuccessfully treated by specialists: (A) an individual-based, one-week withdrawal with prophylactic medication from the start and restricted intake of symptomatic medications and (B) a two-month drug-free period where patients followed a multidisciplinary educational programme on MOH in groups.
Methods
Patients
Patients from DHC were recruited consecutively. DHC is a specialized, tertiary headache centre and all patients referred to the centre have already been seen by their general practitioner and most patients by at least one neurologist and/or other specialist (13). Inclusion criteria were: MOH fulfilling the criteria defined by the International Headache Society in 2006 (3): headache present on ≥15 days/month, regular overuse for >3 months of ergotamine, triptans, opioids or combination analgesics on ≥10 days/month, or simple analgesics or any combination of ergotamine, triptans, combination analgesics or opioids on ≥15 days/month. Further, patients should have received treatment for MOH by a specialist in neurology or neurosurgery, either as a structured detoxification programme or, as a minimum, advice on stopping medication overuse, but should have failed to withdraw from medication overuse. Patients were considered ineligible if they failed to complete a baseline headache diary or were deemed unable to undergo outpatient detoxification by the examining physician, either because they were overusing substances that presented too great a medical risk for outpatient withdrawal or they had significant co-morbidities expected to complicate outpatient withdrawal. Approximately 80% of all MOH patients referred to DHC are treated in such an outpatient programme (14).
Patients were enrolled in programme A from 1 August 2008 to 31 January 2009 and in programme B from 1 February 2009 to 31 November 2009 after providing informed consent. Patients not wishing to participate were offered an individual treatment approach consisting of two sessions with the headache nurse and a two-month drug-free period, after which prophylactic therapy was initiated if needed according to the general practice at DHC.
Study design
Figure 1 illustrates the study design.
Flowchart of the treatment programmes regarding planned contacts and headache-diary keeping. Shading: headache-diary keeping; P: planned contact with physician; N: planned contact with headache nurse; PT: planned contact with physiotherapist; PS: planned contact with psychologist.
At a baseline visit before entering the detoxification programme, patients were interviewed and generally and neurologically examined by a physician experienced in headache. Patients who met inclusion criteria were then referred to one of the treatment programmes. For patients referred to programme A, the type of prophylactic therapy was decided at this visit by the physician according to the patients’ primary headache, efficacy and side effects of previous treatments, co-morbid disorders and preferences.
Programme A
On Day −28 (i.e. four weeks before detoxification) patients were informed individually about MOH and the treatment programme by a specialized headache nurse, and began keeping a headache diary. On Day −1, the headache nurse checked the diaries and finally enrolled the patients who still fulfilled inclusion criteria, and handed out prescriptions for prophylactic medication, rescue medication and symptomatic medications. On Day 1, the overused drugs were abruptly discontinued and prophylactic medication prescribed by the physician at the baseline visit was initiated. From days 1–7, patients were allowed three days with either naproxen 500 mg up to two times a day or acetaminophen 1 g up to three times a day. Additionally, the patients were allowed to take rescue medication: promethazine 25 mg or levomepromazine 25 mg up to three times a day, and metoclopramide 20 mg up to three times a day (2). From day 8, the patients were allowed two days/week with symptomatic medications prescribed by the physician at the baseline visit. Symptomatic medications included simple analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), triptans, and combination analgesics, all except the previously overused drugs. Prophylactic treatment was prescribed by the physician at the baseline visit according to the patients’ primary headache, efficacy and side effects of previous treatments, co-morbid disorders and preferences, and was started from day 1. After two months, the patients saw the headache nurse, who collected the diary and checked the medication use, and the physician, who evaluated and adjusted the prophylactic and symptomatic medications and initiated physical and behavioural therapies with the multidisciplinary team if needed. Four months after starting detoxification, the patients were interviewed by telephone by a headache nurse, checking that they had not relapsed to medication overuse. They were seen by both the physician and headache nurse six months after detoxification. Patients continued keeping the headache diary until six months after starting detoxification.
Programme B
Patients attended a “headache school”: a multidisciplinary educational programme where, in groups of up to seven patients, they received education on MOH and headache in general from a team of headache nurses, physiotherapists and psychologists in six two-hour sessions on days −28, −1, 14, 21, 35 and 63.
On day −28, the patients received standardised information on MOH and the detoxification programme and started keeping a headache diary. On day −1, the headache nurse checked the diaries and enrolled the patients who still fulfilled inclusion criteria, and handed out prescriptions for rescue medication prescribed by the physician at the baseline visit. All symptomatic medications were stopped on day 1, simultaneously for all patients within the group. For the following two months, the patients were advised to take only sedative and anti-emetic rescue medications—promethazine or levomepromazine 25 mg up to three times a day and/or metoclopramide 20 mg up to three times a day (2)—but no symptomatic medications or prophylactics.
After the two-month drug-free period and again after six months, the patients were seen individually by a physician who initiated prophylactic treatment and physical and behavioural therapy with the multidisciplinary team if needed.
Twelve-month follow-up
All patients in both programmes received a four-week headache diary and a questionnaire 12 months after starting treatment together with a letter requesting them to complete these or to contact DHC if they did not wish to do so. Patients who were unable to keep a headache diary or did not wish to do so were invited to a telephone interview 12 months after starting detoxification.
Between months 6 and 12, all patients were seen regularly by the physician approximately every fourth month and could contact the headache nurses by telephone if needed.
Headache diary
Patients in both groups were requested to keep a headache diary from day −28 until two months after starting detoxification and again for four weeks up to the 12-month follow-up. The diary assessed headache frequency, duration, intensity on a 1–3 scale and characteristics, as well as medication use (15).
Questionnaires
Patients were asked to complete the migraine disability assessment (MIDAS) (16), the WHO quality of life questionnaire (WHO QoL) (17) and the hospital anxiety and depression scale (HADS) (18) at baseline and again at the 12-month follow-up.
The study was approved by the local ethics committee.
Statistics
The primary outcomes were patients cured of MOH, defined as patients no longer overusing medication; responders, defined as patients with ≥50% reduction in headache frequency (19); and relative reduction in headache frequency from baseline to 12 months after withdrawal.
The secondary outcomes were number of responders, patients cured of MOH, drop-outs, and relative reduction in headache frequency after two months; differences from baseline to 2 and 12 months in headache duration, headache intensity, and headache index (intensity × duration), symptomatic and prophylactic medication intake, and number of patients with episodic headache; relapse to MOH between month 2 and 12; use of resources defined as contacts to DHC in the 12 months, and absence from work derived from the MIDAS questionnaire.
As this study was non-randomised, between-group comparisons were made only for explanatory reasons and for the purpose of hypothesis generation.
Data are presented as means and ranges. Drop-outs were excluded from calculations of the primary outcome measures and were included only in the baseline data and for calculating drop-out rates. We used the Student’s t-test for data following a normal distribution. For data with skewed distribution, we used the Mann-Whitney U-test. For dichotomous data we used Fischer’s exact test for samples of less than five and the χ2-test for larger samples. For pair-wise related samples we used the related samples Wilcoxon’s signed ranks test.
Results
Baseline
During the study period, 383 patients were referred to the DHC under the diagnosis of MOH. Of these, the physician considered 71 patients eligible to enter programme A and 67 eligible to enter programme B at the baseline visit. On day −28, 12 patients, four in programme A (5.6%) and eight in programme B (13.1%), did not wish to participate and 28 patients no longer met inclusion criteria, 17 because they no longer overused medication, two because they disclosed a significant co-morbid disorder that had not been apparent at the baseline visit and nine because they had not completed a baseline diary.
From August 2008 to January 2009, we included 51 patients in programme A and from February 2009 to November 2009, we included 47 patients in programme B. Baseline data on the 98 enrolled patients are listed in Table 1; Figure 2 illustrates patient flow throughout the study.
Patient flow throughout the study. MOH: medication-overuse headache. Clinical and demographic data on the 98 included patients. Data are presented as mean (range) unless otherwise stated. Headache index: headache duration × headache intensity. Polyabusers: patients overusing more than one medication type. WHO quality of life ranges from 5–40, 5 indicating the lowest and 40 the highest quality of life.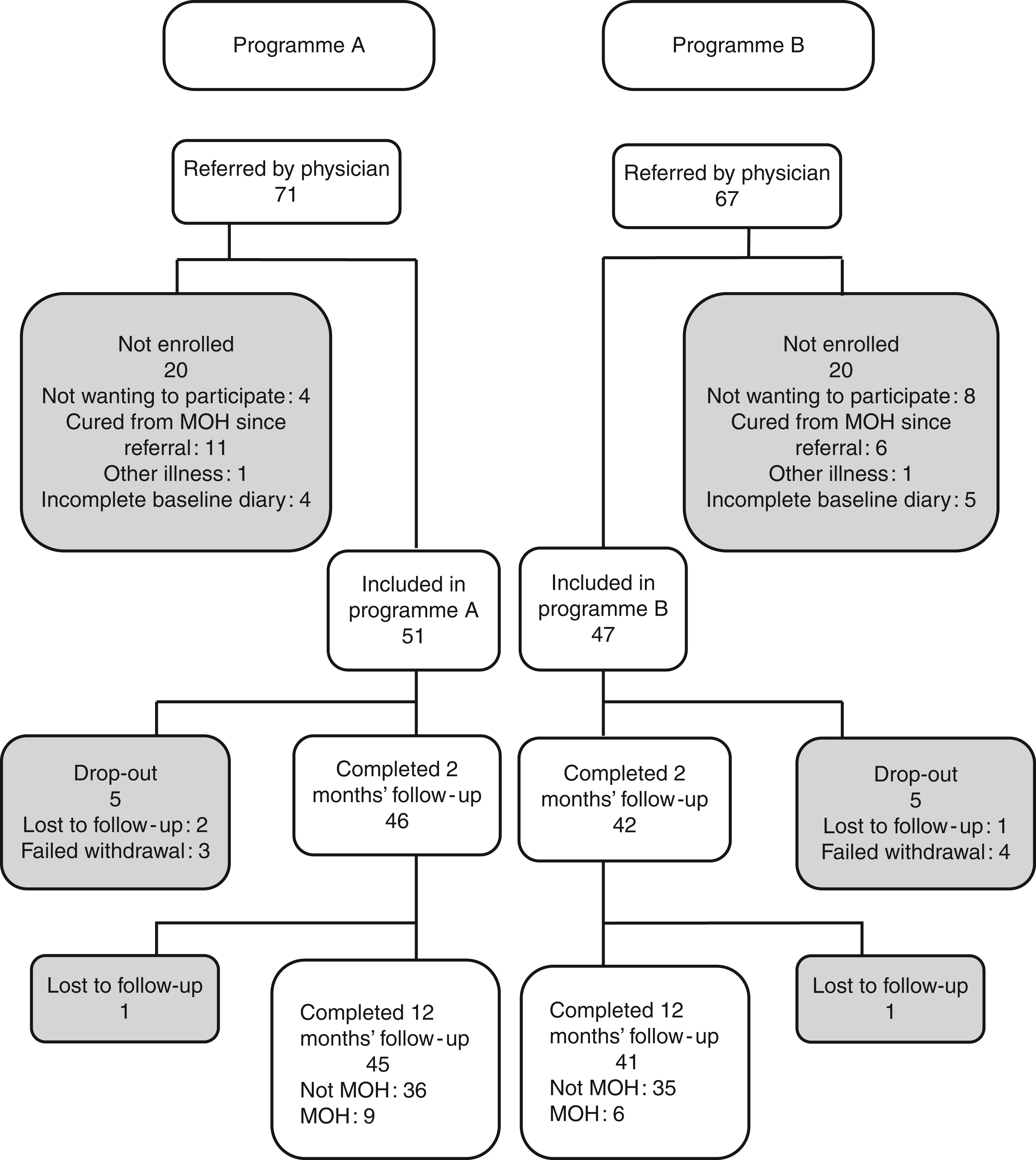

In programme A, 40 patients (78.4%) were started on prophylactic medication from day 1: 13 received a beta-blocker: 11 metoprolol and 2 propranolol; 12 received an antidepressant: 10 amitriptyline and 2 mirtazapine, 11 candesartan, 3 topiramate and 1 flunarizine. The offer of prophylactics was declined by 11 patients (21.6%). All patients were prescribed rescue medication and/or symptomatic medication: 42 patients received levomepromazine, 7 promethazine, 45 a simple analgesic or NSAID and 25 a triptan.
In programme B, 41 patients were prescribed levomepromazine and 5 received promethazine. One patient declined rescue medication, because it had been ineffective in her previous unsuccessful detoxification.
Primary outcomes
Results of the 12-month follow-up.

Data are presented as mean (range) unless otherwise stated.
Cured of medication overuse headache: patients who no longer overuse medication.
Responders: patients with a reduction of more than 50% in headache frequency from baseline.
Headache index: headache intensity × headache duration.
WHO quality of life ranges from 5–40, 5 indicating the lowest and 40 the highest quality of life. Within-group comparisons: ***p < 0.001 compared with baseline; *p < 0.05 compared with baseline.
Secondary outcomes
Results of the two-month follow-up.
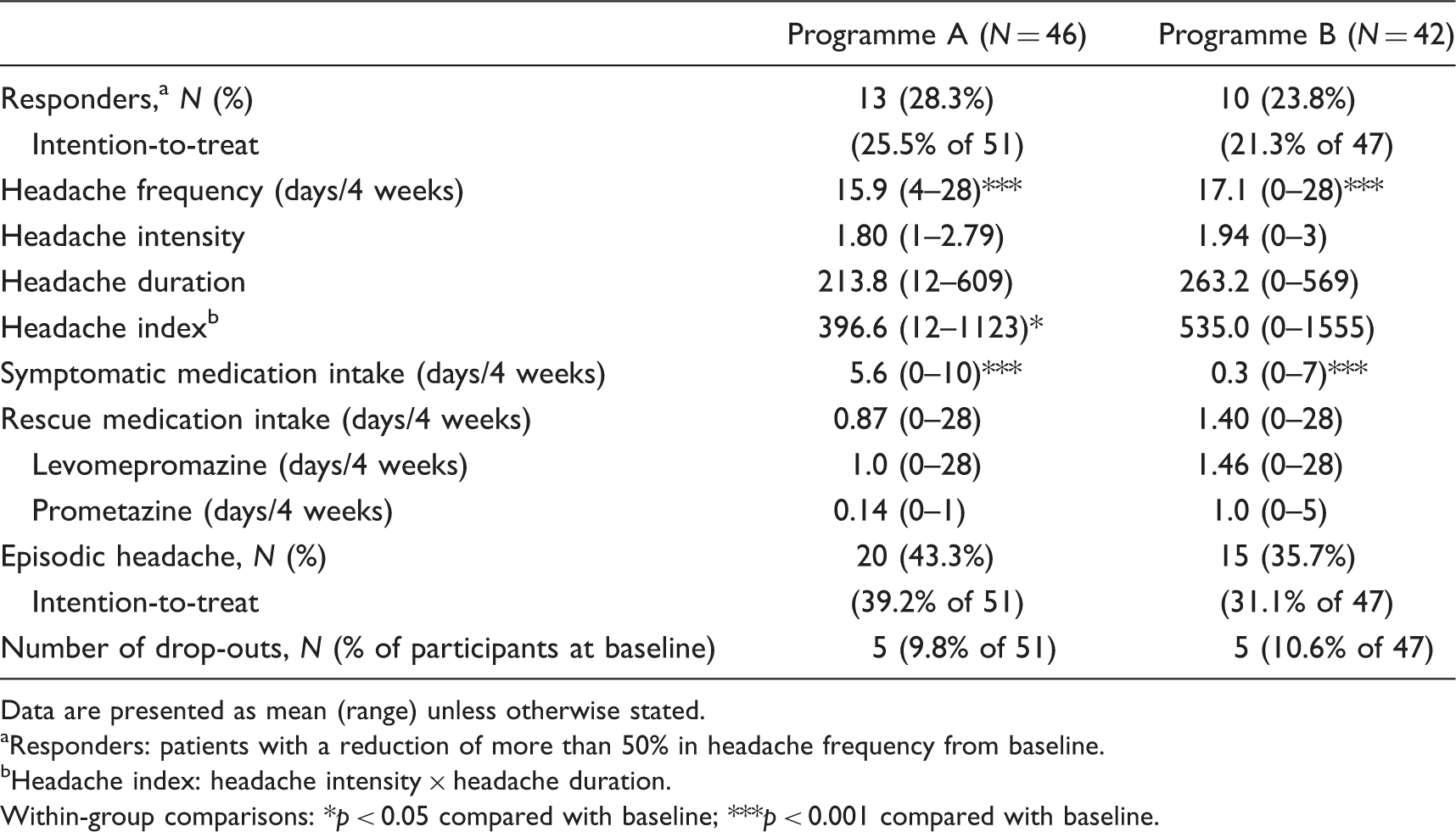
Data are presented as mean (range) unless otherwise stated.
Responders: patients with a reduction of more than 50% in headache frequency from baseline.
Headache index: headache intensity × headache duration.
Within-group comparisons: *p < 0.05 compared with baseline; ***p < 0.001 compared with baseline.
At the 12-month follow-up, 72 patients (83.7%) sent in a complete diary and 14 (16.3%), eight from programme A and six from programme B, were interviewed by telephone, and two, one in each programme, were lost to follow-up. Those two patients were both responders at month 2. Of the 86 remaining patients, nine from group A (19.6%) and six from group B (15.4%) relapsed into MOH. Patients in programme B had a medication intake on 6.5 days/4 weeks, whereas patients in programme A had a medication intake on 8.7 days/4 weeks. Data on the 86 patients who completed the study are listed in Table 2.
Prophylactic medication
In programme A, 40 of the 51 patients (78.4%) received prophylactic medication from day 1. After two months, there were no differences in headache parameters or medication intake between the patients with and without prophylactics in programme A. Of the patients who received prophylactic medication from day 1, 24 (64.8%) continued with unchanged medication, 10 had their medication changed and three discontinued prophylaxis. In total, 34 of the 37 patients who completed the two-month withdrawal (91.9%) continued on prophylactics. Of the nine completing patients not receiving prophylactics in the first two months, five (55.6%) were prescribed prophylactics at the two-month physician visit. In programme B, 26 of the 42 patients who completed the two-month detoxification (61.9%) received prophylactics at the two-month physician visit.
At the 12-month follow-up, 32 patients (88.9%) who had received prophylactics in from day 1 were taking prophylactic medication. For patients not receiving prophylactics from day 1, this number was four (44.4%) in programme A and 23 (56.1%) in programme B.
Resources required for the detoxification programmes
Resources used in the detoxification programmes.

Data are presented as mean (range) unless otherwise stated.
Psychologist and nurse were both at the headache school for 0.5 hours. Thus, the total time used by the multidisciplinary team apart from the physician, was 12.5 and not 12 hours per headache school.
Between-group comparisons for hypothesis generation
There were no statistically significant differences in the baseline data for patients in programme A versus programme B.
We compared all results obtained at the two- and 12-month follow-up of programme A with the results of programme B. Only three outcomes differed significantly between the groups. These were frequency of medication intake at month 12, which was 8.7 days in programme A and 6.5 days/4 weeks in programme B (p = 0.03); use of prophylactic medication at month 12, which was used by 80% in programme A and 56.1% in programme B (p < 0.001); and unplanned contacts to the physicians of the headache centre during the 12-month follow-up, which was 1.09 contacts in programme A and 0.66 in programme B (p = 0.03).
Discussion
In this study of patients with treatment-resistant MOH, two different treatment programmes both showed clinically impressive improvement on all primary efficacy parameters: 80% in programme A and 85.4% in programme B were cured of MOH; 48.9% in programme A and 48.8% in programme B were responders; and headache frequency was reduced by 40.2% in programme A and 38.4% in programme B. Further, the patients had a significant reduction in headache-related disability and an improved quality of life. These data demonstrate the importance of a structured detoxification programme and close follow-up for MOH patients previously regarded as treatment-resistant. Further, the drop-out rates and numbers of patients not wanting to participate were fairly low for both treatment programmes. This indicates a high acceptability from the patients. It has previously been thoroughly debated whether or not to detoxify MOH patients (20,21). This study adds weight to the argument of attempting detoxification even in patients who have previously been regarded as treatment-resistant. The reduction in headache days seen after a two-month drug-free period was comparable to or greater than the effect seen in placebo-controlled studies of prophylactic therapy for MOH (11,12,22,23).
During the first two months, our multidisciplinary team of physicians, headache nurses, physiotherapists and psychologists used less time per patient in programme B compared with programme A, even though those in programme B were seen more often in the first, difficult time of the detoxification. This was possibly due to the low-resource-demanding group design. During the first two months, 95.3% of patients who completed programme B were completely abstinent from symptomatic medication and yet the drop-out rate was similar to that of programme A and lower than in most other MOH studies where both symptomatic and prophylactic medications were allowed during withdrawal (24–26), even though patients in programme B had only anti-emetics and sedatives, to relieve nausea and help them relax. The drug-free period gave the patients an opportunity to discover how to function without symptomatic medication and to evaluate different headache episodes without automatically taking medication and on this background develop alternative coping strategies.
A recent study has shown that MOH patients request education and information about the disease and how they can be active participants in the treatment (27). Patients in programme B attended such an educational programme in groups. In addition to what the patients learned from the multidisciplinary team, the group provided an opportunity to exchange experiences and gain support from patients in the same situation as themselves (28). Apparently, patients in programme B benefitted from the multidisciplinary approach and the group support. The group dynamic and experiences of other patients may have helped these patients gain confidence and the ability to cope with the challenges of a restricted medication intake when suffering from headache. This could explain the fairly low drop-out rate in this strict detoxification programme compared with the drop-out rate of 36% in a previous study of the two-month drug-free period, where only some of patients received multidisciplinary education in groups (2).
We evaluated two different drug regimes as well as the effect of a multidisciplinary education programme in an open-label design. In the feedback from the patients, many expressed appreciation of the education in groups, but none appreciated the drug-free period. Multidisciplinary treatment for headache is increasingly acknowledged (29). We therefore conclude that the multidisciplinary treatment is most likely responsible for the low consumption of symptomatic medication in programme B at month 12 and thus suggest multidisciplinary education in groups but this must be tested in a randomised controlled trial. Randomised studies are needed to finally answer whether use of restricted analgesics should be recommended from the start of withdrawal or after a drug-free period.
We found that prophylactic headache medication was relevant only for less than two-thirds of patients in programme B after two months. Rossi et al. found no difference in headache frequency at two-month follow-up between the prophylactics and the non-prophylactics groups (30). These results indicate that the use of prophylactics in the first two months is irrelevant for quite a number of patients. In contrast, Hagen et al. (8) found a significantly lower headache index in the prophylactics group. But the groups in their study were not directly comparable, as patients in the prophylactics group were allowed free amounts of symptomatic medication and those in the detoxification group were not. Thus, these results are not directly comparable to ours. A recent German study showed that only 35% of patients adhere to the prescribed prophylactic treatment after one year, mainly due to side effects or ineffectiveness (31). In our study, 88.9% of patients receiving prophylactic medication from day 1 continued using prophylactics at month 12. We suggest that some patients receiving prophylactics from the beginning of withdrawal have been exposed to unnecessary medication with the disadvantages of side effects and increased costs. Both patient and physician may have been afraid to discontinue prophylactic medications and risk increasing headache. Patients in both programmes improved significantly from baseline to the two-month follow-up, but as this study is not randomised, we are unable to conclude whether the effect of the prophylactic medication outweighs the risk of overmedication. A randomised, controlled study with or without prophylactics is needed. As a consequence of our results and the results found earlier by Zeeberg et al. (7), we in DHC continue postponing the possible initiation of prophylactic medications to the end of a two-month detoxification period.
As previously recommended, the diagnosis of MOH was made on the basis of both a diary and clinical interview (32). Another strength of our study is the one-year follow-up period (8). Between the two- and 12-month follow-ups, patients further improved on most outcome measures and the relapse rate was low.
For practical reasons, patients were included consecutively, first in programme A then in B. This means that the study was non-randomised, which could theoretically result in bias due to a time effect. A drawback of this study is the lack of a control group. As previously stated (8), this type of study is difficult to design with a control group, as even simple advice will affect patients (30,33). Also, we find it unethical to leave these severely affected patients in a medication-overuse situation that they were clearly unable to terminate even with the help of other specialists. These patients have a long history of MOH, and as other studies show that a large proportion of patients relapse to MOH during the first year after detoxification (7,24–26,34,35,36–38), the results seen in our study are unlikely to be regression towards the mean.
This study was done on a highly selected patient population, who had all previously failed withdrawal one or more times. The types of drugs overused in this study are representative for the drugs used by the Danish headache-patient population, where tranquilizers and barbiturates only very rarely are used (2). Thus, most patients referred to the headache centre can be treated as outpatients (12). Also, Danish patients in general are reluctant to take medication on a daily basis, such as prophylactic medication. This may be different in countries with a different approach to drug consumption.
In conclusion, a structured detoxification programme for patients with previously treatment-resistant MOH is highly effective. For future treatment programmes on MOH we suggest a multidisciplinary educational programme for patients in groups, as this is less time-consuming for staff. In addition, postponing prophylactic medications until two months after the start of withdrawal might reduce unnecessary medication, but further controlled studies are needed. Although the design of this study does not permit conventional comparisons between the two methods, we hope that this study provides inspiration and relevant information that will help in calculating sample sizes for future studies on treating MOH.
Footnotes
Funding
The study was supported by grants from “IMK Almene Fond”. The funding source was not involved in any stage of the study.
Acknowledgements
The authors thank Mrs Annette V Rasmussen for assisting in practicalities in conducting the study and Mrs Hanne Andresen for technical assistance during data collection. The study was supported by grants from IMK Almene Fond. The funding source was not involved in any stage of the study.
Conflict of interest
Signe B Munksgaard has no disclosures. Dr Lars Bendtsen has received honoraria for lectures from MSD and Pfizer and serves on the scientific advisory board for Berlin-Chemie and Allergan. Dr Rigmor H Jensen has received honoraria for lectures and patient leaflets from MSD and Pfizer and serves on medical advisory boards for Allergan, LindeGas, ATI and NeuroCore.
