Abstract
We have shown that in healthy volunteers (HV) one session of 1 Hz repetitive transcranial magnetic stimulation (rTMS) over the visual cortex induces dishabituation of visual evoked potentials (VEPs) on average for 30 min, while in migraineurs one session of 10 Hz rTMS replaces the abnormal VEP potentiation by a normal habituation for 9 min. In the present study, we investigated whether repeated rTMS sessions (1 Hz in eight HV; 10 Hz in eight migraineurs) on 5 consecutive days can modify VEPs for longer periods. In all eight HV, the 1 Hz rTMS-induced dishabituation increased in duration over consecutive sessions and persisted between several hours (n = 4) and several weeks (n = 4) after the fifth session. In six out eight migraineurs, the normalization of VEP habituation by 10 Hz rTMS lasted longer after each daily stimulation but did not exceed several hours after the last session, except in two patients, where it persisted for 2 days and 1 week. Daily rTMS can thus induce long-lasting changes in cortical excitability and VEP habituation pattern. Whether this effect may be useful in preventative migraine therapy remains to be determined.
Keywords
Introduction
Repetitive transcranial magnetic stimulation (rTMS) is able to activate or inhibit the underlying cortex depending on whether high (5–20 Hz) or low (≤1 Hz) frequency stimulations are used (1, 2). We were first to study the effect of rTMS on the visual cortex by using pattern-reversal visual evoked potentials (PR-VEP) in healthy volunteers and migraine patients (3, 4). Besides amplitude of the N1–P1 and P1–N2 components after a number of averagings, which is the classically used measure, we determined PR-VEP habituation, i.e. amplitude reduction, over sequential blocks of averagings, which is deficient or replaced by potentiation in migraineurs between attacks (see 5 for a review). In our initial study (3) we compared the effects of 1 Hz and 10 Hz rTMS on PR-VEP in migraine patients and healthy volunteers. In the latter, PR-VEP amplitude was significantly decreased in the first block of 100 averagings after 1 Hz rTMS and habituation over six sequential blocks significantly attenuated (or dishabituation); 10 Hz rTMS had no significant effect. In migraine patients, 10 Hz rTMS was followed by a significant increase of first block PR-VEP amplitude and potentiation replaced by a normal habituation pattern, while 1 Hz rTMS was ineffective.
We have previously determined the precise duration of the rTMS effect on PR-VEP. One session of 1 Hz rTMS in healthy subjects abolishes habituation of the PR-VEP N1–P1 component on average for 33 min (4), whereas 10 Hz rTMS in migraineurs normalizes habituation for a mean duration of 9 min (3). It has been hypothesized that the interictal habituation deficit may play a pathogenic role in migraine (6), because one of the physiological roles of habituation is to protect against overstimulation (7). Habituation depends on complex neurobiological mechanisms controlling neuronal excitability and inhibition. In migraine patients amplitude and habituation of evoked (8) and event-related potentials (9, 10) undergo marked changes in temporal relation to the attack.
There is evidence that rTMS can induce long-lasting modifications in activity and metabolism of cortical neurons (11–13). This may explain some of the therapeutic effects found with rTMS in neuropsychiatric disorders (14). It remains to be demonstrated that normalizing PR-VEP habituation in migraineurs by high-frequency rTMS is able to prevent attacks. However, before such a strategy can be realistically explored, it seems necessary to obtain rTMS-induced cortical changes that last for more than 9 min. This is why we have now studied the effect on PR-VEP habituation between and after five daily rTMS sessions both in healthy volunteers with the 1 Hz frequency and in migraine patients with the 10 Hz frequency.
Subjects and methods
Subjects
We studied eight healthy control subjects (HV) without personal or family history of primary headaches (three women and five men; mean age 23.2 ± 1.9 years) and eight out-patients with migraine recruited from a specialized headache clinic (five females and three males, mean age 23.3 ± 1.0 years). According to International Headache Society criteria (15), six patients had migraine without aura (MoA, code 1.1) and two migraine with typical aura (MA, code 1.2.1). All subjects were devoid of any other medical condition and had no personal or family history of epilepsy, which is recommended for rTMS studies (16). They had between one and two attacks per month (mean 1.8 ± 0.4) and a history of migraine ranging from 2 to 9 years (mean 5 ± 3.2). None of them received prophylactic antimigraine treatment for at least 3 months before study, nor used ergotamine as acute treatment. All the recordings were made in the headache-free interval, at least 3 days before and after an attack. The latter was verified by a telephone call given 4 days after the recordings. To avoid interference with changes of cortical excitability due to hormonal variations (17, 18), females were recorded at mid-cycle, i.e. 12–16 days after the first day of menses. Written informed consent was obtained from all subjects. The study was approved by the Ethics Committee of the Faculty of Medicine, University of Liège, Belgium.
Recordings of pattern-reversal visual evoked potentials
PR-VEP recordings were performed as described in a previous study (3). Briefly, stimuli were presented as a checkerboard pattern of black and white squares (8 min of arc) at a reversal frequency of 3.1 Hz. During uninterrupted stimulation, six sequential blocks of 100 responses were averaged for a total duration of 3 min. PR-VEP were recorded before and immediately after the rTMS. To evaluate the duration of the rTMS effect beyond 3 min, PR-VEP were monitored for up to 123 min after rTMS according to the following protocol: 3-min PR-VEP recordings alternating with 3-min resting periods up to 15 min, with 7-min resting periods up to 60 min and with 12-min resting periods up to 123 min. We repeated this protocol daily on the five consecutive days of rTMS application. Thereafter, we performed 3-min PR-VEP recordings for another 2 days and then once per week in subjects with lasting rTMS-induced changes.
Transcranial magnetic stimulation
We used a Magstim Rapid® magnetic stimulator (Magstim Co. Ltd, Whitland, Dyfed, UK), connected to a double 7.0-cm figure-of-eight-shaped coil, with a maximal stimulator output of 1.2 T. We first identified the phosphene threshold (PT), using single TMS pulses and the same procedure as previously described (3). For rTMS of the visual cortex, the coil was positioned as for the phosphene detection: vertical position, the handle pointing upwards, on the inion-nasion line, inferior limit 1 cm above the inion. Stimulus intensity was set to the PT. We used two different stimulation frequencies: 1 Hz (low-frequency rTMS) in healthy volunteers without interruption for 15 min, and 10 Hz (high-frequency rTMS) in migraineurs in 18 trains of 5 s with intertrain intervals of 10 s. For both frequencies, the same number of 900 pulses was thus delivered daily for five consecutive days at the same hour. The PT, and thus the stimulation intensity, was redefined at each session, since we have shown that magnetophosphene thresholds can vary by more than 10% when tested on different days (19). Four healthy volunteers chosen at random underwent five consecutive 1 Hz rTMS sham stimulation sessions during which the coil was perpendicular to the skull. Four migraineurs (chosen at random) also underwent five 10 Hz rTMS sham sessions in the same conditions. In the sham situation, there is a subjective sensation and noise similar to that achieved with effective rTMS, but no brain activation occurs, which is not known to the subjects.
Data analysis
We analysed the six consecutive blocks of 100 PR-VEP responses in terms of peak-to-peak amplitudes of the N1–P1 component. The N1 peak was defined as the most negative point between 60 and 90 ms after the stimulus and P1 as the most positive point following N1 between 80 and 120 ms. PR-VEP habituation was defined as the ratio between amplitudes of the first over the sixth block of 100 averaged responses expressed as percentage, a positive value indicating habituation, a negative one absence of habituation or potentiation.
N1–P1 habituation was measured 12 times after each rTMS session for a total duration of 123 min in all subjects and the protocol was repeated daily for five consecutive days. In order to obtain a global view of the poststimulus effect, we have plotted the first/sixth block ratios for each PR-VEP recording (y axis) over the 123-min poststimulus observation period (x axis) and measured the negative area under the curve (AUC) (i.e. potentiation) for each subject and each daily rTMS session. For both subject groups mean AUC and standard deviations (SD) were calculated.
Statistical analysis
Means ± SD were calculated for habituation on the first day of the rTMS session and paired Student's t-tests were used to assess the effect of rTMS on the variable. Means ± SD were calculated for the PT during the five rTMS sessions. A generalized linear mixed model (GLMM) was used to study the evolution of the PT during the 5 days of stimulation. AUC and mean AUC were also analysed with a GLMM. Results were considered significant at the 5% level (P < 0.05). Statistical calculations were carried out with SAS (Version 8.2 for Windows; SAS Institute, Cary, NC, USA) and S-Plus (Version 6.0 for Windows; Insightful Corp., Seattle, WA, USA).
Results
Acute changes induced by the first rTMS session
Before the first rTMS session, mean PR-VEP habituation in the eight healthy volunteers was 14 ± 11%. After this session (first 3 min of recording), this habituation was significantly decreased to −9 ± 19% (P = 0.0028). In migraineurs, habituation before the first rTMS session was replaced by potentiation (−22 ± 18%), which was significantly reversed to 0 ± 17% during the first 3 min of recording after rTMS (P = 0.043). With sham stimulation in healthy volunteers (n = 4) and in migraineurs (n = 4), the mean habituation after the first rTMS session did not significantly change.
Evolution over the 5 days of stimulation
In all eight healthy volunteers, the 1 Hz rTMS effect, i.e. the dishabituation, was still present at the end of the recording session on day 5. In the global group of eight migraineurs, the mean 10 Hz rTMS effect, i.e. the reduction of potentiation, was still present at the end of the recording session on day 5. However, when looking at individual data in migraineurs, one can see that two subjects had no prolongation of the 10 Hz rTMS-induced reduction of potentiation on successive sessions.
The average AUC of poststimulus changes in first/sixth PR-VEP block ratios remained negative between the first and the fifth days of stimulation in both groups of subjects. This illustrates that the 1 Hz rTMS-induced dishabituation persisted in healthy volunteers and the 10 Hz-induced reversal of potentiation remained incomplete in migraineurs for the whole 5-day period (Fig. 1). Quantitatively, however, changes in AUC differed significantly between the two subject groups (P = 0.023). In healthy volunteers the AUC increased significantly (P = 0.0096), while in migraineurs it tended to decrease over time without reaching the level of statistical significance (P = 0.068). This indicates that the dishabituation induced in healthy volunteers by daily 1 Hz rTMS (Fig. 2) and the reduction in potentiation induced in migraineurs by daily 10 Hz rTMS (Fig. 3) accrued between days 1 and 5. The habituation did not significantly change in both sham groups during the 5 days of stimulation.

Evolution over the 5 days of repetitive transcranial magnetic stimulation (rTMS) of the negative area under the curve (mean ± SE) for healthy volunteers (□, n = 8) and migraineurs (•, n = 8).
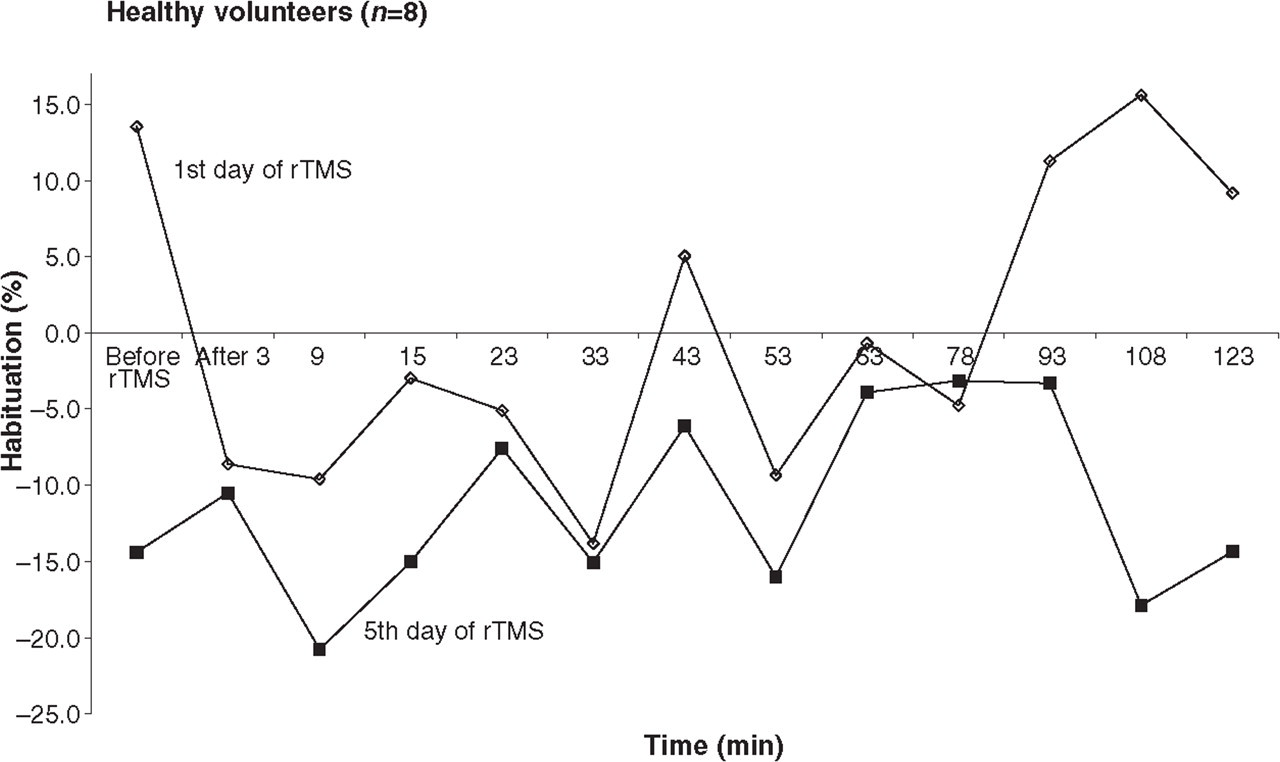
Evolution of N1–P1 habituation in healthy volunteers (n = 8) on days 1 and 5 of 1 Hz repetitive transcranial magnetic stimulation (rTMS).

Evolution of N1–P1 habituation in migraineurs (n = 8) on days 1 and 5 of 10 Hz repetitive transcranial magnetic stimulation (rTMS).
Evolution after the 5 days of stimulation
Four healthy volunteers, who still had a lasting 1 Hz rTMS-induced dishabituation on day 5, recovered their degree of baseline habituation the day after the last rTMS session. In the four others, the rTMS effect persisted for much longer periods. One subject still had PR-VEP dishabituation on day 21 after the last rTMS session, but no more on day 28. In another subject, the effect lasted for 28 days, in the third one for 42 days and in the fourth one for 63 days.
Four migraineurs, who still had a lasting 10 Hz rTMS-induced habituation on day 5, recovered their degree of baseline potentiation the day after the last rTMS session. Only two migraineurs had a 10 Hz rTMS-induced reduction of potentiation (or even habituation) outlasting the last rTMS session. In one of them the effect had disappeared after 2 days, but the other patient, suffering from MA, had a reduction of PR-VEP potentiation for 7 days.
Evolution of the phosphene threshold
PTs were not significantly different between the two groups. During the five successive days of stimulation, mean PT significantly decreased from 74% to 67% in HV, from 75% to 69% in migraineurs (P < 0.05).
Discussion
This study demonstrates for the first time that rTMS applied over the occipital scalp in daily sessions during five consecutive days has a cumulative effect on excitability of the underlying visual cortex in every healthy volunteer and in most migraine patients studied. The effect lasts several weeks in 50% of healthy subjects tested but only several hours in most migraineurs, except in two patients in whom it persisted for 2 and 7 days.
We have assessed visual cortex excitability by measuring habituation in six sequential blocks of 100 averaged PR-VEP responses before and for 123 min after each rTMS session and as long as habituation changes persisted after the fifth session. We also determined TMS thresholds for magnetophosphenes (PT) at each daily rTMS session. rTMS (1 Hz) was used in healthy volunteers, because it caused PR-VEP dishabituation in our previous study (3), while 10 Hz rTMS had no effect; by contrast, 10 Hz rTMS was applied in migraineurs, since it was able to transform PR-VEP potentiation into normal habituation whereas 1 Hz rTMS was ineffective.
In the present study, as in our previous studies (3, 20), we found no difference in PT between HV and migraineurs before rTMS. Although the study was not designed to assess PT changes over time and we have not retested PT after each rTMS session, but only before each session, the PT decrease between the first and fifth rTMS sessions in both groups must be discussed briefly. From the known effects of rTMS on cortical excitability, indeed, one would have expected an increase of PT in HV after the 1-Hz stimulation (21). Interestingly, a PT decrease with 1 Hz rTMS was also reported by Brighina et al. (22) in MA patients and thought to be abnormal, while we found a similar decrease with 10 Hz rTMS. The reason for these discrepancies is not clear. It must be kept in mind, however, that phosphenes are a highly subjective experience which lacks reliability (19) and their detection may be eased on repeated testing by a learning effect. A PT decrease with repeated sessions over time was indeed found in other studies both in controls and in migraineurs (23).
In four healthy subjects 1 Hz rTMS transformed for several weeks the normal pattern of PR-VEP habituation into potentiation which characterizes migraine patients between attacks. Only two out of eight migraineurs had a long-term reduction of the interictal PR-VEP potentiation (2 days and 1 week). The difference in duration of the rTMS effect between healthy volunteers and migraine patients could be due to the different stimulation frequencies used. That low-frequency rTMS may produce longer effects than high-frequency stimulations on the underlying cortex and on distant cortical sites is suggested by several studies of the motor cortex, although the results also depend on the method used to assess cortical excitability. For instance, a decrease of motor evoked potential (MEP) amplitude was observed for 15 min after a 15-min 0.9 Hz rTMS train over the motor cortex (24) and a decrease of intracortical inhibition for 3 min following 15 Hz motor rTMS (25). After 1 Hz rTMS over the premotor cortex, MEP amplitudes were reduced for 15 min (26), whereas changes of intracortical excitability lasted up to 1 h (27). Another obvious factor which might explain the differences in duration of the rTMS effect is that the cerebral cortex of migraine patients has interictally a permanent, though fluctuating, functional abnormality, i.e. dysexcitability with a habituation deficit (5).
Proceeding from the ‘ceiling concept’ of Knott & Irwin (28), we have hypothesized that the habituation deficit in migraine might be due to a reduced preactivation level of sensory cortices rather than to hyperexcitability as such (29). The above-mentioned study (3) which shows that excitatory 10 Hz, but not inhibitory 1 Hz, rTMS normalizes habituation in the visual cortex of migraineurs, favours this hypothesis. We have also speculated that the lack of habituation in cortical information processing could play a pathogenic role in migraine (6), as it might favour lactate accumulation (7, 30), leading to activation of the trigeminovascular system, and thus the attack, because of the interictal reduction in mitochondrial phosphorylation potential demonstrated in the brain of migraineurs (31). None of the healthy subjects developed migraine attacks during the observation period, nor at later stages up to 1 year after termination of the study, despite the long-lasting rTMS-induced dishabituation of PR-VEP. This could be due to the relatively short duration of the cortical effect (3–9 weeks) compared with almost lifetime duration of the habituation deficit in migraine (32). It seems more likely, however, that the abnormality in cortical information processing is only one factor in migraine pathophysiology and as such not sufficient to trigger a migraine attack.
If the habituation deficit plays a role in migraine pathogenesis, one might expect that its total or partial alleviation by high-frequency rTMS could have a preventative effect on attacks. The only migraine patient in whom 10 Hz rTMS reduced the PR-VEP potentiation for 1 week, suffered from migraine with typical aura and both his attack frequency (one attack/month) and the observation period were too small to draw any conclusion about such a therapeutic perspective. In fact, the rather short duration of the 10 Hz rTMS effect in most migraineurs casts doubt on the usefulness and feasibility of such an intervention, unless longer-lasting changes can be induced with higher stimulation frequencies, e.g. 20 Hz, which remains to be determined. Indeed, a very recent rTMS study showed that several 20 Hz rTMS sessions over the prefrontal cortex of chronic migraineurs can ameliorate the attack frequency, the headache index and the number of abortive medications (33).
An interesting feature of the interictal habituation deficit in migraine is that it normalizes spontaneously just before and during the attack (8, 10). This coincided in one study (34) with a decrease in platelet serotonin content and thus an increase in the biologically active compartment of serotonin, which is known to increase the preactivation level in sensory cortices (35). Taken together, these data suggest that the migraine attack is preceded and accompanied by an increase in cortical activation (9, 10). The latter is confirmed by preliminary results from a peri-ictal rTMS study performed by us (36) and could be another potential trigger for the migraine attack, as metabolic demands in the brain are expected to rise significantly just before the attack. If this is correct, a study of the preventative potential of low-frequency rTMS seems worthwhile, the more so that the cortical changes induced by 1 Hz rTMS last much longer than those observed with 10 Hz rTMS, at least in healthy volunteers. The method is testable without tedious daily long-term stimulations because it can be applied prophylactically for shorter durations in predictable attacks, such as those triggered naturally by menses or experimentally by nitroglycerin.
Footnotes
Acknowledgements
This study was supported by grant no. 3.4523.00 from the National Fund for Scientific Research (Brussels, Belgium) and grant no. 125 from the Migraine Trust (London, UK) to J.S. V.B. was a recipient of a Clinical Fellowship of the International Headache Society. This work was also supported by a grant from the ‘Fondation Léon Frédéricq’ from the Faculty of Medecine of the University of Liège to A.F. and M.V.
