Abstract
Introduction: Mood and anxiety disorders are comorbid with migraine and commonly assumed to portend a poor response to preventive migraine therapies. However, there is little evidence to support this assumption.
Method: We examined impact of a mood and/or anxiety disorder diagnosis using American Psychiatric Association Diagnostic and Statistical Manual criteria on response to the three preventative migraine therapies evaluated in the Treatment of Severe Migraine trial (n = 177): β-blocker, behavioral migraine management, or behavioral migraine management +β-blocker. Daily diaries assessed migraine activity for the 16 months of the trial. The Migraine Specific Quality of Life Questionnaire and Headache Disability Inventory assessed headache-related disability at regular intervals. Mixed models for repeated measures examined changes in these three outcomes with preventative migraine therapy in participants with and without a mood or anxiety disorder diagnosis.
Results: Participants with a comorbid mood or anxiety disorder diagnosis recorded larger reductions in migraine days (p < .05) and larger reductions in the Migraine Specific Quality of Life Questionnaire (p < .001) and Headache Disability Inventory (p < .01) than did participants with neither diagnosis.
Discussion: Significantly larger reductions in migraine activity and migraine-related disability were observed in participants with a mood and/or anxiety disorder diagnosis than in participants who did not receive either diagnosis.
Keywords
Introduction
Migraine is a prevalent and disabling disorder characterized by episodes of moderate to severe head pain and associated symptoms such as nausea, photo- and phonophobia (1). Migraine has been observed to be comorbid with mood and/or anxiety disorders (MoADs) (2–4) in both population (5–9) and clinic samples (10–13), raising the question of how the presence of a MoAD influences response to headache treatments. It is commonly assumed that the presence of a MoAD is associated with a poorer response to preventive migraine treatments, both preventive medications and behavioral treatments, for primary (migraine and tension-type) headache disorders in adults (14–17). However, only a handful of studies have directly addressed this question.
To the best of our knowledge, only three studies have used interview-based diagnosis, with Diagnostic and Statistical Manual for Mental Disorders (DSM) diagnostic criteria (18) to determine the presence of MoAD (19–21), although a fourth study (22) used a well-validated interview (Hamilton Rating Scales (23,24)). All four studies reported that the presence of a MoAD was associated with larger, rather than smaller, reductions in headaches and in headache-related disability with preventative headache therapy or reported that the presence of a MoAD diagnosis did not influence response to preventative therapy.
A second set of studies used self-report measures of symptoms of depression or anxiety, rather than an interview-based diagnosis with DSM diagnostic criteria (18). These studies report conflicting results, with individual studies reporting that higher scores on measures of self-reported symptoms of anxiety and depression are negatively associated, positively associated, or unassociated with observed reductions in headache activity with preventative therapies for primary headache disorders (22,25–32). Additional information is clearly needed about the role of MoADs in treatment response to preventative medication and behavioral treatments for primary headache disorders in adults.
In the present study we prospectively evaluate the relationship between the presence of a MoAD diagnosis and changes in migraine days and headache-related disability across the preventative migraine therapies evaluated in the Treatment of Severe Migraine (TSM) trial.
Method
Procedure
The current study is a secondary analysis of data from the TSM trial (33). A pretreatment evaluation included a structured headache interview, neurological and medical examinations, an interview for psychosocial history, and psychosocial and demographic questionnaires. Participants then entered a 5-week run-in of optimized acute therapy (OAT), individually tailored drug therapy for the acute treatment of migraine episodes. Participants who continued to experience three or more migraines with disability during OAT were stratified by sex and randomized to one of three preventative therapies designed to reduce the frequency and severity of migraine episodes, or to OAT alone. The three preventive therapies were β-blocker, behavioral migraine management (BMM), and BMM + β-blocker. Because the focus of the current analysis is on the influence of a MoAD diagnosis on the effectiveness of preventative migraine treatments, which differ notably from acute treatments in therapeutic targets and measurement of treatment response, the group that received only OAT as active treatment was not included in the current analysis. BMM and preventative medication dose adjustment took place during four monthly clinic visits (months 1–4), with three phone contacts in between visits to identify and address problems with medication adherence and/or use of BMM skills. The 12-month evaluation period included clinic visits at months 5, 7, 10, 13, and 16. Each clinic visit included the completion of psychosocial questionnaires. Participants recorded a daily headache diary for 16 months. For further information about BMM and the TSM trial, see Lipchik and colleagues (34) and Holroyd and colleagues (33).
Participants
Inclusion criteria were an International Classification of Headache Disorders diagnosis of migraine with or without aura 1 and ≥3 migraines with disability per 30 days (1). One hundred and seventy-seven severe migraine sufferers then received one of the three preventative treatments. Forty-six participants (26.0%) were diagnosed with a mood disorder (n = 20), anxiety disorder (n = 11), or both (n = 15) at the pretreatment evaluation. The most common mood disorder was major depressive disorder (n = 14) followed by depressive disorder not otherwise specified (n = 11) and dysthymia (n = 10). The most common anxiety disorder was generalized anxiety disorder (n = 12), followed by anxiety disorder not otherwise specified (n = 11) and panic disorder (n = 3). The comorbidity between mood disorders and anxiety disorders mirrored that observed in the US population (35). Thus, 43% (18/42) of participants with a mood disorder also received an anxiety disorder diagnosis and 58% (19/33) of the participants with an anxiety disorder also received a mood disorder diagnosis. The moderate sample size (n = 177) and the substantial comorbidity between MoADs did not allow us to examine the separate or individual effects of MoAD diagnoses on treatment outcome. Thus, participants who met criteria for a MoAD (or both) were compared with participants who did not meet criteria for a MoAD, consistent with the procedure used in other recent studies (20,36).
Demographic Characteristics

Means and standard deviations displayed for continuous variables. Percentages displayed for categorical variables. T-tests used to test for differences with continuous variables. Chi-square used to test for differences with categorical variables. PRIME-MD = Primary Care Evaluation of Mental Disorders; For all variables other than Household Income, Total n = 177; PRIME-MD Diagnosis n = 46; No PRIME-MD Diagnosis n = 131.
Sixty-six participants dropped out by month 10 (leaving 111 participants in the study) and an additional 23 participants dropped out by month 16 (leaving 88 participants who completed the study). Thus, the TSM trial had a low rate of dropout (37% at 10 months) relative to other clinical trials of preventative medication for migraine, which tend to report dropout rates of 50% at 6 months (37–39). Therefore, using a crude average over months, the dropout rate for the TSM trial (≈3% per month) was less than half of the comparative dropout rate for other recent preventative medication trials (≈8% per month). The dropout rate from the TSM trial appears either lower than (40), or comparable to (41,42), those reported in other trials of behavioral headache treatments, although few behavioral treatment studies report comparable dropout rates at 6 and 12 months following behavioral treatment.
Study dropout did not vary significantly by pretreatment MoAD diagnosis or treatment group, p > .50.
All participants provided written informed consent according to procedures approved by the Ohio University Human Subjects Committee.
Treatment
All participants received a 5-week run-in of OAT (individually tailored drug therapy for acute migraine treatment), including a triptan (5-HT1B/D-agonist) and, if judged appropriate, a nonsteroidal anti-inflammatory medication, anti-emetic medication, and/or rescue medications. For all participants, OAT continued throughout the course of the study.
Participants randomized to the β-blocker and BMM + β-blocker treatments received long acting propranolol HCl (or nadolol if propranolol was not well tolerated or ineffective) as preventative drug therapy. Dose adjustment occurred during months 1–4. Preventative drug therapy continued throughout the course of the study.
Participants randomized to the BMM and BMM + β-blocker treatment conditions received a minimal-contact psychological treatment program (34,43) during months 1–4. Project counselors demonstrated migraine management skills at each of the four monthly clinic visits, and participants applied migraine management skills between sessions with homework guided by the BMM patient manual and accompanying audiotapes (43).
Measures
Psychiatric comorbidity
Primary Care Evaluation of Mental Disorders (PRIME-MD)
The PRIME-MD Structured Clinical Interview (44) is designed to implement DSM criteria for the mental disorders most commonly encountered in primary care, including MoADs. A doctoral or masters’ level clinical psychologist experienced with DSM diagnosis administered the PRIME-MD structured interview to all participants, querying participants as needed to clarify diagnosis. The PRIME-MD has demonstrated good sensitivity and specificity for the diagnosis of both MoADs (44).
Outcomes
Headache Disability Inventory
The Headache Disability Inventory (HDI) (45) is a 25-item questionnaire designed to assess headache-related disability. Two rationally developed subscales were proposed by the authors of the HDI: Functional Disability (‘I avoid traveling because of my headaches’) and Emotional Distress (‘I feel desperate because of my headaches’). However, factor analysis suggests the structure of the HDI is best described by a single factor (46). The HDI has demonstrated high internal consistency (subscale score-total score r = 0.89). The measure also demonstrated good short-term (1 week; r = 0.78) and long-term (2-month; r = 0.83) test-retest reliability (45,47).
Migraine Specific Quality of Life Questionnaire
The Migraine Specific Quality of Life Questionnaire (MSQL) (48) is a 16-item questionnaire designed to assess migraine-related disability. The questionnaire can be divided into three subscales: Role Function-Restrictive, Role Function-Preventative, and Emotional Function. Sample items include ‘In the past 4 weeks, how often have migraines interfered with how well you dealt with family, friends, and others who are close to you?’ (Role Function-Restrictive), ‘In the past four weeks, how often have you had to cancel work or daily activities because you had a migraine?’ (Role Function-Preventative), and ‘In the past four weeks how often have you felt fed up or frustrated because of your migraines?’ (Emotional Function). The subtests have demonstrated adequate reliability (.79–.85) and are highly correlated (.84–.89) (48). Higher scores on the MSQL indicate higher levels of disability (e.g. higher impairment in quality of life).
Migraine days per 30 days
Participants entered headache activity into a Palm III hand-held computer (3 Com Corp.) throughout the course of the study (49). The hand-held computer prompted participants to record a headache activity entry each day. Information collected included whether or not the participant had a headache, the type of headache, time of day and length of headache, the severity of headache, sensitivity to light and sound, and presence of nausea or vomiting. A migraine day was coded as any day during which the participant recorded a migraine. The data were uploaded at each clinic visit starting with the treatment visit at which diary recordings for the OAT run-in were uploaded; therefore, 16 months of daily diary recordings were accumulated.
Demographic variables
Participants completed a questionnaire containing demographic information collected at the pretreatment evaluation, including age, gender, ethnicity, years of education and annual income. Ethnicity was a categorical variable, including ‘White, non-Hispanic’, ‘Black, non-Hispanic’, ‘Asian or Pacific Islander’, ‘Hispanic’, ‘American Indian or Alaskan Native’, and ‘Other’.
TSM trial outcomes
The TSM trial revealed clinically significant decreases in migraines per 30 days, migraine days per 30 days and MSQL scores with all four treatments (33). However, the addition of BMM + β-blocker and OAT yielded significantly larger improvements in each of the three outcome variables than β-blocker, BMM, or OAT alone, which did not differ from one another. See Holroyd and colleagues (33) for further details.
Analysis
Mixed models for repeated measures were used to examine whether a PRIME-MD structured interview diagnosis of a MoAD at pretreatment influenced changes in headache-related disability (HDI and MSQL) or migraine activity (migraine days per 30 days) with preventative treatment.
The HDI, the MSQL, and migraine days per 30 days each served as outcome variables in separate mixed models for repeated measures analyses. Each model included presence of a MoAD diagnosis, month, and their interaction. An exponential covariance structure was determined to be the best-fitting covariance structure using Akaike’s information criterion. Month was modeled using the natural log of month because changes in migraine days per 30 days and headache-related disability were expected to be curvilinear. A significant MoAD × time would indicate that change differed significantly in participants with and without a MoAD diagnosis on the outcome measure of interest. Where the above analysis revealed that treatment response differed significantly between participants with and without a MoAD diagnosis, t-tests were used to compare participants by MoAD diagnosis at the primary endpoint (month 10) and at the longer term follow-up (month 16). All significant tests were two-tailed. All analyses were conducted using SAS 9.2.
Mixed model analysis is preferable to last observation carry forward, an earlier method used for handling missing data in clinical trials, because the latter procedure yields unbiased parameter estimates only under the most restrictive assumption that data are missing completely at random: that is, missing observations are unrelated to both the observed (before dropout) and the unobserved (following dropout) outcome measure values. Moreover, the direction of bias when the missing completely at random assumption is violated is not easy to predict. In contrast, mixed model analysis, which uses all available data to estimate missing values, can yield unbiased parameter estimates when missing observations are related to observed (but not unobserved) outcome values (missing at random assumption). Thus, in the present study, mixed models analysis has the potential to yield unbiased estimates of missing values even if dropout were to differ by an observed variable (i.e. the presence of a MoAD). Moreover, mixed model analysis performs surprising well even on simulated clinical trial data that includes both missing at random observations and observations missing not at random (50–55). Of course, analysis of only the subsample of trial completers (i.e. no estimation of missing values), as conducted in most studies in this area, has the problems of bias inherent in the last observation carry forward procedure, plus additional problems.
Sensitivity analyses repeated the above analyses, with the additional inclusion of the three preventive therapy conditions (β-blocker, BMM, BMM + β-blocker) and all two- and three-way interactions in the analyses described above. These analyses allowed us to examine whether results of the primary analysis were robust when any differences among the three preventive treatments was controlled for. A lack of statistical power did not allow us to reliably test for differences in effectiveness among the three preventative treatments as a function of MoAD. Visual evaluation of the slopes for participants with and without a MoAD separately for each of the three preventive treatments revealed that the slope (improvement) tended to be larger for participants with a MoAD than for participants without a MoAD for each of the three preventive treatments. Thus, the direction of the effect, if not the magnitude of the effect, did not seem to vary across the three preventive treatments.
Overall, the conclusions of the primary analysis and results of the sensitivity analysis did not differ. The only difference of note was that the p value for the MoAD × time interaction for migraine days per 30 days increased by .01 (p < .05 to p = .06) in the sensitivity analysis. This probably reflects the reduced power available for the sensitivity analyses.
Results
Pretreatment differences in outcome variables by psychiatric comorbidity
Consistent with previous findings, a pretreatment MoAD diagnosis was associated with higher HDI scores, t(229) = 5.26, p < .001 and Migraine Specific Quality of Life scores, t(230) = 4.06, p < .001 (Table 1). Participants with and without a pretreatment MoAD diagnosis did not differ in number of migraine days per 30 days during the OAT run-in, p > .50.
Mixed models analyses
Headache Disability Inventory
For the HDI, a significant MoAD × month interaction, F (1, 858) = 9.3, p < .01, qualified the significant main effects of month, F(1, 858) = 199.8, p < .001 and MoAD, F(1, 858) = 22.91, p < .001. Participants with a pretreatment MoAD diagnosis showed greater decreases in HDI scores than participants without a MoAD diagnosis. It can be seen in Figure 1A (and Table 1) that participants with a MoAD diagnosis began treatment with higher HDI scores than participants without a MoAD diagnosis but showed a larger reduction in HDI scores with treatment. Thus, although participants with and without a MoAD diagnosis differed on the HDI before treatment, they were indistinguishable at month 10, t(80.5) = 1.4, p > .10, and month 16, t(57.1) = 0.8, p > .20.
Estimated changes in the Headache Disability Inventory scores (A), Migraine Specific Quality of Life Questionnaire scores (B) and migraine days per 30 days (C) with preventative migraine treatments as a function of a Primary Care Evaluation of Mental Disorders (PRIME-MD) diagnosis of a mood and/or anxiety disorder at pretreatment. For both disability measures, higher scores indicate greater headache-related disability.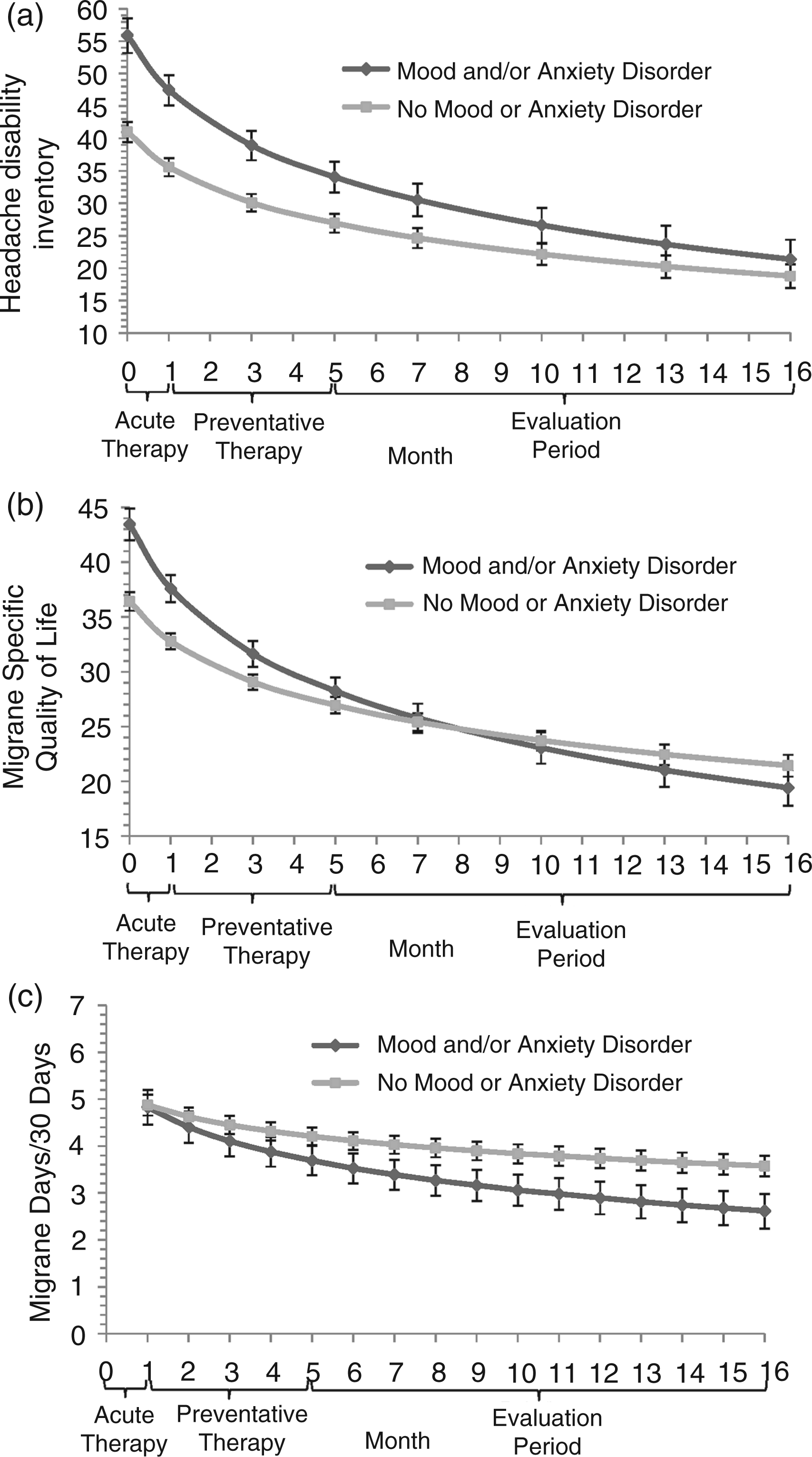
Migraine Specific Quality of Life Questionnaire
For the MSQL, a significant MoAD × month interaction, F(1, 857) = 16.1, p < .001, qualified significant main effects of month, F(1, 857) = 298.6, p < .001, and MoAD, F(1, 857) = 17.8, p < .001. The same findings were observed for each of the three subscales of the MSQL, all p < .01 or smaller. Again, participants with a pretreatment MoAD diagnosis showed larger decreases in MSQL scores (indicating decreases in headache-related disability) than participants without a MoAD diagnosis. It can be seen in Figure 1B (and Table 1) that participants with a MoAD diagnosis began treatment with higher MSQL scores than participants without a MoAD diagnosis but showed a larger reduction in MSQL scores with treatment. Thus, although participants with and without a MoAD diagnosis differed on the MSQL before treatment, they were indistinguishable at month 10, t(80.9) = −0.4, p > .20, and month 16, t(81.0) = −1.1, p > .20.
Migraine days per 30 days
For migraine days per 30 days, a significant MoAD × month interaction, F(1, 1651) = 4.17, p < .05, qualified a significant main effect of month, F(1, 1651) = 61.0, p < .001. Again, participants with a pretreatment MoAD diagnosis experienced greater decreases in migraine days per 30 days than participants without a MoAD diagnosis. It can be seen in Figure 1C (and Table 1) that participants with a MoAD diagnosis did not differ from participants without a MoAD diagnosis in migraine days per 30 days at the beginning of treatment but showed a larger reduction in migraine days per 30 days with treatment. Thus, participants with a MoAD diagnosis reported marginally fewer migraine days per 30 days than participants without a MoAD diagnosis at month 10, t(79.8) = −1.99, p < .06, and statistically significantly fewer migraine days per 30 days at month 16, t(79.5) = −2.3, p < .05.
Discussion
Significantly larger reductions in both migraine activity and in migraine-related disability were observed in participants with a MoAD diagnosis than in participants who did not receive either diagnosis. Thus, participants with a MoAD diagnosis recorded larger reductions in migraine days and fewer migraine days during the evaluation period than did participants with no MoAD diagnosis. Participants with a MoAD diagnosis also showed larger reductions in headache-related disability than participants with no MoAD diagnosis. Participants with a MoAD diagnosis participants showed higher levels headache-related disability before treatment than participants with no MoAD diagnosis, a finding also reported in previous studies (11,56–60). However, this difference was effectively eliminated with preventive treatment, so that participants with and without a MoAD diagnosis showed similar low levels of headache-related disability in the evaluation period.
Previous studies
In addition, two observational studies evaluated treatment (mostly preventive medication) for primary headache disorders in subspecialty headache clinics (19,61). In the first study, participants in each of three subgroups of people with a MoAD diagnosis – mood disorder only (n = 46), anxiety disorder only (n = 29), and both MoAD (n = 72) – showed observably larger improvements in relevant outcome variables (headache days per month and headache-related disability) in 8 of 9 comparisons with participants with no MoAD (n = 75) (19). The separation of MoAD diagnoses into separate subgroups coupled with the high comorbidity between MoADs limited statistical power for these analyses. The second observational study similarly found that patients with a DSM diagnosis of major depression recorded observably larger reductions in headache frequency than did non-depressed headache sufferers at a three-month evaluation of treatment response (mean group improvements of 5.0 vs. 3. 3 headaches per month; Table 2 in that study) (61).
A fourth relevant study did not obtain DSM diagnoses, but did use validated structured interviews (Hamilton Rating Scales for Depression and Anxiety (23,24)) to assess depression and anxiety (22). This open-label trial of nefazodone for chronic daily headaches reported significantly larger reductions in headache activity (headache index) in depressed (Hamilton Depression ≥13) than in non-depressed participants. Larger reductions in headache activity were also observed in anxious (Hamilton Anxiety ≥18) than in non-anxious participants. Of course, given the high comorbidity typically observed between anxiety and depression, many anxious participants may also have been depressed (35), so these two findings may not be independent.
Two recent studies have examined the impact of self-reported symptoms of depression and/or anxiety on outcomes of preventative headache medication (21,22); in the stronger of the two studies (22), people with chronic daily headache who scored a 14 (at least mild depression) or higher (mean = 25) on the Beck Depression Inventory 2nd edition (72) showed significantly larger improvements in headache activity over the course of nefazodone treatment than participants who scored less than a 14 (mean = 6) on this measure of depressive symptoms. Participants with scores in the depressed range also appeared to record lower headache index scores at 12-week evaluation than did participants with scores in the non-depressed range.
Summary and future directions
The present study and previous studies that have examined the impact of an interview-based DSM MoAD diagnosis on response to preventative medication and/or behavioral treatment of primary headache disorders in adults have found that headache sufferers with a MoAD show as large, or larger, response to headache treatments than headache sufferers with no MoAD diagnosis. Moreover, these findings do not appear to necessarily reflect higher baseline headache activity, and thus greater room to change when a MoAD is present, as can be seen in the present results (Figure 1C) and in other results (22).
Studies that have assessed self-reported symptoms of anxiety or depression have yielded conflicting results. These studies are also more difficult to interpret because the low to moderate elevations on self-report measures that have sometimes predicted response to preventive therapy do not necessarily indicate presence of a MoAD. Thus, these studies do not yield unambiguously different results from studies that used an interview-based DSM diagnosis.
Research is needed to identify the variables that determine when the presence of a comorbid MoAD is associated with larger, versus smaller, reductions in headache activity and headache-related disability with preventative headache treatments. We can speculate that any number of variables could influence the effect of a MoADs on response to preventative headache treatment, including the type of headache treatment, the type of headache disorder, the type and the severity of psychiatric comorbidity, 3 or the length of follow-up (e.g. initial treatment response versus long-term maintenance). A practical first step in clarifying this issue would be to generate a methodologically strong body of findings in which the presence of a comorbid MoAD is clearly associated with smaller improvements in headache activity and headache-related disability with preventive medication or behavioral treatment in adults with primary (migraine or tension-type) headache disorders. To the best of our knowledge, such studies are not currently available. A comparison of the two sets of studies with opposite findings would be likely to yield productive directions for future research.
To the best of our knowledge, the present study evaluated the largest sample with a homogenous (migraine) headache diagnosis, used the longest (12-month) evaluation period, and is the only study to use electronic daily diary recordings or to continuously monitor headache activity throughout the 12-month evaluation period, of all studies examining the impact of psychiatric comorbidity on preventative headache treatment outcome. The present study also has a number of other strengths not shared by most studies in this area, including the diagnosis of MoADs by DSM criteria using a validated structured clinical interview, the diagnosis of ICHD-2 criteria to diagnose headache disorders, and the use multiple outcome measures.
The present study also has a number of limitations, generally shared by other studies in this area. Although there was sufficient power for our primary analyses, we lacked the statistical power to reliably test for differences in the effectiveness among the three preventative treatments as a function of the presence or absence of a comorbid MoAD. Unfortunately, no other migraine trial has been adequately powered to reliably test for such differences either. Sensitivity analyses and examination of the three individual treatment slopes provided no indication that the direction of the above MoAD effects differed among the three preventative treatments. Of course, the magnitude of the MoAD effects on preventive treatments outcomes may have differed across the three preventive treatments. An answer to the latter question will require a significantly larger trial. The moderate sample size (n = 177) and the high rates of comorbidity among MoADs also required that MoADs be collapsed into a single MoAD diagnosis category as has been done successfully in other studies (20). A much larger sample would be required to reliably compare effects of specific MoADs on preventive therapy outcomes. In addition, the current study focused on frequent severe episodic migraine, so the findings may not generalize to other primary headache disorders. However, similar results have been observed in chronic tension-type headache, indicating these results are not specific to migraine (20) and thus may generalize more widely. Although the current results with frequent migraines and so may not apply to less frequent migraine, preventive therapies are less likely to be warranted in the treatment of infrequent migraines.
In summary, we observed larger reductions in migraine activity and migraine-related disability with preventative treatment in participants with a MoAD diagnosis than in participants who did not receive either diagnosis. Specification of the conditions that determine when a larger or smaller response to preventative headache treatments is likely to be observed in the presence, rather than absence, of a comorbid psychiatric disorder would be of both clinical and theoretical value.
Footnotes
Acknowledgements
This trial was supported by the National Institutes of Health [grant NS-32374 (to KAH)]. Merck Pharmaceuticals and GlaxoSmithKline Pharmaceuticals donated triptans (5-HT1B/D agonists) for acute migraine therapy, which was their only involvement.
Declaration of conflict of interests
EKS reports no conflict of interest. KAH has received support from the National Institutes of Health (NINDS; NS32375) and received an investigator-initiated grant from ENDO Pharmaceuticals.
