Abstract
Tonabersat is a novel putative migraine prophylactic agent with an unique stereospecific binding site in the brain. Tonabersat has been shown, in animal models, to inhibit experimentally induced cortical spreading depression, the likely underlying mechanism for migraine aura, and cerebrovascular responses to trigeminal nerve stimulation. The aim was to study the potential for tonabersat as a migraine preventive. A randomized, double-blind, placebo-controlled, multicentre, parallel group study recruited patients with migraine with and without aura experiencing between two and six migraine attacks per month. After a 1-month baseline they received tonabersat 20 mg daily for 2 weeks and 40 mg daily for a further 10 weeks. The primary end-point was the change in mean number of migraine headache days between the third month and the baseline period in the intention-to-treat population comparing the placebo (n = 65) and tonabersat (n = 58) groups. At the primary end-point there was a 1.0-day (95% confidence interval −0.33, 2.39; P = 0.14) difference in reduction in migraine days between tonabersat and placebo. There were 10 secondary efficacy end-points, of which two were statistically significant. In month 3 of treatment, the responder rate, defined as a 50% reduction in migraine attacks, was 62% for tonabersat and 45% for placebo (P < 0.05), and the rescue medication use was reduced in the tonabersat group compared with placebo by 1.8 days (P = 0.02). Placebo responses were particularly high for all end-points. At least one treatment-emergent adverse event was reported in the tonabersat group in 61% of patients compared with 51% in the placebo group; none was worrisome. Placebo responses were unexpectedly high in this trial, complicating straightforward interpretation of the study results. The good tolerability and promising efficacy results support further exploration of higher doses of tonabersat in larger controlled trials.
Keywords
Introduction
Migraine is a highly disabling (1) neurological condition (2) that has been estimated to be the most costly neurological disorder in the European Community at more than €27 billion (£22 billion) per year (3). It is reasonably clear that the most disabled group of patients have frequent attacks, which drives the economic costs of the disorder (4). Whereas the last decade has seen increased use of triptans, serotonin 5-HT1B/1D receptor agonists (5), in the acute treatment of migraine, developments in preventive strategies have been less forthcoming, but no less a need for many migraineurs.
An important recent epidemiology study sets the background for developments in preventive treatments. The American Migraine Prevalence and Prevention (AMPP) Study confirmed estimates of 1-year migraine prevalence for women at 17.1% and for men at 5.6% (6), comparable to previous estimates from the USA (7) and other countries (8, 9). About one-third of patients in the AMPP Study had three or more attacks a month and more than one-half had severe levels of disability during attacks. The authors determined that whereas 13% of patients were receiving preventive treatment, up to 38% of migraine sufferers could potentially benefit from it (6). Thus, the development of new preventives is a highly desirable goal.
Tonabersat ((-)-cis-6-acetyl-4S-(3-chloro-4-fluoro-benzoylamino)-3,4-dihydro-2,2-dimethyl-2H-benzo [b]pyran-3S-ol) is a novel benzoylamino benzopyran with anticonvulsant properties in experimental animal models that has been studied in laboratory models of the trigeminovascular system (10). Tonabersat inhibits cortical spreading depression (CSD), the animal homologue of human aura (11), in the cat (12). The effect on CSD involves inhibition of nitric oxide synthase production (13), a substance implicated in migraine (14). Sumatriptan, in contrast, alters neither migraine aura (15) nor cyclic guanosine monophosphate level changes in CSD, the latter being blocked by tonabersat (16). Tonabersat has no myocardial or coronary effects on isolated human tissue (17), but can inhibit trigeminal-induced craniovascular responses (18) as triptans do (19).
The present study was designed to test the hypothesis that tonabersat, in doses up to 40 mg/day, would have a greater preventive action in migraine than placebo. The primary efficacy end-point was reduction of migraine frequency between month 3 of treatment and baseline comparing the placebo and tonabersat groups. The work has been reported in preliminary form at the 13th Congress of the International Headache Society (Stockholm, Sweden July 2007 (20)).
Methods
The study was a multicentre, double-blind, randomized, parallel-group, controlled trial conducted in Denmark, Hungary and South Africa. In total 160 patients were enrolled and 124 were randomized, 59 to tonabersat and 65 to matching placebo; this was the safety population. The intention-to-treat population, who had taken treatment and provided usable efficacy data (21), was 123 patients, 58 in the tonabersat arm and 65 in the placebo arm (Fig. 1).

CONSORT diagram of patient disposition.
Design
Patients who screened eligible entered a 4-week baseline assessment period prior to randomization. Thereafter, patients who met inclusion criteria entered a 12-week treatment phase. During the first 2 weeks they received tonabersat 20 mg (one tablet) or matching placebo. After 2 weeks the dose was increased to 40 mg daily (two tablets) or two matching placebos. Depending on tolerability during the treatment phase, they could reduce back to one tablet on one occasion during the treatment phase. After completion of the 12-week treatment phase there was a 1-week follow-up period (Fig. 2).
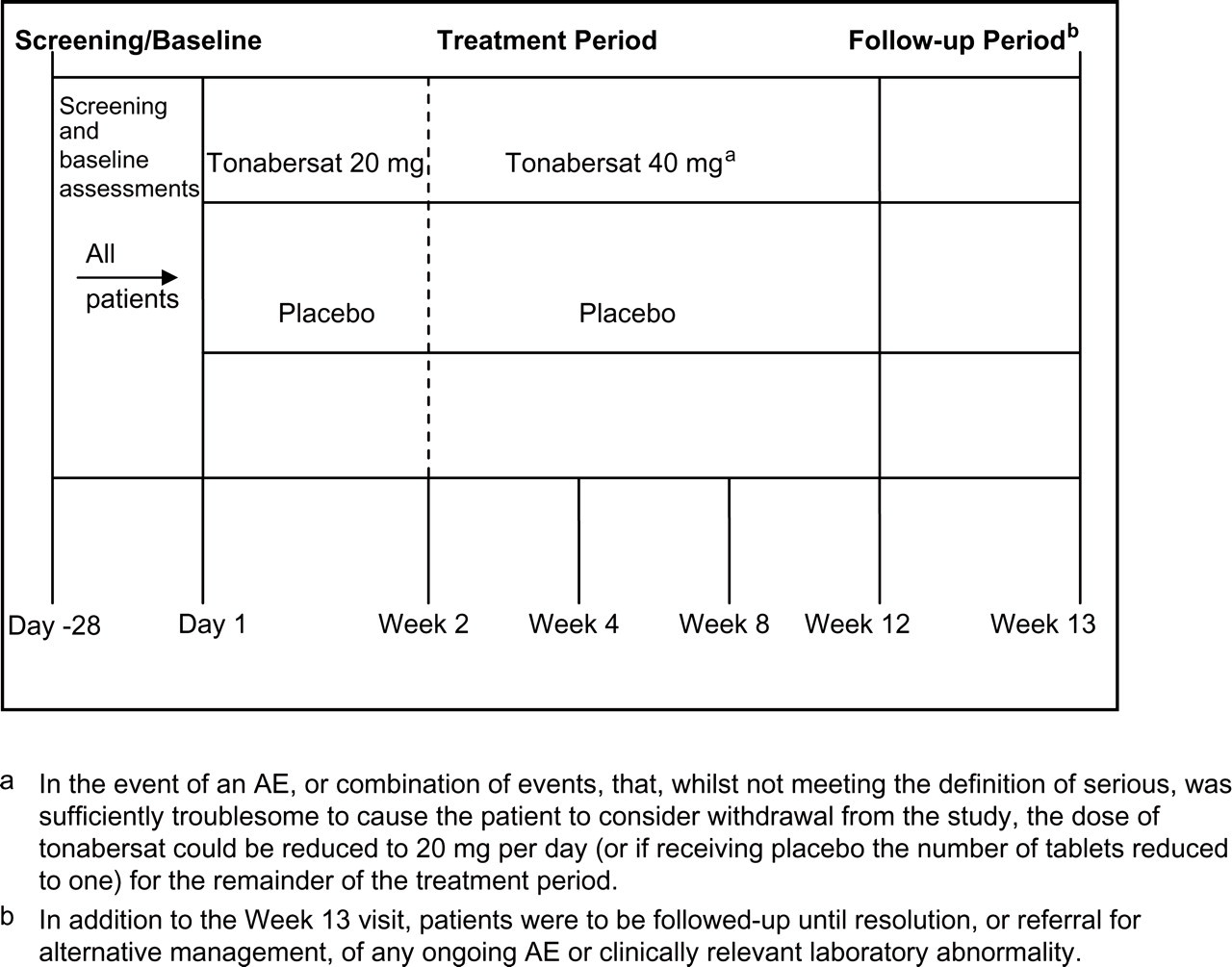
Schematic of study design.
Inclusion criteria
We wished to include male or female patients, aged 18–55 years, in good general health. They should have migraine, with or without aura, by the definition of the second edition of the International Classification of Headache Disorders (22), for ≥ 1 year. They should be experiencing between four and 14 migraine headache days per month as two to six migraine attacks per month. All women of childbearing potential who were not surgically sterilized had to have a documented negative pregnancy test done at the screening visit and be practising a medically acceptable contraceptive method for ≥ 3 months prior to enrolment with contraception maintained for ≥ 7 days after the last dose of trial medication.
Exclusion criteria
Experiencing headache on ≥ 15 days per month.
Patients with pure menstrual migraine defined as patients in whom migraine attacks occur exclusively on day 1 ± 2 (i.e. days −2 to +3) of menstruation in at least two out of three menstrual cycles and at no other times of the cycle.
Patients with other significant central nervous system disorders in the opinion of the investigator.
Failure to respond to more than two adequately dosed (i.e. recommended total daily dose and of sufficient duration) migraine prophylactic medications.
Overuse of acute migraine treatments defined as ≥ 15 medication days per month with analgesics and opioids or 10 medication days per month of ergots or triptans.
Prophylactic treatment within 2 months prior to entry to the trial.
Patients taking any of the following medications: β-blockers, tricyclic antidepressants, antiepileptic drugs, calcium channel blockers, or monoamine oxidase inhibitors (all during the last 2 months), or daily use of oral non-steroidal anti-inflammatory drugs, paracetamol, high-dose magnesium supplements (600 mg/day), multivitamin preparations containing > 10 mg riboflavin, oral corticosteroids or herbal preparations (e.g. feverfew, butterwort and St John's Wort). Parenteral administration of botulinum toxin was also to be excluded. Patients taking other medications used as prophylaxis for migraine, including methysergide, anti-spasticity agents (e.g. tizanadine) and the new generation anti-psychotics (e.g. olanzapine) currently or within the previous 2 months, were also to be excluded.
Patients who, in the opinion of the investigator, had significant cerebrovascular disease, for example, transient ischaemic attacks, stroke.
Patients who, in the opinion of the investigator, had clinically significant cardiovascular disease.
Patients suffering from a current clinical diagnosis of major depressive disorder or schizophrenia.
Patients with renal dysfunction, defined as a serum creatinine > 125% of the upper limit of normal for their age group.
Patients with hepatic dysfunction defined as a liver function test (aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, bilirubin) of greater than twice the upper limit of normal for their age group.
Patients with known alcohol or other substance abuse.
Failure to complete the diary card during the baseline period.
Participation in another clinical trial in the previous 4 weeks.
Any woman who was pregnant, lactating or not using medically acceptable contraception.
End-points
The primary end-point was the change in mean monthly number of migraine headache days from the baseline period to month 3.
Secondary end-points
Change in the mean monthly number of migraine headache days from the baseline period to across the whole treatment period.
Change in the mean monthly number of migraine attacks from the baseline period to month 3.
Change in the mean monthly number of migraine attacks from the baseline period to across the whole treatment period.
Proportion of patients defined as a responder, that is, those with a reduction of ≥ 50% in the mean monthly number of migraine headache days in the third month of treatment and over the whole treatment period.
Proportion of patients defined as a responder, that is, those with a reduction of ≥ 50% in the mean monthly number of migraine attacks in the third month of treatment and over the whole treatment period.
Change in the mean monthly consumption of rescue medication from the baseline period to month 3.
Change in the mean monthly consumption of rescue medication from the baseline period to across the whole treatment period.
Speed of effect of treatment.
Overall severity of migraine attacks occurring during the treatment period.
Overall response to the question ‘How satisfied are you with the trial medication?’.
Safety
Safety was evaluated by adverse events (AEs), serious AEs and AEs leading to withdrawal of trial medication, clinical laboratory tests, vital signs, 12-lead electrocardiogram, physical examination and concomitant medications. Safety summaries were presented on this population.
Statistical considerations
The calculation of sample size and definition of a clinically relevant difference was based on the data reported from recent studies with topiramate (23). A sample size of 64 patients per treatment group was required to detect a difference in mean reduction in monthly migraine headache days of 1.25 with 80% power. This would assume a two-sided test was conducted with a significant level of 5% and assuming a common standard deviation of 2.5 for tonabersat and placebo treatment groups. We estimated that 170 patients would need to be enrolled for 128 to be evaluable based on 25% of patients not being eligible for randomization. The efficacy analysis was carried out on the intention-to-treat population as defined above. The changes in mean monthly migraine headache days from baseline to month 3 and from baseline to across the whole treatment period were compared using an analysis of covariance (ANCOVA) model, with explanatory variables of treatment group and the number of migraine headache days during the baseline period.
The difference between active treatment and placebo in the proportion of patients defined as a responder in terms of migraine headache days was analysed using the Cochran–Mantel–Haenszel test with stratification by the number of migraine headache days experienced during the baseline period (≤ 9 migraine headache days or > 9 migraine headache days). For the corresponding analysis of responders in terms of number of migraine attacks, the χ2-test was used, unless the expected cell counts were low, in which case Fisher's exact test was used. The change in the mean monthly number of migraine attacks from baseline to month 3, the change in the mean monthly number of migraine attacks from baseline to the treatment period, the change in the mean monthly consumption of rescue medication from baseline to month 3 and the change in the mean monthly consumption of rescue medication from baseline to the treatment period were analysed using an ANCOVA model.
Speed of effect was to be determined using a step-down procedure on the primary variable. If there was a significant difference between the treatment groups at month 3, the analysis was to be repeated for month 2, and if there was a significant difference in this analysis then the analysis was to be repeated again for month 1. From these analyses, it would be determinable how quickly the reduction in migraine headache day frequency occurred. Overall severity and overall response to the question ‘How satisfied are you with the trial medication?’ were compared across treatment groups using the χ2-test. Significance was assessed at the P < 0.05 level. Summary data are presented as mean ± S.E.M. unless otherwise indicated.
Results
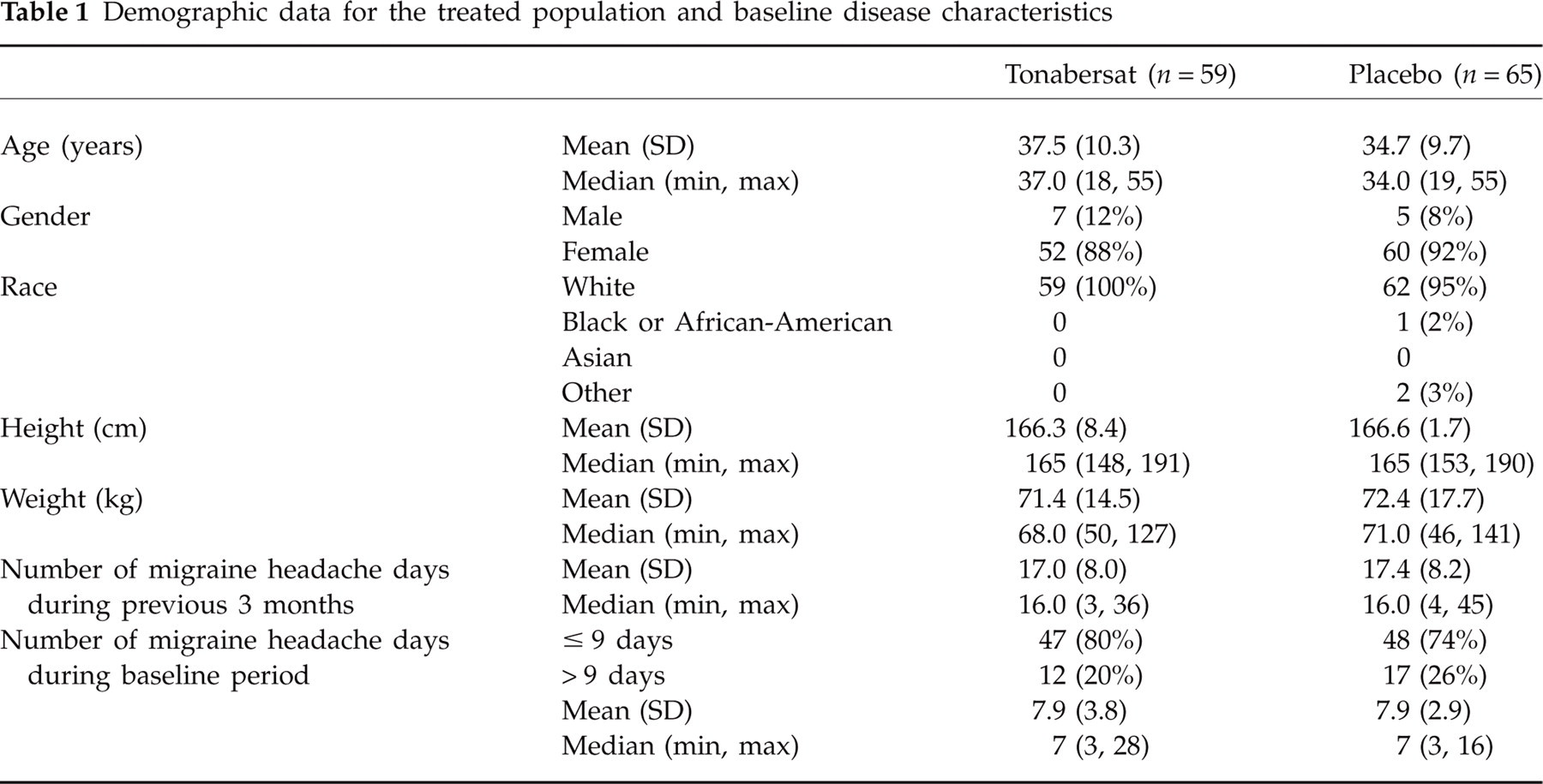
In total, 160 patients were enrolled for a 4-week baseline observation period, after which 124 were randomized to either tonabersat (n = 59) or placebo (n = 65) (Fig. 1). The intention-to-treat analysis, where patients treated and had at least one data point, gave 58 patients in the tonabersat group and 65 in the placebo group. The study population demographics were typical of migraine clinical trials and did not differ significantly between the two treatment groups (Table 1).
Demographic data for the treated population and baseline disease characteristics
Primary end-point
The primary efficacy variable was the change in the mean monthly number of migraine headache days from the baseline period to month 3. The baseline number of days was 7.9 ± 0.5 in the tonabersat group and 7.9 ± 0.4 in the placebo group. At month 3 reductions in mean monthly headache days observed in the tonabersat group were −4.4 ± 0.5 days compared with −3.7 ± 0.6 days in the placebo group. The difference derived from the analysis of covariance was 1.0 [95% confidence interval (CI) −0.33, 2.39]. This difference was not statistically significant (P = 0.14).
Secondary end-points
Change in attack/day rates
The reduction in mean monthly migraine headache days from baseline across the whole treatment period was −3.6 ± 0.5 for tonabersat and −3.1 ± 0.5 for placebo (difference 0.8, 95% CI −0.32, 1.91; P = 0.16). The reduction in mean monthly migraine attacks comparing baseline to month 3 of treatment was −2.4 ± 0.4 for the tonabersat group and −1.4 ± 0.5 for the placebo group (difference 1.0, 95% CI −0.29, 2.20; P = 0.13), whereas comparable rates for the entire treatment period were −1.9 and −1.4, respectively.
Responder rates
The overall 50% responder rate comparing the number of headache days during baseline with month 3 of treatment for tonabersat was 59% compared with placebo at 49% (P = 0.22). Comparing the baseline with the entire treatment period the tonabersat rate was 47% and the placebo rate 39% (P = 0.29). The overall 50% responder rate for reduction in the number of migraine attacks during month 3 of treatment was 62 and 45% for tonabersat and placebo treatment groups, respectively (P < 0.05). Comparable figures for the entire treatment period were 41 and 37% (P = 0.69).
Other analyses
The consumption of rescue medication was reduced significantly from baseline in the tonabersat group by −3.7 ± 0.5 compared with −2.0 ± 0.6 in the placebo group (difference 1.8, 95% CI 0.26, 3.41; P = 0.02). For the overall treatment period the reduction in the tonabersat group was −2.9 ± 0.5 compared with −2.1 ± 0.4 in the placebo group (difference 1.0, 95% CI −0.08, 2.05; P = 0.07). Attacks were reported to be of moderate severity during the treatment period in 45% of patients on tonabersat and 42% of patients on placebo (P = 0.87). In answer to the question, ‘How satisfied are you with the trial medication?’, 76% of patients in the tonabersat group and 63% in the placebo group expressed satisfaction (P = 0.14).
Safety and tolerability—adverse events
Tonabersat was well tolerated in patients with migraine headache during the 12-week treatment period.
Adverse events
Adverse events in this trial were generally mild to moderate in severity, and were rarely treatment-limiting. At least one treatment-emergent AE (TEAE) was reported for 61% of patients in the tonabersat treatment group compared with 51% receiving placebo. Nausea (11% of patients), upper respiratory tract infection (7% of patients) and dizziness and urinary tract infection (each reported in 7% of patients) were most commonly reported. A higher proportion of patients in the tonabersat group compared with the placebo group reported nausea (17% vs. 6% of patients), dizziness (9% vs. 5% of patients) and headache (7% vs. 2% of patients). Vertigo was reported only by patients in the tonabersat group (9% of patients). TEAEs considered to be related to treatment were more common in the tonabersat group compared with the placebo group: 39 and 15% of patients, respectively. Only one patient in each treatment group was reported with at least one serious TEAE. There was no indication of any difference in AE profile between the 20-mg and 40-mg doses of tonabersat.
Withdrawal from the study
Adverse events leading to withdrawal of treatment were seen in two patients receiving tonabersat, one with severe drowsiness and one with nausea and dizziness, and one patient receiving placebo with dizziness, hypoaesthesia and memory impairment.
Dose reductions
Adverse events led to a dose reduction in two patients in the tonabersat treatment group, with both patients experienced nausea and one patient also reporting headache.
Safety testing
There was no clinically relevant difference between tonabersat and placebo groups in haematology, biochemistry or urinalysis parameters across the treatment period, and no clinically relevant changes in vital signs were observed.
Discussion
In this study, tonabersat failed to demonstrate a significantly greater reduction of migraine headache days than placebo on the primary end-point. However, tonabersat was numerically better than placebo for all secondary end-points and was well tolerated. Although the study failed in its primary goal, it may be argued that the data justify further studies of the utility of tonabersat in the preventive management of migraine. Issues such as dosing, time of exposure and, in this study particularly, the high placebo rate need some exploration.
Rationale for preventive efficacy of tonabersat
The site of action of tonabersat is unknown. However, an effect on CSD seems one way to screen potential antimigraine preventives. It is reasonably well accepted that CSD is the experimental expression of migraine aura (24) and that migraine preventives, despite coming from different pharmacological classes, all share the ability to attenuate CSD in animal models (25, 26). It must be said, in reverse, that no compounds that failed to demonstrate antimigraine efficacy have been tested for their CSD-blocking capacity. Tonabersat has been shown to block CSD (12). Given further the effect on trigeminal-stimulated craniovascular responses (18), the general basis for the current study seems reasonable.
Was the dose correct?
One principle problem with migraine prevention is to understand how to dose new chemical entities in this indication. It is said there are no models of prevention, although indeed it is not clear whether the models used to study trigeminovascular nociceptive mechanisms can be used to study preventive strategies. As an example, it can be shown that both the preventive treatment propranolol (27) and the acute treatment naratriptan (28) can alter trigeminovascular nociceptive processing in the ventroposteromedial thalamus. Moreover, topiramate can attenuate trigeminovascular nociceptive traffic when administered acutely (29) and is clearly an established preventive treatment (23). These models are, however, unable to provide precise dosage predictions in humans. The fact that tonabersat was very well tolerated in this study may imply it was under-dose. Certainly, given its tolerability profile, extending the dose range in a subsequent study would seem appropriate.
High placebo rate
The placebo response rate in, for example, the 50% responder end-point, at 49% was more than double the 23% seen in comparable parallel-group preventive studies conducted with topiramate, a well-established migraine prophylactic agent with antiepileptic and CSD-blocking properties (30, 31). Given that the absolute response rate is likely to have a ceiling, the substantial placebo rate may have significantly compromised the outcome of the present study. Reducing placebo response rates is related to expectations (32), and this issue needs addressing in future studies. Recent studies have reported very modest placebo response rates in chronic migraine (33) so that the goal seems possible. Perhaps enrolling somewhat more experienced patients with more challenging problems might mitigate expectations and thus placebo rates.
In conclusion, although failing on the primary end-point, the promising results for the secondary end-points, the possible methodological explanation for this, and the good tolerability support further exploration of tonabersat in the prevention of migraine attacks.
Footnotes
Acknowledgements
The study protocol was developed jointly by the sponsor and the authors. P. J. G. wrote the initial draft of the paper. The study was sponsored by Minster Pharmaceuticals.
Contributing centres
