Abstract
The objective of this study was to evaluate whether the quality of sleep and the degree of fatigue and daytime sleepiness are related to migraine. We investigated 489 subjects comprising 97 patients with eight or more, 77 patients with five to seven and 196 patients with one to four migraine days per month, and 119 migraine-free controls with fewer than six headache days per year. The patients were recruited via articles in newspapers not stressing the subject of the study. All participants underwent a semistructured interview and completed the Pittsburgh Sleep Quality Index (PSQI), the Fatigue Severity Scale (FSS), the Epworth Sleepiness Scale (ESS) and the Self-rating Depression Scale and the Self-rating Anxiety Scale. For statistical analysis we used two way MANOVAs, post hoc univariate two-way ANOVAs and Hochberg's GT2 tests as well as three-way mixed design ANOVAs. The PSQI total score was highest in patients with frequent migraine (5.9 ± 4.3) and lowest in controls (4.3 ± 2.5, P = 0.04). Four subscores of the PSQI showed similar statistically significant differences. The FSS and ESS scores did not differ in the four study groups. Analysing depression and anxiety revealed a significant impact on PSQI, FSS and ESS, but did not demonstrate interactions with migraine, thus suggesting that the impact of migraine is similar in patients without and with psychiatric comorbidity. In conclusion, the quality of sleep is decreased in patients with migraine, whereas fatigue and daytime sleepiness do not differ from healthy controls. The decreased quality of sleep in migraineurs is also a consequence of migraine itself and cannot be explained exclusively by comorbidity with depression or anxiety.
Introduction
Migraine is one of the most common and debilitating neurological disorders, with a 1-year prevalence of approximately 18% in women and 6% in men (1, 2). Impairment of the patient's quality of life is caused not only by the attacks themselves, but also by comorbid disorders such as depression and anxiety. Additional problems such as sleep disorders, excessive fatigue and daytime sleepiness might also affect the quality of life in migraineurs. The aim of the present study was to further examine these later complaints.
Sleep disorders are common in the general population, affecting up to one-third of adults (3). Considering the high prevalence of sleep disturbances and the close relation between sleep and life quality, one can regard sleep quality as an important indicator of quality of life (3).
Fatigue is a frequent complaint in every day life and occurs in many medical conditions. Severe idiopathic fatigue is the hallmark of chronic fatigue syndrome (4). In clinical practice, terms such as sleepiness, tiredness, generalized weakness, loss of strength and loss of interest are often confused, and used to represent fatigue. Fatigue also interferes with activities of daily living and the patient's quality of life.
Excessive daytime sleepiness is reported by up to 31% of the adult population (5) and its consequences can be significant, including accidents, negative economic and public health outcomes, reduced work and school performance, and impaired psychosocial functioning. In the absence of sleep deprivation, daytime sleepiness is almost inevitably caused by an identifiable and treatable sleep disorder.
Migraine patients frequently cite sleep problems as a trigger factor of attacks (6). Analysis of 39 headache diaries revealed decreased sleep duration and sleep quality in the night before a migraine attack. Approximately two-thirds of migraine attacks began when the patients woke up (7). Furthermore, chronically shortened sleep patterns were associated with more frequent and more severe migraine (8). Rothrock et al. (9) identified a greater prevalence of insomnia among patients with chronic vs. episodic headache. Notably, previous or current history of depression did not differ among the groups. However, in a large prospective study, including 327 patients, sleep was not associated with an increased risk of occurrence of headache and migraine in migraineurs (10). Finally, Rhode et al. found that the frequency of restless legs syndrome was significantly higher in migraine patients than in control subjects (17.3% vs. 5.6%, P < 0.001) (11).
The importance of tiredness and fatigue in migraine patients has been stressed by several authors (6, 10, 12–15). Giffin et al. (15) reported that tiredness was the most common premonitory symptom in a prospective diary study. In another prospective study (10), tiredness was associated with an increased risk of occurrence of headache and migraine in migraineurs. Among patients with chronic migraine, fatigue (defined by a Fatigue Severity Scale score > 27) was found in > 80%, and two-thirds met the diagnostic criteria for chronic fatigue syndrome (16).
In an uncontrolled study (17), 37% of patients with migraine had excessive daytime sleepiness. The degree of daytime sleepiness was positively correlated with the frequency of migraine attacks as well as with fatigue. In a controlled study (18), excessive daytime sleepiness was more frequent in migraineurs than in controls, but the frequency was much lower (14% vs. 5%; odds ratio 3.1; 95% confidence interval 1.1–8.9).
Even though potential relations between migraine and sleep have been established in several studies (6, 10, 12–15), there are only few controlled studies on the quality of sleep in migraine patients (19–21). Controlled studies on fatigue are limited to the period before and during an attack (10, 15). Daytime sleepiness has been examined in only one open and one controlled study (16, 18). Considering this lack of information, the aim of the present study was to investigate whether the quality of sleep and the degree of fatigue and daytime sleepiness are related to migraine. In detail, we wanted to clarify whether the quality of sleep decreases and fatigue as well as daytime sleepiness increase with an increasing frequency of migraine attacks. In addition, we wanted to examine the impact of depression and anxiety on these findings.
Patients and methods
Patients
We screened 415 patients after response to an article in newspapers about trigger factors of migraine not focusing on the precise aim of this study. Details on patient recruitment have been published elsewhere (10). In addition, we screened 126 control subjects named by patients themselves (excluding family members and relatives) or contacted by one of the authors (T.H.). We included a total of 489 subjects comprising 97 patients with eight or more, 77 patients with five to seven and 196 patients with one to four migraine days per month, and 119 migraine-free controls with fewer than six headache days per year. All patients had migraine without aura, migraine with aura or probable migraine according to the 2nd edition of the International Classification of Headache Disorders (ICHD-II) (22). Patients with a history of medication overuse, medication overuse headache, any other primary or secondary headaches, and recent changes in headache characteristics were excluded, as were patients and controls with clinically relevant other diseases. However, we did not exclude patients with migraine and additional tension-type headache. All subjects gave written informed consent.
Methods
We recorded biographical data, medical history and headache history using a semistructured questionnaire to establish the headache diagnoses according to ICHD-II. In addition, we used the Headache Impact Test (HIT-6) (23), the Pittsburgh Sleep Quality Index (PSQI) (24), the Fatigue Severity Scale (FSS) (25), the Epworth Sleepiness Scale (ESS) (26, 27), the Self-rating Depression Scale (SDS) (28) and the Self-rating Anxiety Scale (SAS) (29).
The HIT-6 is a short questionnaire that includes six questions relevant to headache severity, frequency and disability. Ratings of ‘never,’ ‘rarely,’ ‘sometimes,’ ‘very often’ and ‘always’ are weighted with a certain number of points so that the total score lies between 36 and 78. A total score of ≥ 60 indicates that the headache is having a severe impact on the patient's life (30, 31).
The PSQI measures the quality and patterns of sleep in the preceding 4 weeks. It differentiates ‘poor’ from ‘good’ sleep by measuring seven areas: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication and daytime dysfunction over the last 4 weeks. Scoring of answers is based on a 0–3 Likert scale, whereby 3 reflects the negative extreme. A total score of ≥ 5 indicates a ‘poor’ sleeper (3, 24).
The FSS is composed of nine items with a 7-point response format (never = 1, rare = 2–3, occasionally = 4, often = 5–6, always = 7). The total score is expressed by the average of all subscores. Scores ≥ 4 were considered to indicate excessive fatigue.
The ESS is also a self-rating instrument to evaluate the tendency to doze off during daytime. It consists of eight items concerning everyday situations. Reponses to each item are ranked from 0 to 3 according to the probability for dozing off during a task (0 = never, 1 = low probability, 2 = moderate probability, 3 = high probability). A score ≥ 10 indicates excessive daytime sleepiness.
The SDS is a 20-item self-report measure of the symptoms of depression. Subjects rate each item according to how they felt during the preceding 7 days. Item responses are ranked from 1 to 4. The sum of the 20 items produces a score ranging between 20 and 80, and a cut-off value of ≥ 40 suggests clinically relevant depression (28).
The SAS measures affective and somatic symptoms of an anxiety disorder. The structure of the SAS is like that of the SDS. It also consists of 20 questions, which refer to the last 7 days. The result of the SAS is obtained by summing up the scores. A cut-off value of ≥ 40 suggests the presence of a clinically relevant anxiety disorder (29).
Data were analysed using the
Results
Biographic data and medical history
There were no statistically significant differences between the four study groups regarding age, sex or number of children. The percentage of persons with a partner was higher among patients than among controls (P = 0.03). The percentage of subjects with ≥ 13 years of education was lower in patients than in controls (P = 0.001). In all four groups, the majority of the subjects reported no other medical disorders (range 62.7–73.2%), and there was no statistically significant difference. Oral contraceptives were used more often by patients than by controls (P = 0.003).
Post hoc analysis did not show any statistically significant relation between marital status, education or oral contraceptives and the total scores of PSQI, FSS and ESS in the four study groups.
Headache
In the three patient groups, the prevalence of migraine without aura, migraine with aura and probable migraine ranged between 72.2 and 88.0%, 8.0 and 11.6% and 3.5 and 5.2%, respectively; among patients with eight or more migraine days per month 12.6% had chronic migraine.
Coexisting tension-type headache was present in 24.9% of patients, differing statistically significantly between the three study groups. Post hoc analysis did not show any statistically significant relation between the presence of tension-type headache and the total scores of PSQI, FSS and ESS in the three patient groups.
Headache-related quality of life was assessed with the HIT-6. The scores were 65.0 ± 3.9, 65.3 ± 4.7 and 63.4 ± 4.6 in patients with eight or more, five to seven and one to four migraine days per month, respectively, and 39.7 ± 5.0 in the control group. The difference between the four study groups was statistically highly significant (P < 0.001). Furthermore, the HIT-6 score was significantly lower in patients with one to four migraine days than in those with five to seven and eight or more migraine days per month (P = 0.021 and P = 0.043, respectively).
Quality of sleep
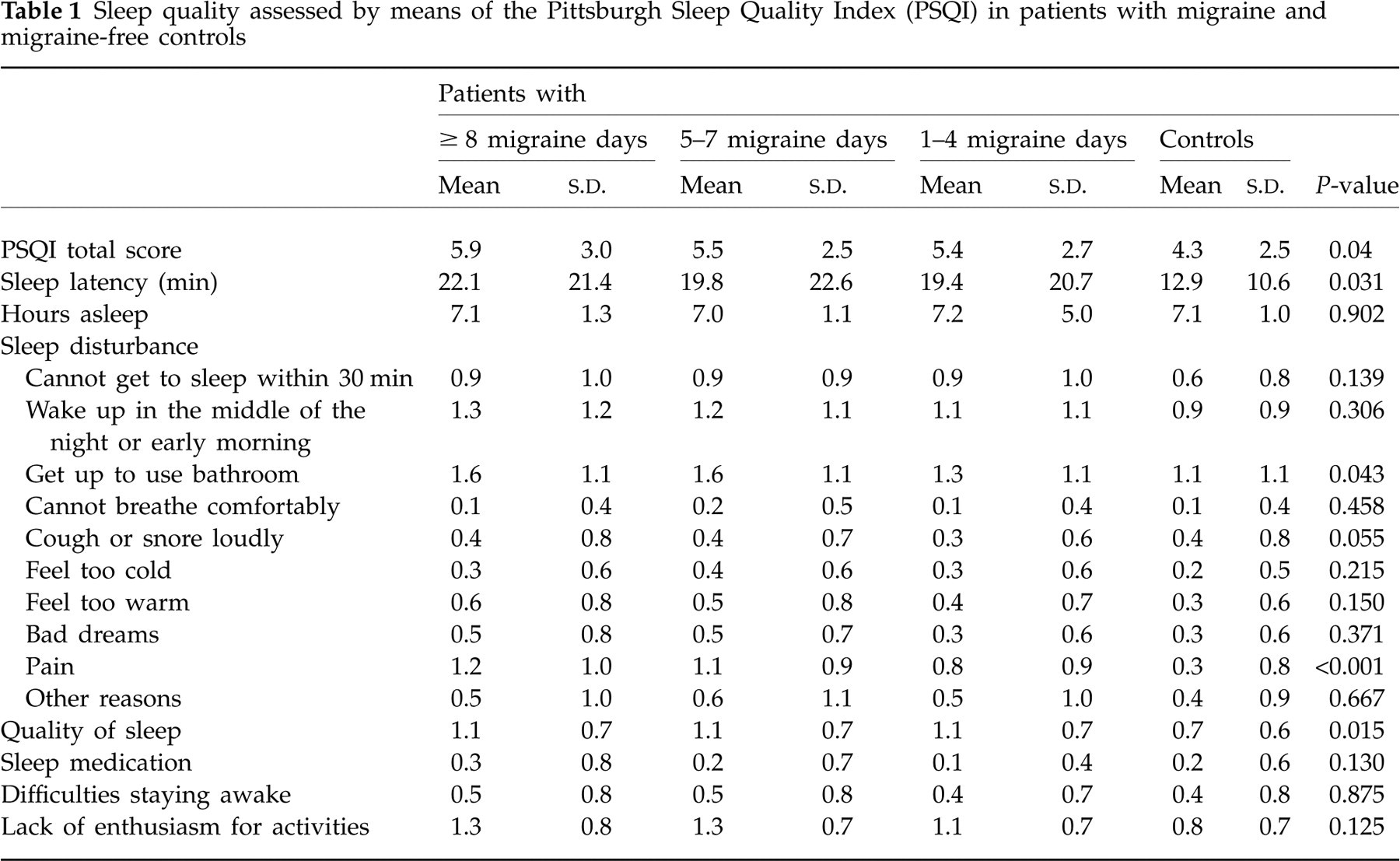
The prevalence of poor sleepers defined by a PSQI total score of ≥ 5 was 64.9, 64.0, 57.4 and 34.9% in patients with eight or more, five to seven and one to four migraine days per month and controls, respectively. Similarly, the PSQI total score as well as almost all items of the PSQI were highest in patients with eight or more migraine days and lowest in controls (Table 1). Post hoc Hochberg tests showed that controls differed from each of the three patient groups (one to four, five to seven, eight or more migraine days per month) regarding the PSQI total score (P = 0.003, P = 0.014 and P < 0.001, respectively), and the subscores sleep disturbance because of pain and sleep quality (for both variables and comparison of controls with each patient group, P < 0.001). In addition, controls differed from patients with one to four and eight or more migraine days per month regarding sleep latency (P = 0.037 and P = 0.005, respectively) and from patients with eight or more migraine days regarding sleep disturbance because of getting up to use the bathroom (P = 0.046). Comparing the three patient groups with each other showed no differences in the PSQI total score and revealed only one significantly different subscore, i.e. sleep disturbance because of pain, which was more common in patients with eight or more migraine days than in those with one to four migraine days per month.
Sleep quality assessed by means of the Pittsburgh Sleep Quality Index (PSQI) in patients with migraine and migraine-free controls
Fatigue and daytime sleepiness
Fatigue and daytime sleepiness were more prominent in patients than in controls, but the differences did not reach the level of statistical significance. Fatigue was present in 39.1, 37.3, 33.8 and 23.7% of patients with eight or more, five to seven and one to four migraine days per month and controls, respectively. Similarly, the FSS total score was highest in patients with eight or more migraine days per month and lowest in controls (32.7 ± 11.4, 31.9 ± 11.8, 31.2 ± 10.9, 28.2 ± 9.4). In patients with eight or more, five to seven and one to four migraine days and controls, excessive daytime sleepiness was diagnosed in 32.6, 34,8, 33.7 and 22.8%, respectively, and the ESS score was 7.9 ± 4.0, 7.8 ± 3.6, 8.0 ± 4.0 and 7.7 ± 4.3.
Depression and anxiety
The prevalence of depression and/or anxiety as well as the SDS and SAS scores decreased statistically significantly from patients with eight or more migraine days per month to controls (Table 2).
Fatigue assessed by means of the Fatigue Severity Scale in patients with migraine and migraine-free controls


SDS, Self-rating Depression Scale; SAS, Self-rating Anxiety Scale.
To analyse the impact of psychiatric comorbidity we compared patients without depression and anxiety with those with depression and/or anxiety. Comparisons were performed for the total scores of HIT-6, PSQI, FSS and ESS and those items of the PSQI that showed statistically significant differences in the entire study population. Statistically significant differences between subjects without and with psychiatric comorbidity were found for the total scores of the PSQI (4.7 ± 2.5 vs. 6.4 ± 2.7, P < 0.001), FSS (28.4 ± 9.6 vs. 36.5 ± 11.5, P < 0.001) and ESS (7.5 ± 4.0 vs. 8.6 ± 3.9, P = 0.014), for sleep disturbance because of pain (0.7 ± 0.9 vs. 1.1 ± 1.0, P = 0.009) and sleep quality (0.8 ± 0.6 vs. 1.3 ± 0.7, P < 0.001), whereas HIT-6, sleep latency and getting up to use the bathroom did not differ. Statistically significant interactions between psychiatric comorbidity and migraine were not seen in any of these comparisons, suggesting that the impact of migraine on these variables is similar in patients without and with psychiatric comorbidity.
Discussion
In this study, we have analysed the quality of sleep as well as the degree of fatigue and daytime sleepiness in patients with migraine and healthy controls differentiating three patient groups according to the days with migraine per month. In addition, we have examined the impact of depression and anxiety. Migraine was related to several aspects of sleep quality, but was not associated with fatigue and daytime sleepiness. In the entire study population, the PSQI total score was highest, sleep latency was longest and the quality of sleep was poorest in subjects with eight or more migraine days per month. In addition, these patients most often reported sleep disturbances because of getting up to use the bathroom and pain. In subjects without depression and anxiety, migraine still had an unfavourable effect on the PSQI total score, sleep latency, sleep quality, and sleep disturbances because of nocturnal use of the bathroom and pain. Depression and anxiety unfavourably affected PSQI, FSS and ESS total scores as well as sleep disturbances because of pain and sleep quality. In addition, psychiatric comorbidity had a statistically significant impact on almost all other subscores of the PSQI and on all subscores of the FSS (data not shown).
All four study groups were very similar regarding their biographic data and medical history. Groups differed significantly in only three of 12 items after Bonferroni correction. Migraine patients lived more often with a partner and had undergone significantly fewer years of education. In addition, significantly more female migraineurs used oral contraceptives compared with female controls. There is no evidence in the literature that the living with or without a partner or the level of education have an effect on sleep quality, fatigue or daytime sleepiness in patients with migraine. A relation with the use of oral contraceptives is unlikely. However, we could not definitely exclude bias in favour of the control group. Therefore, we performed a post hoc analysis revealing no relation between these variables and the total scores of PSQI, FSS and ESS.
Apart from that, the prevalence of co-existing tension-type headache differed in the three patient groups. The effect of migraine and tension-type headache on sleep has been previously compared in children only (32) without revealing any significant differences between the two headache disorders. Post hoc analysis of our data did not show any relation between coexisting tension-type headache and the total scores of PSQI, FSS and ESS.
The present study is the first to provide a detailed analysis of the PSQI and its subscores in patients with migraine. Only one previous study (21) has applied the PSQI in patients with migraine. Similar to our findings, the prevalence of poor sleepers was higher and the sleep latency was longer in migraineurs than in controls, but, in contrast to our results, these parameters were not related to the frequency of migraine attacks (21). Niederberger and co-workers (33) found a worsened sleep quality in the nights before migraine attacks. In another study, including chronic headache sufferers, without differentiating between migraine and tension-type headache (34), the number of estimated hours of sleep at night was lower in the headache than in the control group. It also took headache sufferers longer to fall asleep and to fall back asleep after waking up at night. However, considering the different headache diagnoses a comparison with our findings is not feasible (34).
Analysing fatigue in patients with chronic migraine, Peres et al. (16) reported elevated FSS scores in 84.1% of 63 patients in an uncontrolled study. We found increased scores in 33.8–39.1% of patients and in 23.7% of controls. The cut-off point, however, was different. We defined excessive fatigue by average subscore of ≥ 4, whereas Peres et al. used a total score of > 27. Applying the latter cut-off the percentages of subjects with excessive fatigue increased to 54.2–62.3% in patients and 48.5% in controls. In the chronic headache sufferers investigated by Spierings et al. (34), patients did not feel tired more often than controls, but the intensity of their fatigue, as measured on a 10-cm visual analogue scale, was significantly higher.
Using the Subjective Sleepiness Scale and the Dutch version of the Stanford Sleepiness Scale, Stronks and colleagues (35) found higher levels of sleepiness among migraineurs compared with a headache-free population. It seems noteworthy that nowadays the ESS is most widely used in the assessment of excessive daytime sleepiness. An uncontrolled study by Peres et al. (17) showed excessive daytime sleepiness indicated by an ESS score ≥ 10 in 37.0% of all people with migraine, in 32.4% of those with episodic migraine, and in 39.8% of those with chronic migraine. Barbanti and colleagues (18) found an ESS score of ≥ 10 in only 14% of all migraine patients in a case–control study. In our patients, the prevalence of ESS scores ≥ 10 was similar to that in the study of Peres et al. (17).
The well-known comorbidity of migraine, depression and anxiety is also reflected in the present study. Almost 40% of the patients had increased SDS and/or SAS scores. Comparing patients without and with psychiatric comorbidity showed a decrease in sleep quality and an increase in fatigue and daytime sleepiness, but no interaction with migraine frequency, thus indicating that the impact of migraine on sleep quality is similar in patients without and with psychiatric comorbidity. Two previous studies (16, 21) have shown a correlation between sleep quality and excessive daytime sleepiness on the one hand and depression and anxiety on the other. In the study on sleep quality (21), 50% of migraine patients without depression, but 85% of those with depression were poor sleepers. The prevalence rates of poor sleepers in the control group and in subjects without and with anxiety were not given, however. In the study on excessive daytime sleepiness in migraineurs the ESS scores were directly correlated with levels of depression and anxiety (18).
The strengths of the present study are the large number of subjects, the controlled study design, patient recruitment via newspapers avoiding the limitations of patient selection at a tertiary headache centre, the use of the ICHD-II criteria and of validated questionnaires, the consideration of comorbid depression and anxiety, the similarity of the study groups regarding biographical data and general medical history, and the statistical analysis. Limitations of the study are its cross-sectional design, exclusion of patients with infrequent migraine attacks and the fact that the study was not population-based.
In conclusion, the quality of sleep is decreased in patients with migraine, whereas fatigue and daytime sleepiness do not differ from healthy controls. The decreased quality of sleep is also a consequence of migraine and cannot be explained exclusively by comorbidity with depression or anxiety.
