Abstract
Objectives: To examine the association of genetic variants in the syntaxin 1A gene (STX1A) with common forms of migraine, and perform a combined analysis of the data from the current study and previously published reports.
Methods: We investigated the parent-to-offspring transmission of rs6951030, rs4363087 and rs2293489 in 191 family trios, each with a proband with childhood-onset migraine, and performed a case-control analysis between the probands and 223 unrelated controls. In addition, we performed a combined data analysis with an overall sample of 567 migraine patients and 720 unrelated controls and performed a migraine-specific gene-network analysis.
Results: The transmission disequilibrium test revealed significant transmission distortion of rs4363087 in migraine overall (OR = 1.56, p = 0.006; p = 0.01 after correction for multiple testing) and migraine without aura (OR = 1.58, p = 0.01; corrected p = 0.04). Two-marker haplotype analysis revealed transmission distortion of A-G (rs6951030-rs4363087; OR = 1.47, p = 0.01) and A-C (rs4363087-rs2293489; OR = 0.66, p = 0.01). Combined analysis showed significant association of rs941298 with migraine overall (OR = 1.28, p = 0.004) and migraine without aura (OR = 1.3, p = 0.008). Network analysis identified 24 genes relating STX1A to other migraine candidate genes, including KCNK18 (TRESK channel) involved in the cytoplasmatic calcium signalling together with syntaxin 1A.
Conclusion: Our results provide support for the hypothesis that STX1A represents a susceptibility gene for migraine.
Introduction
Migraine is a disabling neurological disorder with a current global prevalence higher than 10% (1) and a complex pathophysiology. Despite family and twin studies suggesting a strong genetic component for the disease (40–65% heritability) (2–4), candidate genes and linkage approaches have failed to identify robustly replicated susceptibility genes, with the exception of familial hemiplegic migraine (FHM), a rare subtype of migraine with aura showing Mendelian inheritance. Genetic variants in three genes (CACNA1A, ATP1A2, SCN1A) are clearly associated with FHM (5–7), but their involvement in common migraine has not been convincingly demonstrated (8,9); however, because the three genes are all implicated in ion transport, these findings suggest that ionic disturbances could also play a role in the pathogenesis of the common forms of migraine (10).
This hypothesis has been confirmed with the identification of a frameshift mutation, F139WfsX24, in the KCNK18 gene that segregates perfectly with the migraine with aura phenotype in a multigenerational pedigree (11). KCNK18 encodes the TRESK channel (TWIK-related spinal cord potassium channel), a tandem pore potassium channel (K2p) primarily expressed in the nervous system, involved in the regulation of neuronal excitability (12–14). Syntaxin 1A, a protein part of the SNARE complex (15), physically interacts with different presynaptic K+ channels, modulating their ability to shape presynaptic action potentials (16,17); moreover, in vitro studies have revealed a direct interaction of the protein with the α1A subunit of the P/Q-type calcium channel encoded by CACNA1A, the first FHM gene identified (18,19).
The syntaxin 1A gene (STX1A) at chromosome 7q11.23 was recently associated with the common forms of migraine in a case-control association study from Spain, reporting association with the single-nucleotide polymorphism (SNP) rs941298 and the allelic combination A-T-G (rs6951030-rs941298-rs4363087) (20). These findings have been subsequently supported by a Portuguese study analysing the contribution to migraine of three SNPs in STX1A, two of which were analysed in the original report (21).
In an attempt to confirm the involvement of the STX1A gene in migraine susceptibility, we investigated three SNPs in the STX1A locus (rs6951030, rs4363087 and rs2293489), using a case-control and family-based association study design. In addition, we performed a combined analysis of the data from the present study and the two previously published reports, and through bioinformatic data mining, we built a migraine-specific protein–protein interaction network, including STX1A and the other genes previously associated with migraine.
Subjects and methods
Subjects
One hundred and ninety-one family trios (father, mother and affected proband) were recruited through the Headache Outpatient Centre, Department of Child and Adolescent Psychiatry, Medical University of Vienna, Austria. Of those admitted during the period between February 2004 and December 2005 who were diagnosed with migraine with (MA) or without aura (MO), patients and parents were asked to participate in the study. Eighty-nine patients were not included in the study because they showed comorbidity with other disorders such as asthma, epilepsy, allergies or tension-type headache.
Forty-eight per cent of the probands were male and 52 were female. Age ranged from 6 to 19 years, with a mean age of 10.8 ± 2.8 years. The diagnoses were established by an experienced child neurologist based on the criteria of the second edition of the International Classification of Headache Disorders (ICHD-II) (22,23). Of the 191 probands, 137 met ICHD-II criteria for MO and 43 for MA, while the remaining 11 were not classified. The sample also included 223 unrelated healthy controls, who were recruited from high schools in Vienna and were matched for age and sex. They completed a structured headache symptoms questionnaire to exclude migraine symptomatology. The study was approved by the Ethics Committee of the Medical University of Vienna. Informed written consent was obtained from all patients, parents and control individuals.
For the combined analysis, we used an overall sample of 567 migraine patients and 720 unrelated controls, obtained by combining our case-control sample (191 migraine patients and 223 unrelated controls) with the case-control samples of the two prior reports, the Spanish study (210 migraine patients and 210 unrelated controls) and the Portuguese study (188 migraine patients and 287 unrelated controls). To focus the analysis exclusively on common forms of migraine, we excluded from the Spanish sample the 22 patients with FHM.
Genotyping
DNA was prepared using established methods from buccal swabs as described previously (24,25). The samples were taken by the subjects themselves and were either immediately left at the clinic or sent to the Vienna centre by mail. The samples were coded before DNA extraction and stored at the Social Genetics and Developmental Psychiatry Centre molecular genetics laboratory of the Institute of Psychiatry. The genotyping assay adopted (TaqMan) allowed us to genotype only two of the three SNPs analysed in the original study. For the third SNP, rs941298, we were unable to design a suitable custom assay, consequently we selected a perfect proxy SNP, rs2293489 (r2 = 1 with rs941298), located in the STX1A locus (WBSCR22 gene), using the SNAP program (26). All SNPs (rs6951030, rs4363087 and rs2293489) were genotyped with a TaqMan SNP genotyping assay on an ABI Prism 7900HT Sequence Detection System (Applied Biosystems, Foster City, CA). Genotyping was performed using a standard protocol with ABsolute QPCR Rox Mix (Thermo Fisher Scientific, Waltham, PA) and predesigned assays (Applied Biosystems; rs6951030: C_29194787_10; rs4363087: C_31464141_10; rs2293489: C_15971044_10). Data evaluation was carried out using the SDS 2.1 software (Applied Biosystems).
Study selection and data extraction for the combined analysis
The two previous reports investigating the involvement of the STX1A gene in migraine susceptibility were identified through a PubMed search, using the keywords ‘syntaxin 1A’, ‘STX1A’, ‘migraine’, ‘migraine without aura’, ‘migraine with aura’. The search originally produced four results.
The study eligibility criteria included: (i) publication in peer-reviewed journals; (ii) report of original and independent data; (iii) investigation of patients with migraine diagnosed according to the International Classification of Headache Disorders (ICHD); (iv) case-control or family-based association study design, providing sufficient data to calculate odds ratio (OR) and p-value. Only two of the four studies identified met the above criteria and were thus included in the combined analysis, the Spanish study (20) and the Portuguese study (21). The other two studies identified by the keyword search were excluded as they did not perform genetic association analysis of migraine with STX1A (PMID: 20080591; PMID: 17666428).
The data extracted from each study were: title and authors, journal, year of publication, ethnicity of the population investigated, study design, number of cases and controls, diagnosis of subgroups, polymorphisms genotyped, allele and genotype frequencies.
Statistical analysis
The program package UNPHASED was used to test for association between the individual marker locus and the hypothetical disease locus for both the family trios and the case-control sample (http://www.mrc-bsu.cam.ac.uk/personal/frank/software/unphased/) (27). The transmission disequilibrium test (TDT) analysis for the family trios was performed using the TDTPHASE application of UNPHASED. All analyses were performed in both the migraine sample overall and the MO and MA subgroups. Pairwise linkage disequilibrium calculations and haplotype estimations were performed using the Haploview 4.2 software (http://www.broadinstitute.org/haploview/haploview) (28).
Because two of the markers analysed were in significant LD in our sample, a correction for multiple testing using the Bonferroni method would have resulted in an overly conservative test, and so allele frequencies were adjusted for multiple comparisons using the correlation matrix-based method, which takes into account the linkage disequilibrium between markers (29). The effective number of independent SNPs (Meff) was determined using the software SNPSpD (http://gump.qimr.edu.au/general/daleN/SNPSpD/), with the method described by Li and Ji (29,30), and the threshold for significance was calculated as αcorr = 0.05/Meff. Based on this method, we obtained a Meff estimate of 2, and we therefore set the significance threshold to αcorr = 0.025 (αcorr = 0.05/2). In addition, in order to correct for testing the associations also in the MO and MA subgroups, allele frequencies in these groups were further adjusted for multiple comparisons using the Bonferroni method, and the significance threshold was lowered to αcorr = 0.0125 (αcorr = 0.025/2). Haplotype frequencies were corrected for multiple comparisons using a permutation procedure (10,000 permutations).
The combined analysis for rs6951030 and rs941298 was performed using the software CATMAP, an R package that conducts fixed-effects and random-effects meta-analyses of case-control or family-based genetic data (31). We performed the Q statistical test to assess possible heterogeneity among the individual studies and CATMAP was configured in order to apply the random-effects model in the presence of heterogeneity and the fixed-effects model in the absence of heterogeneity. The allelic forest plots for the two SNPs were made using the STATA software, version 10 (32).
Power analysis
Power analysis was performed using the genetic power calculator (GPC) (http://pngu.mgh.harvard.edu/~purcell/gpc/) (33), assuming a migraine prevalence of 14%, genotype relative risks of 1.5 (heterozygotes, Aa) and 2.25 (homozygotes, AA) and a significance level (α) of 0.05. For the risk allele of rs6951030 (allele frequency 14.6%), the migraine sample overall showed a non-centrality parameter (NCP) of 6.7 and 73.6% power, whereas the MO and MA subgroups revealed NCP of 5.7 and 2.6, and power of 66.5% and 36.6%. For the risk allele of rs4363087 (allele frequency 43.7%), the total migraine sample showed NCP of 11.4 and 92% power, while the MO and MA subgroups revealed NCP of 9.4 and 4.03, and power of 86.6% and 51.9%. Finally, for the risk allele of rs2293489 (allele frequency 34.3%), the migraine sample overall showed NCP of 10.9 and 91% power, whereas the MO and MA subgroups revealed NCP of 9.1 and 3.99, and power of 85.5% and 51.5%.
For the combined analysis, with the effect sizes detected (rs941298: OR = 1.28; rs6951030: OR = 1.12), power calculation revealed that samples of 383 (rs941298) and 2510 (rs6951030) cases would be required to achieve 80% power.
Bioinformatic analysis
The SNAP program was used to select a perfect proxy SNP (r2 = 1) for rs941298. SNAP is a bioinformatics query tool for the retrieval of linkage disequilibrium proxy SNP results given input of one or more query SNPs and based on empirical observations from the International HapMap Project (http://www.broadinstitute.org/mpg/snap/) (26).
Network analysis was performed using MetaCore version 6.5 (GeneGo Inc., St Joseph, MI), an integrated database and software suite for pathways and networks analysis, based on a curated database of human protein–protein, protein–DNA and protein–compound interactions. Networks were generated using Dijkstra’s shortest paths building algorithm, in order to obtain a network containing all the candidate genes connected with the shortest possible path. Functional and binding interactions, also with unspecified effect, were included in the network building parameters.
Ontology enrichment analysis was performed for each network, in order to identify the pathways and biological functions most represented or ‘enriched’ by the input data.
Results
The genotyping success rate was 99.14% for rs6951030 and rs4363087, 99.48% for rs2293489. For each SNP, genotype frequencies were in Hardy–Weinberg equilibrium in both cases and controls (cases/controls: rs6951030: p = 0.94/p = 0.89; rs4363087: p = 0.13/p = 0.71; rs2293489: p = 0.053/p = 0.82). Pairwise linkage disequilibrium calculations revealed significant LD (r2 = 0.7) between the SNPs rs4363087 and rs2293489 in the case-control sample (eFigure 1).
Forest plot of odds ratio (OR) and overall OR with 95% CI for the T allele of the SNP rs941298 in migraine overall using the fixed-effects model. CI indicates confidence interval.
Family-based and case-control association results
Family-based association results (transmission disequilibrium test)

MO: migraine without aura; MA: migraine with aura; T: transmitted; NT: not transmitted; OR: odds ratio; CI: confidence interval.
Uncorrected.
Significant after correction for multiple testing.
Three-marker haplotype analysis results (transmission disequilibrium test)
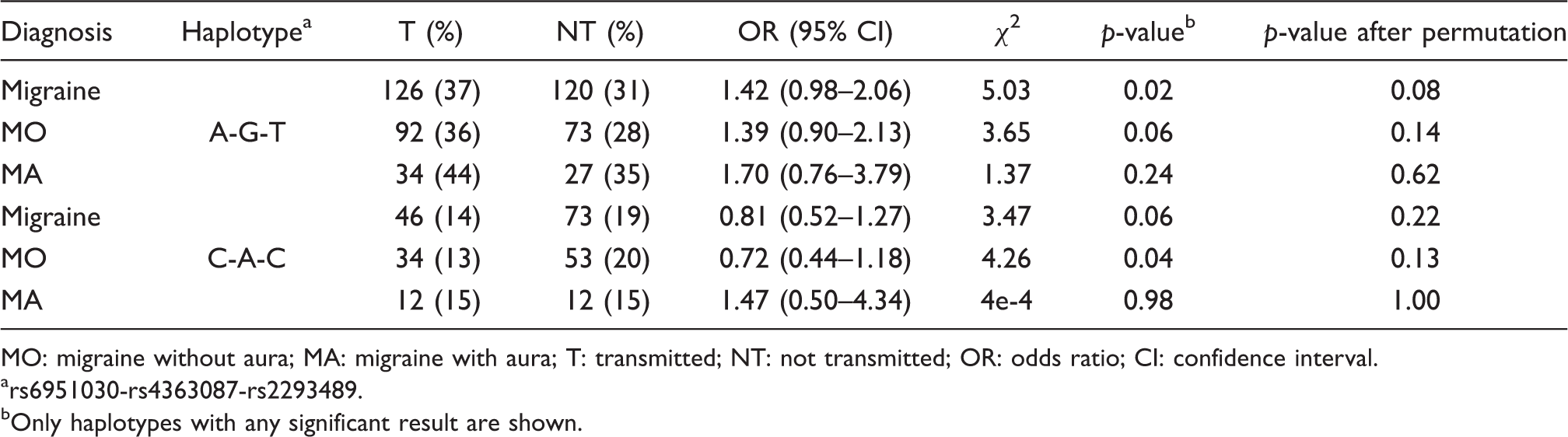
MO: migraine without aura; MA: migraine with aura; T: transmitted; NT: not transmitted; OR: odds ratio; CI: confidence interval.
rs6951030-rs4363087-rs2293489.
Only haplotypes with any significant result are shown.
Two-marker haplotype analysis results (transmission disequilibrium test)

MO: migraine without aura; MA: migraine with aura; T: transmitted; NT: not transmitted; OR: odds ratio; CI: confidence interval.
rs6951030-rs4363087.
rs4363087-rs2293489.
rs6951030-rs2293489.
Only haplotypes with any significant result are shown.
Case-Control Genotype and Allele Frequencies

MO = migraine without aura. MA = migraine with aura.
Combined analysis
In order to provide a more comprehensive evaluation of the involvement of the STX1A gene in the aetiology of the common forms of migraine, we performed a combined analysis of the case-control data from the present study and the published data of the two precedent reports, for a total sample of 567 migraine patients and 720 unrelated controls.
The combined analysis was performed only for the SNPs rs6951030 and rs941298, which were genotyped across all three studies. Rs941298 showed a significant allelic association with the migraine sample overall (OR = 1.28; 95% CI, 1.08 to 1.51; p = 0.004). Subdividing the pooled sample into the MO and MA subgroups, the association remained significant in the MO subgroup (OR = 1.3; 95% CI, 1.07 to 1.57; p = 0.008) but there was only a trend toward significance in the MA subgroup (OR = 1.2; 95% CI, 0.98 to 1.57; p = 0.07). The allelic forest plot for the migraine sample overall is shown in Figure 1. The SNP rs6951030 did not show any association with the migraine sample overall (OR = 1.12; 95% CI, 0.8 to 1.6; p = 0.51) and with both the MO and MA subgroups (eFigure 2). The Q statistical test for heterogeneity was not significant for rs941298 (Q test p = 0.49) but it was significant for rs6951030 (Q test p = 0.05), revealing the presence of between-study heterogeneity for the second marker. The combined analysis data tables can be found in the online supplement (eTables 1 and 2).
Migraine-specific protein–protein interaction network including isoflurane (blue circle), syntaxin 1A (green circle) and the four migraine-related proteins (red circles). Lines between nodes represent different types of interaction, including activating (green), inhibiting (red) and unspecified (grey) effects. Detailed description of the symbols can be found at http://www.genego.com/pdf/MC_legend.pdf.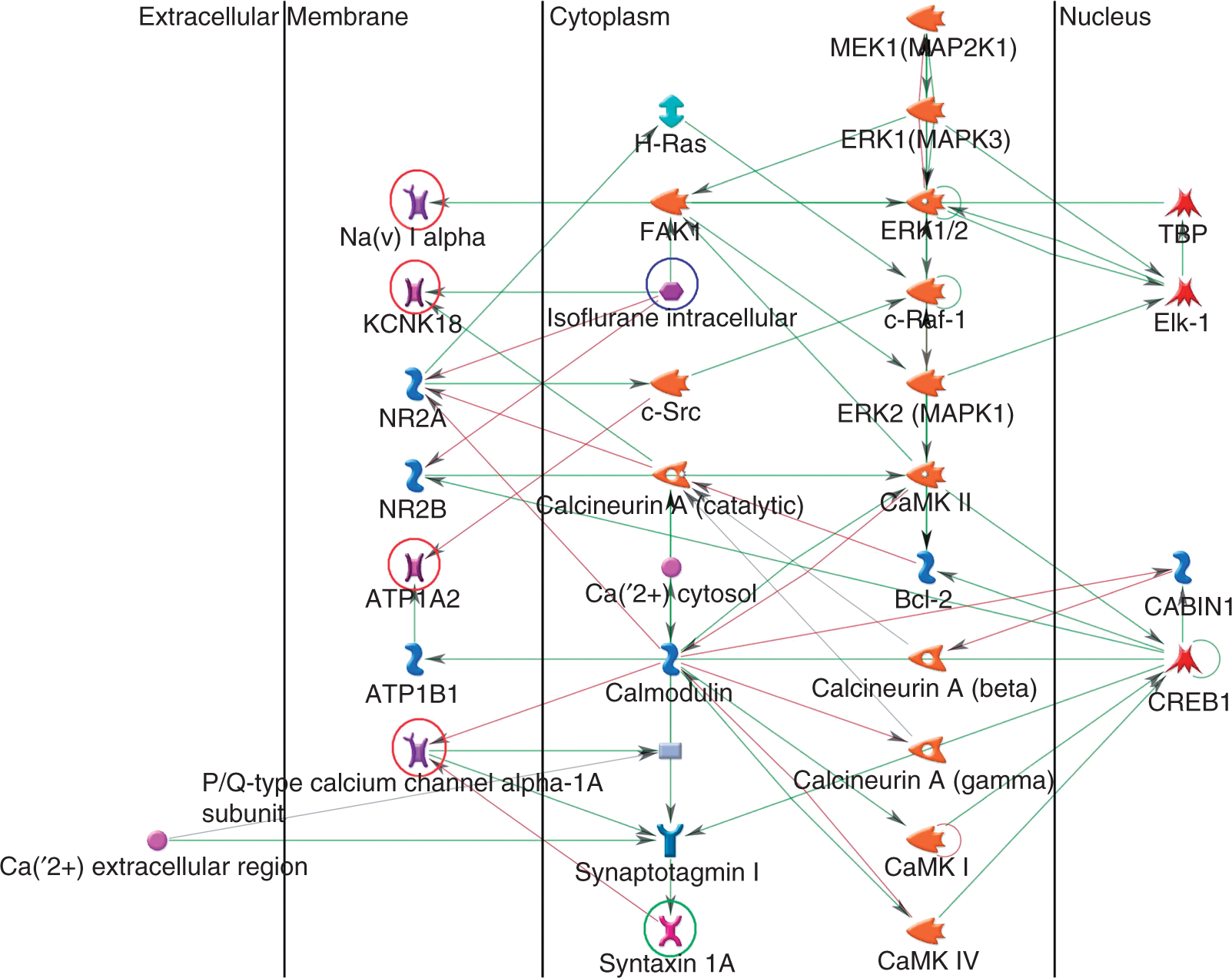
Network analysis
In order to investigate the biological relationship between STX1A and the other genes previously associated with both common migraine (KCNK18) and FHM (CACNA1A, ATP1A2, SCN1A), we built a migraine-specific protein–protein interaction network using the bioinformatic tool MetaCore (GeneGo Inc., St Joseph, MI). To focus our attention on the interactions most closely tied to the candidate genes and thus most likely to be involved in migraine pathophysiology, we selected the ‘shortest paths’ building option. Increasing the maximum path length of the shortest paths algorithm to four edges, we obtained a network consisting of 27 nodes (24 genes), expressed primarily in brain (p = 1.3E-07), which included both syntaxin 1A and the products of the four migraine-related genes (eFigure 3).
Referring to the common forms of migraine, the network highlighted an important relationship between syntaxin 1A and the KCNK18 (TRESK) protein, which are both related to cytoplasmatic calcium signalling; in particular, syntaxin 1A physically interacts with the α1A subunit of the P/Q-type calcium channel and inhibits the channel activity, while the TRESK channel is regulated by calcineurin, a Ca2+/calmodulin-dependent phosphatase activated by increases in intracellular Ca2+ concentration.
Performing an ontology enrichment analysis, the network showed enrichment of proteins for gene ontology (GO) processes such as transport (p = 2.4E-09) and transmission of nerve impulse (p = 9E-09), which is strongly consistent with the hypothesis that ionic disturbances may play a role in the pathogenesis of common migraine; moreover, the network was over-represented for genes associated with diseases such as mental disorders (p = 2.8E-09) and central nervous system diseases (p = 7.2E-08), which is in keeping with the well documented high level of comorbidity between migraine and several psychiatric and neurological disorders such as depression and epilepsy (34).
Because the TRESK channel has recently been identified as an important target for volatile anaesthetics such as isoflurane (35,36), which is also thought to inhibit the neurotransmitter release machinery by interaction with the syntaxin 1A protein (37,38), we decided to test the impact of isoflurane on our migraine-specific network adding the anaesthetic to the network objects.
The resulting network (33 nodes), expressed primarily in brain (p = 6.1E-10) and containing all the five candidate genes, revealed a direct interaction between isoflurane and four proteins, including the TRESK channel (KCNK18), the N-methyl D-aspartate receptors subtype 2A and 2B (NR2A-NR2B) and the protein tyrosine kinase 2 (FAK1) (Figure 2).
Performing an ontology enrichment analysis, this second network also showed strong enrichment of proteins for gene ontology (GO) processes such as transmission of nerve impulse (p = 7.1E-12), transport (p = 1.5E-11) and synaptic transmission (p = 3.2E-11); moreover, the network confirmed a significant statistical over-representation of genes associated with mental disorders (p = 5E-10) and central nervous system diseases (p = 1.2E-09).
Discussion
In an attempt to confirm the involvement of the STX1A gene in migraine susceptibility, we investigated three SNPs (rs6951030, rs4363087 and rs2293489), in the STX1A locus, in a sample of 191 family trios (137 MO, 43 MA) and 223 unrelated controls. Performing the TDT analysis for the single markers, we observed association between rs4363087 and both the migraine sample overall and the MO subgroup. Rs2293489 also showed association with the migraine sample overall, replicating the findings of both the previous studies with a perfect proxy of rs941298. Performing a three-marker haplotype analysis we observed association between the allelic combination A-G-T, the equivalent to the significant haplotype in the original report, and migraine overall. However, we did not find any association between this haplotype and either MO or MA, which may relate to statistical power, which decreases when the migraine sample is subdivided, especially for MA, because of the small number of MA trios.
Thus, the current study supports the role of the STX1A gene as putative risk factor for the common forms of migraine suggested in two previous reports (20,21). A case-control association study investigating the contribution to the genetic susceptibility to migraine of three SNPs (rs6951030-rs941298-rs4363087) in the STX1A gene reported association between migraine and both rs941298 and the allelic combination A-T-G (rs6951030-rs941298-rs4363087) (20). This preliminary evidence is supported by a Portuguese study investigating the contribution to the genetics of migraine of three SNPs (rs3793243-rs941298-rs6951030) in STX1A, two of which were analysed in the original report. The study replicated the association between migraine and rs941298, and it also reported association with both rs6951030 and the haplotype T-C-T (rs3793243-rs941298-rs6951030) (21).
Because individual studies may not have sufficient statistical power to detect a modest effect of the polymorphisms on the risk of migraine, we also performed a combined analysis of the data from the present study and the published data of the two precedent reports. The combined analysis revealed a significant association between rs941298 and both the migraine sample overall and the MO subgroup, providing further support for the involvement of this polymorphism in the genetic predisposition to migraine.
Syntaxin 1A is a presynaptic transmembrane protein primarily expressed in neuronal and secretory cells, part of a molecular complex (SNARE) that controls the docking of synaptic vesicles and their fusion with the presynaptic membrane (15). Presynaptic ion channels play an important role in the regulation of neurotransmitter release thanks to their ability to shape presynaptic action potentials; however, many ion channels contribute to the regulation of the neurotransmitter release process also through direct interaction with components of the exocytotic apparatus, and the syntaxin 1A protein represents one of the most important. Syntaxin 1A physically interacts with a wide range of ion channels involved in the regulation of neuronal excitability, such as many members of the voltage-gated K+ channel (Kv) superfamily (16,17).
The TRESK channel (TWIK-related spinal cord potassium channel) (12,13) is a tandem pore potassium channel (K2P) recently associated with MA in a large collaborative study analysing 150 ion channel genes, which identified a frameshift mutation (F139WfsX24) in the KCNK18 gene that was found to co-segregate precisely with the MA phenotype in a large pedigree (11). The K2P channels are background K+ channels that help to set the resting membrane potential and are thus involved in the regulation of neuronal excitability together with the voltage-gated K+ channels (14).
In order to analyse the biological relationship between STX1A and the other genes previously associated with both common migraine (KCNK18) and FHM (CACNA1A, ATP1A2, SCN1A) and in order to put these candidate genes into context, seeking the possibility to generate new gene–gene interaction hypotheses, we built a migraine-specific protein–protein interaction network using the bioinformatic tool MetaCore (GeneGo Inc., St Joseph, MI). Assuming that the genes most likely to be involved in migraine pathophysiology are those most closely interacting with the five candidate genes, we used the ‘shortest paths’ building option, which generates a network containing all the candidate genes connected with the shortest possible path.
The network obtained highlighted an important relationship between syntaxin 1A and the TRESK channel, which are both involved in the cytoplasmatic calcium signalling; effectively, TRESK is regulated by different physicochemical factors in cells such as the cytoplasmatic Ca2+ concentration, which modulates the channel activity with a calcineurin-mediated mechanism (39). The syntaxin 1A protein, instead, plays a crucial role in the regulation of calcium levels in the presynaptic nerve terminals by direct interaction with different voltage-gated calcium channels such as the N-type and P/Q-type Ca2+ channels, the two major Ca2+ channel subtypes found at the nerve terminals, which physically bind syntaxin 1A respectively with the α1B (CACNA1B) and α1A (CACNA1A) subunits (18,19).
Because the TRESK channel has recently been identified as an important target for volatile anaesthetics such as isoflurane (35,36), which is also thought to inhibit the neurotransmitter release machinery by direct interaction with the syntaxin 1A protein (37,38), we decided to test the impact of isoflurane on our migraine-specific network and we observed a direct interaction of the anaesthetic with different network objects.
Because the mechanism of action of general anaesthetics in the nervous system is thought to be primarily mediated by ion channels (40), the above observation, together with the evidence that our network model is strongly over-represented for proteins involved in GO processes such as transport and transmission of nerve impulse, provides support to the hypothesis that ion channel disturbances may play a role in the pathogenesis of common migraine, suggesting a possible linkage between the disorder and the Mendelian channelopathies such as FHM.
Finally, it is important to highlight that our findings provide new significant validation to the involvement of STX1A in the genetic predisposition to common migraine, in that, unlike the prior studies, for the first time we investigated the association in a sample of family trios with children affected with migraine (mean age of 10.8 ± 2.8 years). Migraine occurs in about 8% of children and adolescents (41), which is lower than the prevalence of migraine typically reported in the adult population (>10%) (1). In children under 14 years, the sex ratio is also equal, as seen in the present sample, whereas migraine is more common in adult females than males (1,41). A paediatric sample with an equal sex ratio may be an advantage, as it may allow a better utilisation of the power of the TDT, and may represent a phenotype less tainted by confounding environmental stressors. All this together makes the present study more than a simple replication study, as we also provide new information on childhood migraine, which is a subtly different phenotype compared to migraine in adults.
In summary, our results provide support for the hypothesis that STX1A represents a susceptibility gene for the common forms of migraine. We observed a new significant association of rs4363087 with migraine overall and MO, and we replicated the finding of the two precedent reports for rs941298. In addition, in an attempt to overcome the power limitations due to the relatively small size of our sample, we performed a combined data analysis which confirmed the association between rs941298 and both migraine overall and MO; the MA subgroup, instead, showed just a trend toward significance for the SNP, which could be explained by the relatively small number of MA cases. Replication will be needed to further validate our findings and to provide new insights into the role of syntaxin 1A in migraine pathophysiology.
Footnotes
Acknowledgements
We would like to thank all families and controls for their collaboration. We are grateful to Prof. Cathryn M. Lewis for statistical advice.
Funding
This study was supported by the Jubiläumsfonds of the Austrian National Bank given to Ç. W-B. (grant no. 10645).
Conflict of interest
Christian Wöber has received honoraria and travel funding from AstraZeneca (Austria), Linde Gas (Austria), A. Menarini Pharma GmbH (Austria) and Pfizer (Austria). All other authors declare no financial interests or potential conflicts of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
