Abstract
Objective: The aim was to identify and describe migraine trigger factors in patients with familial hemiplegic migraine (FHM) from a population-based sample.
Methods: 127 FHM patients were sent a questionnaire listing 16 trigger factors. Distinction was made between attacks of hemiplegic migraine (HM) and migraine with aura (MA) or without aura (MO) within each patient.
Results: The response rate was 59% (75/127) of whom 57 (76%) had current HM attacks. Sixty-three per cent (47/75) reported at least one factor triggering HM, and 36% (27/75) reported at least one factor that often or always caused HM. Twenty per cent (15/75) reported only HM, whereas FHM in combinations with MA and MO were reported by 80% (60/75). Stress (with attacks either following or during the stress), bright light, intense emotional influences and sleeping too much or too little were the trigger factors mentioned by most.
Conclusion: Many FHM patients report trigger factors and one-third reported at least one trigger factor often or always triggering FHM. The typical triggers are the same as for MA. Patients should be educated to avoid these factors. The role of trigger factors in the onset of new or first attacks of FHM remains unknown.
Introduction
A migraine trigger is any factor that on exposure or withdrawal leads to the development of a migraine attack (1). Many migraine patients report trigger factors for their migraines but the mechanisms by which external migraine triggers exert their effect is not clarified. Familial hemiplegic migraine (FHM) is a rare dominantly inherited subtype of migraine with aura, characterized by fully reversible half sided weakness and other aura symptoms preceding or accompanying a migraine headache (2). Mutations in at least three different genes cause the FHM phenotype (3–5). Migraine aura is likely to be the symptom of cortical spreading depression (CSD), a wave of electrophysiological hyperactivity followed by neuronal inhibition (6). A model has been proposed that links FHM mutations with a propensity to CSD (7). Functional studies of a range of FHM mutations in animals suggest that impaired ion transport and cellular signalling could be a key factor in determining brain susceptibility to migraine aura and headache (7,8). Environmental triggers factors for FHM have previously not been investigated. To date, no specific therapy is available for FHM patients and therefore identification and avoidance of trigger factors might be important preventive strategy in clinical practice.
The aim of the present study was twofold: to identify environmental trigger factors in patients with familial hemiplegic migraine from a population-based sample and to compare the pattern of migraine trigger factors in FHM with that of the common types of migraine; with typical aura (MA) and without aura (MO).
Methods
Patients
Our study population comprised FHM patients from 44 families recruited from a population-based sample (9). All patients had previously a physical and neurological examination by a physician and had been diagnosed with FHM according to the International Classification of Headache Disorders 2nd Edition (2). All patients were genetically tested and sequence analysis revealed exonic mutations in the CACNA1A and ATP1A2 genes in six families (10). Of the original 147 eligible patients, 7 were deceased, 13 were non-contactable and 52 did not participate (Figure 1).
Study population. FHM, Familial hemiplegic migraine; MA, migraine with aura; MO, migraine without aura.
Standard protocol approvals, registrations and patient consents
The study was presented to the Ethics Committee of the County of Copenhagen that deemed no formal approval was necessary. We received written informed consent from all patients participating in the study.
Data
We sent the questionnaire regarding trigger factors to all 127 eligible patients. Those not responding within 1month were mailed a reminding letter. The questionnaire was similar to that used in a previous MA study (11) and consisted of 16 listed trigger factors, and participants were asked to grade these factors on a 5-point scale depending on their likelihood of precipitating an attack of FHM (0 = never, 1 = infrequent, 2 = approximately half of the time, 3 = often and 4 = always). The patients were also asked to fill out whether they suffered from MA and MO. If so they should fill out the questionnaire for each type of migraine separately. It was possible to add up to three additional trigger factors and grade them as well. Participants were asked to state their current frequency of attacks of FHM, MA and MO per year.
Statistical analyses
The Spearman's correlation, Mann Whitney U or ANOVA, test were used as appropriate. All p-values are two-sided. p-values < 0.05 were considered significant. All analyses were performed with SPSS for Windows 17.0 (Chicago, Illinois, USA).
Results
Clinical characteristics of the patients (median, range)
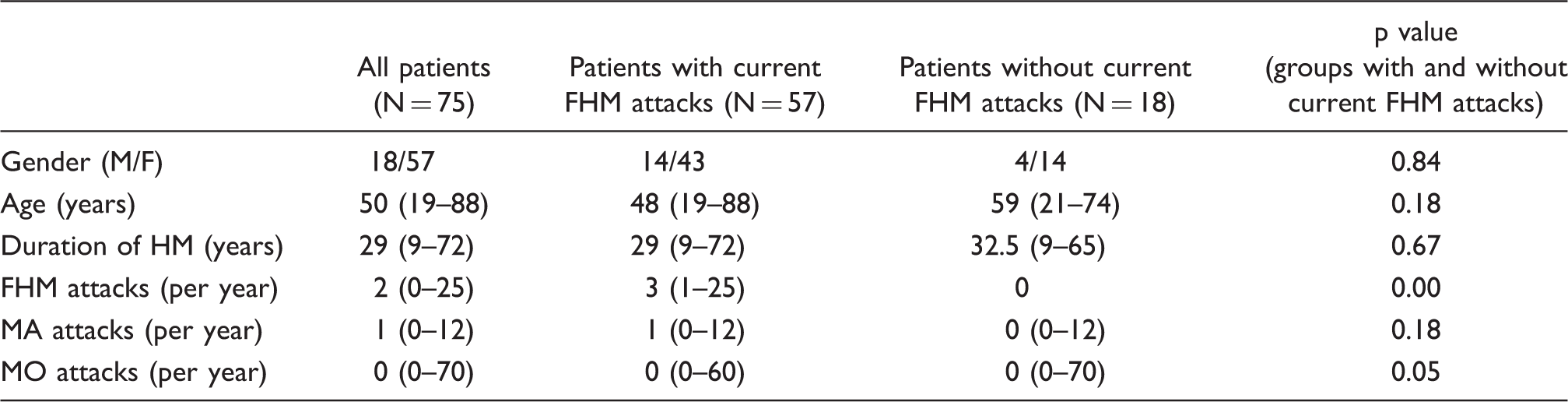
Number of FHM patients with other types of migraines, attack frequency (median, range) and the three most frequent reported triggers for FHM

A total of 57 (76%) patients had current attacks of FHM (at least one attack within the last year) whereas 18 (24%) had had no attacks for a year or more (no attacks). No statistically significant difference was found in age between the groups with current FHM attacks and no attacks (p = 0.18), nor in the duration of FHM (p = 0.67).
Overall 63% (47/75) of patients reported at least one trigger factor. Of patients with current attacks, 79% (45/57) reported one or more trigger factors. Among participants with no attacks for the last year, only 11% (2/18) reported one or more trigger factors (p < 0.001).
The median number of trigger factors for FHM was 2 (range 0–12) (Figure 2). The group with current FHM attacks reported significantly more trigger factors, median 4 (range 0–12), than the group without current attacks, median 0 (range 0–4, p < 0.001).
Percentage of patients according to the number of factors triggering an attack of FHM (n = 75).
A total of 36% (27/75) of patients reported at least one trigger factor that often or always precipitated an FHM attack, and in the group with current FHM attacks this percentage was 56% (25/45, p = 0.014). Seventeen percent (13/75) reported a trigger factor that could always precipitate an FHM attack, with no difference between groups with and without current FHM attacks (p = 0.16).
Individual trigger factors
The whole cohort
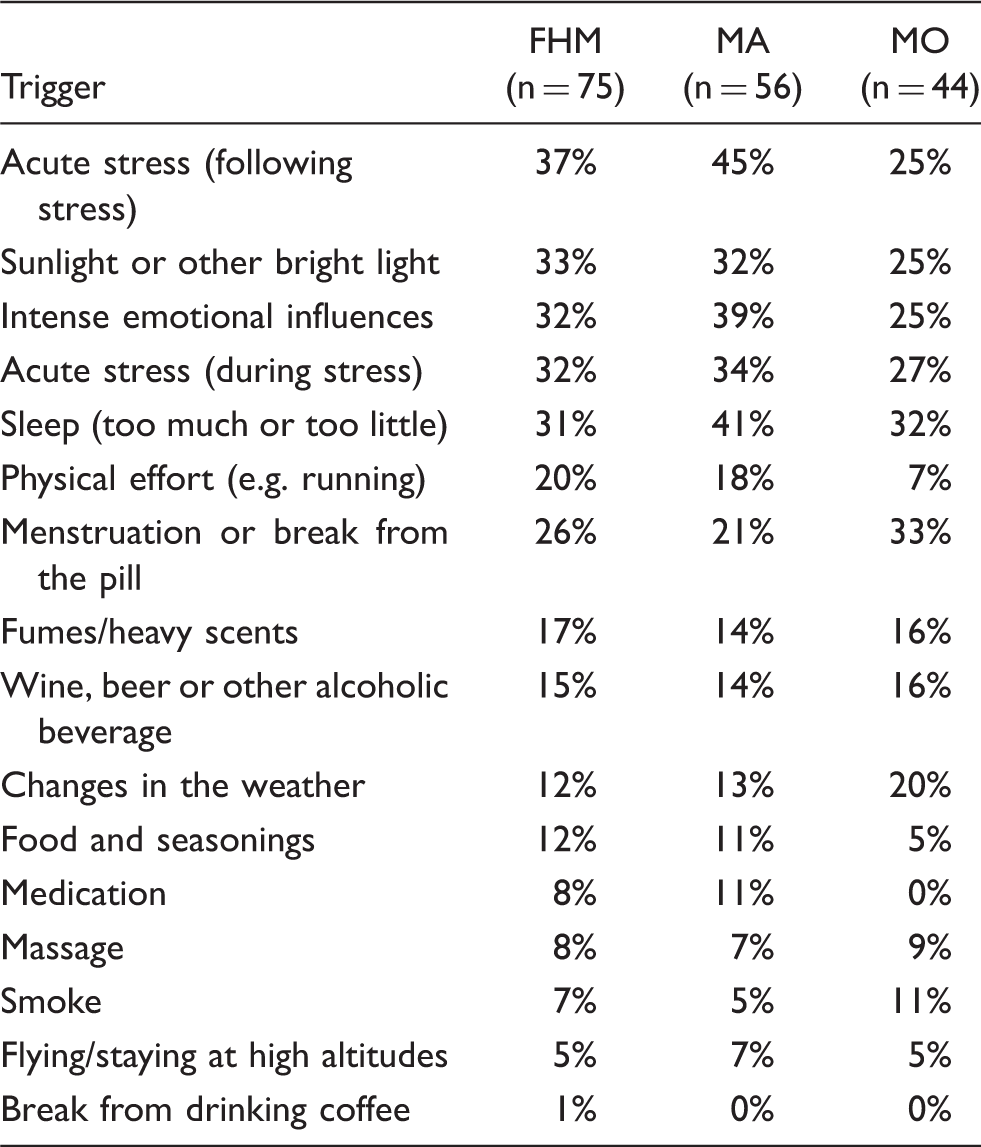
Acute stress (with the attack following the stress) was a trigger factor for FHM in 37% (28/75) of participants, followed by sunlight or other bright light in 33% (25/75), acute stress (with the attack occurring during the stress) in 32% (24/75), intense emotional influences in 32% (24/75). Sleeping too much or too little was a trigger factor for FHM in 31% (23/75) and menstruation or break from the pill was reported in 26% of the women (15/57) (Table 3). The triggers reported to often or always precipitate an FHM attack were acute stress (following stress) in 17% (13/75) followed by sunlight or other bright light in 13% (10/75), acute stress (during stress) in 12% (9/75), intense emotional influences in 12% (9/75), sleeping too much or too little in 9% (7/75), and menstruation or break from the pill in 9% of the women (5/57) (Figure 3).
Number of patients with current FHM attacks and at least one trigger factor reporting each individual trigger factor. Grey bars indicate the total number of patients who have reported the individual trigger. Black bars indicate the number of patients who have indicated that the specific trigger factor often or always triggers an attack (n = 45). Percentage of patients (in the whole cohort) reporting trigger factors for hemiplegic migraine (FHM) as well as for migraine with aura (MA) and migraine without aura (MO) if applicable
Patients with current FHM attacks and at least one trigger factor
Acute stress (following stress) was a trigger factor for FHM in 58% (26/45) of participants, followed by sunlight or other bright light in 55% (25/45), acute stress (during stress) in 51% (23/45), intense emotional influences in 51% (23/45), and sleeping too much or too little in 51% (23/45) (Figure 4).
Percentage of FHM patients with current FHM attacks and at least one trigger factor reporting each individual trigger factor (grey bars). For comparison the percentage of MA (black bars) and MO patients (white bars) reporting each individual trigger factor is included. MA data adapted from (11) and MO data adapted from (16,25). FHM and MA data were collected using the same questionnaire; MO data represent several studies, not differentiating between stress phases.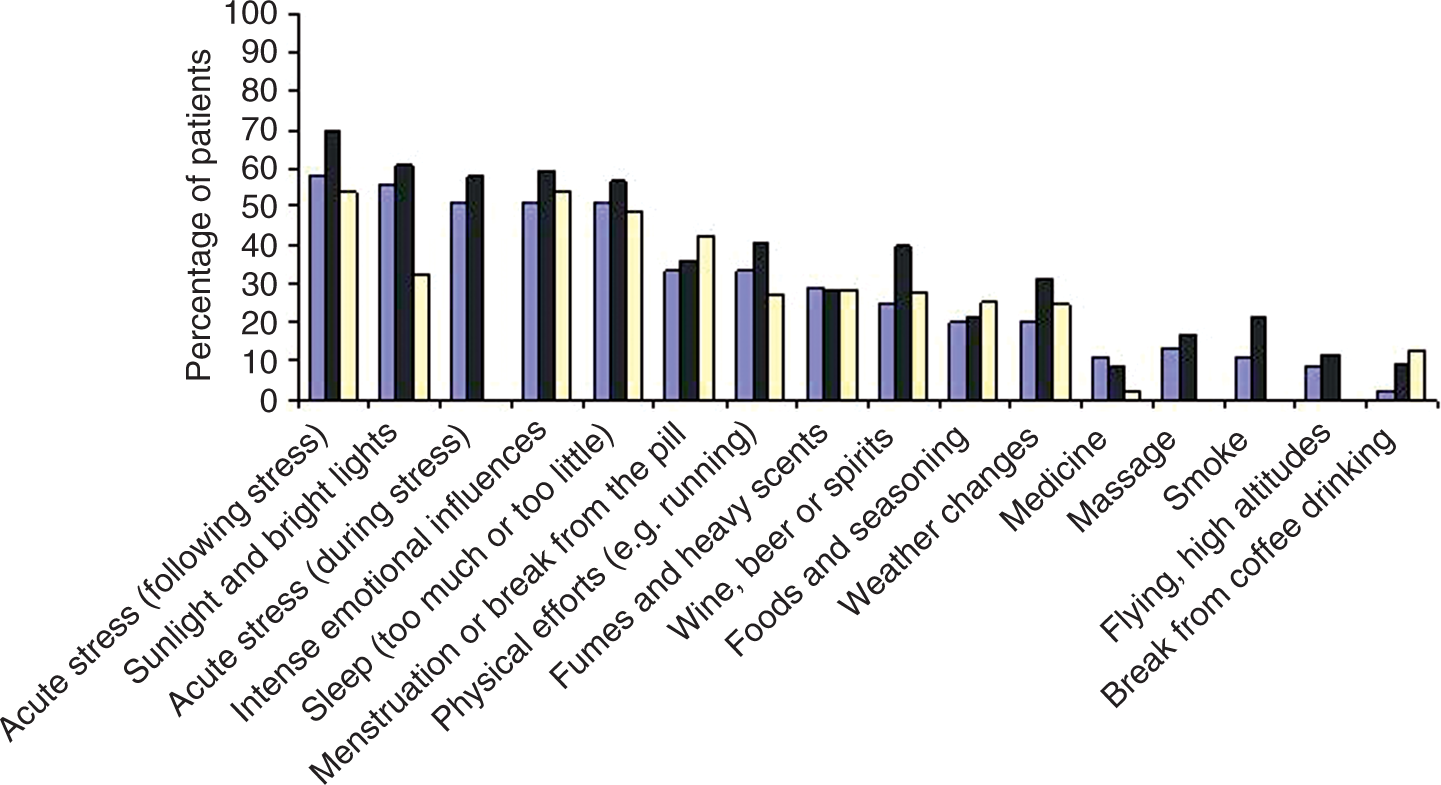
The triggers reported to often or always precipitate an FHM attack were acute stress (following stress) in 24% (11/45) of participants followed by sunlight or other bright light in 22% (10/45), acute stress (during stress) in 18% (8/45), intense emotional influences in 18% (8/45), sleeping too much or too little in 15% (7/45), and menstruation or break from the pill in 14% of the women (5/35).
The importance of migraine co-morbidity
Of patients with coexisting migraine with typical aura (MA) or migraine without aura (MO), 65% (39/60), and in the group with the pure FHM phenotype, 53% (8/15), reported one or more trigger factors (p = 0.25). The median number of trigger factors for FHM with coexisting migraine was 2 (range 0–12) compared with 1 (range 0–12) in the group with the pure FHM phenotype (p = 0.43). In the group with current attacks of FHM, MA and MO (n = 29) we found no difference in the number of trigger factor for MO (median 0, range 0–12) compared with MA (median 2, range 0–9) (p = 0.9). Comparison of the individual trigger factors for attacks of FHM and combinations of coexisting MA and MO are shown in Table 3.
The importance of mutation status
In 25% of the patients (19/75), mutations in the CACNA1A or ATP1A2 genes had been identified. Of these patients 53% (10/19) reported one or more trigger factors, compared with 66% (37/56) and in the group in which no mutation was found (p = 0.11). The median number of trigger factors for FHM patients with known mutation was 1 (range 0–10), and 2.5 (range 0–12) in the group in which no mutation was found (p = 0.16).
Self reported trigger factors
‘Other factors’ were reported only by a minority (n = 12) and encompassed skipped meals in three patients, anticipation and motion sickness in two patients and dietary nitrites in one patient. None of these factors were rated higher than two on the five point scale.
Correlations and group comparisons
The number of trigger factors was positively correlated with frequency of FHM attacks (p < 0.01), but there was no correlation between age and number of trigger factors (p > 0.10) or FHM duration and number of trigger factors (p = 0.25).
Discussion
The different types of migraine, FHM, MA and MO, are often presented as part of a spectrum of migraine disorders with shared neurobiology (12,13). FHM is a very interesting migraine variant, because it is considered a valid genetic model to study the pathogenic mechanisms in both FHM and common types of migraine (13,14). The present study of FHM patients from a population-based sample shows that many patients report factors that trigger FHM attacks. A number of factors are even reported to often or always precipitate an attack of FHM.
Trigger factors in migraine
Many migraine patients report specific, and to a large degree similar, trigger factors for their migraines (15). This has been reported across the migraine spectrum, in patients with MO (16), MA (11) and from mixed populations (17,18). The proportion of migraine patients reporting trigger factors seems similar across geographic, cultural, ethnic or racial boundaries (19). The mechanisms by which external migraine triggers exert their effect is not clarified, but it has been suggested that different migraine triggers activate a wide variety of brain areas, ultimately resulting in vasodilation and local release of inflammatory molecules that activate meningeal nociceptors (20).
Differences between MO and MA
In a large study (17) the presence of aura was more important for the difference in triggers than other phenotypic traits, and in a large population-based survey MA was more frequently associated with precipitating factors than MO (21). In contrast, two Danish studies found that precipitating factor were present in 61–65% of MA and 79–90% of MO patients (22,23). MA patients with current attacks of both MA and MO also reported a significantly higher number of trigger factors for MO than for MA (11). In contrast, in FHM patients with coexisting MA and MO, we found no difference in the number of trigger factors for MA and MO.
FHM and MA/MO
FHM is a rare disease and most patients are found in clinic-based samples. Our patients were recruited from a large population-based cohort (9) that has been genetically characterized (10). It has been suggested that subjects from the population experience migraine trigger factors less often than clinic patients (24). In our cohort, however, the percentage of patients not reporting any trigger factors was similar to that in a large clinic-based sample (17). To ensure comparability, and to avoid recall bias, we did a sub-group analysis in patients with current attacks and at least one trigger. In this group 79% (45/57) of FHM patients reported one or more trigger factors compared with 80% (278/347) in MA patients with current attacks (11). In our group of FHM with current attacks, 56% indicated that they were aware of at least one factor often or always giving rise to an attack of FHM, compared with 67% in the MA study (11). The most prevalent triggers in FHM are the same as in MA and MO, apart from bright light, which appears to have only a minor role for MO (25); see also Figure 4.
Based on previous reports, FHM patients seem less sensitive to pharmacological migraine triggers (26–29) than patients with typical migraine (30–32). The present data suggest, however, that FHM and MA, and to a large extent MO, share environmental migraine triggers.
FHM mutations and triggers
From animal studies a possible link between mutations in the FHM gene and sensitivity to external triggers has been suggested because in tottering mice carrying mutations of the calcium channel gene CACNA1A, stress, caffeine and ethanol reliably elicited attacks (33).
We could not establish any difference in the pattern of FHM triggers between patients in whom mutations have been found and those in whom mutations were absent. This fits with the clinical observation that migraine features of FHM with and without identified mutations are similar (34).
Some differences between common types of migraine and FHM
In two recent reports from clinic populations (19,35), the most prevalent trigger was emotional stress (59–95%), similar to our FHM sample. Odours were also were quite frequent (46–63%), whereas only a minority of FHM patients (17%) reported this. The reason for this difference is not known, but cerebral activation patterns during olfactory stimulation might be abnormal in a sample of predominantly MO patients (36). It could be speculated that FHM patients have a different olfactory threshold. In MA patients, Hauge et al. (11) found that vigorous physical exercise was a migraine trigger in 41%. This was reported by 33% in the present FHM patients, and even fewer (27%) in MO patients (Figure 4).
Methodological considerations
Even though patients might tend to forget about previous attacks, the dramatic FHM phenotype helps the patients to remember possible triggers. Even then, the group with current FHM attacks reported significantly more trigger factors than the group without current attacks. In an earlier report on the Danish cohort, 9% reported that minor head trauma was a trigger of FHM attacks (37). No responders, however, reported this in the present questionnaire. To compensate for recall-bias, we singled out the subgroup of FHM patients with current attacks and at least one trigger factor, to allow comparisons with previous MA/MO data. To evaluate the role of migraine triggers in the onset of new or first attacks of FHM, and to determine whether avoidance of trigger factors improves migraine status, prospective cohort studies are required.
Importance for treatment
Triptan treatment seems effective irrespective of trigger factors in migraine patients (38). Triptans are, however, currently contraindicated in FHM-patients even though they are probably effective (39). Until effective treatment of migraine aura becomes available (40), educating patients to pay attention to and avoid relevant trigger factors described in the present study is a particularly important part of the treatment of FHM patients, especially in patients with frequent attacks.
Footnotes
Funding
This work was supported by grants from the University of Copenhagen, The Danish Headache Society and through the Lundbeck Foundation Center for Neurovascular Signalling (LUCENS).
Conflicts of interest
None of the authors have any potential financial interests in the subject for this study. MA has received grant support and honoraria for lecturing from Merck and honoraria for lecturing from Pfizer, GlaxoSmithKline and AstraZeneca and is a consultant and/or scientific advisor for Merck and BTG International. JO has within the past 2 years received grants and research support from and/or has been on the speakers’ bureau of Merck and Company, Pfizer, Minster Pharmaceuticals, RoxRo Pharma, MAP Pharmaceuticals, NeurAxon and Lundbeck.
Acknowledgements
The authors thank all participating patients.
