Abstract
Objective: The study was designed to investigate the differences in salivary cortisol (hypothalamic-pituitary-adrenocortical [HPA] axis), immunoglobulin A (IgA) (immune system) concentrations and α-amylase (sympathetic nervous system [SNS]) activity between children with chronic tension-type headache (CTTH) and healthy children.
Methods: Thirty-six children, 10 boys and 26 girls (age: 9 ± 2 years) with CTTH and 36 age- and sex-matched healthy children were recruited. Salivary cortisol, α-amylase activity, salivary flow rate, IgA concentration and IgA rate were collected from non-stimulated saliva. A headache diary was used for collecting data on intensity, frequency and duration of headache for four weeks.
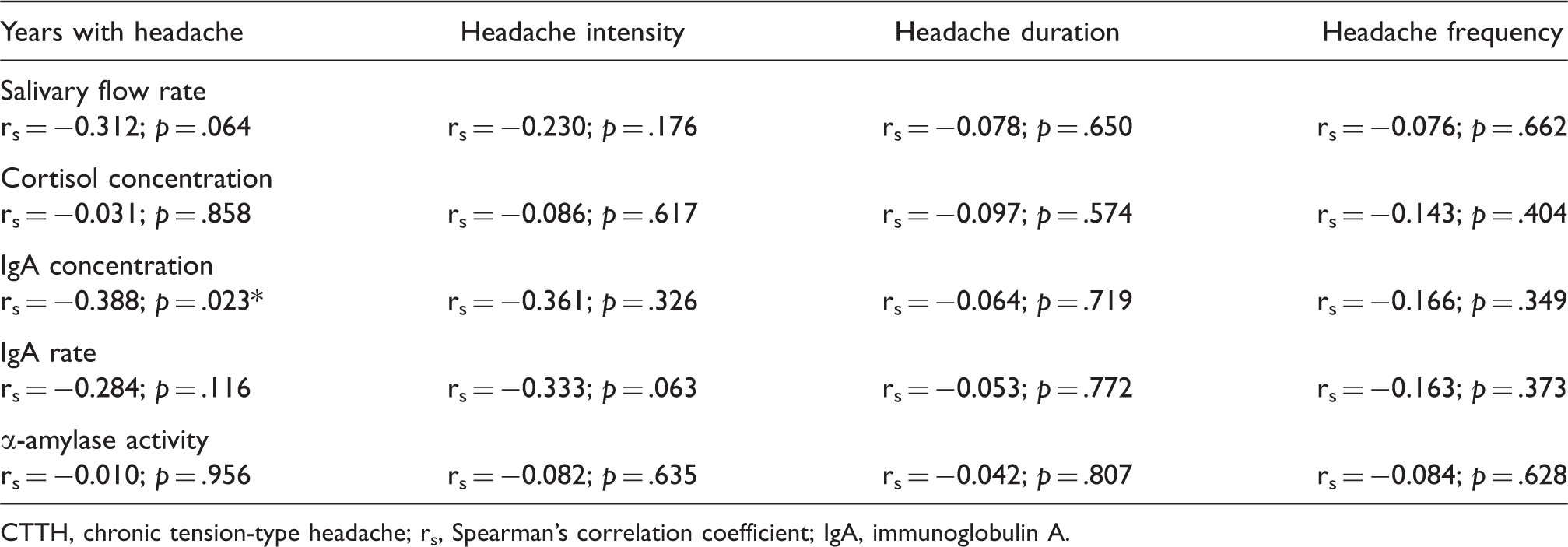
Results: Children with CTTH showed lower IgA concentration (p = .008) and IgA rate (p = .039), but not lower cortisol concentration (p = .447), salivary flow rate (p = .289) or α-amylase activity (p = .559), as compared to healthy children. Neither age (p > .582) nor gender (p > .227) influenced salivary markers. A significant association between the number of years with headache and IgA concentration (rs = − 0.385; p = .023) was found: the greater the number of years with headache, the lower the IgA concentration.
Conclusions: These results suggest that children with CTTH present with deficits in the immune system, but not dysfunction in the HPA axis or SNS. Future studies are needed to elucidate the direction of these relationships.
Introduction
Tension-type headache (TTH) is the most common form of head pain in both adults (1) and children (2). As TTH is problematic for children, new studies are warranted (3,4). Although there has been an increase in knowledge regarding the pathogenesis of TTH pain, the true patho-anatomical mechanisms remain unknown (5).
Various studies have reported that stress can play a relevant role in the development of TTH in adults (6) and children (7,8). It has been demonstrated that stress activates the hypothalamic-pituitary-adrenocortical (HPA) axis and sympathetic nervous (SNS) system (9). Dysfunction of the HPA axis has been identified in patients with chronic pain (10,11) or fibromyalgia syndrome (12). Nevertheless, studies investigating the activation of the HPA axis by salivary markers in headache are scarce. Patacchioli et al. found that women with chronic migraine showed increased cortisol levels as compared to healthy women (13). A recent study demonstrated that salivary α-amylase activity was lower during migraine pain attacks as compared to headache-free periods and to healthy controls (14). Leistad et al. suggested that patients with TTH have an abnormal dynamic activation of the HPA axis during mental stress (15), although basal concentrations of cortisol have not been found to be significantly different between TTH and controls (15,16). Therefore, as dysfunction in the HPA axis and SNS may be present in TTH, further studies are needed to investigate the HPA axis in children with TTH.
Additionally, Heesen and Engler suggested that immune system abnormalities may be present in some primary headaches (17). It is possible that immune system changes could be present in TTH. In fact, an old study found higher concentrations of IgA in individuals with TTH as compared to healthy controls, suggesting that humoral immune system activation might be related to TTH pain (18). Nevertheless, further studies are needed because immunological abnormalities have not been investigated in children with TTH.
To the best of the authors’ knowledge, no study has previously investigated HPA axis function, SNS and immune activity in children with headache. The aims of the current study were (i) to assess the differences in salivary cortisol levels (HPA axis function), α-amylase activity (SNS) and IgA (humoral immune system) concentrations between children with chronic TTH (CTTH) and healthy children; (ii) to analyze the relationship between salivary markers and headache intensity, frequency or duration; and (iii) to determine if salivary cortisol levels, α-amylase activity and IgA levels were related to gender in both groups. We hypothesized that children with CTTH have different cortisol levels, α-amylase activity and IgA concentrations as compared to healthy children.
Materials and methods
Subjects
Consecutive children diagnosed with CTTH by an experienced neuropediatrist from the Neurology Department of Hospital Quirón were screened for eligibility criteria. In all children headache features, temporal profile and family history were assessed. To be included children had to describe all the characteristics typical of CTTH per the criteria of the International Classification of Headache Disorders, second edition (ICHD-II) (19). Only either photophobia or phonophobia was permitted. No children reported vomiting or reported nausea during headaches. Other primary headaches and medication overuse headaches were excluded. No evidence of any secondary headaches could be present. None of the participating children were taking prophylactic drugs at the time of the study.
Additionally, age- and sex-matched children without history of head or neck pain symptoms were recruited from volunteers who responded to a local announcement. Ethical approval was granted by Local Ethics Committee. Informed consent was obtained from both children and parents and procedures were conducted according to the Declaration of Helsinki.
Self-reported measures
Children, with the help of their parents, completed a headache diary for four weeks in order to complement the diagnosis (20). An 11-point numerical pain rating scale (21) (NPRS; range: 0 = no pain, to 10 = maximum pain) was used to assess headache intensity. The headache diary was used to calculate the following variables: (i) headache intensity, calculated from the mean of the NPRS of the days with headache; (ii) headache frequency, calculated by dividing the number of days with headache by the number of the analyzed weeks (days/week); and (iii) headache duration, calculated by dividing the sum of the total hours of headache by the number of days with headache (hours/day).
The Beck Depression Inventory (BDI-II) a 21-item self-report measure assessing affective, cognitive and somatic symptoms of depression, was also used (22). Children, with the help of the parents, were asked to choose from a group of sentences which best described how they had been feeling in the preceding two weeks. For example, to assess sadness, they could choose either: “I do not feel sad”, “I feel sad much of the time”, “I am sad all the time”, or “I am so sad or unhappy that I can not stand it”. The BDI-II has shown good internal consistency (α = 0.86), with higher scores indicating higher levels of depressive symptoms (23,24).
Saliva sample collection
Non-stimulated saliva samples were collected from each child for assessment of HPA axis, SNS and immune system functions according to standardized procedures (25). Saliva collections were made with children seated, leaning forward and with their heads tilted down. Non-stimulated whole saliva was collected into the collection tube (passive drooling technique). The process was done for five minutes. All saliva sampling was performed between 9 and 11
Salivary cortisol and IgA concentrations and α-amylase activity were calculated using a commercial luminescence immune assay (Salimetrics, State College, PA, USA), reading the luminescence units with an automatic luminometer (Sunrise, TECAN Group, Männedorf, Switzerland). IgA was expressed as concentration and also as the range of secretion (IgA concentration * salivary flow rate). All saliva samples were analyzed in a single batch to eliminate inter-assay variance and were measured in duplicate. In fact, adequate intra-assay accuracy was obtained with a coefficient of variance between 5.5% and 7%.
Statistical analysis
Data were analyzed with the R Project for Statistical Computing software (version 2.9.2). Means and standard deviations (SD) and 95% confidence intervals (95% CI) of the data were calculated for each variable. The Kolmogorov-Smirnov test was used to analyze normal distribution of the variables (p > .05). Quantitative data without a normal distribution (IgA rate, IgA concentration and pain history, headache intensity, frequency, duration) were analyzed with non-parametric tests, whereas data with a normal distribution (salivary flow rate, cortisol, α-amylase activity) were analyzed with parametric tests. Differences in salivary flow rate, cortisol concentration and α-amylase activity between groups were assessed with the unpaired student t-test. Differences in IgA concentration and rate were assessed with non-parametric Mann-Whitney U test. Gender was included in the main analysis as a covariate. In addition pair-wise comparisons between genders were also performed with the Mann-Whitney U test in either group. The Spearman’s (rs) correlation coefficient was used to assess the association between endocrine and immunological outcomes and clinical variables. When necessary, data were log transformed to get homogeneity of variance. A p value <.05 was considered statistically significant.
Results
Seventy-five consecutive children presenting with headache between September 2009 and March 2010 were screened for eligibility criteria. Thirty-nine (52%) were excluded: migraine (N = 15), frequent episodic TTH (N = 5), hemicranial headache (N = 2), high levels (BDI-II < 9 point) of depression and anxiety (N = 7). Finally, a total of 36 children, 10 boys and 26 girls, aged 6 to 12 years old (mean: 9 ± 2 years) satisfied inclusion criteria and agreed to participate. In our sample, the headache history was 1.9 years (95% CI 1.6–2.2 years), the mean intensity per episode was 5.0 (95% CI 4.5–5.4), mean headache period per day was 4.1 hours (95% CI 3.4–5.0 hours) and the number of days per week with headache was 4.5 (95% CI 4.1–5.0 days/week). No significant associations between headache intensity, frequency and/or duration (p > 0.529) were found. The BDI-II score of the current sample was 3.2 (95% CI 2.8–3.5). No significant associations between headache parameters and BDI-II were found (p > 0.672). In addition, 36 age- and sex-matched children without headache, 10 boys and 26 girls, aged 6 to 12 years (mean: 9 ± 2 years old) also participated (p = .976).
Differences in salivary flow rate, cortisol concentration, IgA rate, IgA concentration and α-amylase activity between children with CTTH and healthy children

IgA, immunoglobulin A; CTTH, chronic tension-type headache.
Vales are expressed as mean ± standard deviation (95 % confidence interval) for the mean scores and as mean ± standard deviation for difference scores.
Statistically significant differences between groups (student t-test, p < .05).
Within CTTH group a significant negative association between years with headache and IgA concentration (rs = − 0.385; p = .023; Figure 1) was found: the greater the number of years with headache attack, the lower the IgA concentration. No other significant correlations between salivary markers and headache clinical parameters were found (Table 2).
Scatter plots of relationship between duration of CTTH pain and IgA concentrations (N = 36). Note that some points are overlapping. A negative linear regression line is fitted to the data. CTTH, chronic tension type headache; IgA, immunoglobulin A; rs, Spearman’s correlation coefficient. Linear negative correlations between headache clinical parameters and salivary markers in children with CTTH CTTH, chronic tension-type headache; rs, Spearman’s correlation coefficient; IgA, immunoglobulin A.
Salivary flow rate, cortisol concentration, IgA rate, IgA concentration and α-amylase activity in both groups, by gender

CTTH, chronic tension-type headache; IgA, immunoglobulin A.
Values are expressed as mean ± standard deviation.
Discussion
The current study showed lower concentrations of salivary IgA but not different cortisol concentrations or α-amylase activity in children with CTTH as compared to healthy children. IgA concentration was negatively associated with headache history: that is, the greater the years with headache, the lower the IgA concentrations. Finally, neither gender nor age influenced IgA, cortisol or α-amylase activity.
Cortisol concentration has been used for investigating the HPA axis in different widespread pain conditions (10–12), migraine (13), and TTH (15,16) in adults. The current study is the first one, to the authors’ knowledge, analyzing salivary cortisol levels in children with CTTH. We found that cortisol concentrations were not different between children with CTTH and healthy children, a finding that is in agreement with those of previous studies conducted in adults with TTH, where serum plasma cortisol levels, instead of salivary concentration levels, were used (15,16). Salivary cortisol has been found to be a convenient method of assessment which provides a valid and reliable correlation with serum/plasma free diurnal cortisol levels (26,27). In fact, salivary markers of cortisol concentration and α-amylase activity is commonly used to investigate the activation of HPA axis and the SNS (28).
Nevertheless, our study investigated static function of the HPA axis as basal cortisol concentrations were assessed. Previous studies of human cortisol in pain conditions had focused on circadian variations of cortisol release or cortisol responses to acute-induced psychosocial stress. Therefore, future studies investigating changes in cortisol levels as a response to induced potential stress in children with CTTH should be conducted. In fact, a recent study has demonstrated the existence of subgroups of patients with chronic pain who respond with slower salivary cortisol responses to acute-induced psychosocial stress, showing that HPA axis activity may be different in salivary cortisol response (29). We do not know if children with CTTH also exhibit different cortisol responses with induced stress.
Additionally, we did not find significant differences in salivary flow rate and α-amylase activity between children with CTTH and healthy children, suggesting that no impairments in the SNS are present in children with CTTH. Further, no gender differences in α-amylase activity were found, in concordance with previous results (30). These results conflict, however, with those recently found in adults with migraine, where lower α-amylase activity during head pain attacks was found (14). Discrepancies between studies may be related to the headache condition (CTTH versus migraine), age of the sample (children versus adults) and status of the headache during the salivary collection (pain-free versus migraine attack). This study found changes in α-amylase activity depending on the presence or absence of migraine (14). In fact, lower α-amylase activity was related to higher intensity of migraine (14), which is expected because salivary α-amylase activity was correlated with the intensity of the pain (31). We do not know if α-amylase activity would show dynamic fluctuations in children with CTTH, although this would be unexpected, as TTH attacks usually are of less intensity than migraine. In addition, a similar salivary flow rate between both study groups further supports the hypothesis about the absence of an alteration of the SNS in children with CTTH.
The most important finding of the current study was that children with CTTH showed lower concentrations of IgA, which was associated with a longer history of headache. This finding would support the presence of deficits in the immune system in children with CTTH. Our results conflict with those previously found in an earlier study, where higher concentrations of IgA were found in adults with TTH (18). It is possible that differences in sample population (age, no depressive or anxiety symptoms and defined CTTH) are responsible for discrepancies between studies.
Nevertheless, a cause-and-effect relationship cannot be inferred from the current study. In fact, lower IgA concentrations may be a risk factor for development of CTTH or a consequence of the pain (17). For instance, if lower IgA concentration were found to be a risk factor for the development of CTTH, this would explain the high co-morbidity between headache and asthma (32,33), and the fact that the majority of children with headache who attend a pediatric emergency department also suffer from respiratory infectious diseases (34). However, this hypothesis seems unlikely as no children in the current study suffered from asthma. On the other hand, it is more plausible that chronic pain tends to induce immunological defences, which is supported by the fact that IgA concentrations were lower in those children with a longer history of headache. Lower IgA concentrations induced by headache would predispose to higher incidence of respiratory illness and asthma (35,36). Future longitudinal studies are required to further elucidate the causal relationship between changes in the immune system and CTTH in children.
Limitations of the current study should be recognized. First, we included a subgroup of children with CTTH without depressive symptoms (BDI-II < 4). We do not know if these results would be similar in children with migraine or in other subgroups of patients with frequent episodic TTH. In addition, it would be interesting to investigate if the presence of depression or symptoms of anxiety would change our results. However, this is unlikely as it has been demonstrated that abnormalities of the HPA axis function are not explained by accompanying psychological stress as levels of psychological distress did not explain the relationship with salivary cortisol levels in chronic widespread pain (37). Secondly, the design of this study does not allow conclusions to be drawn about the causality of the associations between CTTH and changes found in the immune system. Prospective longitudinal studies are now needed to elucidate this relationship.
Conclusion
We found lower salivary concentrations of IgA but not different cortisol levels or α-amylase activity in children with CTTH as compared to healthy children. IgA concentration was negatively associated with headache history: that is, the greater the number of years with headache, the lower the IgA concentration. Finally, neither gender nor age influenced IgA, cortisol or α-amylase activity. These results suggest that children with CTTH present deficits in the immunological system, but not dysfunction in the HPA axis or the SNS. Prospective longitudinal studies are needed to elucidate these relationships.
Footnotes
Conflict of interest
All authors certify they have no conflicts of interest for this manuscript.
Acknowledgements
This study received funds from a grant conceded by the Universidad Rey Juan Carlos and Comunidad de Madrid (reference CCG10-URJC BIO-5011).
