Abstract
Objective: We pharmacologically characterized pituitary adenylate cyclase–activating polypeptides (PACAPs), vasoactive intestinal peptide (VIP) and the VPAC1, VPAC2 and PAC1 receptors in human meningeal (for their role in migraine) and coronary (for potential side effects) arteries.
Methods: Concentration response curves to PACAP38, PACAP27, VIP and the VPAC1 receptor agonist ([Lys15,Arg16,Leu27]-VIP[1-7]-GRF[8-27]) were constructed in the absence or presence of the PAC1 receptor antagonist PACAP6-38 or the VPAC1 receptor antagonist, PG97269. mRNA expression was measured using qPCR.
Results: PACAP38 was less potent than VIP in both arteries. Both peptides had lower potency and efficacy in meningeal than in coronary arteries, while mRNA expression of VPAC1 receptor was more pronounced in meningeal arteries. PACAP6-38 reduced the Emax of PACAP27, while PG97269 right-shifted the VIP-induced relaxation curve only in the coronary arteries.
Conclusion: The direct vasodilatory effect of VIP and PACAP might be less relevant than the central effect of this compound in migraine pathogenesis.
Introduction
Migraine is a neurovascular disorder associated with activation of the trigeminal cervical complex, release of vasoactive peptides and dilatation of cephalic blood vessels. This vasodilatation seems to be principally caused by calcitonin gene-related peptide (CGRP), which is released after activation of perivascular trigeminal sensory nerves (1,2). Interestingly, other neuropeptides released after activation of the trigeminovascular complex, including pituitary adenylate cyclase–activating polypeptides (PACAPs) and vasoactive intestinal peptide (VIP) (3) have recently been described also to induce headache, and might play a role in the pathophysiology of migraine (4–7).
pKi values of PACAP38 and VIP for the three receptors subtype

pIC50 value. For references, see the database of the International Union of Basic and Clinical Pharmacology (IUPHAR) Committee on Receptor Nomenclature and Drug Classification (http://www.iuphar-db.org/DATABASE).
PACAP38 and VIP have been shown in vivo in humans to decrease mean flow velocity in the middle cerebral artery and to induce vasodilatation in the superficial temporal artery in a similar magnitude (4–7). Moreover, PACAP38 induced migraine-like headache in migraine patients and headache in healthy volunteers (7). However, VIP induced only mild headache in migraine patients and healthy volunteers (5,6). Because PACAP38 displays a higher affinity for the PAC1 receptor, activation of this receptor may result in migraine-like headaches and, accordingly, antagonism of the PAC1 receptor may be a putative target for migraine treatment. On this basis, and in an attempt to extend the knowledge of the mechanisms of these neuropeptides in migraine pathophysiology, the present study set out to investigate in both human meningeal and coronary arteries: (i) the vasorelaxation induced by VPAC/PAC receptor agonists in the absence or presence of VPAC and PAC receptor antagonists; and (ii) the mRNA expression of VPAC/PAC receptors.
Materials and methods
Isolated human arteries
Human meningeal arteries (seven male, six female, age 42–75 years, internal diameter 500–750 µm) were obtained perioperatively from patients undergoing neurosurgical procedures at Erasmus Medical Center, Rotterdam. The right proximal (five male, four female, age 32–63 years, internal diameter 2–3 mm) and distal (16 male, eight female, age 17–61 years, internal diameter 500–1500 µm) coronary arteries were obtained from heart-beating organ donors who died of noncardiac disorders less than 24 hours before the tissue was taken to the laboratory. The hearts were provided by the Rotterdam Heart Valve Bank after donor mediation by Bio Implant Services Foundation/Eurotransplant Foundation (Leiden, The Netherlands) following removal of the aortic and pulmonary valves for homograft valve transplantation (large and small epicardial arteries). Medical histories of the patients or donors were not available due to ethical restrictions. Blood vessels were placed in Krebs bicarbonate buffer solution for meningeal arteries (composition in mM: NaCl 119, KCl 4.7, CaCl2 1.25, MgSO4 1.2, KH2PO4 1.2, NaHCO3 25 and glucose 11.1; pH 7.4) or for coronary arteries (composition in mM: NaCl 118, KCl 4.7, CaCl2 2.5, MgSO4 1.2, KH2PO4 1.2, NaHCO3 25 and glucose 8.3; pH 7.4) aerated with 5% CO2 in O2 (carbogen) and transported to the laboratory for investigation.
Functional experiments
The arteries were cut in small segments of about 2–4 mm each and suspended on stainless-steel hooks in 15–ml organ baths (proximal coronary segments) or Mulvany myographs (meningeal and distal coronary segments) containing oxygenated Krebs bicarbonate solution at 37°C. After equilibration for at least 30 minutes, with change of solution twice at 15–minute intervals, changes in tension were recorded with a Harvard isometric transducer. The vascular segments were stretched to a stable tension of about 15 mN for the proximal segments (the optimal tension as determined in previous studies), or to a tension normalized to 90% of l 100 for the distal segments (the diameter when transmural pressure equals 100 mmHg (14)). The segments were exposed to 30 mM KCl once (distal segments) or twice (proximal segments). After washout, the tissue was exposed to 100 mM KCl to determine the maximum contractile response to KCl.
The relaxant effect of human PACAP38, PACAP27 (a C-truncated 27 amino acid version of PACAP38), VIP and the VPAC1 receptor agonist, ([Lys15,Arg16,Leu27] –VIP[1-7]-GRF[8-27]), were examined by cumulative application of increasing concentrations of the peptide in the absence or presence of the PAC1 receptor antagonist, PACAP6-38 or the VPAC1 receptor antagonist, PG97269. Each segment was precontracted with 30 mM KCl before the agonists were added. Segments were exposed to a single cumulative concentration-response curve and a matched pair’s protocol was used, where one segment acted as control (no antagonist present) while in other segment response to the agonist were measured in presence of the antagonist. Agonist responses were assessed in the presence of precontraction induced by 30 mM KCl following equilibration (20–30 min) with 1 µM of the antagonist. After washout the functional integrity of the endothelium was verified by analysing relaxation to substance P (1 nM) after precontraction with prostaglandin F2α (PGF2α, 1 µM) in proximal coronary artery segments, and by relaxation to substance P (10 nM) after precontraction with the thromboxane A2 analog U46619 (10 nM) in distal coronary and meningeal artery segments as described previously (15,16). Although relaxations to substance P were assessed with different concentrations in proximal (1 nM) and distal (10 nM) coronary artery segments, these different concentrations do not account for the relaxation effect, as determined in previous studies from our group (17).
Compounds
VIP, PACAP38, PACAP27 and PACAP6-38 were obtained from NeoMPS (Strasbourg, France). The VPAC1 receptor agonist ([Lys15,Arg16,Leu27]-VIP[1-]-GRF[8-27]) and the VPAC1 receptor antagonist (PG97-269) were obtained from Phoenix Europe (GmbH, Karlsruhe, Germany). All test substances were dissolved in distilled water and stored at −20°C. Prior to using, the samples were diluted in isotonic saline to the desired concentration, which is thousand-fold higher than the final concentration in the organ bath.
Data presentation and statistical evaluation
The vasodilator responses to the agonists were expressed relative to the precontraction evoked by KCl ( = 100%). For each segment the maximum vasodilator effect (Emax) was calculated. The concentration response curves for all agonists were analysed using nonlinear regression analysis and the potency of agonists was expressed as pEC50 (i.e. negative logarithm of the molar concentration of agonist inducing half the maximum response) using Graph Pad Prism 4.0 (Graph Pad Software, Inc., San Diego, CA, USA). The blocking potency of the antagonists was estimated by calculating EC50 ratios and plotting a Schild plot (18) using linear regression to get the slope value. Because only one concentration of antagonist was studied, the “apparent pKB” values were calculated, constraining the Schild slope to unity. Data are expressed as mean ± standard error of the mean (SEM) and N refers to the number of patients from whom the vessels were collected. Statistical testing was performed using unpaired Student’s t-test, unless indicated otherwise. p values of .05 or less were assumed to denote significant changes.
mRNA expression studies
Primers and target sequences used for the mRNA expression studies

Results
Vascular responses in human meningeal arteries to PACAP38 and VIP in the absence or presence of PACAP6-38 or PG97269
The contraction induced by 100 mM KCl was 11 ± 2 mN (N = 9) and the average endothelium-dependent relaxant response to substance P (10 nM) was 64 ± 5% of the precontraction induced by 10 nM U46619. PACAP38 and VIP both induced relaxation in human meningeal arteries with an Emax of 34 ± 12% of precontraction with 30 mM KCl (N = 7, Figure 1; left panel) and 40 ± 10% (N = 6, Figure 1; right panel) respectively. The pEC50 values of PACAP was significantly less ( < 6.9 ± 0.1) than that of VIP (7.4 ± 0.2). The concentration response curves to PACAP38 and VIP in the presence of PACAP6-38 or PG97269 did not differ from the control responses (Figure 1).
Relaxant effect of PACAP38 (left) and vasoactive intestinal peptide (VIP) (right) on human meningeal arteries that were precontracted with 30 mM KCl in the absence or presence of 1 µM of PACAP6-38 or PG97269. Values given represent mean ± standard error of the mean (SEM), N = 6. PACAP = pituitary adenylate cyclase–activating polypeptide.
Vascular responses in human coronary arteries to PACAP38, PACAP27, VIP and the VPAC1 receptor agonist in the absence or presence of PACAP6-38 or PG97269
In proximal coronary segments, the contraction induced by 100 mM KCl was 47 ± 6 mN and the average endothelium-dependent relaxant response to substance P (1 nM) was 14 ± 6% of the precontraction induced by 1 µM PGF2α (N = 4). VIP induced a relaxation with an Emax value of 17 ± 6% of precontraction with 30 mM KCl (N = 7) in the proximal coronary arteries (data not shown), whereas the other agonists used in this study had no effect.
In distal coronary segments the contraction induced by 100 mM KCl was 11 ± 1 mN (N = 14) and the average endothelium-dependent relaxant response to substance P (10 nM) was 78 ± 3% of the precontraction induced by 10 nM U46619.
PACAP38 induced a relaxation with an Emax of 75 ± 6% of precontraction with 30 mM KCl (N = 5, Figure 2; left upper panel), which was similar to that of PACAP27 and VIP (Emax: 77 ± 9% and 87 ± 7%, respectively; N = 6, Figure 2). In contrast, the relaxation to the VPAC1 receptor agonist (41 ± 9%, N = 6) was significantly lower than that of the other agonists tested. The pEC50 value of VIP (8.4 ± 0.1, N = 6) was significantly higher than that of PACAP38, PACAP27 and the VPAC1 receptor agonist (7.1 ± 0.2, 7.7 ± 0.2 and 7.5 ± 0.2, respectively, N = 4–6).
Relaxant effect of PACAP-38 (upper left), PACAP27 (lower left), vasoactive intestinal peptide (VIP) (upper right) and VPAC agonist (lower right) on human distal coronary arteries that were precontracted with 30 mM KCl in the absence or presence of 1 µM of PACAP6-38 or PG97269. *PACAP6-38 reduced the Emax of the relaxation to PACAP27 (p = .05). # PG97269 induced a significant rightward shift of the concentration response curve to VIP (p = .05). Values given represent mean ± standard error of the mean (SEM), N = 6. PACAP = pituitary adenylate cyclase–activating polypeptides. VPAC = vasoactive pituitary adenylate cyclase. M = Molar.
The PAC1 receptor antagonist PACAP6-38, but not the VPAC1 receptor antagonist PG97269, significantly reduced the Emax of the relaxation to PACAP27 (Figure 2: left lower panel). In contrast, PG97269, but not PACAP6-38, produced a significant reduction of the potency of the concentration response curve to VIP (apparent pKB value of 5.9 ± 0.1; Figure 2; right upper panel). In addition, the relaxation induced by the VPAC1 receptor agonist seemed also to be blocked by PG97269; however, this was not significant (p = .50, Figure 2; right lower panel). Both antagonists did not affect the relaxation induced by PACAP38 (Figure 2; left upper panel) and did not affect the vascular tone per se (data not shown).
Detection of VPAC1, VPAC2 and PAC1 receptor mRNA in human meningeal and coronary arteries
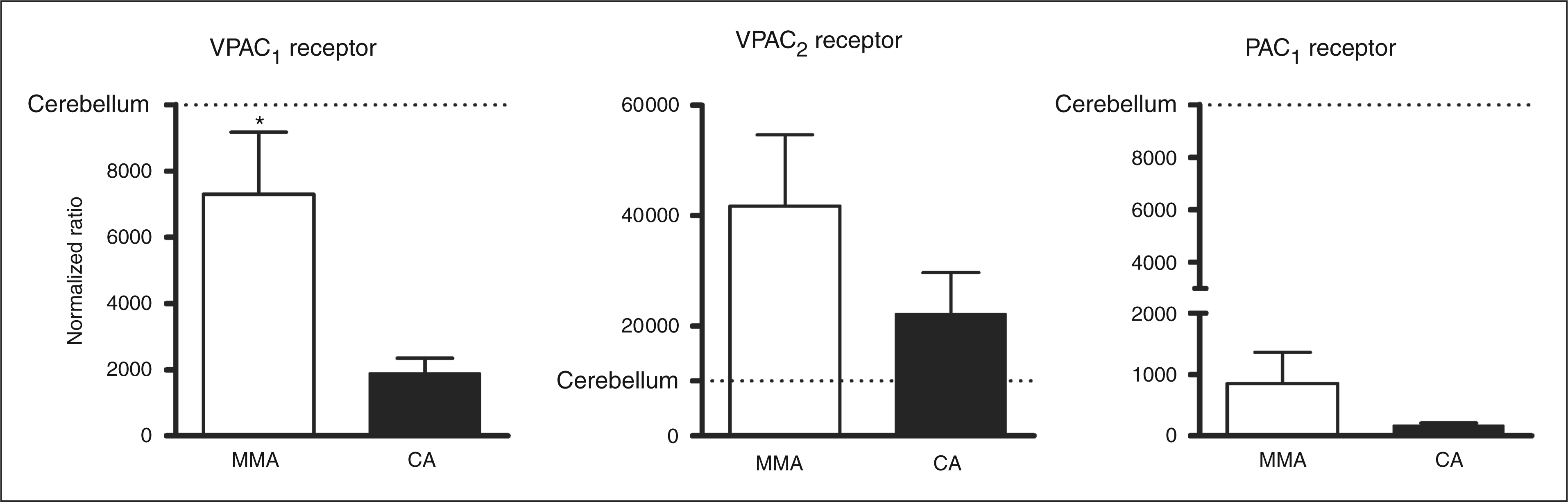
qPCR products were verified by agarose gel analysis, which showed that each qPCR product yielded only one band (Figure 3). The relative mRNA expression of the VPAC1, VPAC2 and PAC1 receptor in human meningeal and coronary arteries was analysed with qPCR, with cerebellum as a reference tissue (Figure 4). The overall expression profile of the three receptors is similar in the two arteries, with the VPAC1 receptor being the only one present in significantly higher quantities in human meningeal arteries (Figure 4; left panel). There seems to be a tendency of higher mRNA expression of the VPAC2 and PAC1 receptor in the human meningeal artery compared to human coronary artery, but due to high individual variability, the difference did not reach statistical significance (Figure 4; middle and right panel). The mRNA expression of VPAC1 receptor in the human meningeal artery was comparable with the cerebellum, while in the human coronary artery, VPAC1 receptor mRNA expression was much lower than the cerebellum (Figure 4; left panel). Similarly, The PAC1 receptor mRNA expressions of both arteries are also lower than that of the cerebellum (Figure 4; right panel). In contrast, mRNA expression of VPAC2 receptor was higher in the analysed arteries than in the cerebellum (Figure 4; middle panel).
qPCR products verified by agarose gel analysis, from the left: 1: β-actin, 2: 100 BP ladder, 3: PAC1, 4: VPAC2, 5: VPAC1, 6: 50 BP ladder. qPCR = quantitative polymerase chain reaction. PAC = pituitary adenylate cyclase. VPAC = vasoactive pituitary adenylate cyclase. bp = base pairs. mRNA expression of VPAC1 (left panel), VPAC2 (middle panel) and PAC1 receptor (right panel) in human middle meningeal arteries (MMA, N = 10) and human coronary arteries (CA, N = 9) expressed as normalized ratios. Values given represent mean ± standard error of the mean (SEM) *p = .05. VPAC = vasoactive pituitary adenylate cyclase.
Discussion
Several lines of evidence have recently suggested the potential role of PACAPs and VIP in the pathophysiology of migraine (3,7,11). It is to be highlighted that PACAP27 and PACAP38 interact with VPAC1, VPAC2 and PAC1 receptors, whereas VIP and the VPAC1 receptor agonist do not interact with PAC1 receptors. Since PACAP38, but not VIP, induces migraine-like headaches in migraine patients (6,7), and only PACAPs interact with the PAC1 receptor, it could be suggested that the PAC1 receptor is involved in migraine pathophysiology. In an attempt to shed further light on this hypothesis, the present study investigated in human meningeal arteries for the first time (i) the vasodilator responses to VPAC/PAC receptor agonists in the absence or presence of VPAC and PAC receptor antagonists; and (ii) the mRNA expression of VPAC/PAC receptors. Although it has been suggested that cranial blood vessel dilatation during migraine is just an epiphenomenon (19,20), there is also ample evidence for a prominent role of cranial blood vessels in the pathophysiology of migraine (21–23). Futhermore, meningeal arteries are of special interest in migraine research as sensitization of dural afferent nociceptors has been proposed to be a pain mechanism during migraine attacks (24).
In the current study, similar experiments were carried out in human coronary arteries, which allowed us to investigate the potential cardiac side effects of antagonists blocking the VPAC/PAC receptors, as investigated for other antimigraine drugs (16).
Apart from the implications discussed below, our study shows that all VPAC/PAC receptor agonists induced vasorelaxation with different efficacy and potency in the human meningeal and coronary arteries. In the proximal coronary artery, only VIP was able to induce a small dilatation of the artery, which is in accordance with a previous study in human epicardial coronary artery (25), while in the distal artery, all the tested agonists induced vasodilatation. Such a difference between proximal and distal coronary artery in VIP-, but not PACAP-induced vasodilatation has also been observed in the rabbit coronary artery (26). Moreover, in this study, PACAP38 and VIP both induced vasodilatation with a lower potency and efficacy in meningeal arteries than in coronary arteries. Possible explanations for these regional differences in vascular responses are tissue-dependent factors such as the levels of receptor protein expression and coupling efficiency of the receptors (8). For example, the potency of PACAP and VIP in meningeal artery is considerably lower than that described earlier in human lenticulostriate and posterior cerebral artery (27). Different vasodilator responses to PACAP and VIP between several tissues were also described in the rabbit posterior cerebral artery and coronary artery (26), as well as in the rat basilar artery and middle cerebral artery (28). The VPAC1 receptor has been suggested to be one of the main receptors responsible for vasodilation elicted by VIP and PACAPs in different vascular beds (29,30). In our hands, both VIP and PACAP were more potent and more efficacious in coronary arteries than in meningeal arteries; hence we expected a higher expression of VPAC1 receptor mRNA in coronary than in meningeal arteries. In contrast, our mRNA expression data show a significantly higher mRNA expression of the VPAC1 receptor in the human meningeal than in the coronary artery. In this context, it should obviously be kept in mind that mRNA expression of a given receptor does not necessarily correlate with its receptor protein expression. Unfortunately, the antibodies we tried were not selective in our hands; therefore we were not able to study protein expression of the receptors involved.
VIP was more potent than PACAP38 in both blood vessels. This observation points to VPAC1 and VPAC2 receptor activation as being primarily responsible for vasodilatation in these arteries, a finding consistent with the low mRNA expression level of the PAC1 receptor. Moreover, lower potency of PACAP on vasodilatation was also found in the rabbit posterior cerebral artery (26) and the rat basilar artery (28), while this difference was not seen in the rat middle meningeal artery (28). This supports the regional differences in vascular responses induced by PACAP and VIP, may probably be assigned to species differences and/or expression of the receptors in vasculature.
In the antagonists experiments, the VPAC1 receptor antagonist PG97269 and the PAC1 receptor antagonist PACAP6-38 did not affect the relaxation to PACAP38 and VIP in human meningeal arteries; this may suggest that such responses are not primarily mediated by VPAC1 and PAC1, but rather via VPAC2 receptors, as is the case in pig urinary bladder neck (31). However, the VPAC2 receptor antagonist, PG99465, did not affect vasorelaxation to VIP on the dural artery of rat in an intravital model (29) and to both peptides in cranial arteries in vitro (28), which may suggest limited vascular involvement of the VPAC2 receptor in cranial arteries. Moreover, PG99465 has also been described as a VPAC1 receptor agonist (9), which limits the use to investigate the effect of the VPAC2 receptor. Another theoretical possibility could be that if two receptors (that is, VPAC/PAC) are involved in the vasorelaxant responses and only one receptor was blocked in each experiment, then activation of the other (unblocked) receptor would overshadow the blockade produced by one antagonist. However, since the potency of these peptides to induce relaxation via the VPAC/PAC receptors is very low in the meningeal arteries compared to the potent vasodilator CGRP (32), blockade of both these receptors will not be very effective in affecting vasorelaxation of the meningeal arteries in migraine.
In the human coronary artery, PG97269 blocked the relaxation to VIP and slightly attenuated the relaxation to the VPAC1 receptor agonist. This suggests that the vasodilatation response is partly mediated via the VPAC1 receptor. The PAC1 receptor antagonist PACAP6-38 did not affect the relaxation to PACAP38, VIP or the VPAC1 receptor agonist, but it reduced the maximal response by PACAP27 at a concentration of 1 µM. Reduction of maximal response suggests that PACAP6-38 might act as a non-competitive or irreversible antagonist on the PAC1 receptor. However, this is unlikely since PACAP6-38 did not reduce the maximal response induced by PACAP38. This discrepancy might be explained by the fact that that vasodilatation induced by PACAP27 and PACAP38 is probably not via the same VPAC/PAC receptor. It has been shown that N-terminus of PACAP has a higher affinity for the VPAC2 receptor than the VPAC1 receptor (33), while the C-terminus of PACAP has a higher affinity for the VPAC1 receptor than the VPAC2 receptor (34). This suggests that PACAP38 generally have a preference to act on the VPAC2 receptor and PACAP27 on the VPAC1 receptor. Since the vasodilatation induced by PACAP27 is blocked by PACAP6-38 and not by PG97269, might suggest that vasodilatation induced by PACAP27 in human coronary arteries is mediated mainly via the PAC1 receptor. In addition, PACAP38 induced vasodilatation is not blocked by PACAP6-38 and not by PG97269, suggesting that the vasodilatation effect of PACAP38 in human coronary artery is most likely mediated via the VPAC2 receptor. More selective and potent agonists and antagonists would be helpful in unequivocal characterization for these receptors.
Based on the above discussion, it can be suggested that vasodilatation induced by PACAP and VIP differ not only in species but also in region of the arteries from the same species. This is probably due to the selective activation of the three type of the VPAC/PAC receptor, which might be tissue dependent. Moreover, several splice variants of the PAC1 receptor, which are to a certain extent tissue specific, has been described to affect cellular function by altering receptor pharmacology and signaling (8). This supports the evidence that the effect of PACAP and VIP differ in tissues. It is unlikely that differences between the VPAC1, VPAC2 and PAC1 receptors between species do account for the different responses, since these receptors are quite well conserved. The homology in amino acid identity between rat and human receptors is: 83.9%, : 84.9% and : 86.7% for VPAC1, VPAC2 and PAC1 receptors, respectively, with consensus position figures being 89.5% for VPAC1, 89.3% for VPAC2 and 90.5% for PAC1. Such homology is considered as highly identical sequences, major differences in functionality would be unlikely.
As previously described, PACAPs and VIP are less potent in human meningeal arteries as compared to CGRP (pEC50 = 8.7 ± 0.1, (32)). This low potency of both PACAP and VIP seems not to be in line with the observation that PACAP38, but not VIP, induced migraine-like headaches in migraine patients (6,7). Furthermore, following PACAP38 infusion migraine-like attacks occur several hours after the peak vasodilatation, and therefore migraine development does not seem to be well correlated with dilatation of cephalic vessels.
Thus, this suggests that the vasodilator properties of the PACAPs, VIP and their receptors’ vascular action may be less relevant in migraine. And that the migraine-like headache induced by PACAP38 may involve other types of modulation in the perivascular space, rather than a vascular mechanism, as also previously suggested (7,19). It has been suggested that migraine-like headache induced by PACAP38 involves sensitization of peripheral sensory trigeminal fibers, since throbbing headache, which had been related to sensitization, have been reported after PACAP38 infusion (7). Moreover, activation of peripheral sensory trigeminal fibers by mast cell degranulation and direct sensitization of central and second order trigeminal neurons have been discussed as a possible mechanism for the migraine-like headache induced by PACAP38 (7). It is noteworthy that the mRNA expression levels of the VPAC2 receptor are higher, whereas those of the PAC1 receptor are much lower in the analyzed arteries in comparison to cerebellum, used as a calibrator. This may indicate that PAC1 receptors are primarily expressed in neuronal tissues, while both VPAC receptors are of greater importance in blood vessels.
Nevertheless, it is still possible that PACAP would indirectly induce vascular effects mediated via activation of perivascular nerves, as PACAP is found in these nerves (11,12). Consistent with these views, the PAC1 receptor antagonist PACAP6-38 but not the VPAC receptor antagonists blocked the vasodilatation induced by neurogenic dural stimulation (35). Moreover, neuronal firing in the trigeminal cervical complex after salivatory nucleus stimulation is also blocked by PACAP6-38, which points to a peripheral effect (35). Taken together, it may be suggested that the PACAP- and VIP-induced vasodilatation seen in healthy volunteers and migraine patient (5–7) is probably mediated by perivascular nerve activation, since direct stimulation with these peptides in human isolated meningeal artery segments induced only minor relaxations. Moreover, PACAP38-induced migraine-like headache is less likely to involve cranial vasodilatation, since only PACAP38, and not VIP, induced migraine-like headache in migraine patients, while both peptides induced cranial vasodilatation (19). Thus, the PAC1 receptor may play a role in activating central mechanisms involved in migraine. Interestingly, PACAP38 infusion in humans causes prolonged dilatation of superficial temporal artery and all the subjects experienced flushing, especially on the face and trunk, which lasted up to 24 hours. Therefore, it cannot be totally ruled out that PACAP38 activates other processes, including vascular mechanisms,
In conclusion, since the potency of the PACAPs and VIP in human meningeal arteries is lower than that of CGRP, the vasodilator properties of the PACAPs and VIP (and their vascular receptors), may be less relevant in migraine. In addition, the migraine-like headaches induced by PACAP38 may not involve meningeal vasodilatation, but may involve central effects of the PAC1 receptor. This study extends the knowledge of PACAP and VIP as well as their receptors in the vascular component of migraine pathogenesis.
